Abstract
Aspiration pneumonia is thought to be associated with a poor outcome in patients with community acquired pneumonia (CAP). However, there has been no systematic review regarding the impact of aspiration pneumonia on the outcomes in patients with CAP. This review was conducted using the MOOSE guidelines: Patients: patients defined CAP. Exposure: aspiration pneumonia defined as pneumonia in patients who have aspiration risk. Comparison: confirmed pneumonia in patients who were not considered to be at high risk for oral aspiration. Outcomes: mortality, hospital readmission or recurrent pneumonia. Three investigators independently identified published cohort studies from PubMed, CENTRAL database, and EMBASE. Nineteen studies were included for this systematic review. Aspiration pneumonia increased in-hospital mortality (relative risk, 3.62; 95% CI, 2.65–4.96; P < 0.001, seven studies) and 30-day mortality (3.57; 2.18–5.86; P < 0.001, five studies). In contrast, aspiration pneumonia was associated with decreased ICU mortality (relative risk, 0.40; 95% CI, 0.26–0.60; P < 0.00001, four studies). Although there are insufficient data to perform a meta-analysis on long-term mortality, recurrent pneumonia, and hospital readmission, the few reported studies suggest that aspiration pneumonia is also associated with these poor outcomes. In conclusion, aspiration pneumonia was associated with both higher in-hospital and 30-day mortality in patients with CAP outside ICU settings.
Similar content being viewed by others
Introduction
The incidence of pneumonia and pneumonia mortality are both greater in the elderly population1,2, and as human life expectancy continues to increase, it is anticipated that pneumonia deaths will also increase. The most common type of pneumonia in the elderly is aspiration pneumonia3,4. Aspiration pneumonia can develop after the inhalation of pathogenic bacteria into the lower respiratory tract, and it can present as ventilator-associated pneumonia, hospital-acquired pneumonia, or as community acquired pneumonia (CAP)5. The prevalence rate of aspiration pneumonia is estimated to be 5% to 24% in subjects with CAP6,7,8,9.
Aspiration pneumonia can be defined as pneumonia in patients who have aspiration risk. Risk factors for aspiration include impaired consciousness, weakness, swallowing difficulties, esophageal dysfunction or mechanical obstruction, and sedation4,10,11. However, overt aspiration is generally not witnessed12,13, and aspiration alone cannot fully explain the development of pneumonia14. Most healthy subjects passively aspirate oropharyngeal secretions during night, but their cough reflex, mucociliary clearance, and immune system usually prevents the development of pneumonia15.
Healthcare-associated pneumonia (HCAP) was defined in the 2005 American Thoracic Society/Infectious Diseases Society of America guidelines to identify patients at increased risk for infection due to multidrug-resistant pathogens in CAP16. While broad spectrum antibiotic therapy has not been shown to improve the outcome of patients with HCAP17,18,19, other host factors including age, comorbidities and aspiration risk are associated with higher mortality17,20,21,22,23. In this systematic review, we selected studies using defined diagnostic criteria for aspiration pneumonia to survey the prevalence of aspiration pneumonia and evaluate the relationship between aspiration pneumonia and mortality, recurrent pneumonia, and repeat hospital admission in subjects with CAP.
Methods
This systematic review was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines24.
Definitions and inclusion criteria
Aspiration pneumonia is defined as pneumonia in subjects with aspiration risk signified by impaired conscious25, neurological disease26,27, esophageal dysfunction or esophageal mechanical obstruction28,29, or aspiration witnessed during eating or vomiting4,10,11. We included studies in adults aged 15 years and older with specific outcomes that included mortality, recurrent pneumonia, or hospital readmission and we compared these in subjects with “non-aspiration” pneumonia defined as CAP in subjects without aspiration risk. In all but one of the studies we identified, aspiration pneumonia was studied as a risk factor in all subjects with CAP including those with HCAP. We could neither perform a multivariate-analysis on a single study, nor could we compare these results with those that combine HCAP with non-HCAP subjects, so this study was excluded from the analysis.
Search criteria
We identified published cohort studies written in English from the PubMed database using the search terms: “community acquired pneumonia [All Fields] OR healthcare associated pneumonia [All Fields] AND aspiration [All Fields]”, from the Cochrane Central Register of Controlled Trials (CENTRAL) database using the search terms: “community acquired pneumonia AND aspiration” and “healthcare associated pneumonia AND aspiration”, and from the EMBASE using the search terms: “community acquired pneumonia AND aspiration” or “healthcare associated pneumonia AND aspiration” (accessed on August 31, 2016). Studies published only in abstract form were excluded because the methods and results could not be fully assessed. Full texts of articles were further evaluated by three investigators (KK, TA and JK).
Data extraction
We extracted the following information from included studies: study design, sample size, inclusion and exclusion criteria, subject demographics, prevalence rate of aspiration pneumonia, type of outcome, type of statistical analysis and other significant predictive factors for each outcome.
Assessing risk of bias
The risk of bias in the included studies was assessed according to the recommendations outlined in the Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. and MOOSE guidelines for the following items: selection, performance, detection, attrition, and publication bias24. Each study included in this review was assessed for quality as good, moderate, or poor based on biases using the modified Hayden’s criteria30. Disagreements among the investigators were resolved by review of the assessments to reach consensus.
Data analysis
We conducted meta-analysis by the outcomes of studies as follows: in-hospital mortality, and 30-day mortality. ICU mortality, long-term mortality, readmission and recurrence, and treatment failure. Outcomes were pooled using Mantel-Haenszel risk ratios, and the precision of the estimates was expressed as the 95% confidence interval (CI). Statistical heterogeneity was assessed using the Higgins I2 tests. A random-effects model was used when significant heterogeneity was found. Publication bias was assessed by examination of funnel plots31. Statistical significance was defined by a P value < 0.05 for all analyses. The meta-analysis was performed with the Review Manager ver. 5.3 software program (The Nordic Cochrane Centre, The Cochrane Collaboration).
Results
Database search and characteristics of included studies
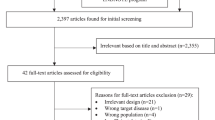
We identified 1065 studies through PubMed, CENTRAL database, EMBASE, and additional studies from review articles. We then excluded 1020 studies as the abstract did not meet the inclusion criteria. We excluded 26 of the remaining 45 records after retrieving and inspecting the full text. The reasons for exclusion were: the population included inappropriate subjects (n = 5)32,33,34,35,36, mismatched outcomes (n = 7)23,37,38,39,40,41,42, no description of aspiration pneumonia or the definition of aspiration pneumonia did not meet a priori criteria (n = 14)43,44,45,46,47,48,49,50,51,52,53,54,55,56. The process of the study selection is shown in Fig. 1.
We finally included 19 studies in this systematic review: a second analysis of a multicenter retrospective international database of patients with CAP using propensity score analysis57, a secondary analysis of a prospective observational study58, four prospective observational studies59,60,61,62, ten retrospective observational studies21,63,64,65,66,67,68,69,70,71, a combined a retrospective derivation cohort and a prospective validation cohort72, two other combined a retrospective and a prospective observational studies73,74. These studies were published from the USA (n = 5), France (n = 4), Japan (n = 4), Spain (n = 3), United Kingdom (n = 1), Canada (n = 1) and Switzerland (n = 1). They were assessed quality based on the modified Hayden’s criteria: good (n = 3)57,58,72, moderate (n = 11)21,61,62,63,64,66,67,69,71,73,74 or poor (n = 5)59,60,65,68,70. The percentage of subjects with aspiration pneumonia ranged from 1.5 to 50.4%. All studies diagnosed aspiration pneumonia on clinical grounds, and 9 studies also required radiographic evidence of involvement of a dependent pulmonary segment21,40,60,62,63,67,68,72,73. Some studies performed video fluoroscopy58,59,71. The wide range reported for the prevalence rate of aspiration pneumonia was, no doubt, influenced by subject selection criteria, how “aspiration pneumonia” was clinically defined, and by the judgment of the clinicians involved in making the diagnosis. Outcomes of these studies were classified as follows: 30-day mortality (n = 5)21,58,60,65,66, in-hospital mortality outside of the ICU (n = 7)57,59,63,64,69,70,71, ICU mortality (n = 4)67,72,73,74, 90-day mortality (n = 1)68, 1-year mortality (n = 2)58,69, all cause readmission (n = 2)58,66, recurrent pneumonia (n = 3)58,61,63, and treatment failure (n = 1)62. Three of 19 studies evaluated multiple outcomes50,58,63. Although all studies except for two57,68 described bacteriological findings, none required the detection of anaerobic bacteria for the definition of aspiration pneumonia.
In-hospital mortality
Seven studies (69,129 subjects) evaluated in-hospital mortality outside of the ICU57,59,63,64,69,70,71. Mortality in patients with aspiration was significantly higher than that in non-aspiration pneumonia (Table 1). Meta-analysis indicated that aspiration risk increased in-hospital mortality (relative risk, 3.62; 95% CI, 2.65–4.96; P < 0.00001; I2 = 86%) as shown in Fig. 2. Most of the included studies individually assessed the impact of aspiration pneumonia using multivariate analysis (Table 1). Three of these studies found that aspiration pneumonia independently increased in-hospital mortality after multivariate adjustments. Hayashi et al. showed that the CURB-65 score; a scoring system for predicting mortality in CAP (HR 1.617; 95% CI 1.236–2.117), and Eastern Cooperative Oncology Group performance status (ECOG PS) (1.476; 1.042–2.090) were associated with higher in-hospital mortality and after adjusting for these, aspiration pneumonia was no longer a risk factor63.
30-day hospital mortality
Five studies (6,042 subjects) analyzed 30-day mortality21,58,60,65,66. The mortality in patients with aspiration risk was significantly higher than that in non-aspiration pneumonia in four of five studies (Table 2). Fernandez et al. included only patients aged over 80 years60. In the meta-analysis including this study, aspiration risk increased in-hospital mortality (relative risk, 3.57; 95% CI, 2.18–5.86; P < 0.00001; I2 = 85%) as shown in Fig. 3. When the study was excluded, the relative risk increased to 4.15 (95% CI, 2.48–7.01; P < 0.00001; I2 = 86%). While two studies indicated that aspiration risk significantly increased 30-day mortality21,65, the other studies did not retain statistical significance after adjustment58,60,66. CURB-65 score (HR 1.495; 95% CI 1.033–2.163)21, moderate or severe liver disease (9.21; 3.16–26.86) and leukocytosis (4.47; 2.27–8.82) were additional risk factors for 30-day mortality65. In studies that showed that aspiration pneumonia was not associated with 30-day mortality in multivariate analysis58,60, congestive heart failure (OR 2.05; 95% CI 1.31–3.20), pneumonia severity index (PSI) score (2.64; 2.01–3.45), and ECOG PS (1.57; 1.34–1.84)58, shock (10.69; 1.33–86.27), respiratory failure (3.50; 1.03–11.96), renal failure (5.83; 2.32–14.68), and Gram-negative pneumonia (20.27; 1.01–410.59)60 were associated with higher 30-day mortality.
ICU mortality
Five studies including 1,644 subjects analyzed ICU mortality67,72,73,74. These studies were all published from the same research group in France. Paradoxically, the mortality in subjects with aspiration pneumonia was significantly lower than that in those with non-aspiration pneumonia in two of these studies (Table 3), and aspiration pneumonia was associated with decreased ICU mortality (relative risk, 0.40; 95% CI, 0.26–0.60; P < 0.00001) (Fig. 4). One of these studies showed that aspiration risk was a better prognostic factor for ICU survival compared to non-aspiration pneumonia in the multivariate analysis72. This study also identified other prognostic factors for ICU mortality including ineffective antimicrobial therapy (prognostic score resulted from validation cohort, +1.5), immunosuppression (+1.38), and higher organ system failure score (OSFS) (+0.64)75.
90-day mortality and one year mortality
One study evaluated 90-day mortality68, and two assessed one year mortality after discharge58,69. Aspiration pneumonia increased one year mortality (40.4% in aspiration pneumonia vs 22.1% in non-aspiration pneumonia, p < 0.001)69. The other studies showed that aspiration pneumonia was significantly associated with 90-day mortality (HR 3.09; 95% CI 1.90–5.03)68 or one-year mortality (HR 1.73; 95% CI 1.15–2.58)58 in each multivariate analysis. Mortensen et al.68 also found that age (1.64; 1.39–1.93), hypothermia (1.90; 1.03–3.49), liver disease (3.88; 1.18–12.70), white blood cell count <400/uL (2.99; 1.12–8.00), serum urea nitrogen level >30 mg/dL (2.44; 1.62–3.68), and arterial oxygen tension <60 mHg (1.99; 1.32–3.00) were associated with increased 90-day mortality.
All cause readmission and recurrent pneumonia
We identified no study specifically evaluating on readmission for pneumonia, but 2 studies assessed all cause readmission rate58,66 and 3 studies analyzed recurrent pneumonia58,61,63 (Table 4). Two studies showed that aspiration risk was associated with increased readmission rate after multivariate analysis, and 2 studies identified that aspiration risk was associated with a higher rate of recurrent pneumonia58,63. One study showed that age (OR, 2.182; 95% CI 1.370–3.475), lack of pneumococcal vaccination (1.909; 1.302–2.798), COPD (1.534; 1.021–2.303) and corticosteroid therapy >20 mg/day (1.971; 1.047–3.713) but not aspiration risk were associated with recurrent pneumonia61.
Treatment failure
One research group analyzed the association between aspiration risk and treatment failure; defined as fever for more than 3 days with clinical deterioration necessitating a change in initial empiric antibiotic therapy, the occurrence of a severe side effect, or death occurring after at least 48 h of antibiotic treatment26. The rate of treatment failure was higher in subjects with aspiration risk than that in non-aspiration pneumonia (48.6% vs 19.1%, p < 0.001), and was associated with aspiration pneumonia (OR, 2.97; 95% CI 1.29–6.86) with neoplasia (3.25; 1.11–9.56), neurological disease (2.34; 1.07–5.13), and elevated monocytes (0.40; 0.20–0.80).
Publication bias
There appeared to be funnel plot asymmetry for in-hospital mortality (Fig. 5) suggesting the possibility of publication bias. Due to small number of studies included in each meta-analysis, Sterne’s test31 was not appropriate for detecting funnel plot asymmetry.
Discussion
This systematic review suggests that aspiration risk is associated with greater in-hospital and 30-day mortality in subjects with CAP except, perhaps, in the ICU setting. Although there are insufficient data to perform a meta-analysis on long-term mortality, recurrent pneumonia, and hospital readmission, the few reported studies suggest that aspiration pneumonia is also associated with these outcomes.
Although aspiration pneumonia was significantly associated with in-hospital mortality when all studies were combined, multivariate analysis in individual studies suggested greater variability (Table 1). For example, Hayashi et al. reported that aspiration pneumonia did not remain as a significant risk factor for in-hospital mortality after multivariate analysis63, but that CURB-65 and ECOG PS increased in-hospital mortality in the same analysis. The CURB-65 includes confusion as a scoring item and the ECOG PS includes weakness as a severity of disability so these scoring systems may, in part, encompass aspiration risk factors. The Fujiki study reported the highest mortality of aspiration pneumonia at 59.1% with an especially strong association with in-hospital mortality (OR 49.9; 95% CI 6.23–398.94)59. Differences in study size, study population, and in the definition of what constitutes aspiration pneumonia may account for these markedly divergent results.
The risk ratio for 30-day mortality (3.57; 95% CI, 2.18–5.86) was similar to that for in-hospital mortality (4.67, 95% CI, 2.59–8.41) most likely because these outcomes are similar. Fernandez et al. focused on patients aged over 80 years and in these subjects, gram-negative pneumonia was the main risk factors for 30-day mortality60. HCAP patients, many of whom are elderly, may be at greater risk of death from aspiration pneumonia, but only one such study has been reported. In that study, aspiration pneumonia in subjects with HCAP was associated with an increased 30-day mortality compared to those with pneumonia but no aspiration (14.8% vs 4.3%, p = 0.025)32.
Paradoxically, in the studies that analyzed mortality in the ICU, aspiration risk was associated with a lower risk of death67,72,73,74. It was hypothesized that the younger study population, the use of intravascular volume repletion, the earlier appreciation and treatment of pneumonia in the ICU and close monitoring might improve prognosis74. It is also possible that patients with pneumonia who are admitted to the ICU are, on the whole, sicker and with comorbidities that might minimize any influence of aspiration on outcomes.
Aspiration risk was associated with all cause readmission, perhaps because many of these subjects were debilitated and had comorbidities58,66. In subjects with weakness or neurologic dysfunction (e.g. stroke), dysphagia and aspiration often continues even with careful medical management. In general, recurrent pneumonia within 3 to 5 years of an episode of CAP occurs in 9 to 12% of subjects with a median time to recurrence of 123 to 317 days and mortality ranging from 4 to 10%76. While some risk factors for aspiration pneumonia result from the natural course of aging77, a multidisciplinary approach might reduce the risk of aspiration pneumonia. El Solh et al. reviewed ways to prevent aspiration pneumonia and concluded that few data were available to guide an evidence-based approach to the prevention using drugs such as angiotensin-converting enzyme inhibitors or capsaicin78. The evidence relating to non-pharmacologic approaches; swallowing rehabilitation, thickening feeds, oral hygiene, gastroesophageal reflux management, and a head-up position are also limited, but the combination approach using pharmacologic and non-pharmacologic methods may be of value in high risk patients79,80.
To summarize our results, aspiration risk is associated with in-hospital and 30 day mortality outside the ICU, with long term mortality, all cause readmission, and recurrent pneumonia. Most of the studies demonstrated that aspiration pneumonia is an independent risk factor for these outcomes. There are several limitations to interpreting these data. All studies were observational and most are retrospective. This has the potential to introduce selection, measurement, and possibly publication bias. Due to the small number of publications for each outcome, we could not conduct Stem’s test or meta-regression analysis for confirm funnel plot asymmetry as a measure of publication bias, however, visual examination of the funnel plots suggested the possibility of bias for in-hospital mortality but not for other outcomes. Although we have provided specific ad hoc definitions for pneumonia and for aspiration risk; these are still somewhat subjective making case ascertainment challenging. If uniform criteria to establish aspiration risk can be developed and accepted for future studies, this will enable well controlled and appropriately powered studies to determine if interventions that can decrease aspiration risk, will also affect morbidity and mortality in this population.
Additional Information
How to cite this article: Komiya, K. et al. Prognostic implications of aspiration pneumonia in patients with community acquired pneumonia: A systematic review with meta-analysis. Sci. Rep. 6, 38097; doi: 10.1038/srep38097 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Chong, C. P. & Street, P. R. Pneumonia in the elderly: a review of severity assessment, prognosis, mortality, prevention, and treatment. South Med J 101, 1134–1140 (2008).
Mylotte, J. M., Goodnough, S. & Naughton, B. J. Pneumonia versus aspiration pneumonitis in nursing home residents: diagnosis and management. J Am Geriatr Soc 51, 17–23 (2003).
Teramoto, S. et al. High incidence of aspiration pneumonia in community- and hospital-acquired pneumonia in hospitalized patients: a multicenter, prospective study in Japan. J Am Geriatr Soc 56, 577–579 (2008).
Marik, P. E. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med 344, 665–671 (2001).
Ottosen, J. & Evans, H. Pneumonia: challenges in the definition, diagnosis, and management of disease. Surg Clin North Am 94, 1305–1317 (2014).
Reza Shariatzadeh, M., Huang, J. Q. & Marrie, T. J. Differences in the features of aspiration pneumonia according to site of acquisition: community or continuing care facility. J Am Geriatr Soc 54 (2006).
Torres, A. et al. Severe community-acquired pneumonia. Epidemiology and prognostic factors. Am Rev Respir Dis 144, 312–318 (1991).
Moine, P., Vercken, J. B., Chevret, S., Chastang, C. & Gajdos, P. Severe community-acquired pneumonia. Etiology, epidemiology, and prognosis factors. French Study Group for Community-Acquired Pneumonia in the Intensive Care Unit. Chest 105, 1487–1495 (1994).
Marrie, T. J., Durant, H. & Yates, L. Community-acquired pneumonia requiring hospitalization: 5-year prospective study. Rev Infect Dis 11, 586–599 (1989).
Hu, X., Lee, J. S., Pianosi, P. T. & Ryu, J. H. Aspiration-related pulmonary syndromes. Chest 147, 815–823 (2015).
van der Maarel-Wierink, C. D., Vanobbergen, J. N., Bronkhorst, E. M., Schols, J. M. & de Baat, C. Risk factors for aspiration pneumonia in frail older people: a systematic literature review. J Am Med Dir Assoc 12, 344–354 (2011).
Kikuchi, R. et al. High incidence of silent aspiration in elderly patients with community-acquired pneumonia. Am J Respir Crit Care Med 150, 251–253 (1994).
Rubin, B. K. “The cruelest lies are often told in silence”. Chest 140, 567 (2011).
Perry, L. & Love, C. P. Screening for dysphagia and aspiration in acute stroke: a systematic review. Dysphagia 16, 7–18 (2001).
Gleeson, K., Eggli, D. F. & Maxwell, S. L. Quantitative aspiration during sleep in normal subjects. Chest 111, 1266–1272 (1997).
Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171 (2005).
Ewig, S., Welte, T. & Torres, A. Is healthcare-associated pneumonia a distinct entity needing specific therapy? Curr Opin Infect Dis 25, 166–175 (2012).
Ewig, S., Welte, T., Chastre, J. & Torres, A. Rethinking the concepts of community-acquired and health-care-associated pneumonia. Lancet Infect Dis 10, 279–287 (2010).
Grenier, C. et al. Impact of guideline-consistent therapy on outcome of patients with healthcare-associated and community-acquired pneumonia. J Antimicrob Chemother 66 (2011).
Chalmers, J. D. et al. Epidemiology, antibiotic therapy, and clinical outcomes in health care-associated pneumonia: a UK cohort study. Clin Infect Dis 53, 107–113 (2011).
Komiya, K. et al. Impact of aspiration pneumonia in patients with community-acquired pneumonia and healthcare-associated pneumonia: a multicenter retrospective cohort study. Respirology 18, 514–521 (2013).
Chalmers, J. D., Rother, C., Salih, W. & Ewig, S. Healthcare-associated pneumonia does not accurately identify potentially resistant pathogens: a systematic review and meta-analysis. Clin Infect Dis 58 (2014).
Carratala, J. et al. Health care-associated pneumonia requiring hospital admission: epidemiology, antibiotic therapy, and clinical outcomes. Arch Intern Med 167, 1393–1399 (2007).
Stroup, D. F. et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. J Am Med Assoc 283, 2008–2012 (2000).
Adnet, F. & Baud, F. Relation between Glasgow Coma Scale and aspiration pneumonia. Lancet 348, 123–124 (1996).
Priefer, B. A. & Robbins, J. Eating changes in mild-stage Alzheimer’s disease: a pilot study. Dysphagia 12, 212–221 (1997).
Thomas, F. J. & Wiles, C. M. Dysphagia and nutritional status in multiple sclerosis. J Neurol 246, 677–682 (1999).
O’Neill, O. M., Johnston, B. T. & Coleman, H. G. Achalasia: a review of clinical diagnosis, epidemiology, treatment and outcomes. World J Gastroenterol 19, 5806–5812 (2013).
Enzinger, P. C. & Mayer, R. J. Esophageal cancer. N Engl J Med 349, 2241–2252 (2003).
Hayden, J. A., Cote, P. & Bombardier, C. Evaluation of the quality of prognosis studies in systematic reviews. Ann Intern Med 144, 427–437 (2006).
Sterne, J. A., Egger, M. & Smith, G. D. Systematic reviews in health care: Investigating and dealing with publication and other biases in meta-analysis. BMJ 323, 101–105 (2001).
Fukuyama, H., Yamashiro, S., Tamaki, H. & Kishaba, T. A prospective comparison of nursing- and healthcare-associated pneumonia (NHCAP) with community-acquired pneumonia (CAP). J Infect Chemother 19, 719–726 (2013).
Oshitani, Y., Nagai, H., Matsui, H. & Aoshima, M. Reevaluation of the Japanese guideline for healthcare-associated pneumonia in a medium-sized community hospital in Japan. J Infect Chemother 19, 579–587 (2013).
Giannella, M. et al. Pneumonia treated in the internal medicine department: focus on healthcare-associated pneumonia. Clin Microbiol Infect 18, 786–794 (2012).
Park, D. R. et al. The etiology of community-acquired pneumonia at an urban public hospital: influence of human immunodeficiency virus infection and initial severity of illness. J Infect Dis 184, 268–277, doi: 10.1086/322040 (2001).
Falcone, M., Corrao, S., Licata, G., Serra, P. & Venditti, M. Clinical impact of broad-spectrum empirical antibiotic therapy in patients with healthcare-associated pneumonia: a multicenter interventional study. Intern Emerg Med 7, 523–531 (2012).
Artero, A. et al. Ertapenem therapy for pneumonia requiring hospital admission in elderly people. Rev Esp Quimioter 29, 8–14 (2016).
Morimoto, K. et al. The burden and etiology of community-onset pneumonia in the aging Japanese population: a multicenter prospective study. PLoS One 10 (2015).
Ishida, T. et al. Clinical characteristics of nursing and healthcare-associated pneumonia: a Japanese variant of healthcare-associated pneumonia. Intern Med 51, 2537–2544 (2012).
Garcia-Vidal, C. et al. Low incidence of multidrug-resistant organisms in patients with healthcare-associated pneumonia requiring hospitalization. Clin Microbiol Infect 17 (2011).
Skull, S. A. et al. Hospitalized community-acquired pneumonia in the elderly: an Australian case-cohort study. Epidemiol Infect 137, 194–202 (2009).
Mody, L., Sun, R. & Bradley, S. F. Assessment of pneumonia in older adults: effect of functional status. J Am Geriatr Soc 54, 1062–1067 (2006).
Ishida, T. et al. Clinical characteristics of pneumonia in bedridden patients receiving home care: a 3-year prospective observational study. J Infect Chemother 21, 587–591 (2015).
Kurashi, N. Y., al-Hamdan, A., Ibrahim, E. M., al-Idrissi, H. Y. & al-Bayari, T. H. Community acquired acute bacterial and atypical pneumonia in Saudi Arabia. Thorax 47, 115–118 (1992).
Karalus, N. C. et al. Community acquired pneumonia: aetiology and prognostic index evaluation. Thorax 46, 413–418 (1991).
Torres, O. H. et al. Outcome predictors of pneumonia in elderly patients: importance of functional assessment. J Am Geriatr Soc 52, 1603–1609 (2004).
Kothe, H. et al. Outcome of community-acquired pneumonia: influence of age, residence status and antimicrobial treatment. Eur Respir J 32, 139–146 (2008).
Zalacain, R. et al. Community-acquired pneumonia in the elderly: Spanish multicentre study. Eur Respir J 21, 294–302 (2003).
Marrie, T. J. & Wu, L. Factors influencing in-hospital mortality in community-acquired pneumonia: a prospective study of patients not initially admitted to the ICU. Chest 127, 1260–1270 (2005).
Kaplan, V. et al. Pneumonia: still the old man’s friend? Arch Intern Med 163, 317–323 (2003).
El-Solh, A. A., Sikka, P., Ramadan, F. & Davies, J. Etiology of severe pneumonia in the very elderly. Am J Respir Crit Care Med 163, 645–651 (2001).
Fine, M. J. et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 336, 243–250 (1997).
Conte, H. A., Chen, Y. T., Mehal, W., Scinto, J. D. & Quagliarello, V. J. A prognostic rule for elderly patients admitted with community-acquired pneumonia. Am J Med 106, 20–28 (1999).
Lim, W. S. & Macfarlane, J. T. Defining prognostic factors in the elderly with community acquired pneumonia: a case controlled study of patients aged > or = 75 yrs. Eur Respir J 17, 200–205 (2001).
Rello, J., Rodriguez, R., Jubert, P. & Alvarez, B. Severe community-acquired pneumonia in the elderly: epidemiology and prognosis. Study Group for Severe Community-Acquired Pneumonia. Clin Infect Dis 23, 723–728 (1996).
Simonetti, A. et al. Timing of antibiotic administration and outcomes of hospitalized patients with community-acquired and healthcare-associated pneumonia. Clin Microbiol Infect 18, 1149–1155 (2012).
Lanspa, M. J. et al. Characteristics associated with clinician diagnosis of aspiration pneumonia: a descriptive study of afflicted patients and their outcomes. J Hosp Med 10, 90–96 (2015).
Taylor, J. K., Fleming, G. B., Singanayagam, A., Hill, A. T. & Chalmers, J. D. Risk factors for aspiration in community-acquired pneumonia: analysis of a hospitalized UK cohort. Am J Med 126, 995–1001 (2013).
Fujiki, R., Kawayama, T., Ueyama, T., Ichiki, M. & Aizawa, H. The risk factors for mortality of community-acquired pneumonia in Japan. J Infect Chemother 13, 157–165 (2007).
Fernandez-Sabe, N. et al. Community-acquired pneumonia in very elderly patients: causative organisms, clinical characteristics, and outcomes. Medicine (Baltimore) 82, 159–169 (2003).
Garcia-Vidal, C. et al. Aetiology of, and risk factors for, recurrent community-acquired pneumonia. Clin Microbiol Infect 15, 1033–1038 (2009).
Genne, D. et al. Analysis of factors that contribute to treatment failure in patients with community-acquired pneumonia. Eur J Clin Microbiol Infect Dis 25, 159–166 (2006).
Hayashi, M. et al. Clinical features and outcomes of aspiration pneumonia compared with non-aspiration pneumonia: a retrospective cohort study. J Infect Chemother 20, 436–442 (2014).
Marrie, T. J., Carriere, K. C., Jin, Y. & Johnson, D. H. Factors associated with death among adults <55 years of age hospitalized for community-acquired pneumonia. Clin Infect Dis 36, 413–421 (2003).
Lanspa, M. J., Jones, B. E., Brown, S. M. & Dean, N. C. Mortality, morbidity, and disease severity of patients with aspiration pneumonia. J Hosp Med 8, 83–90 (2013).
Jaoude, P., Badlam, J., Anandam, A. & El-Solh, A. A. A comparison between time to clinical stability in community-acquired aspiration pneumonia and community-acquired pneumonia. Intern Emerg Med 9, 143–150 (2014).
Leroy, O. et al. A five-year study of severe community-acquired pneumonia with emphasis on prognosis in patients admitted to an intensive care unit. Intensive Care Med 21, 24–31 (1995).
Mortensen, E. M. et al. Causes of death for patients with community-acquired pneumonia: results from the Pneumonia Patient Outcomes Research Team cohort study. Arch Intern Med 162, 1059–1064 (2002).
Hsu, J. L., Siroka, A. M., Smith, M. W., Holodniy, M. & Meduri, G. U. One-year outcomes of community-acquired and healthcare-associated pneumonia in the Veterans Affairs Healthcare System. Int J Infect Dis 15, e382–387 (2011).
Riquelme, R. et al. Community-acquired pneumonia in the elderly: A multivariate analysis of risk and prognostic factors. Am J Respir Crit Care Med 154, 1450–1455 (1996).
Nakagawa, N. et al. Comparison of clinical profile in elderly patients with nursing and healthcare-associated pneumonia, and those with community-acquired pneumonia. Geriatr Gerontol Int 14, 362–371 (2014).
Leroy, O. et al. Severe community-acquired pneumonia in ICUs: prospective validation of a prognostic score. Intensive Care Med 22, 1307–1314 (1996).
Georges, H. et al. Epidemiological features and prognosis of severe community-acquired pneumococcal pneumonia. Intensive Care Med 25, 198–206 (1999).
Leroy, O. et al. Community-acquired aspiration pneumonia in intensive care units. Epidemiological and prognosis data. Am J Respir Crit Care Med 156, 1922–1929 (1997).
Knaus, W. A., Draper, E. A., Wagner, D. P. & Zimmerman, J. E. Prognosis in acute organ-system failure. Ann Surg 202, 685–693 (1985).
Dang, T. T., Majumdar, S. R., Marrie, T. J. & Eurich, D. T. Recurrent pneumonia: a review with focus on clinical epidemiology and modifiable risk factors in elderly patients. Drugs Aging 32, 13–19 (2015).
Komiya, K. et al. Physicians’ attitudes toward the definition of “death from age-related physical debility” in deceased elderly with aspiration pneumonia. Geriatr Gerontol Int 13, 586–590 (2013).
El Solh, A. A. & Saliba, R. Pharmacologic prevention of aspiration pneumonia: a systematic review. Am J Geriatr Pharmacother 5, 352–362 (2007).
Teramoto, S., Yoshida, K. & Hizawa, N. Update on the pathogenesis and management of pneumonia in the elderly-roles of aspiration pneumonia. Respir Investig 53, 178–184 (2015).
Puisieux, F. et al. Swallowing disorders, pneumonia and respiratory tract infectious disease in the elderly. Rev Mal Respir 28, e76–93 (2011).
Acknowledgements
This systematic review was performed in the process of composing Japanese Respiratory Society Practical Guideline for Pneumonia. We would like to express our appreciation for all the member of the Committee of the Guideline.
Author information
Authors and Affiliations
Contributions
K.K., B.K.R. and J.K. reviewed and prepared the manuscript. K.K., J.K., H.M., H.M., T.A., N.A., H.T., S.N., N.S. and S.K. contributed to the protocol design. K.K. T.A. and J.K. performed manuscript screening, data extraction and analyses. O.T. designed the data extraction form. K.K. and J.K. are guarantors of this manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Komiya, K., Rubin, B., Kadota, Ji. et al. Prognostic implications of aspiration pneumonia in patients with community acquired pneumonia: A systematic review with meta-analysis. Sci Rep 6, 38097 (2016). https://doi.org/10.1038/srep38097
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep38097
This article is cited by
-
Advancing healthcare through thoracic ultrasound research in older patients
Aging Clinical and Experimental Research (2023)
-
Factors associated with gravity-dependent distribution on chest CT in elderly patients with community-acquired pneumonia: a retrospective observational study
Scientific Reports (2022)
-
High-resolution computed tomography features associated with differentiation of tuberculosis among elderly patients with community-acquired pneumonia: a multi-institutional propensity-score matched study
Scientific Reports (2022)
-
An uncommon etiological factor for aspiration pneumonitis caused by spontaneous sphenoid sinus meningoencephalocele with cerebrospinal fluid rhinorrhea: a case report
BMC Pulmonary Medicine (2021)
-
Pneumonia
Nature Reviews Disease Primers (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.