Abstract
Pooled DNA based GWAS to determine genetic association of SNPs with visual acuity (VA) outcome in anti-vascular endothelial growth factor (anti-VEGF) treated neovascular age-related macular degeneration (nAMD) patients. We performed pooled DNA based GWAS on 285 anti-VEGF treated nAMD patients using high density Illumina 4.3 M array. Primary outcome was change in VA in Early Treatment Diabetic Retinopathy Study (ETDRS) letters after 6 months of anti-VEGF treatment (patients who lost ≥5 ETDRS letters classified as non-responders and all remaining classified as responders). GWAS analysis identified 44 SNPs of interest: 37 with strong evidence of association (p < 9 × 10−8), 2 in drug resistance genes (p < 5 × 10−6) and 5 nonsynonymous changes (p < 1 × 10−4). In the validation phase, individual genotyping of 44 variants showed three SNPs (rs4910623 p = 5.6 × 10−5, rs323085 p = 6.5 × 10−4 and rs10198937 p = 1.30 × 10−3) remained associated with VA response at 6 months. SNP rs4910623 also associated with treatment response at 3 months (p = 1.5 × 10−3). Replication of these three SNPs in 376 patients revealed association of rs4910623 with poor VA response after 3 and 6 months of treatment (p = 2.4 × 10−3 and p = 3.5 × 10−2, respectively). Meta-analysis of both cohorts (673 samples) confirmed association of rs4910623 with poor VA response after 3 months (p = 1.2 × 10−5) and 6 months (p = 9.3 × 10−6) of treatment in nAMD patients.
Similar content being viewed by others
Introduction
Age-related macular degeneration (AMD) is a common complex progressive neurodegenerative disease in the elderly, which can lead to irreversible severe vision loss1. Vision is threatened when AMD advances to its late sequelae of either geographic atrophy (GA) or choroidal neovascularization (CNV) also known as nAMD2. One of the most important regulators of the neovascularization process is vascular endothelial growth factor A (VEGF-A)3. Currently, the most effective treatment for nAMD is inhibition of VEGF through the use of recombinant, humanised anti-VEGF monoclonal antibodies such as ranibizumab (Lucentis), aflibercept (Eylea) or off-label bevacizumab (Avastin). These drugs have been shown in multiple studies4,5,6,7,8 to be efficacious in improving vision, but a varying response to anti-VEGF treatment has been observed. Approximately 10% of patients showed no improvement in visual acuity (VA) (loss of >15 ETDRS letters), and exhibited a continuous decline in VA over two years of treatment similar to that previously reported for both ANCHOR and MARINA trials5,6. This range of variable VA response may in part be explained by genetic predisposition.
It is well established that genetic factors are associated with risk of developing AMD9. Several of the genes associated with AMD are also genes encoding components of the neovascularization pathway and have previously been investigated in variation to anti-VEGF treatment response studies8,10,11,12. However, conflicting findings have been reported thus far.
The current study aimed to investigate associations with genetic variants in a hypothesis-free manner using a genome-wide association study (GWAS). We investigated whether genetic factors influencing ranibizumab treatment outcomes in nAMD patients could be identified through the initial use of a GWAS pooling strategy followed by a technical validation and subsequent replication in an independent cohort.
Results
The objective of this study was to investigate whether genetic variants could be identified that might influence the treatment outcome after anti-VEGF treatment in nAMD patients. The demographic characteristics of patients of the Melbourne discovery cohort and the replication cohort are shown in Table 1. A total of 297 patients from the Melbourne discovery cohort (285 individuals used initially in the pooled GWAS, plus an additional 12 ranibizumab-treated AMD patients were included, giving a total of 297 individuals) and 376 patients from the replication cohort met the study inclusion criteria with a mean age of 79.2 years and 77.1 years, respectively. A mean baseline VA (at the time of first injection) of 51 ETDRS letters was observed in the Melbourne discovery cohort and 52.6 ETDRS letters in the replication cohort (Table 1). In the Melbourne discovery cohort, sex, smoking, type of lesion, size of CNV and number of injections up to 6 months of treatment, showed no significant association with change in VA at 3 and 6 months of treatment (p > 0.05). Baseline VA showed a consistent negative association with change in VA at 3 and 6 months (p < 0.001) in both the Melbourne and replication cohort. In addition, age at the time of nAMD treatment was associated with change in VA at 3 months (p < 0.001) but not at 6 months in the replication cohort (p > 0.05) (Supplementary Table S1). Both the discovery and replication cohort have similar demographic and clinical characteristics except for the size of the CNV.
First Phase- Genome wide association Study
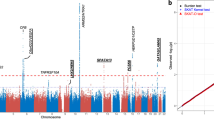
First phase results from the pooled GWAS are shown in a Manhattan plot in Fig. 1. A total of 44 SNPs from the pooled GWAS were selected for technical validation through independent individual genotyping on the basis of a genome-wide significant p-value (37 SNPs, p < 9 × 10−8), SNPs in genes involved in drug resistance response (‘pharmagenes’) (2 SNPs, p < 5.0 × 10−6) and SNPs leading to a missense change in a coding region of a gene (5 SNPs, p < 1.0 × 10−4) (Table 2).
Manhattan plot of SNPs tested in a pooled GWAS for response to anti-VEGF treatment.
The X-axis indicates the chromosomal position of the SNPs and the Y-axis shows their corresponding p-value (−log10). The red line indicates the threshold for genome-wide significance (p < 5 × 10−8) and the blue line indicates the threshold for suggestive association (p < 1 × 10−5).
Second Phase- Validation
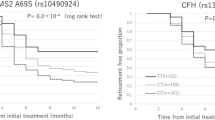
In the second phase, all 44 SNPs from the first phase were genotyped in 297 patients (the same cohort as in phase 1 but with the addition of 12 extra patient samples who became available between the time of phase1 and phase2). This individual genotyping confirmed the 3 SNPs (rs4910623, rs323085, rs10158937) to be significantly associated (p < 0.05) with response at 6 months of ranibizumab treatment after Bonferroni correction for 44 independent tests. SNP rs4910623 showed the highest significance (p = 5.7 × 10−5), in which the ‘G’ risk allele led to a worse response (OR = 2.58 [95% CI = 1.63–4.10]). While the C allele of both rs323085 and rs10158937 (p = 6.5 × 10−4 and p = 1.30 × 10−3, respectively) appeared protective (OR = 0.16 [95% CI = 0.06–0.46] and OR = 0.32 [95% CI = 0.17–0.65], respectively) (Table 3).
Since a loading dose of three monthly injections is prescribed to all patients, we determined if the effect of these three selected SNPs in treatment outcome could be seen at this time point. The G allele of rs4910623 showed an association with worse outcome at three months of treatment (p = 1.5 × 10−3, OR = 2.23 [95% CI = 1.36–3.66]).
SNP rs10158937 also showed an association with the C allele leading to a better response after 3 months (p = 9.0 × 10−3, OR = 0.36 [95% CI = 0.17–0.77]), consistent with the six months result. However rs323085 did not show significant association at three months (Table 3).
Third Phase- Replication
In the third phase, we analysed the association of the three SNPs with treatment outcome in a replication cohort of European descent, consisting of 215 ranibizumab-treated AMD patients following pro re nata treatment protocol. The effect of rs4910623 replicated at six months of treatment (p = 3.5 × 10−2, OR = 1.71 [95% CI = 1.04–2.80]). At the 3 month treatment time point all replication patients were included in the analysis (n = 376) as they were all treated in a similar manner to the discovery cohort. The effect of this SNP on treatment response was also similar at this time point (p = 2.4 × 10−3, OR = 1.8 [95% CI = 1.24–2.71]). SNPs rs323085 and rs10158937 were not associated with treatment response in the replication cohort (Table 4).
The frequency of rs4910623 risk allele (G) in the Melbourne discovery cohort was 71% and 72% for the non-responder group (3 and 6 months respectively) in comparison to 51% and 49% in the responders group (3 and 6 months, respectively) (Table 3). A similar change in allele frequency was also seen in the replication cohort with the frequency of the risk allele G being 62% and 38% in the non-responder group compared to 48% and 52% in the responders group (3 and 6 months (pro re nata patients), respectively) (Table 4). The overall distribution of change in VA corresponding to each genotype of rs4910623 in both cohorts is shown in Fig. 2.
Meta-Analysis
A meta-analysis of SNP rs4910623 in both the Melbourne discovery cohort and the replication cohort showed differences in allele distribution between responders and non-responders at 3 months (p = 1.21 × 10−5) and 6 months (p = 9.3 × 10−6) of treatment (only pro re nata patients from the replication cohort were included) indicating the influence of rs4910623 on outcome of ranibizumab treatment in AMD patients (Table 5).
Discussion
We sought to identify genetic variants that influence the ranibizumab treatment response in AMD. Using a pooled DNA GWAS approach, we identified SNP rs4910623 in the promoter region of the OR52B4 gene as being associated with a worse response to ranibizumab treatment. The association was validated in the same cohort through individual genotyping and confirmed in an independent replication cohort of AMD patients of European descent. Individuals carrying the risk allele G did not respond as well to ranibizumab treatment in either cohort after 3 or 6 months of treatment compared to those with the A allele. In the replication cohort we observed significant association of rs4910623 with change in VA in patients on pro re nata treatment regimen at 6 months. However, 47 patients from Montreal were not included in this analysis as they followed a treat and extend regimen. We undertook an additional sensitivity analysis where these 47 patients were included back into the 6 month analysis time point to provide a total of 262 patients. A similar trend for association of the G allele of rs4910623 was seen (p = 4.6 × 10−2, OR = 1.53 [95% CI = 1.00–2.30]). As this group consisted of only 47 patients it was too small to draw any conclusive conclusions about whether this treatment protocol significantly affected the association of the G allele of rs4910623 with change in VA. In the meta-analysis we found significant association of rs4910623 patients with change in VA at both 3 and 6 months of treatment.
Two other SNPs, rs323085 and rs10158937, located 202 kb 3′ of LOC100287225and 42 kb at 3′ of LEPR gene respectively, did not remain associated with change in VA after replication. The inconsistency in the results for these two SNPs could indicate that they are false positive findings.
The baseline characteristics of both the Melbourne discovery and replication cohort did not reveal significant association with change in VA except for the baseline VA at 3 and 6 months of treatment. Baseline VA is negatively associated with change in VA, thus lower baseline VA will lead to a greater VA gain after treatment and vice versa. Therefore our findings are in agreement with the ANCHOR and MARINA trials where higher VA at baseline was associated with a smaller gain in VA at 12 and 24 months of treatment, respectively13. This in-turn reflects a floor and ceiling effect of baseline VA14. Based on definition of response in our study it may have floor effect of baseline VA where individuals with higher baseline VA will have limited chance to gain vision after the treatment and vice versa. On the other hand, the baseline VA is also reported as one of the predictors of treatment outcome for ranibizumab15. To overcome these potential issues we incorporated baseline VA as a covariate into our analysis. However, there was a difference in baseline CNV size in the discovery and replication cohort. This is probably due to differences in measurements between the cohorts with the Melbourne discovery cohort being based on one optic-disc area is equal to 2.54 mm2, using an optic disc diameter of 1.8 mm whereas the replication cohort was based on each patient’s optic disc area.
Using the HaploReg database16, SNP rs4910623 is predicted to alter the GATA and TCF4 regulatory motifs 22 bp upstream of the OR52B4 gene. We also analysed the surrounding variants within its linkage disequilibrium (LD) block from the 1000 Genomes Project (r2 = 0.8, 250 Kb). A number of the identified SNPs in this LD block are predicted to alter transcriptional binding sites (Supplementary Table S3). Thus, this region may play an important role in transcriptional regulation of this gene.
The OR52B4 protein (Q8NGK2, UniprotKB accession number) consists of 314 amino acids (aa) and is a member of the G-coupled protein receptor (GPCR) family, which is located in the plasma membrane of the cell. Interestingly we find the conserved domain of Q8NGK2 17 is a 7-transmembrane domain (7tm) region which is highly conserved with the 7tm_4 super family domain of all the olfactory receptors (33–321 aa) and also the 7tm_1 domain of the 7 transmembrane receptor (rhodopsin family) (43–294 aa) as indicated using the NCBI Conserved Domain Database (CCD). (http://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi?seqinput=NP_001005161.2). Rhodopsin is also member of GPCR family and it functions to capture photons of light and trigger the intra-cellular photo-transduction cascade, forming the molecular basis of vision18. GPCRs are highly selective and expressed in specific tissues and cells, making them particularly important as potential drug targets as >50% of therapeutic agents in the market target these receptors19.
To assess expression of the OR52B4 gene in human eye tissue, we used online resource: “The Ocular Tissue Database”20. Expression of the OR52B4 gene was present in the retina, optic nerve and choroid (Supplementary Table S4). The identification of olfactory gene expression in the eye is relatively new, but not surprising, as21 recently published expression profiling of a number of olfactory receptors (olfr) in mouse cornea, retina and choroid. Their analysis of eye tissue expression confirmed the presence of many olfr, with one of them, olfr547 representing a homolog of the human OR52B4 gene (85% identity). Interestingly, olfr547 is a close relative of olfr78, which has recently been shown to function in regulation of blood pressure in smooth muscle cells22,23. Expression of genes in the olfr pathway in mouse ocular tissues is mediated by the olfaction specific Gα protein, localised in the nuclear layer of the retina and previously thought to be only expressed in the olfactory epithelium21. Moreover, olfactory genes have been shown to be associated with cancer and other blood diseases, suggesting expression and function of olfactory proteins in a number of different pathways23,24,25. Interestingly, another olfactory receptor (OR10J5) was recently reported by Kim and colleagues26 to control cell migration and phosphorylation of signaling during angiogenesis in mice. However the functional role of OR52B4 gene in angiogenesis needs to be elucidated and it has so far not previously been reported to be associated with pharmacogenetic response to AMD.
Previous studies investigating genetic factors in anti-VEGF treatment outcome have assessed known AMD risk-associated genes and genes encoding components of the neovascularization pathway8,10,11,12. However, most of these studies have produced variable results; for example, the Y402H variant in the CFH gene has been associated with poor VA outcome in some studies but in other studies has been reported to show no association with VA. Similarly, inconsistent results of association were reported for ARMS2/HTRA1 11,27. Our pooled GWAS findings did not identify genome-wide significance for SNPs in the previously associated known AMD risk genes CFH, VEGF-A, HTRA1/ARMS2 (Supplementary Table S5). These findings are in agreement with our previous study28 and the CATT study, the largest current cohort in which major AMD risk alleles have been investigated for treatment outcome following treatment with bevacizumab, where no association with the CFH, VEGF, VEGFR2 and HTRA1/ARMS2 genes was identified with anti-VEGF treatment response11,12. To date, only one prior GWAS has been undertaken to identify genetic risk factors for treatment response to AMD. That study was conducted on 65 patients and reported the SNP rs7607942 near the ERBB4 gene to be associated with poor VA outcome after 6 months of treatment, although this SNP did not reach genome-wide significance (uncorrected p = 6.692 × 10−6)29. Major drawbacks of that study were the small sample size and lack of replication of the reported association in an independent ranibizumab-treated AMD cohort. We did not find any association of this SNP in the current study (p > 0.05).
We chose to use a pooled DNA GWAS strategy to investigate response to anti-VEGF treatment in AMD as this provided a cost-effective and efficient approach to identify genetic associations23,30,31,32,33. We were able to technically validate ~70% of the 44 SNPs that were identified in the pooling strategy at a nominal significance level. Whilst most SNPs replicated, the level of significance of the SNPs diminished following technical validation. Possible explanations for the diminished significance may be due to experimental error during pool construction, array-specific errors resulting from the use of a limited number of arrays per pool, variation in allele frequency due to small sample size in some of the pools, allele-specific bias skewing the results for some SNPs, and/or small differences in the set of samples used in the validation stage34,35.
For the replication cohort, the number of injections at 6 month of treatment was not available, however it is interesting to note that a variation in treatment regime through change in number of injections between 3 and 6 months does not appear to ameliorate the genetic effect seen in the current study. However, after individual genotyping, replication and meta-analysis, SNP rs4910623 remained associated with worse functional outcome (change in VA) after the anti-VEGF treatment in AMD. Currently there is much interest in the role of anatomical features such as fluid clearance as a measure of treatment response. However, it was not considered in this analysis but this would be useful to examine in the future as to whether this SNP also affects fluid clearance and central macular thickness (CMT) on OCT following anti-VEGF treatment. To our knowledge, SNP rs4910623 and the 2 other SNPs associated with good response in the discovery phase have not previously been reported as associated with anti-VEGF treatment outcome in any type of cancer, thus it will be interesting to investigate the role of these SNPs in cancer following anti-VEGF treatment. Furthermore, much work needs to be undertaken to examine the biological basis of this phenomenon as well as the functional role of OR52B4 gene in models of angiogenesis, such studies could lead to the development of other therapies for AMD patients that can be used as an adjunct to existing treatment. This, in turn, offers the prospect of personalising treatment, based on genotype and opens up the route for exploring other drug treatment opportunities. In conclusion, we report for the first time a pooled DNA based GWAS on pharmacogenetic response to ranibizumab treatment in AMD that identifies association of variant rs4910623 in the OR52B4 gene. This finding was replicated in an independent replication cohort, suggesting that this gene may be involved in the response to anti-VEGF treatments in AMD patients of European decent. Finally, our data suggests that the SNP rs4910623 could be used as a diagnostic genetic marker before anti VEGF treatments for AMD.
Methods
Patient Recruitment and Data Collection
Melbourne Discovery Cohort
The study was approved by the Human Research and Ethics Committee of the Royal Victorian Eye and Ear Hospital (RVEEH) and performed in accordance to the tenets of the Declaration of Helsinki (7th revision). Written informed consent was obtained for all study participants before participation. Patients were included in the study when they were >50 years of age, presented with an active sub-foveal CNV secondary to AMD, and received intravitreal anti-VEGF injections. Neovascularization was confirmed by fundus photography (Canon CR6-45NM; Canon Saitama, Japan), fundus fluorescein angiography (FFA) using IMAGEnet 2000 (Topcon Corporation, Tokyo, Japan), and optical coherence tomography (OCT) with Stratus OCT version 5.0.1 (Carl Zeiss Meditec, Dublin, CA) or Cirrus HD-OCT version 6.0.0.599 (Carl Zeiss Meditec). Exclusion criteria were nAMD associated with non-AMD conditions such as degenerative myopia, angioid streaks and hereditary retinal disorders. In addition bilateral nAMD patients who had different VA response between both eyes and treatment with either laser photocoagulation or photodynamic therapy before anti-VEGF treatment as previously reported were also excluded36. Initially a total of 315 nAMD patients were retrospectively recruited from the medical retinal clinic of the RVEEH. A total of 297 patients meet the eligibility criteria consisting of 277 patients with unilateral nAMD and 20 patients with bilateral nAMD who had the same (either gain or loss) VA response in both eyes. A total of 18 bilateral patients did not meet the eligibility criteria because they had differing VA response in both eyes.
VA was measured using an Early Treatment Diabetic Retinopathy Study (ETDRS) chart at 4-metre distance at each visit. The majority of nAMD patients were treated with monthly ranibizumab injections (Lucentis; Novartis Pharma AG, Basel, Switzerland) for 3 months of initial dosing except for 27 (9.0%) patients who first received a single injection of bevacizumab (Avastin; Roche, Basel, Switzerland) who then went on to receive normal dosing using ranibizumab. After 3 monthly injections, pro re nata treatment regimen were adopted for follow-up injections, that was based on evaluation by a retinal specialist, who considered loss of VA ≥ 5 EDTRS letters, the presence of fluid on an OCT scan or presence of chronic or new retinal haemorrhage as criteria for treatment. Data on demographics and clinical history (including VA and treatment regimens) were collected for all participating patients.
Replication Cohort
Ethical approval for the replication cohort was obtained from the local ethic committees of the Radboud university medical center, University of Cologne, and McGill University Health Centre. Written informed consent was acquired from all participants.
The replication cohort consisted of 376 treatment naïve patients of European descent with choroidal neovascularization secondary to AMD. Patients were diagnosed by retinal specialists based on ophthalmic examination, spectral-domain OCT (Spectralis HRA + OCT, Heidelberg Engineering, Heidelberg, Germany) or fluorescein angiography (FA) (Spectralis HRA + OCT, Heidelberg Engineering, Heidelberg, Germany; or Imagenet, Topcon Corporation, Tokyo, Japan). All included patients were over the age of 50 years, had not undergone any previous ophthalmic surgery, except for cataract removal, and did not have other retinal disorders besides AMD.
A total of 144 patients were treated at the Department of Ophthalmology of the Radboud university medical centre, Nijmegen, the Netherlands and 182 at the University of Cologne, Germany. These patients were included in the European Genetic Database (EUGENDA), a multicenter database for the clinical and molecular analysis of AMD. The remaining 50 patients were treated at the McGill University Health Center, Montreal, Canada. All patients received the loading dose of three consecutive monthly intravitreal injections of 0.5 mg ranibizumab (Lucentis; Novartis Pharma AG, Basel, Switzerland). Afterwards, they were followed up monthly and treated on a pro re nata regimen at the clinics of Nijmegen and Cologne and on a treat-and-extend regimen at the clinic of Montreal. In Nijmegen and Cologne (pro re nata regimen), in cases of persistence or recurrence of choroidal neovascularization, which was defined as loss of VA of ≥5 ETDRS letters, leakage seen on FA, fluid seen by OCT, or new macular hemorrhage or fluid, three consecutive monthly ranibizumab injections were administered. VA was assessed before treatment and after the three loading injections for all patients. VA was also recorded after 6 months of treatment for 262 patients. For 303 patients, Snellen VA measurements were recorded retrospectively and 73 patients were followed up prospectively using ETDRS VA. Baseline variables were collected using questionnaires or retrieved from the patient files. One eye was selected per patient; if both eyes received treatment, the first eye treated was selected and if treatment started simultaneously, the study eye was chosen randomly.
First phase - Pooled DNA Based GWAS in the Melbourne Discovery Cohort
In the first phase of the study, a GWAS using a DNA pooling approach was conducted (Macgregor et al.37). A peripheral blood sample was collected from each patient and DNA was extracted (Qiagen, Victoria, Australia). Genomic DNA was quantified by Nanodrop Specrophotometer (Thermo Scientific) for a DNA concentration ≥175 ng/μl. A total of 285 samples were selected for the pooling cohort (12 samples were excluded due to a low DNA concentration required for pooling). Each pool was given a unique ID number, based on presenting VA. Non-responders from each pool were classified as those who showed loss of ≥5 EDTRS letters VA following 6 months of ranibizumab treatment and all remaining patients were classified as responders. Six equimolar DNA pools were generated with a pool size varying from 9 to 133 samples (Supplementary Table S2). An equal amount of DNA from each sample (varying from 0.2 μg to 0.6 μg) was added to the appropriate pool, with a minimum volume of 5 μl per sample to reduce pipetting errors at low volume. Pooled genomic DNAs were assayed using an Illumina Human Omni5-Quad Bead Chip that captured 4.3 million SNP markers. We randomised the pooled samples on the genotyping arrays to avoid spurious associations that may arise from array position. Approximately 4,600 indels or copy number variants were removed from the analyses. We applied stringent quality control (QC) as previously described (Lu et al.38). Briefly, we excluded SNPs with minor allele frequency (MAF) <1% from the reference panel of CEU (Utah residents with ancestry from northern and western Europe) samples in the 1000 Genome Project. Thus, the number of SNPs was reduced to 2.5 million. Further QC criteria for excluding SNPs from the analysis were: (1) SNPs with more than 10% negative probe scores; (2) a sum of the mean raw red and green intensity values less than 1200 for each array (to ensure calibration); (3) SNPs with a small number of expected probes in pools (SNPs with MAF between 1–5% required at least half of the expected number of probes, and SNPs with MAF > 5% required at least a third; the expected number of probes was calculated as the average number of probes per pool per SNP multiplied by the number of samples in each pool); (4) non-autosomal SNPs; (5) SNPs where the variance of the estimated allele frequency was significantly different between all pools. After applying these QC filters, a total of 1,940,408 SNPs were retained for further analysis. A GWAS was carried out between responders and non-responders from three different baseline VA categories and then results were combined in a meta-analysis, which included 225 responders and 60 non-responders.
Second Phase - Technical Replication in the Melbourne Discovery Cohort
In the 2nd phase, the most interesting associations found in the pooled DNA GWAS were validated by undertaking individual genotyping of the original 285 samples, plus the inclusion of the additional 12 patient samples that subsequently became available whose initial concentration was not enough for the pooling GWAS. These samples were genotyped individually for 44 SNPs using the MassArray platform (Agena Bioscience, San Diego, CA) and performed as previously described36.
Third Phase - Replication in an Independent Cohort of European Descent
In the third phase of the study, SNPs that showed association (p < 0.05 after correction for multiple testing) in the 2nd phase of the study were analysed in an independent ranibizumab-treated AMD cohort of European descent. DNA was extracted and quantified in Nijmegen, the Netherlands. Subsequently, DNA samples of a total of 376 AMD patients were genotyped in Melbourne using the MassArray platform (Agena Bioscience, San Diego, CA) as previously described36.
Statistical analysis
Linear regression was undertaken to assess the role of non-genetic factors (potential cofounders), including age, sex, smoking status, CNV size, lesion type, number of injections and baseline VA, on the dependent variable “change in VA” at 3 and 6 months respectively.
The GWAS of pooled DNA was undertaken at the Queensland Institute of Medical Research (QIMR) as previously described37. A customised pipeline developed at QIMR was used for data analysis37,38. Briefly, a GWAS was run within each pool comparison, i.e. a linear model based approach was used for association between SNP and allele frequency difference in responder compared to non-responder pools accounting for pooling errors. A meta-analysis was performed summarising the GWAS results from the multiple pool comparisons, weighted by inverse variances. Finally, we applied post-analysis checks to the meta-analysis results in order to reduce the chance of false positive findings, filtering any SNP that had results from only one of the multiple pool comparisons and those with large discrepancies in association results of the SNP itself and its proxies.
For individual genotyping data, logistic regression was used to assess differences in allele frequency between the responder and non-responder groups (defined in the same manner as for the GWAS) and adjusted for baseline VA in the discovery cohort and baseline VA and age at first injection in the replication cohort. In this replication cohort, different treatment regimens were administered after the 3 initial monthly injections. Therefore, we stratified the analysis for the replication cohort into two groups; pro-re-nata and treat-and-extend. These groups consisted of 215 pro re nata (Nijmegen and Cologne) and 47 treat-and-extend (Montreal) patients respectively. Results are reported as odds ratios (OR), 95% confidence intervals (95% CI) and p value with statistical significance being defined as p < 0.05 following Bonferroni correction for multiple testing. For the analyses, the statistical software Plink V1.07 (http://pngu.mgh.harvard.edu/~purcell/plink/) and SPSS IBM SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, New York, USA) were used. To prevent any possible confounding, the analyses performed in the both the Melbourne discovery and the replication cohort were corrected for baseline VA.
Meta-analysis of the discovery and replication cohorts was performed using Metal software39. For 3 month meta-analysis all the patients from Melbourne discovery and the replication cohort were included because they all were treated with 3 initial doses of anti-VEGF injections while for the 6 months analysis we considered the fact that the Melbourne discovery cohort was treated using a pro re nata strategy. Therefore, we included only the 215 pro re nata treated patients from Nijmegen and Cologne and excluded the 47 patients from Montreal who were treated with a treat-and-extend regimen.
Additional Information
How to cite this article: Riaz, M. et al. GWAS study using DNA pooling strategy identifies association of variant rs4910623 in OR52B4 gene with anti-VEGF treatment response in age-related macular degeneration. Sci. Rep. 6, 37924; doi: 10.1038/srep37924 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Gehrs, K. M., Anderson, D. H., Johnson, L. V. & Hageman, G. S. Age‐related macular degeneration—emerging pathogenetic and therapeutic concepts. Annals of medicine 38, 450–471 (2006).
Ferris, F. L. et al. Clinical classification of age-related macular degeneration. Ophthalmology 120, 844–851 (2013).
Ferrara, N. Vascular endothelial growth factor: molecular and biological aspects. Current topics in microbiology and immunology 237, 1–30 (1999).
Avery, R. L. et al. Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology 113(363–372), e365, doi: 10.1016/j.ophtha.2005.11.019 (2006).
Heier, J. S. et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119, 2537–2548, doi: 10.1016/j.ophtha.2012.09.006 (2012).
Rosenfeld, P. J. et al. Ranibizumab for neovascular age-related macular degeneration. The New England journal of medicine 355, 1419–1431, doi: 10.1056/NEJMoa054481 (2006).
Schmidt-Erfurth, U. et al. Efficacy and safety of monthly versus quarterly ranibizumab treatment in neovascular age-related macular degeneration: the EXCITE study. Ophthalmology 118, 831–839, doi: 10.1016/j.ophtha.2010.09.004 (2011).
Finger, R. P., Wickremasinghe, S. S., Baird, P. N. & Guymer, R. H. Predictors of anti-VEGF treatment response in neovascular age-related macular degeneration. Survey of ophthalmology 59, 1–18, doi: 10.1016/j.survophthal.2013.03.009 (2014).
Fritsche, L. G. et al. A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nature genetics 48, 134–143, doi: 10.1038/ng.3448 (2016).
Lorés-Motta, L. et al. A genetic variant in NRP1 is associated with worse response to ranibizumab treatment in neovascular age-related macular degeneration. Pharmacogenetics and genomics 26, 20–27 (2016).
Fauser, S. & Lambrou, G. N. Genetic predictive biomarkers of anti-VEGF treatment response in patients with neovascular age-related macular degeneration. Survey of ophthalmology 60, 138–152, doi: 10.1016/j.survophthal.2014.11.002 (2015).
Hagstrom, S. A. et al. VEGFR2 Gene Polymorphisms and Response to Anti-Vascular Endothelial Growth Factor Therapy in Age-Related Macular Degeneration. Ophthalmology 122, 1563–1568, doi: 10.1016/j.ophtha.2015.04.024 (2015).
Tsilimbaris, M. K., López-Gálvez, M. I., Gallego-Pinazo, R., Margaron, P. & Lambrou, G. N. Epidemiological and Clinical Baseline Characteristics as Predictive Biomarkers of Response to Anti-VEGF Treatment in Patients with Neovascular AMD. Journal of ophthalmology 2016 (2016).
Tufail, A. et al. The Neovascular Age-Related Macular Degeneration Database: Multicenter Study of 92 976 Ranibizumab Injections. Ophthalmology 121, 1092–1101 (2014).
Kaiser, P. K. et al. Ranibizumab for predominantly classic neovascular age-related macular degeneration: subgroup analysis of first-year ANCHOR results. American journal of ophthalmology 144(850–857), e854 (2007).
Ward, L. D. & Kellis, M. HaploReg: a resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic acids research 40, D930–D934 (2012).
Marchler-Bauer, A. et al. CDD: NCBI’s conserved domain database. Nucleic acids research, gku1221 (2014).
Baylor, D. A. & Burns, M. E. Control of rhodopsin activity in vision. Eye 12(Pt 3b), 521–525, doi: 10.1038/eye.1998.140 (1998).
Flower, D. R. Modelling G-protein-coupled receptors for drug design. Biochimica et biophysica acta 1422, 207–234 (1999).
Wagner, A. H. et al. Exon-level expression profiling of ocular tissues. Experimental eye research 111, 105–111, doi: 10.1016/j.exer.2013.03.004 (2013).
Pronin, A. et al. Expression of olfactory signaling genes in the eye. PloS one 9, e96435 (2014).
Pluznick, J. L. et al. Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proceedings of the National Academy of Sciences of the United States of America 110, 4410–4415, doi: 10.1073/pnas.1215927110 (2013).
Sherva, R. et al. Genetic modifiers of Hb E/beta0 thalassemia identified by a two-stage genome-wide association study. BMC medical genetics 11, 51, doi: 10.1186/1471-2350-11-51 (2010).
Solovieff, N. et al. Fetal hemoglobin in sickle cell anemia: genome-wide association studies suggest a regulatory region in the 5′ olfactory receptor gene cluster. Blood 115, 1815–1822 (2010).
Wei, P., Tang, H. & Li, D. Insights into pancreatic cancer etiology from pathway analysis of genome-wide association study data. PloS one 7, e46887 (2012).
Kim, S. H. et al. Expression of human olfactory receptor 10J5 in heart aorta, coronary artery, and endothelial cells and its functional role in angiogenesis. Biochemical and biophysical research communications 460, 404–408, doi: 10.1016/j.bbrc.2015.03.046 (2015).
Leveziel, N. et al. Genotypic influences on severity of exudative age-related macular degeneration. Investigative ophthalmology & visual science 51, 2620–2625 (2010).
Abedi, F. et al. Variants in the VEGFA gene and treatment outcome after anti-VEGF treatment for neovascular age-related macular degeneration. Ophthalmology 120, 115–121 (2013).
Francis, P. J. The influence of genetics on response to treatment with ranibizumab (Lucentis) for age-related macular degeneration: the Lucentis Genotype Study (an American Ophthalmological Society thesis). Transactions of the American Ophthalmological Society 109, 115 (2011).
Pearson, J. V. et al. Identification of the genetic basis for complex disorders by use of pooling-based genomewide single-nucleotide–polymorphism association studies. The American Journal of Human Genetics 80, 126–139 (2007).
Schrauwen, I. et al. A genome-wide analysis identifies genetic variants in the RELN gene associated with otosclerosis. American journal of human genetics 84, 328–338, doi: 10.1016/j.ajhg.2009.01.023 (2009).
Skibola, C. F. et al. Genetic variants at 6p21. 33 are associated with susceptibility to follicular lymphoma. Nature genetics 41, 873–875 (2009).
Earp, M. A. et al. Genome-wide association study of subtype-specific epithelial ovarian cancer risk alleles using pooled DNA. Human genetics 133, 481–497 (2014).
Macgregor, S., Visscher, P. M. & Montgomery, G. Analysis of pooled DNA samples on high density arrays without prior knowledge of differential hybridization rates. Nucleic Acids Research 34, e55–e55 (2006).
Macgregor, S. Most pooling variation in array-based DNA pooling is attributable to array error rather than pool construction error. European Journal of Human Genetics 15, 501–504 (2007).
Abedi, F. et al. Genetic influences on the outcome of anti-vascular endothelial growth factor treatment in neovascular age-related macular degeneration. Ophthalmology 120, 1641–1648, doi: 10.1016/j.ophtha.2013.01.014 (2013).
Macgregor, S. et al. Highly cost-efficient genome-wide association studies using DNA pools and dense SNP arrays. Nucleic acids research 36, e35 (2008).
Lu, Y. et al. Genome-wide association study for ovarian cancer susceptibility using pooled DNA. Twin Research and Human Genetics 15, 615–623 (2012).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191, doi: 10.1093/bioinformatics/btq340 (2010).
Gregori, N. Z., Feuer, W. & Rosenfeld, P. J. Novel method for analyzing snellen visual acuity measurements. Retina 30, 1046–1050 (2010).
Acknowledgements
We thank all the clinicians, nurses and research staff who participated in patient recruitment. We acknowledge funding from the National Health and Medical Research Council of Australia (NHMRC) project grant1008979 and Senior Research Fellowship 1028444 (PNB). Melbourne International research Scholarship and Melbourne International fee remission scholarship from the University of Melbourne Australia (MR). The Centre for Eye Research Australia (CERA) receives Operational Infrastructure Support from the Victorian Government. This project has received funding from the European Union’s Seventh Framework Programme for research, technological development and demonstration under grant agreement no. 317472 (EyeTN). R.K. Koenekoop is supported by the Foundation Fighting Blindness Canada and the Canadian Institutes for Health Research.
Author information
Authors and Affiliations
Contributions
P.N.B., R.H.G.: designed the study, M.R., L.L.M., A.J.R., S.M., L.Y., G.M., and K.B. performed experiments and data analysis, R.H.G., A.O., R.K.K., J.C., P.M., L.A., T.S., S.F., D.S., F.V.A., E.K.d.J., C.B.H. Sample collection, M.R., L.L.M., A.J.R., E.K.d.J., S.M., L.Y., R.H.G., A.I.d.H., P.N.B. manuscript preparation and proof reading.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Riaz, M., Lorés-Motta, L., Richardson, A. et al. GWAS study using DNA pooling strategy identifies association of variant rs4910623 in OR52B4 gene with anti-VEGF treatment response in age-related macular degeneration. Sci Rep 6, 37924 (2016). https://doi.org/10.1038/srep37924
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37924
This article is cited by
-
Phenotypic variation and quantitative trait loci for resistance to southern anthracnose and clover rot in red clover
Theoretical and Applied Genetics (2022)
-
Genetics and genetic testing for age-related macular degeneration
Eye (2018)
-
Identification of novel alleles associated with insulin resistance in childhood obesity using pooled-DNA genome-wide association study approach
International Journal of Obesity (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.