Abstract
Mosquito oviposition site selection is essential for vector population dynamics and malaria epidemiology. Irrigated rice cultivations provide ideal larval habitats for malaria mosquitoes, which has resulted in increased prevalence of the malaria vector, Anopheles arabiensis, in sub-Saharan Africa. The nature and origin of the cues regulating this behaviour are only now being elucidated. We show that gravid Anopheles arabiensis are attracted and oviposit in response to the odour present in the air surrounding rice. Furthermore, we identify a synthetic rice odour blend, using electrophysiological and chemical analyses, which elicits attraction and oviposition in laboratory assays, as well as attraction of free-flying gravid mosquitoes under semi-field conditions. This research highlights the intimate link between malaria vectors and agriculture. The identified volatile cues provide important substrates for the development of novel and cost-effective control measures that target female malaria mosquitoes, irrespective of indoor or outdoor feeding and resting patterns.
Similar content being viewed by others
Introduction
Oviposition site selection by malaria mosquitoes is a behaviour that needs to be exploited in future control strategies. Although existing indoor control methods, including indoor residual spray (IRS) and long lasting insecticidal nets (LLINs), have been effective in reducing indoor malaria transmission1, we are now seeing a reduction in that efficacy2, likely due to the emergence of insecticide resistance3,4,5, the resurgence of vectors6,7, and the shift from indoor to outdoor vector behaviour7,8,9,10 leading to a proportional increase in outdoor malaria transmission11,12,13,14. In many areas, this change is associated with one of the primary malaria vectors, Anopheles arabiensis6. Besides larval control, which has largely been disbanded in favour of IRS and LLINs15 in contemporary sub-Saharan Africa, there are no effective control strategies that target malaria mosquito populations outdoors. Gravid malaria mosquitoes, irrespective of indoor or outdoor feeding patterns, must seek for suitable aquatic habitats to lay their eggs. Identifying and manipulating cues from such habitats could provide important insights toward novel mosquito control tools.
Selection of oviposition sites for vectors of malaria is dependent on a number of physical and chemical factors. Gravid malaria mosquitoes are attracted to water bodies by water vapour16, as well as olfactory cues from conspecific larvae17,18, microorganisms19 and soil20 associated with the sites. To date, the only compound confirmed as an oviposition attractant for malaria mosquitoes is cedrol, a soil-associated compound20. While habitat cues associated with the sites potentially attract mosquitoes over a longer distance, these have not been fully explored. Emergent and other vegetation may provide such potential cues, as grasses and other short vegetation often emerge from and surround natural larval habitats of malaria mosquitoes21,22.
Governmental and non-governmental initiatives are systematically increasing the regions under irrigated cultivation throughout sub-Saharan Africa23. Although irrigated cultivations of cereal crops, including rice, maize and sugarcane, provide food security, they also provide ideal larval habitats for malaria mosquitoes24,25. This generally creates a spatial link between the vector and the people, thereby increasing the risk of malaria transmission. The cereal crops provide the larvae with shelter from abiotic and biotic threats, as well as nutrients directly from shed pollen and indirectly from accumulated detritus and associated microorganisms25,26. Irrigated rice agro-ecosystems, in particular, can create permanent mosquito larval habitats27, thereby increasing the abundance of malaria vectors compared with other cultivated cereals28,29,30. Moreover, the agronomic activities in rice, including the addition of fertilizers and insecticides31,32, can also affect larval density by increasing available nutrients33 and creating predator-free habitats34, respectively. Thus, female mosquitoes that select breeding sites within irrigated rice and other cereal cultivations have the potential to increase their own fitness by providing their offspring with selective advantages. We show that gravid An. arabiensis, one of the predominant mosquito species in rice cultivation35,36, are attracted to the odour of rice, irrespective of the phenological stage of the plant, and identify a synthetic blend of rice-volatiles that acts as a long-range attractant and elicits oviposition under laboratory and semi-field conditions. The manipulation of the odour-mediated oviposition site selection behaviour of gravid adults has distinct potential as a tool in the future control of malaria vectors. Here, we take a significant step toward the development of a long-sought-after control tool for gravid malaria mosquitoes, by identifying the first blend of odours to be used as a synthetic lure.
Results
Gravid malaria mosquitoes are attracted to rice odour
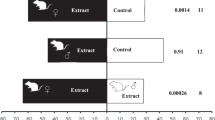
Searching female mosquitoes make choices at increasingly refined scales, from habitat finding to resource acceptance37. Here, we investigate the nature and origin of the cues regulating the oviposition site selection in rice cultivations by An. arabiensis. To investigate whether An. arabiensis respond behaviourally to the odour compounds present in the air surrounding rice, we collected the headspace odour from two available rice cultivars, MR1 and MR3, at three different phenological stages: tillering, booting and flowering. Gravid mosquitoes showed a significant dose-dependent attraction to the headspace odour extracts of both MR1 and MR3 cultivars in a two-port olfactometer bioassay (Fig. 1a). This finding was independent of phenological state, when compared to the hexane controls (Supplementary Fig. 1; Supplementary Table 1) and the headspace odour extract of the breeding water collected from natural larval habitats of An. arabiensis (Supplementary Fig. 2; Supplementary Table 2), and indicates that rice volatiles can attract these mosquitoes over the short range. Similarly, gravid mosquitoes preferred to lay their eggs in water treated with the headspace odour extracts of all phenological stages of the MR1 and MR3 cultivars over controls (Supplementary Figs 3 and 4; Supplementary Tables 3 and 4) in a two-choice oviposition assay (Fig. 1b).
Gravid Anopheles arabiensis respond to rice odour.
Diagrams of the two-port olfactometer (a) and oviposition (b) assays. Attraction (c,e) and oviposition (d,f) preference of mosquitoes to headspace volatiles of the MR1 and MR3 rice cultivars compared to hexane control, respectively (c)χ2 = 11.87, P = 0.0006; (d), χ2 = 16.21, P < 0.0001; (e), χ2 = 8.378, P = 0.0038; (f), χ2 = 14.05, P = 0.0002). The headspace of the MR3 rice cultivar significantly attracted (g) and elicited oviposition (h) of mosquitoes over that of the MR1 cultivar (g, χ2 = 7.080, P = 0.0078; h, χ2 = 5.822, P = 0.0158). (i) Schematic of the combined gas chromatograph (GC) and electroantennographic detection (EAD) analysis. FID, flame ionization detector (courtesy of Dr Majid Ghaninia). (j) EAD traces depict voltage changes (mV) in response to the bioactive compounds in the headspace of the MR3 rice cultivar, eluting from the GC and registered by the FID. Note that the intensity of the response to sulcatone was variable and denoted here by an asterisk in the GC-EAD trace. The identity and release rate of the bioactive compounds are shown at the left. A synthetic blend composed of the bioactive compounds identified, in their natural ratio (j), elicited attraction (k) and stimulated oviposition (l) in gravid mosquitoes in a dose-dependent manner (k, χ2 = 18.34, P < 0.0001; l, χ2 = 32.93, P < 0.0001). Error bars represent standard errors of the mean. Statistical significance was tested using nominal logistic regression (likelihood ratio test). Ten replicates of 10 mosquitoes each were used in each behavioural experiment. All diagrammatic representations, other than in i, are courtesy of Pixabay, an open source image database.
Since no significant differences were observed among the behavioural responses to the headspace of the phenological stages of either of the cultivars (Supplementary Figs 1–4; Supplementary Tables 1–4), the headspace extracts from all stages were pooled for each cultivar to use in subsequent behavioural assays. While the pooled headspace of each cultivar was dose-dependently attractive and elicited oviposition when compared to hexane (Fig. 1c–f; Supplementary Table 5) and breeding water controls (Supplementary Figs 5 and 6; Supplementary Tables 6 and 7), the pooled MR3 headspace extract was preferred by gravid mosquitoes over that of MR1 in both assays (Fig. 1g, and Supplementary Fig. 6c; Supplementary Tables 5 and 7). The behavioural observations showed that gravid mosquitoes are both attracted and oviposit in response to rice odour, and capable of discriminating between odour profiles emitted by different rice cultivars.
A complex odour blend drives mosquito oviposition behaviour
As olfactory cues are detected mainly by the antenna in mosquitoes, we next tested the response of gravid mosquitoes to individual rice odour compounds of the preferred MR3 pooled headspace extract using combined gas chromatography and electroantennographic detection (GC-EAD; Fig. 1i). Of the eight compounds eliciting antennal responses (Fig. 1j), ß-caryophyllene, decanal, sulcatone (6-methyl-5-hepten-2-one) and limonene were previously identified in other rice cultivars38. The antennal response to sulcatone was variable across trials (* in Fig. 1j), but was included in further behavioural experiments to validate its role as an active component of the natural blend. The overall volatile release rate was 24 ng min−1, with limonene and nonanal as the most abundant compounds (Fig. 1j).
A synthetic blend of all the eight GC-EAD-active compounds identified in the MR3 headspace, in their natural ratio (Fig. 1j), elicited short-range attraction and oviposition in gravid An. arabiensis at the intermediate doses tested (Fig. 1k,l; Supplementary Table 5). In contrast, at the highest doses, the mosquitoes were either indifferent to, or avoided, the treatment. The release rates and ratio of individual compounds within the blend, in each assay, were found to be consistent with the synthetic blend, as determined by headspace odour analysis (Supplementary Fig. 7). Subtraction of individual components from the full blend, released at the lowest effective dose tested (10 ng), did not significantly reduce the attraction of gravid females in the two-port olfactometer against a pentane control, when compared to the attraction response to the full blend, with the exception of the removal of nonanal (Supplementary Fig. 8; Supplementary Table 8). However, nonanal by itself was unable to elicit attraction equivalent to that of the full blend (Supplementary Fig. 8). In contrast, the oviposition response of gravid females was significantly reduced in the two-choice oviposition assay with a pentane control, when compared to the oviposition response to the full blend, for all the subtractive blends, except ß-pinene (Supplementary Fig. 9; Supplementary Table 9). Thus, to induce the full behavioural repertoire, the complete blend is required.
Gravid mosquitoes show long-range attraction to the rice blend
An evaluation of the synthetic MR3 odour blend, in large (10.8 m × 6.7 m × 2.4 m) outdoor screened-in enclosures (Fig. 2a) in Kenya, revealed that oviposition site-seeking An. arabiensis were attracted over the long-range and caught in a dose-dependent manner in recessed BG sentinel traps39 (Supplementary Fig. 10). Based on this experiment, the most cost-effective release rate tested (3 ng min−1, based on the release rate of α-pinene) was used in further trials. Gravid An. arabiensis were significantly attracted to the full synthetic blend (Fig. 2b) when compared against the solvent control (heptane). No significant difference was found between the solvent controls when tested in a pairwise fashion (Fig. 2c).
Long-range attraction of gravid Anopheles arabiensis to the synthetic rice blend.
Diagram of the semi-field screened-in enclosure and the recessed BG-Sentinel traps (a) used to attract and capture free-flying mosquitoes. Using a randomised complete block design, a significantly higher number of mosquitoes were found to be attracted to traps baited with the synthetic blend compared to a heptane control (b, Pr = 0.74, 95% CI 0.68–0.79). No significant difference was observed between heptane controls (c, Pr = 0.51, 95% CI 0.44–0.58). Semi-field data (b,c) were analysed with generalized linear model using a quasi-binomial distribution. The experiments were conducted over 12 nights each with 200 gravid An. arabiensis released per trial.
Discussion
Gravid An. arabiensis are attracted and stimulated to oviposit by rice plant volatiles under laboratory and semi-field conditions. This has the potential of strengthening the spatial association between the malaria vector and the agricultural landscape. Additional experiments are, however, required to verify that malaria mosquitoes use rice odours to locate appropriate oviposition sites in the wild. Female mosquitoes were attracted to volatiles emitted by all stages of the rice plant, which concurs with field observations showing that the larvae are present in rice cultivation throughout the growing season (s), with varying abundance27,33. Rice volatiles thus can play a significant role in the maintenance of vector populations, along with other aspects of rice cultivation that modulate female choice, including water availability, insecticide- and fertiliser-use, and vegetation density creating obstruction and shade24,27,33,34,35,36. The selective advantage for this association is yet to be conclusively determined. The enhanced availability of food in rice fields due to microbial growth in response to nitrogen surplus and water turbidity following fertiliser application33, along with the reduction of competition and predators, by insecticide treatment34, are possible drivers for this adaptive selection.
Mosquitoes use volatiles originating from plants as attractants and stimulants for oviposition40. While gravid anopheline mosquitoes display preferences for volatiles associated with living plants41 and microbes associated with their larval habitats19,20, culicines prefer those associated with the microbial fermentation of plant tissues25,42. Here, we have identified a long-range plant odour-based attractant blend of readily available and affordable organic compounds for gravid malaria mosquitoes, providing the integrated vector management community with new perspectives for developing a tool that targets gravid anophelines regardless of feeding and resting preferences. The revelation that rice plant volatiles attract gravid anophelines, together with the suitability of irrigated cereal cultivations as larval habitats20,21,22, strongly indicates that further analysis of the oviposition ecology of anophelines within these agro-ecological habitats is likely to produce additional targets for the development of odour-based attractants. While the identified blend consists of numerous ubiquitous plant compounds, previously shown to drive host plant selection in herbivorous43,44, the composition and ratio of this blend is novel, and it is this combination that results in the efficacy of this blend affecting malaria mosquito behaviour. The current malaria vector control strategies in widespread use, the IRS and LLINs, which focus on indoor feeding and resting mosquitoes, may benefit greatly from the addition of a complementary odour-based tool targeted against gravid females. As the next step following this pre-field study, we intend to test the rice odour blend under field conditions to evaluate its efficacy in relation to natural odours in the landscape.
Materials and Methods
Animal model
Anopheles arabiensis, Mwea and Dongola strains, were used for behavioural (International Centre of Insect Physiology and Ecology, Kenya; ICIPE) and electrophysiological (SLU, Sweden) analysis, respectively. The mosquitoes were maintained at 27 ± 2 °C, 75 ± 5% relative humidity under a 12 h light/12 h dark photoperiod. For laboratory experiments, larvae were reared in distilled water and fed Tetramin® fish food (Tetra, Melle, Germany). Alternatively, for semi-field experiments, larvae were reared in water from Lake Victoria and fed on ground Go-Cat® food (Nestlé Purina Petcare company, Nairobi, Kenya). Adults were maintained in cages (30 cm × 30 cm × 30 cm; custom made or Bugdorm, MegaView Science, Talchung, Taiwan), kept under laboratory (27 ± 2 °C, 75 ± 5% relative humidity) or ambient outdoor conditions (ICIPE, Mbita, Kenya), and provided with 10% honey or sucrose solution ad libitum. Five days post-emergence, females were offered sheep blood from an artificial feeder (SLU, Hemotek, Discovery Workshops, Accrington, UK), rat blood (ICIPE, Nairobi) or the arm of a volunteer (ICIPE, Mbita) once every day for two days, 15–30 min per day. For all experiments, gravid females, 3 days post-blood feeding, were selected by visually inspecting the enlarged pale white abdomen and used for bioassays.
Headspace volatile collections
Headspace volatiles of MR1 and MR3 rice cultivars (150 replicates per stage) were collected in Saudi Star Agriculture and Irrigation Project in Gambella, Ethiopia. These available cultivars have desirable agronomic characters, including high yield, aeration seeding, moderate resistance to lodging, and are able to grow under rain-fed and irrigated conditions. The above-ground parts of intact rice plants in the field, at tillering, booting and flowering stages were enclosed in polyamide bags (Toppits, Cofresco, Germany). In addition, the headspace volatiles of water from natural breeding sites (200 replicates) were collected from 1 l poured into a Teflon bag. These sites were selected from pools at the edge of Lake Ziway, Ethiopia that contained Anopheles gambiae sensu lato larvae. A charcoal-filtered continuous airstream (1.0 l min−1) was drawn by a Personal Air Sampler (PAS-500, Spectrex, Redwood City, CA, USA) over the rice plant, or by a diaphragm vacuum pump (KNF Neuberger, Freiburg, Germany) over the breeding water, onto an aeration column for 2 h. Aeration columns were made of Teflon tubing (6 cm × 3 mm id), holding 35 mg Super Q (80/100 mesh; Alltech, Deerfield, IL, USA) between polypropylene wool plugs and Teflon stoppers. The columns were rinsed with 1 ml re-distilled n-hexane (LabScan, Malmö, Sweden) before use. Adsorbed volatiles were eluted with 300 μl re-distilled n-hexane, in order not to dilute the sample, or risk evaporation, between aeration and subsequent GC-EAD and GC-MS analyses. Headspace volatile extracts from each cultivar and phenological stage were pooled separately and then stored in glass vials at −80 °C until used for behavioural, electrophysiological and chemical analyses.
Two-port olfactometer
A two-port still air olfactometer was used to test the mosquito attraction preference for the headspace volatiles collected from the different phenological stages of the MR1 and MR3 cultivars, and from natural breeding water. All assays were conducted between 18:00 and 21:00 local time under red light (200 lx) conditions (icipe, Nairobi), the peak oviposition activity as determined in pilot experiments. For each replicate, 10 gravid females were allowed to acclimatize for 5 min in a custom-made cage (22 cm × 30 cm × 12 cm; L:D:H) constructed of clear vinyl for easy viewing. Thereafter, two dental-wick odour dispensers (4 cm × 1 cm; L:d; DAB Dental AB, Upplands Väsby, Sweden) were simultaneously introduced into the cylindrical vinyl arms (13 cm × 9 cm; L:d) positioned at opposite ends of the cage. The ends of the cylindrical arms were covered by mesh. Mosquito attraction preference to the following treatments was analysed: (a) headspace volatiles of breeding water vs. hexane control, (b) headspace volatiles of each phenological stage of MR1 or MR3 vs. hexane, (c) headspace volatiles of each phenological stage of MR1 or MR3 vs. headspace volatiles of breeding water, and (d) headspace volatiles of the pooled phenological stages of MR1 vs. headspace volatiles of the pooled phenological stages of MR3. No significant difference in attraction preference was found between the headspace breeding water when compared directly against the hexane control. The behavioural response to the two rice cultivars was analysed to increasing release rates of the headspace volatile extract from all phenological stages of MR1 and MR3. After five minutes, the period after which there was no net increase in mosquitoes in each arm, the behavioural responses of the mosquitoes were scored by counting the number of mosquitoes in each port after five minute. Ten replicates per treatment and per dose were performed. Between each trial the bioassay was cleaned with 70% ethanol and the position of the treatments changed to avoid bias.
Oviposition bioassay
The oviposition preference of gravid mosquitoes was analysed in a two-choice assay. Custom-made metal wire framed cages (30 cm × 30 cm × 30 cm) covered with white nylon mosquito netting were used. Two 100 ml polypropylene cups (Qingdao Ori-Color Industry and Commerce Co., Ltd., China), placed in opposite corners of the cages, and filled to the brim with distilled water or field collected breeding water, served as the oviposition substrate, as indicated. The position of the cups was exchanged between experiments. Treatment cups were conditioned by dispensing the headspace volatile extracts of the three phenological stages of MR1 and MR3 as described above directly onto the oviposition substrate; hexane was used as a control. An additional comparison between the headspace volatiles of the pooled phenological stages of MR1 vs. headspace volatiles of the pooled phenological stages of MR3 conditioned breeding water was performed for the oviposition assays. No significant difference in oviposition preference was found between the breeding and distilled water when compared directly against each other. Ten gravid mosquitoes were transferred from the maintenance cage at dusk (18:00), and the numbers of eggs in the two cups were counted on the following day (09:00). All experiments were replicated ten times.
Electrophysiological analysis
Antennal responses of gravid female An. arabiensis to the pooled headspace extract of the three phenological stages of the MR3 rice cultivar were analysed using combined gas chromatography and electroantennographic detection (GC-EAD). An Agilent Technologies 6890 GC (Santa Clara, CA, USA) was equipped with a HP-5 column (30 m × 0.25 mm id, fused silica, 0.25 μm film thickness, Agilent Technologies), and hydrogen was used as the mobile phase at an average linear flow rate of 45 cm s−1. Each sample (2 μl) was injected in splitless mode (30 s, injector temperature 225 °C). The GC oven temperature was programmed from 35 °C (3 min hold) at 10 °C min−1 to 290 °C (10 min hold). At the GC effluent, 4 psi of nitrogen was added and split 1:1 in a Gerstel 3D/2 low dead volume four way-cross (Gerstel, Mülheim, Germany) between the flame ionization detector and the EAD. The GC effluent capillary for the EAD passed through a Gerstel ODP-2 transfer line, which tracked the GC oven temperature, into a glass tube (10 cm × 8 mm), where it was mixed with charcoal-filtered, humidified air (1.5 l min−1). The antenna was placed 0.5 cm from the outlet of this tube. The antennal preparation was made by using the excised head and inserting the distal end of the antenna, after cutting the distal segment, into a recording glass electrode filled with Beadle–Ephrussi Ringer. The recording electrode was connected to a pre-amplifier probe (10×) and then to a high impedance DC amplifier interface box (IDAC-2; Syntech, Kirchgarten, Germany). The reference electrode, filled with Beadle–Ephrussi Ringer, was inserted into the head capsule, and grounded. Fifteen recordings were performed.
Chemical analysis
The pooled MR3 headspace extract was analysed on a combined gas chromatograph and mass spectrometer (GC-MS; 6890 GC and 5975 MS; Agilent Technologies), operated in the electron impact ionization mode at 70 eV. The GC was equipped with fused silica capillary columns (60 m × 0.25 mm, 0.25 μm film thickness), coated with DB-wax (J&W Scientific, Folsom, CA, USA) or HP-5MS (Agilent Technologies). Helium was used as the mobile phase at an average linear flow rate of 35 cm s−1. Two micro-litres of the sample were injected. The temperature programmes were the same as for the GC-EAD analysis. Compounds were identified according to retention times (Kovat’s indices) and mass spectra, in comparison with custom made and NIST05 libraries (Agilent), and confirmed by co-injection of authentic standards: (±)-α-pinene (CAS no. 7785-70-8; Aldrich, 98%), (−)-β-pinene (CAS no. 18172-67-3; Sigma, 99%), 3-carene (CAS no. 13466-78-9; Aldrich, 90%), (±)-limonene (CAS no. 5989-27-5; Sigma, 97%), nonanal (CAS no. 124-19-6; Aldrich, 95%), decanal (CAS no. 112-31-2; Aldrich, 92%), β-caryophyllene (CAS no. 87-44-5; Sigma, 98.5%) and sulcatone (6-methyl-5-hepten-2-one; CAS no. 110-93-0; Fluka, 96%). For quantification, 100 ng of heptyl acetate (99.8% chemical purity; Aldrich) was added as an internal standard to a 20 μl aliquot out of the total 400 μl headspace extract.
Bioassays with synthetic blend
The assays were carried out in the same two-port olfactometer and oviposition bioassay that were used for the natural extract experiments (n = 10). The synthetic blend mimicked the composition and ratio of compounds in the pooled natural extracts of the MR3 rice cultivar (Fig. 1j). To verify the integrity of the blend, a 2 μl aliquot of the synthetic blend was dispensed as above. The head space was collected on an aeration column (as above) connected to a glass funnel (20 cm d), suspended over the cotton wick and oviposition substrate, through which air was drawn using a diaphragm vacuum pump (KNF Neuberger, Freiburg, Germany), at 50 ml min−1 for 30 min. The head space volatiles were analysed as above (Supplementary Fig. 7). Synthetic blends were prepared at different doses, in half orders of magnitude, between 1 to 1000 ng of α-pinene in pentane. The ratio among the compounds within the blend was maintained as a constant across all doses. Then, dose-response assays were conducted with the full blend against its solvent control. Subtractive bioassays followed in order to determine the relative activity of the identified components, after establishing the effective release rate (Fig. 1k,l).
Semi-field trials with the synthetic blend
Semi-field experiments were conducted in an outdoor screened-in enclosure (10.8 m long × 6.7 m wide × 2.4 m high) at icipe, Thomas Odhiambo Campus, Mbita, Kenya. The ground was covered with sand to a depth of 30 cm for placement of modified BG Sentinel traps (Biogents AG, Regensburg, Germany)45. The traps, containing 4 l of distilled water, were recessed in the corners of the screened house at a distance of 1.5 m from the two adjacent walls. The experimental design used was a randomized complete block design and was performed under ambient environmental conditions. Preliminary trials were conducted to determine the attractiveness of the full synthetic blend dissolved in heptane (Merck, Darmstadt, DE), with release rates between 3 to 100 ng min−1 of α-pinene within the blend with four replications to identify the optimum release rate for the test system. The blend was released by diffusion from a wick dispenser made out of a 2 ml glass vial with a pin hole in the centre of the cap through which a cotton wick encased in Teflon protruded into the air46. The wick dispenser allows for the release of all compounds in constant proportions throughout the experiment. Heptane was used as a solvent to allow for the release of compounds throughout the course of the experiment. Two hundred gravid mosquitoes were released in the evening (18:00) and the number of females recaptured in each catch bag were counted at 08:00. Thereafter, trials were carried out using the most cost-effective release rate of 3 ng min−1 for 12 nights with the same number of gravid mosquitoes. Furthermore, controls vs. control trials were performed in which the attraction to both traps baited with heptane was evaluated, with the same number of mosquitoes and replications as above. Dissection of the females post-assay confirmed that 100% of the mosquitoes caught in the experiments were gravid.
Statistical analysis
The attraction (AP) and oviposition (OP) preference for the laboratory assays were determined using indices generated by (T − C)/(T + C); whereas a proportion (Pr) was calculated for the semi-field bioassays: Pr = T(C)/(T + C). T is the number of mosquitoes or eggs associated with the test odours and C the number of mosquitoes or eggs associated with the control odours. The behavioural responses of gravid An. arabiensis in the two-port olfactometer and oviposition bioassay were analysed using a nominal logistic fit model, in which choice was the dependent variable, weighted by the number of (1) mosquitoes in the attraction assays and (2) eggs laid in the oviposition assays, with dose as the independent fixed effect and replicate as a random effect (JMP® Pro 12.0.1. SAS Institute Inc., Cary, NC, USA). Here, we report the χ2 and p-value from the Likelihood Ratio Test. The data from semi-field experiments were analysed by using R software (version 3.00) with generalized linear model (GLZ) using a quasi-binomial distribution in R statistical software version 2.1347.
Additional Information
How to cite this article: Wondwosen, B. et al. Rice volatiles lure gravid malaria mosquitoes, Anopheles arabiensis. Sci. Rep. 6, 37930; doi: 10.1038/srep37930 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
O’Meara, W. P. et al. Changes in the burden of malaria in sub-Saharan Africa. Lancet Infect Dis 10, 545–555 (2010).
Ranson, H. & Lissenden, N. Insecticide resistance in African Anopheles mosquitoes: A worsening situation that needs urgent action to maintain malaria control. Trends Parasitol 32, 187–196 (2016).
Trape, J.-F. et al. Malaria morbidity and pyrethroid resistance after the introduction of insecticide-treated bednets and artemisinin-based combination therapies: a longitudinal study. Lancet Infect Dis 11, 925–932 (2011).
Ranson, H. et al. Pyrethroid resistance in African anopheline mosquitoes: what are the implications for malaria control? Trends Parasitol 27, 91–98 (2011).
Toé, K. H. et al. Increased pyrethroid resistance in malaria vectors and decreased bed net effectiveness, Burkina Faso. Emerg Infect Dis 20, 1691–1696 (2014).
Zhou, G. et al. Changing patterns of malaria epidemiology between 2002 and 2010 in Western Kenya: the fall and rise of malaria. PloS One 6, e20318 (2011).
Sinka, M. E. et al. Modelling the relative abundance of the primary African vectors of malaria before and after the implementation of indoor, insecticide-based vector control. Malar J 15, 1 (2016).
Riehle, M. M. et al. A cryptic subgroup of Anopheles gambiae is highly susceptible to human malaria parasites. Science 331, 596–598 (2011).
Killeen, G. F. et al. Eliminating malaria vectors. Parasit Vectors 6, 172 (2013).
Sougoufara, S. et al. Biting by Anopheles funestus in broad daylight after use of long-lasting insecticidal nets: a new challenge to malaria elimination. Malar J 13, 10.1186 (2014).
Kitau, J. et al. Species shifts in the Anopheles gambiae complex: do LLINs successfully control Anopheles arabiensis? PLoS One 7, e31481 (2012).
Russell, T. L. et al. Linking individual phenotype to density-dependent population growth: the influence of body size on the population dynamics of malaria vectors. Proc R Soc Lond [Biol], rspb20110153 (2011).
Mwangangi, J. M. et al. Shifts in malaria vector species composition and transmission dynamics along the Kenyan coast over the past 20 years. Malar J 12, 1–9 (2013).
Ojuka, P. et al. Early biting and insecticide resistance in the malaria vector Anopheles might compromise the effectiveness of vector control intervention in Southwestern Uganda. Malar J 14, 1 (2015).
Fillinger, U. & Lindsay, S. W. Larval source management for malaria control in Africa: myths and reality. Malar J 10, 10.1186 (2011).
Okal, M. N. et al. Water vapour is a pre-oviposition attractant for the malaria vector Anopheles gambiae sensu stricto. Malar J 12, 365 (2013).
Ogbunugafor, C. B. & Sumba, L. Behavioral evidence for the existence of a region-specific oviposition cue in Anopheles gambiae sensu stricto. J Vector Ecol 33, 321–324 (2008).
Rejmánková, E. et al. Volatile substances from larval habitats mediate species-specific oviposition in Anopheles mosquitoes. J Med Entomol 42, 95–103 (2005).
Sumba, L. A. et al. Mediation of oviposition site selection in the African malaria mosquito Anopheles gambiae (Diptera: Culicidae) by semiochemicals of microbial origin. Int J Trop Insect Sci 24, 260–265 (2004).
Lindh, J. M. et al. Discovery of an oviposition attractant for gravid malaria vectors of the Anopheles gambiae species complex. Malar J 14, 119 (2015).
Ndenga, B. A. et al. Productivity of malaria vectors from different habitat types in the western Kenya highlands. PLoS One 6, e19473 (2011).
Minakawa, N. et al. Habitat characteristics of Anopheles gambiae sensu stricto larvae in a Kenyan highland. Med Vet Entomol 18, 301–305 (2004).
FAO. Irrigation management transfer. FAO Water Reports 32 (2007).
Mwangangi, J. M. et al. Survival of immature Anopheles arabiensis (Diptera: Culicidae) in aquatic habitats in Mwea rice irrigation scheme, central Kenya. Malar J 5, 114 (2006).
Ye-Ebiyo, Y., Pollack, R. J. & Spielman, A. Enhanced development in nature of larval Anopheles arabiensis mosquitoes feeding on maize pollen. Am J Trop Med Hyg 63, 90–93 (2000).
Merritt, R., Dadd, R. & Walker, E. Feeding behavior, natural food, and nutritional relationships of larval mosquitoes. Ann Rev Entomol 37, 349–374 (1992).
Mwangangi, J. M. et al. Anopheles larval abundance and diversity in three rice agro-village complexes Mwea irrigation scheme, central Kenya. Malar J 9, 10.1186 (2010).
Ijumba, J. & Lindsay, S. Impact of irrigation on malaria in Africa: paddies paradox. Med Vet Entomol 15, 1–11 (2001).
Diakité, N. R. et al. Spatial and temporal variation of malaria entomological parameters at the onset of a hydro-agricultural development in central Côte d’Ivoire. Malar J 14, 1–11 (2015).
Mboera, L. E. et al. Spatial abundance and human biting rate of Anopheles arabiensis and Anopheles funestus in savannah and rice agro-ecosystems of Central Tanzania. Geospat Health 10 (2015).
Reid, M. C. & McKenzie, F. E. The contribution of agricultural insecticide use to increasing insecticide resistance in African malaria vectors. Malar J 15, 1 (2016).
Darriet, F., Rossignol, M. & Chandre, F. The combination of NPK fertilizer and deltamethrin insecticide favors the proliferation of pyrethroid-resistant Anopheles gambiae (Diptera: Culicidae). Parasite 19, 159–164 (2012).
Mutero, C. M. et al. Ammonium sulphate fertiliser increases larval populations of Anopheles arabiensis and culicine mosquitoes in rice fields. Acta Trop 89, 187–192 (2004).
Service, M. Mortalities of the immature stages of species B of the Anopheles gambiae complex in Kenya: comparison between rice fields and temporary pools, identification of predators, and effects of insecticidal spraying. J Med Entomol 13, 535–545 (1977).
Jarju, L. et al. Agriculture and the promotion of insect pests: rice cultivation in river floodplains and malaria vectors in The Gambia. Malar J 8, 170 (2009).
Muturi, E. J. et al. Mosquito species diversity and abundance in relation to land use in a riceland agroecosystem in Mwea, Kenya. J Vector Ecol 31, 129–137 (2006).
Takken, W. & Knols, B. G. Odor-mediated behavior of Afrotropical malaria mosquitoes. Ann Rev Entomol 44, 131–157 (1999).
Fujii, T., Hori, M. & Matsuda, K. Attractants for rice leaf bug, Trigonotylus caelestialium (Kirkaldy), are emitted from flowering rice panicles. J Chem Ecol 36, 999–1005 (2010).
Okal, M. N. et al. Analyzing chemical attraction of gravid Anopheles gambiae sensu stricto with modified BG-Sentinel traps. Parasit Vectors 8, 301 (2015).
Afify, A. & Galizia, C. G. Chemosensory cues for mosquito oviposition site selection. J Med Entomol 52, 120–130 (2015).
Torres-Estrada, J. L. et al. Vegetation-derived cues for the selection of oviposition substrates by Anopheles albimanus under laboratory conditions. J Am Mosq Control Assoc 21, 344–349 (2005).
Herrera-Varela, M. et al. Habitat discrimination by gravid Anopheles gambiae sensu lato–a push-pull system. Malar J 13, 1 (2014).
Knudsen, J. T., Tollsten, L. & Bergström, L. G. Floral scents – a checklist of volatile compounds isolated by head-space techniques. Phytochemistry 33, 253–280 (1993).
Dudareva, N. et al. Plant volatiles: recent advances and future perspectives. Crit Rev Plant Sci 25, 417–440 (2006).
Okal, M. N. et al. Analysing the oviposition behaviour of malaria mosquitoes: design considerations for improving two-choice egg count experiments. Malar J 14, 1–17 (2015).
Birgersson, G. et al. Pheromone production, attraction, and interspecific inhibition among four species of Ips bark beetles in the Southeastern USA. Psyche 2012 (2012).
Team, R. C. R. : a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. URL http://www.R-project.org (2013).
Acknowledgements
This study was funded by the Swedish International Development Cooperation Agency and the Swedish Research Council (RI), and by a grant from the Organisation of Women in Science for the Developing World (OWSD) (BW). We would like to thank the Saudi Star Agriculture and Irrigation Project in Gambella, Ethiopia, for access to the rice varieties and Dr Majid Ghaninia for the illustration of the GC-EAD setup.
Author information
Authors and Affiliations
Contributions
B.W., S.R.H., E.S., U.F. and R.I. designed the study. S.R.H., E.S., H.T., U.F., B.T. and R.I. supervised and B.W. conducted the laboratory and semi-field work. G.B. contributed his expertise to the chemical analyses. B.W., S.R.H. and R.I. wrote the manuscript, while G.B., H.T., U.F. and B.T. critically reviewed the manuscript. All authors read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wondwosen, B., Birgersson, G., Seyoum, E. et al. Rice volatiles lure gravid malaria mosquitoes, Anopheles arabiensis. Sci Rep 6, 37930 (2016). https://doi.org/10.1038/srep37930
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37930
This article is cited by
-
Asymptomatic and submicroscopic malaria infections in sugar cane and rice development areas of Ethiopia
Malaria Journal (2023)
-
Distance from human dwellings differentially affects the efficacy of a synthetic cattle urine odour lure to trap malaria vectors
Malaria Journal (2023)
-
Odour-mediated oviposition site selection in Aedes aegypti depends on aquatic stage and density
Parasites & Vectors (2023)
-
Manure and mosquitoes: life history traits of two malaria vector species enhanced by larval exposure to cow dung, whilst chicken dung has a strong negative effect
Parasites & Vectors (2022)
-
Malaria mosquitoes acquire and allocate cattle urine to enhance life history traits
Malaria Journal (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.