Abstract
Accumulated capsaicinoid content and increased fruit size are traits resulting from Capsicum annuum domestication. In this study, we used a diverse collection of C. annuum to generate 66,960 SNPs using genotyping by sequencing. The study identified 1189 haplotypes containing 3413 SNPs. Length of individual linkage disequilibrium (LD) blocks varied along chromosomes, with regions of high and low LD interspersed with an average LD of 139 kb. Principal component analysis (PCA), Bayesian model based population structure analysis and an Euclidean tree built based on identity by state (IBS) indices revealed that the clustering pattern of diverse accessions are in agreement with capsaicin content (CA) and fruit weight (FW) classifications indicating the importance of these traits in shaping modern pepper genome. PCA and IBS were used in a mixed linear model of capsaicin and dihydrocapsaicin content and fruit weight to reduce spurious associations because of confounding effects of subpopulations in genome-wide association study (GWAS). Our GWAS results showed SNPs in Ankyrin-like protein, IKI3 family protein, ABC transporter G family and pentatricopeptide repeat protein are the major markers for capsaicinoids and of 16 SNPs strongly associated with FW in both years of the study, 7 are located in known fruit weight controlling genes.
Similar content being viewed by others
Introduction
The pepper genus Capsicum originated in Bolivia and consists of 25 to 30 species; five are domesticated: C. annuum, C. baccatum, C. chinense, C. frutescens, and C. pubescens1,2. C. annuum is the most popular and was first introduced from the West Indies to Europe in March 1493, with the first travels of Christopher Columbus3,4. Trade routes between Europe, Middle-East and Asia promoted additional introductions and reciprocal exchanges, so multiple introductions were rapidly cultivated in most tropical, Mediterranean and temperate regions of the world. In these secondary diversification centers, thousands of landraces have been selected for 4 to 5 centuries by growers to fit new environments and local consumption habits and trade, which has resulted in wide phenotypic diversity of pepper cultivars5,6,7.
For many crop species, identification and utilization of single nucleotide polymorphisms (SNPs) have become economical because of the availability and abundance of various high-throughput technologies. Recently, three whole-genome sequence (WGS) drafts of hot pepper were released for public use, which opened up unique opportunities for pepper research. The WGS for C. annuum cv. CM334 is 650.2 Gb (186.6× genome coverage)8. WGS for Zunla-1 (C. annuum L.) and its wild progenitor Chiltepin (C. annuum var. glabriusculum) were made available by Qin et al.9. An examination of population structure, diversity at the molecular level, linkage disequilibrium (LD) distribution across various chromosomes and quantitative trait loci (QTL) estimated by model-based association mapping would provide insights into the evolution of various traits among C. annuum cultivars7. LD distribution information across the C. annuum genome will help group SNPs into haplotypes and, their use in genome analysis can lead to understanding the consequences of selection and breeding histories across the collections of C. annuum L.
Genome-wide association study (GWAS) with SNPs generated by genotyping by sequencing (GBS) has been widely used in all major crops including maize, rice, barley, tomato, wheat, sorghum, soybean, watermelon and several other important plant species10,11,12,13,14 and found effective for mining new genes; however, the population structure must be resolved accurately to reduce spurious associations because of confounding effects of subpopulations. The current research aims to identify genomic segments linked to various fruit traits and capsaicin accumulation in diverse collections of C. annuum. Despite several QTL studies of pepper15,16,17,18,19,20, the current study is unique in that it utilizes WGS drafts for anchoring SNPs and systematic GWAS pipelines to identify SNP markers for various fruit-related traits with special reference to capsaicinoid content and fruit weight (FW). It aims to identify genome-wide effects on capsaicinoid content and fruit weight (FW) in C. annuum populations by using SNPs identified by GBS.
Results
SNP development
A total of 250,493,661 reads were available for analysis after quality trimming and 56.0% of these were aligned to unique positions and 24% to multiple positions on the physical map. Accession-wise reads are presented in Table S2. A total of 66,960 SNPs were identified from the reads obtained for the 94 diverse C. annuum accessions studied; 7678, 7751, 8201, 6285, 4724, 6087, 5230, 3873, 3826, 4279, 3768 and 5268 SNPs were mapped to the WGS draft (Kim et al.8) and located on chromosomes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 and 12, respectively. When screened for minor allele frequency (MAF) of 0.05% and call rate > 95%, total SNPs were reduced to 7,331 with 839, 683, 1007, 459, 566, 715, 464, 423, 479, 641, 543 and 512 on chromosomes 1 to 12, respectively. In addition, we identified 2,521 SNPs (containing MAF 0.05) located in various exons. SNPs in exons of various genes were 288, 295, 346, 131, 183, 231, 162, 153, 154, 218, 164 and 196 on chromosomes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 and 12, respectively. We noted presence of one SNP at every 40.7 kb across the genome, with average gap size of 26.08 kb, and one SNP at every 78.8 kb in the coding regions. After excluding for MAF present at 0.05%, we noted one SNP at every 172.2 kb across the whole genome and one SNP every 386 kb in the exons. Heterozygosity (Table S3) ranged from 18% to 3% among various accessions. Sangria, Jyothi, Hungarian semi hot, Tormenta hot, Costeno Amarillo, Cayenne Purple, PI439355, Watermelon, Numex R Naky, Jalapeno pepper, Prik ki nu and Red Rocket were the most highly heterozygous, from 18% to 9%. Mean heterozygosity was 6%, so the accessions in the study were generally homozygous at most of the polymorphic loci and hence suitable for GWAS analysis. The mean MAF was 1892 (range 2711 [Tepin Guatemala] to 1115 [Pimento Sweet Apple]).
Population stratification and effect of capsaicin content and FW
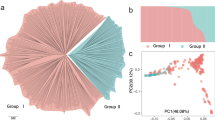
We used PCA of the 7,331 SNPs to classify domesticated and wild C. annuum peppers. Two PCA figures based on capsaicin content (CA) and FW classifications were created to understand the relationships of accessions. This analysis produced a close cluster of hot peppers on CA-PCA and bell peppers as separate cluster (Fig. 1). Sangria, Bellingrath Gardens, Prik ki nu, Pepperoncini, Red Rocket, Hot Red Rocket, Jwala, PI 439355, Jyothi, Hinkelhatz pepper, PI 636424, De Arbol, Goat’s Weed, Costeno Amarillo, Cayenne Purple and Yellow Peter were clustered around the wild accessions. Similarly, three serrano peppers, namely Mexican serrano, Serrano pepper and Serrano Tampiqueno, were grouped together and distant from the rest of the accessions. PCA placement of the various accessions can be noted from the eigen values presented in Table S4. To validate the results of PCA, we used a model-based approach for population structure analysis to analyze the entire panel of 94 C. annuum accessions grouped by CA and FW (Fig. 2). Use of Structure Harvester provided Delta K values, which showed K-4 as the most appropriate.
Principal component analysis (PCA) showing distribution of global Capsicum annuum accession collections with 7, 331 single nucleotide polymorphisms (SNPs).
(A) PCA with accessions grouped by capsaicin content. (B) PCA with accessions grouped by fruit weight. See Table S2 for a list of accessions and for respective eigen values to locate individual accessions on the graph.
Admixture in subpopulations resolved using 7, 331 single nucleotide polymorphisms (SNPs) along with capsaicin content (high, medium and low) (B) and SNPs and Fruit weight (large, medium and small) among C. annuum accession collections by population structure, a model-based approach.
(A) K4 had the highest peak (based on Delta K distribution), so four clusters sufficiently define C. annuum population structure based on capsaicin content and fruit weight.
We examined allele sharing across the panel by calculating identity by state (IBS) coefficients among all pairs of accessions (Fig. 3). Allele sharing clearly tracks subpopulation ancestry as identified by PCA and Structure outputs. The mean observed IBS sharing was greatest for large-sized sweet peppers (0.97) as compared with small-sized hot peppers (0.85), with relatively little IBS sharing between high and low capsaicin-containing peppers (0.54 ± 0.94). The highest IBS sharing among high and low capsaicin content types was between Burning Bush and Chimayo (0.94) and the lowest between Tepin Guatemala and Black Hungarian (0.55). For FW, the mean IBS for small and large types was 0.70 ± 0.06. Most of the admixture occurred within small-sized hot peppers and large bell peppers indicating strong artificial selection for fruit size.
Population differentiation and signals of positive selection
The allele fixation index (FST) between domesticated and wild accessions was 0.10. The FST between low and high, low and medium, and medium and high capsaicin types was 0.08, 0.02 and 0.03, respectively, and that between small and large, small and medium, and medium and large FW types was 0.15, 0.03 and 0.07, respectively. The highest FSTwas noted for SNPs on chromosomes 1, 2, 4, 6, 7 and 8, so these regions are under positive selection. We annotated 122 regions showing strong positive selection and identified important genes for FW (Table S5). When comparing significant (P < 0.001) pairwise FST distribution of large and small FW accessions across the genome, we identified genomic areas with selective sweep signatures that are important for FW (Figs 4 and 5). A segment of 177 Mb was under strong selection sweep on chromosome 11. This sweep was from 34,758,394 to 211,919,750 Mb. In total, 659 genes located in this sweep area have important roles in transmembrane transport of lipids, intercellular carbohydrate transport, carbohydrate metabolic process and carbohydrate transport, ATP binding, flavonoid biosynthesis/auxin response, RNA splicing, nucleic acid binding protein, transcription regulation, glutathione metabolic process, positive regulation of GTPase activity, and other functions (Tables S6 and S7).
Analysis of LD
Haplotype distribution is important in comparing common and unique patterns of genetic variation of C. annuum gene pools and has a wide range of applications. The two major processes that shape haplotype structure are the domestication process and breeding history. We used “Minimize historical recombination”, a block-defining algorithm developed by Gabriel et al.21 to define haplotypes of various lengths. The upper confidence bound was set to 0.98 and the lower bound was set to 0.70. SNPs below MAF of 0.05 were skipped. Maximum block length was set to 160Kb. The EM (Expectation Maximization) algorithm was used for haplotype estimation with convergence tolerance 0.0001 and frequency threshold of 0.01. Maximum EM iterations were set to 50. The current study identified 1189 haplotypes containing 3413 SNPs with a range of 9 to 2 SNPs per haplotype (Table S8).We conducted an extensive LD analysis on the entire dataset of 94 C. annuum collections, on all adjacent marker pairs within a chromosome or within a haplotype block. The results provided values for both the expectation-maximization (EM) algorithm22 and composite haplotype method (CHM)23. R2 (squared-allele frequency correlations) and D’ (LD estimate) values for the EM and CHM methods are in Table S9. We created LD plots using marker-pair associations of adjacent SNPs within a chromosome, within a haplotype block, and within genes (Fig. 6A, B, and C). Length of individual LD blocks varied along chromosomes, with regions of high and low LD interspersed (Fig. 7). Pairwise LD was estimated by r2 and we compared the pattern of decay at different levels. First, with pair-wise analysis considering SNPs across chromosomes, we noted LD decay on average, with an average block size of 139 kb (Fig. 6A). Second, analysis based on adjacent SNPs within haplotypes revealed LD decay within 28 kb (Fig. 6B). We performed genomewide haplotype analysis and identified 1209 haplotypes (Table S10) Third, analysis of SNPs located in exons revealed mean LD decay within 1 kb (Fig. 6C). For LD analysis on chromosome 11, we noted that an entire region under sweep was also under high LD (Fig. 8). Genes under sweep and high LD included CA11g07400 (AP-1 complex subunit gamma-1 with the biological process of intracellular protein transport), with LD 10.2 kb, followed by CA11g09160 (ankyrin-like protein, an acyltransferase), with LD 4.9 kb, and CA11g09970 (flavonol synthase/flavanone 3-hydroxilase), with LD 3.3 kb (Table S7).
GWAS to locate QTL for capsaicin and dihydrocapsaicin content
We used a GWAS with 7,331 SNPs to identify alleles that affect capsaicin and dihydrocapsaicin content (Fig. 7; individual SNP associations along with the details of major and minor allele frequencies and magnitude of associations are in Tables S10, S11, and S12; detailed annotations for all associated SNP markers are in Tables 1 and 2. We found 30 and 56 SNPs associated with capsaicin and dihydrocapsaicin content, respectively; 14 were common to both traits (Fig. 9). Average variation (%) explained per chromosome varied from 11.6 to 17.5 for capsaicin content and 12.9 to 16.9 for dihydrocapsaicin content.
Manhattan plot of the genome-wide association study for fruit weight and capsaicinoids (capsaicin and dihydrocapsaicin).
Chromosome coordinates are displayed along the X-axis, with the negative log-10 of the association P-value for each SNP on the Y-axis. Higher negative log-10 indicates stronger association with the trait. Venn diagrams are of the unique and common significantly associated SNPs for capsaicin and dihydrocapsaicin content and fruit weight in 2011 and 2012.
Significantly associated SNPs for both capsaicin and dihydrocapsaicin content were S1_31111874 (-log10 P = CA 6.6 and DCA 6.9), S3_211558976 -log10 P = CA 4.0 and DCA 3.3), S5_215972421 (-log10 P = CA 3.6 and DCA 3.9), S5_227837981 (-log10 P = CA 3.4 and DCA 3.8), S5_229634509 (-log10 P = CA 3.6 and DCA 4.5), S6_203416571 (-log10 P = CA 3.9 and DCA 3.5), S10_156251204 (-log10 P = CA 3.0 and DCA 5.8), S10_172735351 (-log10 P = CA 3.1 and DCA 4.0), S10_221317647 (-log10 P = CA 4.0 and DCA 3.9), S10_225598553 (-log10 P = CA 3.9 and DCA 3.5), S11_83592400 (-log10 P = CA 3.4 and DCA 3.7), S11_85543247 (-log10 P = CA 4.2 and DCA 6.3), S11_85543251 (-log10 P = CA 3.6 and DCA 5.3), and S11_85543257 (-log10 P = CA 4.2 and DCA 6.3).
GWAS to locate QTL for FW
Despite the high variation noted for FW across the 2 years of the study, 15 common SNPs were found strongly associated (Fig. 8, Table 2, S13 and S14). Our GWAS revealed 28 and 33 SNPs with strong association during 2011 and 2012, respectively. We found an association of S1_178148471 (-log10 P 2011 = 5.2 and 2012 = 4.0) located in sopenicillin N epimerase (AAT_I superfamily) with ADP binding catalytic activity, S1_178214095 (-log10 P 2011 = 5.4 and 2012 = 4.6) of the protein transport protein SEC23-like (zf, MIDAS domain) that regulates Zinc ion binding, S2_169874314 (-log10 P 2011 = 4.0 and 2012 = 6.5) located in the intergenic space of Na+/H+ antiporter and lucose-6-phosphate 1-dehydrogenase, S3_230322338 (-log10 P 2011 = 3.3 and 2012 = 3.9) of the intergenic space between SNF1-related protein kinase and RAD50-interacting protein S3_230372266 (-log10 P 2011 = 3.5 and 2012 = 3.1) located in Ubiquitin-like modifier-activating enzyme 5-like (thiamine synthesis) and S5_131824978 (-log10 P 2011 = 3.9 and 2012 = 3.9) of an unknown protein. We also found an association of S6_202147247 (-log10 P 2011 = 3.9 and 2012 = 3.9), S6_202147285 (-log10 P 2011 = 3.1 and 2012 = 3.2), S6_202147337 (-log10 P 2011 = 3.1 and 2012 = 3.2) and S6_202147420 (-log10 P 2011 = 3.1 and 2012 = 3.2) located in the intergenic space between STYLOSA protein and flavin monooxygenase in both years. We found an association of S6_227195619 (-log10 P 2011 = 3.9 and 2012 = 3.8) of chloroplastic-FANTASTIC FOUR (FAF)-like protein, S8_132459145 (-log10 P 2011 = 6.8 and 2012 = 3.7) of DnaQ-like exonuclease, S9_250224149 (-log10 P 2011 = 6.8 and 2012 = 3.7) of mitochondrial-processing peptidase subunit alpha, S10_229225552 (-log10 P 2011 = 3.1 and 2012 = 5.0) of cell division control protein 45 (CDC45), S11_94177155 (-log10 P 2011 = 3.8 and 2012 = 3.0) of Clathrin assembly protein, and S12_72971688 (-log10 P 2011 = 7.06 and 2012 = 3.9) in the intergenic space between CLAVATA1 receptor kinase and pentatricopeptide repeat. S6_227195619, S6_204246361, S10_229225552 were nonsynonymous SNPs located in isopenicillin N epimerase (AAT_I superfamily), chloroplastic-FAF-like protein, cell division control protein 45 (CDC45) and TRS120 isoform, respectively, which have biological processes involving cell division and meristem organization.
Discussion
Understanding the genetic control of traits influenced by domestication is improving as a result of GWAS for several crops. Domesticated peppers have larger fruits than wild peppers but also have larger leaves, flowers and seeds. An overall increase in size of many different organs could result from an increase in cell number or size or both24. Principal component analysis (PCA), model based population structure and an Euclidean tree built based on identity by state (IBS) indices revealed that the clustering pattern of diverse accessions were in agreement with capsaicin content (CA) and FW classifications indicating the importance of these traits in shaping modern pepper genome. Our study focused on unraveling various mechanisms underlying fruit weight and capsaicin content among C. annuum peppers.
A total of 659 genes located in the sweep of chromosome 11 were identified that influenced FW in C. annuum. In particular, the pentatricopeptide repeat protein and ABC transporter are known genes for tomato FW25, and the ankyrin repeat protein, which functions as an acyltransferase, with a homologue on chromosome 5 highly associated with capsaicin content as well as FW are located in the sweep region. Similarly, recent tomato genome analysis revealed that most of the genes, namely glutathione S-transferase, actin-related protein 2/3 complex, endo-1, 4-beta-glucanase, RCC1 domain-containing protein, ankyrin repeat protein, and xanthine dehydrogenase, were also found in the chromosome sweep important for tomato domestication26. We identified 30 genes in the sweep region with high LD, and our GWAS revealed highly significant markers for FW within the sweep. Qin et al.9 identified 115 regions across the genome containing 511 genes that are important for domestication with strong selective sweep signals by using WGS analysis. Among the genes in the chromosome 11 sweep, ankyrin-like protein, an acyltransferase, showed extended LD, up to 10 kb, which implies its role in pepper fruit weight.
Despite a number of genes involved in the capsaicin synthesis pathway per se, these genes need upstream and trans-regulators for proper spatiotemporal expression during capsaicin synthesis in pepper fruit formation. Our GWAS results showed an association with multiple genes via tightly linked SNP markers. Among the DNA/RNA binding proteins, CCHC zinc-finger, CCCH zinc-finger, GRAS transcription factor, transparent testa 12, and pentatricopeptide repeat protein are the major markers. Because these genes are transporters, transcription factors and catalytic enzymes, they might be regulators of the genes in the capsaicin biosynthesis pathway. Another homologue of ankyrin-like protein on chromosome 5 was also strongly associated with capsaicinoid content. Han et al.27, Stewart et al.28 and Reddy et al.29 demonstrated that Pun1 is responsible for capsaicinoid synthesis; the authors further suggested the presence of an unknown enzyme that reduces vanillin to vanillyl alcohol. Pun1 located on chromosome 2 was noted to encode AT3, an acyl transferase from the BAHD acyl transferase superfamily. How AT3 is related to the acyltransferase on chromosome 11 and 5 needs further investigation. Because its molecular function is also as an acyltransferase, ankyrin repeat-containing protein might function as an additional Pun1 gene, which is yet to be determined, or it might have an accessory role in capsaicin synthesis. Our results demonstrating the influence of multiple chromosomal regions on capsaicinoid content may begin to explain the extensive diversity evident in pepper for capsaicinoid concentration in pungent genotypes.
We identified 16 SNPs strongly associated with FW in both years of the study. The C. annuum progenitor species bear fruit of much smaller size than do the cultivated counterparts. Important findings from our FW GWAS study reveal similar biological function in tomato and other plants (Lin et al.38). We found a strong association of Stylosa protein (S6_202147247 S6_202147285 S6_202147337 S6_202147420) with FW in both seasons of the study; FASCIATED (FAS), encoded by a member of the YABBY family regulating organ polarity, is thought to play an important role in plant growth and development30,31. FAS controls fruit shape in tomato32. Earlier, STYLOSA (STY) was found to regulate floral homeotic meristem and organ identity in Antirrhinum33. Interactions between STYLOSA and YABBY family proteins control Antirrhinum vegetative and reproductive development34. The interaction between STYLOSA (Ca06g14190) and YABBY might be critical for fruit size/shape and weight in pepper.
S6_227195619 causes a nonsynonymous mutation in chloroplastic FAF protein and we found it strongly linked with FW in both seasons of the study. Plastid genes are expressed at high levels in photosynthetically active chloroplasts, including in developing fruits. Chloroplastic-FAF protein regulates shoot meristem size in Arabidopsis. FAF genes are expressed in the center of the shoot meristem, overlapping with the site of WUSCHEL (WUS) expression. Strong interaction between FAF and WUS determine the fate of meristem activity35. Hence, FAF (Ca06g22610) might regulate the size of the shoot meristem as well as FW by modulating the CLV3-WUS feedback loop in pepper. A significantly associated SNP, S9_250224149, was found in the mitochondrial processing peptidase alpha subunit (Ca09g16860), which might be important for fruit development because its ortholog in tomato is specifically and differentially expressed during cell expansion stages in early stages of fruit development36.
S12_72971688 is an intergenic SNP close to CLAVATA 1 receptor kinase that showed a strong association with FW in both seasons. Arabidopsis plants homozygous for mutations at the CLAVATA1 (CLV1) locus, a receptor kinase protein, accumulate excess undifferentiated cells37,38. This gene is critical for shoot and flower meristem size. In our study, Ca12g10020, or CLAVATA1 receptor kinase, might interact with CLV3 as a ligand–receptor pair in a signal transduction pathway coordinating growth between adjacent meristematic regions and controlling the balance between meristem cell proliferation and differentiation.
Fine mapping of the QTL fw3.2 controlling FW in tomato was accomplished recently25 and genes in this QTL range were identified. Among seven putative genes identified in this region, ORF4 encodes a protein with high identity to PNM1 in Arabidopsis PNM1 belongs to the pentatricopeptide repeat containing protein (PPR) family that functions in RNA binding (Hammani et al. 2011). The marker S12_72971688 linked to the locus Ca12g10030 (PPR) might play key roles in determining FW in pepper as QTL fw3.2 does in tomato.
Given our SNP density and sample size, this study is not sufficiently robust to find alleles of small effect among the pepper secondary centers of origin distributed across the world. However, we located SNPs with major effect in highly inbred, diverse and smaller population. Some of the strongest signals are quite far from known capsaicin genes possibly because of ascertainment bias. Also SNPs located in the candidate genes may not be in LD, which hampers their identity in association mapping strategies. Furthermore, stringent control for population stratification and IBS may eliminate useful SNPs located in the candidate genes. Such tradeoffs are part of GWAS, especially in reducing spurious associations39. Zhao et al.39 and Rife et al.40 proposed that SNP identification from transcriptome datasets or spiked GBS, which combines targeted amplicon sequencing with reduced representation GBS, will improve our ability to detect moderate-strength and rare alleles, especially those located in candidate genes. Analogous approaches have been reported39,41,42,43.
Our GWAS for capsaicin and dihydrocapsaicin content and FW identified SNPs located in the candidate genes known to have similar biological functions in tomato and other plants for these domestication related traits. Accessions containing higher minor allele frequencies from this study can be used to generate nested association mapping populations to validate GWAS results and further dissect the complex interaction among genes involved in the pleotropic effects on fruit size and capsaicin content evident in Capsicum.
Materials and Methods
We included 94 accessions of C. annuum belonging to various countries representing a wide geographical area of the world for the molecular diversity analysis (Table S1). These selfed accessions were grown in three replications during two seasons (2011 and 2012) adapting a row-to-plant spacing of 100 × 30 cm. Ten plants per accession were grown in each replication. FW (g), was collected for five plants. Quantitative analysis of capsaicin and dihydrocapsaicin content was performed using greenhouse-grown plants in three replications using the 1200 series HPLC system (Agilent Technologies, Santa Clara, CA) with a degasser, an autosampler, and a binary pump as described7.
Genotyping by sequencing
Genomic DNA was isolated using the DNeasy plant mini kit (QIAGEN, Germany), and GBS was as described44,45. Briefly, genome complexity was reduced by digesting total genomic DNA from individual samples with the ApeKI, a type II restriction endonuclease that recognizes a degenerate 5-bp sequence (GCWGC, where W is A or T), which creates a 5′ overhang (3 bp) and is partially methylation-sensitive (will not cut if the 3′ base of the recognition sequence on both strands is 5-methylcytosine). Digested products were then ligated to adapter pairs with enzyme-compatible overhangs; one adapter contained the barcode sequence and a binding-site Illumina sequencing primer (Illumina Inc., USA). These samples were pooled, purified and amplified with primers compatible with the adapter sequences. Temperature cycling was 72 °C for 5 min, 98 °C for 30 s followed by 18 cycles of 98 °C for 30 s, 65 °C for 30 s, and 72 °C for 30 s with a final Taq extension step at 72 °C for 5 min. Amplified sample pools constituted a sequencing “library”. Libraries were purified and 1 μL was loaded onto an Experion automated electrophoresis station (BioRad, Hercules, CA) for evaluation of fragment sizes. Libraries were considered suitable for sequencing if adapter dimers (~128 bp in length) were minimal or absent and most of the other DNA fragments were between 170 and 350 bp. If adapter dimers were present in excess of 0.5% (based on the Experion output), libraries were constructed again by using a few DNA samples and decreasing adapter amounts. The PCR primers also added 3′ sequences complementary to the solid-phase oligonucleotides that coat the Illumina sequencing flow-cell. After PCR, pooled products were purified; GBS “library” fragment size distributions were checked on a BioAnalyzer (Agilent Technologies, USA). Products were quantified and diluted for sequencing by use of Illumina HiSeq 2500. A bioinformatics pipeline, TASSEL-GBS, designed for efficient processing of raw GBS sequence data into a SNP genotype file46 was used. Barcoded sequence reads were processed and collapsed into a set of unique sequence tags, with one TagCounts file produced per input FASTQ. Chromosomal assignment physical map position of candidate genes and GBS markers, were deduced from the hot pepper WGS draft at http://peppergenome.snu.ac.kr. SNPs were designated on the basis of chromosome number and position (e.g., S10_172735351 indicates SNP located at 172735351 position on chromosome 10).
Data Analysis
Population structure analysis
For quantitative assessment of the number of clusters in the GWAS panel, we used a Bayesian clustering analysis with a model-based approach implemented in STRUCTURE v2.247. This approach involves use of multi-locus genotypic data to assign individuals to clusters or groups (K) without prior knowledge of their population affinities. The program was run with SNP markers for k-values 1–9 (hypothetical number of subgroups), with 100,000 burn-in iterations, followed by 500,000 Markov Chain Monte Carlo (MCMC) iterations for accurate parameter estimates with a high-performance cluster. To verify the consistency of the results, we performed three independent runs for each K. An admixture model with correlated allele frequencies was used. The optimal K value was determined by use of an ad-hoc statistic, ΔK. The number of Ks in each dataset was evaluated by ΔK values estimated and visualized with the software Structure Harvester, (www.taylor0.biology.ucla.edu/structureHarvester)48. To facilitate the interpretation of population-genetic clustering results, we used CLUMPP (CLUster Matching and Permutation Program)49, which groups individuals into populations on the basis of multilocus genotypes, and the output was directly input into a program for cluster visualization DISTRUCT 1.150. In a second approach, we used principle component analysis (PCA) with the SNP & Variation Suite (SVS v8.1.5) (Golden Helix, Inc., Bozeman, MT, USA; www.goldenhelix.com).
Analysis of population differentiation
Fixation index (FST) estimation was based on Wright’s F statistic51 in SVS v8.1.5 (http://goldenhelix.com/). Annotation and gene ontology terms for genes from the selective sweeps were identified with the WGS draft at http://peppergenome.snu.ac.kr.
GWAS mapping
For GBS data, we considered only SNPs successfully mapped to the Capsicum WGS draft, because knowing the chromosome location of SNPs helps prevent spurious LD and thereby unreliable association mapping. Before studying LD decay, haplotype blocks were calculated for all markers using the default settings in SVS v8.1.5. Adjacent and pairwise measurements of LD for GBS data were calculated separately for SNPs in each chromosome. All LD plots and LD measurements and haplotype frequency calculations involved use of SVS v8.1.5 and Tassel 5.0. For GWAS, the population structure Q matrix was replaced by the PC matrix52. The PC matrix and identity by descent (IBD) was calculated from LD-pruned SNPs in SVS v8.1.5. GWAS involved a single-locus mixed linear model developed by the EMMAX method53 and implemented in SVS v8.1.5. We used a PC matrix (first two vectors) to correct for population stratification and the IBD matrix to correct polygenic background. Manhattan plots for associated SNPs were visualized in GenomeBrowse v1.0 (Golden Helix, Inc). The SNP P-values from GWAS underwent sequential Bonferroni correction54 as well as false discovery rate (FDR) analysis55. Annotation and gene ontology terms for the SNP containing sequences were identified with the WGS draft at http://peppergenome.snu.ac.kr.
Additional Information
How to cite this article: Nimmakayala, P. et al. Genome-wide diversity and association mapping for capsaicinoids and fruit weight in Capsicum annuum L. Sci. Rep. 6, 38081; doi: 10.1038/srep38081 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Eshbaugh, W. in New crops (ed Simon, J. E. & Janick, J. ) 132–139 (J Wiley and Sons, New York, 1993).
Pickersgill, B. Relationships between weedy and cultivated forms in some species of chili peppers (genus Capsicum). Evolution, 683–691 (1971).
Andrews, J. The domesticated Capsicum. 163 (University of Texas Press, 1984).
Zewdie, Y., Tong, N. & Bosland, P. Establishing a core collection of Capsicum using a cluster analysis with enlightened selection of accessions. Genet Resour Crop Evol 51, 147–151, doi: 10.1023/b:gres.0000020858.96226.38 (2004).
Pickersgill, B. Genetic resources and breeding of Capsicum spp. Euphytica 96, 129–133, doi: 10.1023/a:1002913228101 (1997).
Nicolaï, M., Cantet, M., Lefebvre, V., Sage-Palloix, A.-M. & Palloix, A. Genotyping a large collection of pepper (Capsicum spp.) with SSR loci brings new evidence for the wild origin of cultivated C. annuum and the structuring of genetic diversity by human selection of cultivar types. Genet Resour Crop Evol 60, 2375–2390, doi: 10.1007/s10722-013-0006-0 (2013).
Nimmakayala, P. et al. Linkage disequilibrium and population-structure analysis among Capsicum annuum L. cultivars for use in association mapping. Mol Genet Genomics 289, 513–521, doi: 10.1007/s00438-014-0827-3 (2014).
Kim, S. et al. Genome sequence of the hot pepper provides insights into the evolution of pungency in Capsicum species. Nat Genet 46, 270–278, doi: 10.1038/ng.2877 (2014).
Qin, C. et al. Whole-genome sequencing of cultivated and wild peppers provides insights into Capsicum domestication and specialization. Proceedings of the National Academy of Sciences of the United States of America 111, 5135–5140, doi: 10.1073/pnas.1400975111 (2014).
Owens, B. F. et al. A Foundation for Provitamin A Biofortification of Maize: Genome-Wide Association and Genomic Prediction Models of Carotenoid Levels. Genetics 198, 1699–1716, doi: 10.1534/genetics.114.169979 (2014).
Iquira, E., Humira, S. & Francois, B. Association mapping of QTLs for sclerotinia stem rot resistance in a collection of soybean plant introductions using a genotyping by sequencing (GBS) approach. BMC Plant Biology 15, 5 (2015).
Zhang, D. et al. Genetic analysis of inflorescence and plant height components in sorghum (Panicoidae) and comparative genetics with rice (Oryzoidae). BMC Plant Biology 15, 107 (2015).
Lin, M. et al. Genotyping-by-sequencing (GBS) identified SNP tightly linked to QTL for pre-harvest sprouting resistance. Theor Appl Genet 1–11, doi: 10.1007/s00122-015-2513-1 (2015).
Poland, J. A., Brown, P. J., Sorrells, M. E. & Jannink, J.-L. Development of high-density genetic maps for barley and wheat using a novel two-enzyme genotyping-by-sequencing approach. PLoS One 7, e32253 (2012).
Rao, G., Chaim, A. B., Borovsky, Y. & Paran, I. Mapping of yield-related QTLs in pepper in an interspecific cross of Capsicum annuum and C. frutescens. Theor Appl Genet 106, 1457–1466 (2003).
Ben-Chaim, A. et al. QTL analysis for capsaicinoid content in Capsicum. Theor Appl Genet 113, 1481–1490 (2006).
Borovsky, Y. & Paran, I. Characterization of fs10. 1, a major QTL controlling fruit elongation in Capsicum. Theor Appl Genet 123, 657–665 (2011).
Blum, E. et al. Molecular mapping of capsaicinoid biosynthesis genes and quantitative trait loci analysis for capsaicinoid content in Capsicum. Theor Appl Genet 108, 79–86 (2003).
Zygier, S. et al. QTLs mapping for fruit size and shape in chromosomes 2 and 4 in pepper and a comparison of the pepper QTL map with that of tomato. Theor Appl Genet 111, 437–445 (2005).
Yarnes, S. C. et al. Identification of QTLs for capsaicinoids, fruit quality, and plant architecture-related traits in an interspecific Capsicum RIL population. Genome 56, 61–74 (2012).
Gabriel, S. B. et al. The structure of haplotype blocks in the human genome. Science 296, 2225–2229 (2002).
Dempster, A. P., Laird, N. M. & Rubin, D. B. Maximum likelihood from incomplete data via the EM algorithm. Journal of the royal statistical society. Series B (methodological) 1–38 (1977).
Weir, B. S. & Cockerham, C. Genetic data analysis II: Methods for discrete population genetic data. Sinauer Assoc. Inc., Sunderland, MA, USA (1996).
Pickersgill, B. Domestication of Plants in the Americas: Insights from Mendelian and Molecular Genetics. Annals of Botany 100, 925–940, doi: 10.1093/aob/mcm193 (2007).
Zhang, N., Brewer, M. T. & van der Knaap, E. Fine mapping of fw3.2 controlling fruit weight in tomato. Theor Appl Genet 125, 273–284 (2012).
Lin, T. et al. Genomic analyses provide insights into the history of tomato breeding. Nature genetics (2014).
Han, K. et al. Biosynthesis of capsinoid is controlled by the Pun1 locus in pepper. Molecular Breeding 31, 537–548, doi: 10.1007/s11032-012-9811-y (2013).
Stewart, C., Jr., Mazourek, M., Stellari, G. M., O’Connell, M. & Jahn, M. Genetic control of pungency in C. chinense via the Pun1 locus. J Exp Bot 58, 979–991 (2007).
Reddy, U. K. et al. Identification of gene-specific polymorphisms and association with capsaicin pathway metabolites in Capsicum annuum L. collections. PLoS One 9 (2014).
Cong, B., Barrero, L. S. & Tanksley, S. D. Regulatory change in YABBY-like transcription factor led to evolution of extreme fruit size during tomato domestication. Nat Genet 40, 800–804 (2008).
Munos, S. et al. Increase in tomato locule number is controlled by two single-nucleotide polymorphisms located near WUSCHEL. Plant Physiol 156, 2244–2254 (2011).
Huang, Z., Van Houten, J., Gonzalez, G., Xiao, H. & van der Knaap, E. Genome-wide identification, phylogeny and expression analysis of SUN, OFP and YABBY gene family in tomato. Mol Genet Genomics 288, 111–129 (2013).
Motte, P., Saedler, H. & Schwarz-Sommer, Z. Stylosa and fistulata: regulatory components of the homeotic control of Antirrhinum floral organogenesis. Development 125, 71–84 (1998).
Navarro, C. et al. Molecular and genetic interactions between STYLOSA and GRAMINIFOLIA in the control of Antirrhinum vegetative and reproductive development. Development 131, 3649–3659 (2004).
Wahl, V., Brand, L., Guo, Y.-L. & Schmid, M. The fantastic four proteins influence shoot meristem size in Arabidopsis thaliana. BMC Plant Biology 10, 285 (2010).
Causse, M. et al. A genetic map of candidate genes and QTLs involved in tomato fruit size and composition. J Exp Bot 55, 1671–1685 (2004).
Schoof, H. et al. The stem cell population of Arabidopsis shoot meristems is maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100, 635–644 (2000).
Clark, S. E. Organ formation at the vegetative shoot meristem. The Plant Cell 9, 1067 (1997).
Zhao, K. et al. Genome-wide association mapping reveals a rich genetic architecture of complex traits in Oryza sativa. Nat Commun 2, 467, doi: 10.1038/ncomms1467 (2011).
Rife, T. W., Wu, S., Bowden, R. L. & Poland, J. A. Spiked GBS: a unified, open platform for single marker genotyping and whole-genome profiling. BMC Genomics 16, 015–1404 (2015).
Huang, X. et al. Genome-wide association study of flowering time and grain yield traits in a worldwide collection of rice germplasm. Nat Genet 44, 32–39, doi: 10.1038/ng.1018 (2012).
Yang, W. et al. Genome-wide association study of rice (Oryza sativa L.) leaf traits with a high-throughput leaf scorer. J Exp Bot 20 (2015).
Han, B. & Huang, X. Sequencing-based genome-wide association study in rice. Curr Opin Plant Biol 16, 133–138 (2013).
Elshire, R. J. et al. A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6, e19379 (2011).
Reddy, U. K. et al. High-Resolution Genetic Map for Understanding the Effect of Genome-Wide Recombination Rate on Nucleotide Diversity in Watermelon. G3: Genes|Genomes|Genetics, doi: 10.1534/g3.114.012815 (2014).
Glaubitz, J. C. et al. TASSEL-GBS: a high capacity genotyping by sequencing analysis pipeline. PLoS One 9, E90346 (2014).
Pritchard, J. K., Stephens, M., Rosenberg, N. A. & Donnelly, P. Association Mapping in Structured Populations. American Journal of Human Genetics 67, 170–181 (2000).
Earl, D. A. Structure harvester: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conservation genetics resources 4, 359–361 (2012).
Jakobsson, M. & Rosenberg, N. A. CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23, 1801–1806 (2007).
Rosenberg, N. A. DISTRUCT: a program for the graphical display of population structure. Molecular Ecology Notes 4, 137–138 (2004).
Weir, B. S. & Cockerham, C. C. Estimating F-statistics for the analysis of population structure. Evolution, 1358–1370 (1984).
Stich, B. et al. Comparison of mixed-model approaches for association mapping. Genetics 178, 1745–1754, doi: 10.1534/genetics.107.079707 (2008).
Kang, H. M. et al. Variance component model to account for sample structure in genome-wide association studies. Nat Genet 42, 348–354, doi: 10.1038/ng.548 (2010).
Holm, S. A simple sequentially rejective multiple test procedure. Scandinavian Journal of Statistics 6, 65–70 (1979).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of royal statistical society 57, 289–300 (1995).
Acknowledgements
The study received funding from USDA-NIFA (2010-02419), NSF-EPSCOR#09-570 - EPS-1003907, NIH Grant P20RR016477 to the West Virginia IDeA Network for Biomedical Research Funding and the Gus R. Douglass Institute (graduate research assistantship to V.A., K.T., L.Y. and B.D.). Doil Choi was supported by the Agricultural Genome Center of Next Generation Biogreen 21 program (PJ011275-01), RDA of Korean government.
Author information
Authors and Affiliations
Contributions
U.R., J.S., G.H., R.H. and P.N. designed the study and drafted the manuscript. P.N., T.S., V.A., G.V., K.T., L.Y., J.D., S.A., A.A., B.D. and M.N. generated field and capsaicinoid phenotyping. P.N., T.S., V.A., G.V., K.T., L.Y., J.D., A.A. and B.D. extracted DNA and assisted to generate genome-wide SNPs. M.P., S.M. and D.C. provided whole genome sequence draft and mapped SNPs to the genome. U.R., P.N., T.S., and V.A. performed GWAS analysis.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Nimmakayala, P., Abburi, V., Saminathan, T. et al. Genome-wide Diversity and Association Mapping for Capsaicinoids and Fruit Weight in Capsicum annuum L. Sci Rep 6, 38081 (2016). https://doi.org/10.1038/srep38081
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep38081
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.