Abstract
Conspecific adults have strong negative effect on the survival of nearby early-stage seedlings and thus can promote species coexistence by providing space for the regeneration of heterospecifics. The leaf litter fall from the conspecific adults, and it could mediate this conspecific negative adult effect. However, field evidence for such effect of conspecific leaf litter remains absent. In this study, we used generalized linear mixed models to assess the effects of conspecific leaf litter on the early-stage seedling survival of four dominant species (Machilus leptophylla, Litsea elongate, Acer pubinerve and Distylium myricoides) in early-stage seedlings in a subtropical evergreen broad-leaved forest in eastern China. Our results consistently showed that the conspecific leaf litter of three species negatively affected the seedling survival. Meanwhile, the traditional conspecific adult neighborhood indices failed to detect this negative conspecific adult effect. Our study revealed that the accumulation of conspecific leaf litter around adults can largely reduce the survival rate of nearby seedlings. Ignoring it could result in underestimation of the importance of negative density dependence and negative species interactions in the natural forest communities.
Similar content being viewed by others
Introduction
Negative conspecific interaction is of important significance for understanding coexistence of species in many communities. It can promote stable coexistence of species when its strength is stronger than the negative interspecific interactions (e.g. interspecific competition)1,2. This interaction becomes more intense among individuals with high density (e.g. negative density dependent effect)3, thus it is commonly expected that conspecific adult trees have widespread and strong negative effect on seedlings surrounding them. This expectation has been confirmed by many common garden experiments using various populations4,5,6,7, but is not widely supported in natural forest communities8,9. In general, the percentage of plant species with significant effect of conspecific adult neighbors on seedlings is only around 36%, even if the effect of other confounding processes like environmental heterogeneity has been rigorously controlled8. Thus, there is a high chance that the effect of conspecific adults on seedlings has been underestimated.
The possible underestimation might rise from our limited knowledge about how adults negatively affect their conspecific seedlings in natural forest communities. Although it is commonly accepted that host-specific natural enemies, such as fungal pathogens and herbivores, are the main cause of massive seedling mortality around conspecific adults10,11, we know few about how these natural enemies accumulate around the adults. For example, where does the increasing host-specific soil biota near the parent trees come from? Is it simply mediated and transported by the roots of adults in the soil? Are there any other important ways to carry host-specific enemies from adults to the nearby seedlings? These uncertainties in turn limit our ability to accurately quantify and estimate the effect of adults on their conspecific seedlings in natural communities.
Here, we hypothesized that conspecific leaf litter could be another important alternative media for the negative effect of conspecific adult neighborhood on seedling survival. Leaves, as the main part of above-ground adult trees, are often attacked by foliar fungal pathogens (intercept most of rain or wind-borne spores)12,13,14. They will finally fall to the nearby ground where most conspecific seedlings distributed. Therefore, the leaf litter may transfer fungal pathogens on adults to seedlings15. García-Guzmán and Benítez-Malvido (2003) have found the seedlings survival rate of Nectandra ambigens was reduced by the leaf-fungal pathogen when they added the leaf litter most from the parent tree. Thus they inferred that leaf-fungal pathogen brought by conspecific leaf litter is the most important reason of the reduced performance of their seedlings. Up to now, there were few studies examined the potential effect of leaf litter-mediated fungal pathogens on seedling establishment in natural forest communities16.
Most conspecific leaf litter covering the seedlings comes from the adult neighbors, so it is commonly assumed that the patterns of conspecific leaf litter and conspecific adult neighborhood are highly correlated. Then commonly applied methods (e.g. neighborhood indices), which simply assume that the negative effect of adult decreases with the distance from adult to the seedlings and is isotropic in each direction17,18,19,20,21,22, would pick up the effect of conspecific leaf litter on the seedling survival. However, when the correlation is weak, the negative effect of conspecific leaf litter on seedlings will not be as simple as described in commonly applied methods. In the natural communities, the distribution of leaf litter is generally decreased with the distance from the parent trees, but is largely influenced by wind speed, direction and forest vertical structure23. These influences will be much stronger for species with light and large leaves24, and will be different in strength on different topological ground. Because the distribution of leaf litter probably largely biased from isotropic shape, the negative effect of adult on seedlings mediated by leaf litter might be anisotropic from the parent tree25,26,27. This ignored fact of the complex spatial intensity of adult effect on conspecific seedlings proposed a unique challenge to robustly evaluate the importance of conspecific adult effect on seedlings by conventional neighborhood indices in natural forest communities.
To overcome this challenge and present the first field examination of whether conspecific leaf litter is an important way to mediate the negative adult effect on conspecific seedlings, observed leaf litter on 561 seedling plots was used to explain the early-stage survival of the seedlings in a subtropical forest. Specifically, we built two types of generalized linear mixed models to address the following questions: (1) Do the amount of conspecific leaf litter significantly affect the early-stage seedling survival of the chosen tree species? (2) If so, can we found this indirect effect by the conventional conspecific adult neighborhood indices? Comparing results of above two analyses finally could reveal whether the negative effect of conspecific adults on seedlings is indeed underestimated in our natural forest community.
Results
The effect of conspecific adult neighbors through leaf litter
The leaf litter from adult neighbors generally had a strong negative effect on the survival of conspecific seedlings (Fig. 1). Except for the Distylium myricoides with the least abundance of newly germinated seedlings among the four examined species, the leaf litter of other three species can significantly reduce the survival rate of their conspecific seedlings. Moreover, the strength of this inhibition from conspecific leaf litter was stronger than the effect from the conspecific seedling neighbors, and became the second strongest factor among seven potential variables in three of four examined species (ranks in Fig. 1). In addition, the negative effect of leaf litter on seedling survival seems to have a moderate species specific trend, because the effect of conspecific leaf litter on seedling survival was more important than heterospecific leaf litter in all examined species.
The relative importance of potential factors (including conspecific and heterospecific leaf litter) influencing on early-stage seedlings survival of four species.
The potential factors were the log-transformed initial height (HT) of seedlings, the amount of conspecific (Lcon) and heterospecific (Lhet) leaf litter, conspecific adult neighborhood indices (Acon) and heterspecifics (Ahet), the density of conspecific (Scon), and heterospecific (Shet) seedling neighbors. Blue bins indicate significant, positive effects of the variables on seedling survival (The model-average estimator  ); while yellow bins indicate significant, negative effects (
); while yellow bins indicate significant, negative effects ( ); gray bars indicate non-significant effects (
); gray bars indicate non-significant effects ( ). The red and brown arrows highlight the variable of conspecific leaf litter and conspecific adult neighborhood indices respectively.
). The red and brown arrows highlight the variable of conspecific leaf litter and conspecific adult neighborhood indices respectively.
The effect of conspecific adult neighbors through neighborhood indices
The conspecific adult neighborhood indices, however, did not have significant, negative effect on seedling survival of the whole chosen species. This result is robust with respect to either exclude or include the variable of conspecific leaf litter (Figs 1 and 2). Meanwhile, the relative importance of conspecific adult neighborhood indices were the lowest among four potential variables of biotic neighbors in three of four examined species (Fig. 1a,b,c), even though the leaf litter variable was removed in the analyses (Fig. 2a,b,c). Therefore, the effect of conspecific leaf litter on seedling survival did not largely included in the effect of conspecific adult neighborhood indices. On the other hand, the conspecific and heterospecific seedling neighborhood indices (density of conspecific and heterospecific seedlings in the same seedling plot as the focal seedling) had significant, negative effects on seedling survival of Machilus leptophylla (Figs 1a and 2a) and Acer pubinerv (Figs 1c and 2c), respectively.
The relative importance of potential factors (excluding conspecific and heterospecific leaf litter) influencing on early-stage seedlings survival of four species.
The potential factors were the log-transformed initial height (HT) of seedlings, conspecific adult neighborhood indices (Acon) and heterospecific (Ahet), the density of conspecific (Scon) and heterospecific (Shet) seedling neighbors. Blue bins indicate significant, positive effects of the variables on seedling survival (The model-average estimator  ); While yellow bins indicate significant, negative effects (
); While yellow bins indicate significant, negative effects ( ); Gray bins indicate non-significant effects (
); Gray bins indicate non-significant effects ( ). The brown arrows highlight the variable of conspecific adult neighborhood indices.
). The brown arrows highlight the variable of conspecific adult neighborhood indices.
Correlation between conspecific leaf litter and adult neighborhood indices
There was substantial difference between the spatial variation of the conspecific leaf litter and the conspecific adult neighborhood indices (Fig. 3). The difference was mostly significant for the Acer pubinerv (Fig. 3c). In addition, the potential explanatory variables of conspecific leaf litter and conspecific adult neighborhood indices did not cooccur in the optimal model groups of Machilus leptophylla, Litsea elongata and Acer pubinerv as the independent variables (Table S1 in appendix). Therefore, the multiple-collinearity of these two potential explanatory variables did not influence the effect of conspecific leaf litter on seedling survival of these three species.
Discussion
Conspecific leaf litter was almost completely ignored in most of previous field studies testing the negative conspecific density dependent effect, thus its potential importance in forest community was still largely unexplored. Our study filled this gap and provided the first empirical support on the contribution of conspecific leaf litter to tree seedling survival. Several of our results consistently indicated that the amount of conspecific leaf litter had strong negative effect on early-stage seedling survival of three species (Machilus leptophylla, Litsea elongata and Acer pubinerve). Because 91.5% of 2458 newly germinated seedlings during the study period belonged to these three species, it is highly probable that the conspecific leaf litter widely mediate the effect of conspecific adult neighborhood on the early-stage seedling survival in our Tiantong plot. The conspecific leaf litter could be an another important media for the negative conspecific density dependence.
Indirect evidences from other studies suggest that the conspecific leaf litter can inhibit the conspecific seedling survival through different ways in multiple species. The diseases of tree species would be caused by species- or genus-specific foliar pathogens (Machilus leptophylla - Pestalotia neglecta, Litsea - Uncinula SP, Acer - Phyllosticta aceris & Uncinula nankinsensis)28,29,30,31. The conspecific leaf litter may play as the carrier of leaf-fungal pathogens which can inhibit the seedling survival of the three tested species. In addition, the leaf of Acer pubinerve (Aceraceae) contains much cis-3-hexenyl acetat32 which are herbivore-induced plant volatiles33. These volatiles would attract natural enemies of the herbivores during the leaf litter decomposition34. Therefore, the seedling of Acer pubinerve would be increasingly fed by natural enemies of the herbivores in the accumulation of conspecific leaf litter.
Except for the biological attraction effect of pathogens and herbivores, conspecific leaf litter can also increase the conspecific seedling mortality through chemical effect35. The leaves of Machilus and Litsea contain much alkaloid36,37 which is an important allelochemical38. The leaf litter of Machilus leptophylla (Lauraceae) and Litsea elongata (Lauraceae) may inhibit the conspecific seedling survival through the autotoxicity. However, it is hard to answer in current study that whether conspecific leaf litter inhibits the seedling survival through the biological process in bitrophic level or the chemical process in unitrophic level. Manipulation experiments are needed to fully understand how conspecific leaf litter affects seedling performance.
The conspecific leaf litter-mediated effect of conspecific adult neighbors on early-stage seedling survival should be detected by the conspecific adult neighborhood indices directly. However, the commonly used conspecific adult neighborhood indices did not have significant effect on seedling survival in our studies (Fig. 2). One possible reason is that this commonly used conspecific adult neighborhood indices fail to capture this indirect negative conspecific adult effect on seedlings, particular in an environment with rough topography and strong wind disturbance. The weak relationship in our study between these conspecific adult neighborhood indices and the amount of conspecific leaf litter partly supports our speculation.
This conspecific adult neighborhood indices was designed to evaluate conspecific neighborhood effect that is homogeneous from the conspecific adult neighbor circumference18,39. However, the leaf dispersal would be inhomogeneous in the circumference of these species in the Tiantong plot. This inhomogeneous pattern of leaf dispersal could be caused by many reasons. The major reason would be the leaf shapes of the chosen species were broad leaf and our study site was influenced by subtropical monsoon40,41. The wind directions would affect broad leaf dispersal23,27. Especially, the most wind-influenced maple leaves of Acer pubinerv (others have leathery leaves)42,43,44 show the largest difference between the distribution of conspecific leaf litter and conspecific adult neighborhood indices (Fig. 3). Meanwhile, the topography of our plot was rough41. The amount of leaf litter would vary in the different microtopography. Because of these difference between the conspecific adult neighborhood indices and distribution of conspecific leaf litter, it is not supprise to find that the conspecific adult neighborhood indices can not detected this conspecific leaf litter- mediated effect of conspecific adult neighbors on seedling survival in the Tiantong plot.
As a consequence, the above weakness of conspecific adult neighborhood indices could result in large underestimation of the importance of negative density dependence in natural forest communities. Considering the importance of conspecific leaf litter on seedling survival offers one possible solution. Majority of previous experiments only tested the soil-mediated effect of adult neighbors on seedling survival6,7,45,46,47, and rarely considered the potential impact of conspecific leaves on seedling demographic rates. In the future, we should design a manipulation experiment to make up this gap. In addition, the habitat preference could be an important mechanism for influencing the early-stage seedling survival48,49,50. In two of four species, the environmental effects explained total variance of seedling survival more than these biotic factors (Table S3 in appendix). This phenomenon indicated that the effect of conspecific leaf litter on seedling survival would be overlaid with the environmental effects in some species (Figure S1 in appendix).
In summary, our study revealed that the accumulation of conspecific leaf litter around adults can reduce the survival rate of nearby seedlings, and provide a certain advantage around adults for the generation of other species. The conspecific leaf litter might be an important media for the negative effect of conspecific adult neighborhood on early-stage seedling survival in Tiantong plot. Ignoring this negative effect of conspecific leaf litter could result in large underestimation of the importance of negative density dependence and negative species interactions in the natural forest communities.
Methods
Data collection
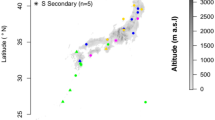
We conducted the study in a 20-ha (500 × 400 m) forest dynamics plot in a subtropical evergreen broad-leaved forest in the Tiantong Forest Park (29°48′N, 121°47′E) (hereafter called Tiantong plot), eastern China41. This region has a subtropical monsoon climate and receives 1374.7 mm mean annual rainfall. The annual average temperature is 16.2 °C. The minimum and maximum monthly mean temperatures are 4.2 °C in January and 28.1 °C in July, respectively. The soil texture is mainly red-yellow soil and pH value of the soil is from 4.5 to 5.040. The topography of the Tiantong plot is rough41, with elevation ranges from 304.26 to 602.89 m. There are two large ridges throughout the plot from south to north (Fig. 4). All free standing trees (DBH ≥ 1 cm) in the Tiantong plot have been mapped, identified to species and measured in 2010. The dominant families of trees in the Tiantong plot are Theaceae, Lauraceae and Fagaceae which are widely distributed in the subtropical evergreen broad-leaved forest41.
To monitor seedling and leaf litter dynamics in the Tiantong plot, 187 census stations were established in two opposite corners of each 20 × 20 m plots in the Tiantong plot (Fig. 4). The census stations were at least 40 m far away from the border of the Tiantong plot to avoid edge effects. Each station consisted of a 0.5 m2 litter trap in the center and three 1-m2 seedling plots surrounded the litter trap. The seedling plots were located 2 m away from the litter trap in three random directions (Fig. 4). In each seedling plot, the newly germinated seedlings were tagged, measured and identified to species in October 2011, April 2012 and October 2012. We analyzed the first-year survival of these newly germinated seedlings. The period of seedling survey for these newly germinated seedlings was from October 2011 to October 2012, from April 2012 to April 2013 and from October 2012 to October 2013 respectively. In addition, other woody plants with DBH ≤ 1 cm in the seedling plot have been tagged, measured and identified to species. Leaf litter in each litter trap was collected every half month from October 2011 to October 2013. After the collection, the leaf litter in each litter trap was identified and grouped into different species immediately in the lab, and dried under 75 °C in an electronic oven for 72 hours. Dry weight of leaf litter for each species in each litter trap was recorded.
A total of 2458 newly germinated seedlings were observed in the 187 seedling plots in the three censuses. Most of them (94.39%) belong to Machilus leptophylla (Lauraceae), Litsea elongata (Lauraceae), Acer pubinerve (Aceraceae) and Distylium myricoides (Hamamelidaceae), with 2051, 116, 82, 71 individuals respectively. Other species only contributed around 5.6% of total newly germinated seedlings, thus were not considered in the analyses of this study to avoid problems of small sample size.
Data analyses
Generalized linear mixed-effects models (GLMMs) with binomial errors were used to examine how the potential factors influenced early-stage seedling survival. The model with the random effects can be specified as:


Where Yijk is 1 if newly germinated seedling i in the seedling plot j of station k alive in the first year and 0 otherwise, πijk is the survival probability of the focal seedling; The random part of equation (2) has two levels, first level is seedling plot j belonging census station k (μj|k) and seconds is census station k (μk)51,52. In the fixed part, α and β refer to an intercept and a vector of coefficients of explanatory variables x, respectively.
The potential explanatory variables in the first type of GLMMs were biotic neighbors (see details in the following paragraph), initial height and leaf litter (divided into conspecific and heterospecific groups). Because, the previous studies showed that the biotic neighbors and initial height were the major factors controlling the early-stage seedling survival18,21,22,53. Meanwhile our study is to examine whether the amount of conspecific leaf litter significantly affect the early-stage seedling survival.
The potential explanatory variable of conspecific leaf litter was defined as the conspecific leaf litter in the litter trap within the same census station as the focal seedling, and calculated by the total weight of the first year from the focal seedling germinated. The heterospecific leaf litter is total weight of the other species’ leaf litter. The potential explanatory variables of biotic neighbors were divided into conspecific and heterospecific seedling and adult neighbors. The seedling neighbors were measured by the density of conspecific or heterospecifc seedlings (DBH ≤ 1 cm) in the same seedling plot as the focal seedling18,20,22,54. The adult neighbors were measured by the adult neighborhood indices. The effects of adult neighbors tend to weaker with the distance from the adults to focal seedlings, but that effects increased with total basal area of these adults18. Therefore adult neighborhood indices were defined as the total of the basal area of conspecific or heterospecific adults (DBH ≥ 1 cm) within 10 m18,22,39 divided by the distance that tree from the center of the focal seedling plot. If the conspecific or heterospecific adults are in the focal seedling plot, the value of distance will be short abnormally. This extremely short distance will cause the adult neighborhood indices large abnormally. Therefore we assumed that trees (in the focal seedling plot) in the edge of the focal seedling plot (1 m × 1 m) and adjusted their distances to the focal seedling plot into 0.5 m.
If the conspecific leaf litter affects early-stage seedling survival negatively, there will be a negative conspecific interaction between the adults and seedlings. To detect this negative conspecific interaction through the common methods, we built the second type of GLMMs by removing potential explanatory variables of leaf litter from the first type of GLMMs. The reason was the effect of leaf litter would disturb the effect of biotic neighbors on seedling survival and almost never be seen in common methods.
We examined the effect and relative importance of potential explanatory variables by model-average estimator and the sum of the weights for each potential explanatory variable respectively55. Firstly, we selected the m (from 0 to n) explanatory variables from the n potential explanatory variables without repeated sampling and combined them as the fixed part of a GLMM, resulting 2n ( ) different GLMMs. Secondly, we calculated the weight of each GLMM (equations S1 and S2) and estimated the relative importance of each potential explanatory variable (equations S3 and S4). Finally, we selected the models whose ΔAICc ≤ 2 as the optimal model groups, and calculated model-average estimator (equations S5, S6, S7 and S8) of each potential explanatory variable55. This process for selecting the models could better reduce the influence of multiple-collinearity among potential explanatory variables in regression models and take into account of all influencing factors comprehensively.
) different GLMMs. Secondly, we calculated the weight of each GLMM (equations S1 and S2) and estimated the relative importance of each potential explanatory variable (equations S3 and S4). Finally, we selected the models whose ΔAICc ≤ 2 as the optimal model groups, and calculated model-average estimator (equations S5, S6, S7 and S8) of each potential explanatory variable55. This process for selecting the models could better reduce the influence of multiple-collinearity among potential explanatory variables in regression models and take into account of all influencing factors comprehensively.
To understand the results of two types of GLMMs, we estimated the adjusted R-squared to evaluate how much the conspecific adult neighborhood indices could explain the distribution of conspecific leaf litter. The response and explanatory variables were sum weight of conspecific leaf litter from October 2011 to October 2013 in each litter trap and the conspecific adult neighborhood indices within 10 m around the focal litter trap respectively.
All of the explanatory variables were subtracted mean of the variable and divided by their own standard deviation (zero-mean normalization). The mean, median and range of the whole potential explanatory variables showed in the Table S4 in appendix. All analyses were conducted in R 3.2.1 (R Development Core Team 2015). The GLMMs were fitted by the “glmer” function of ‘lme4 1.1-9′ package56.
Additional Information
How to cite this article: Liu, H. et al. Conspecific Leaf Litter-Mediated Effect of Conspecific Adult Neighborhood on Early-Stage Seedling Survival in A Subtropical Forest. Sci. Rep. 6, 37830; doi: 10.1038/srep37830 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Tilman, D. Resource Competition and Community Structure. (Princeton, NJ: Princeton University Press 1982).
Chesson, P. Mechanisms of maintenance of species diversity. Annual review of Ecology and Systematics 31, 343–366 (2000).
Terborgh, J. Enemies maintain hyperdiverse tropical forests. The American Naturalist 179, 303–314 (2012).
Bell, T., Freckleton, R. P. & Lewis, O. T. Plant pathogens drive density-dependent seedling mortality in a tropical tree. Ecology Letters 9, 569–574 (2006).
Bagchi, R. et al. Testing the Janzen‐Connell mechanism: pathogens cause overcompensating density dependence in a tropical tree. Ecology letters 13, 1262–1269 (2010).
Mangan, S. A. et al. Negative plant-soil feedback predicts tree-species relative abundance in a tropical forest. Nature 466, 752–755 (2010).
Bagchi, R. et al. Pathogens and insect herbivores drive rainforest plant diversity and composition. Nature 506, 85–88 (2014).
Carson, W. P., Anderson, J. T., Leigh, E. G. & Schnitzer, S. A. Challenges associated with testing and falsifying the Janzen-Connell hypothesis: a review and critique. (Wiley-Blackwell, 2008).
Comita, L. S. et al. Testing Predictions Of The Janzen-Connell Hypothesis: A Meta-Analysis Of Experimental Evidence For Distance- And Density-Dependent Seed And Seedling Survival. Journal of Ecology 102, 845–856 (2014).
Janzen, D. H. Herbivores and the number of tree species in tropical forests. American naturalist 104, 501–528 (1970).
Connell, J. H. On the role of natural enemies in preventing competitive exclusion in some marine animals and in rain forest trees. Dynamics of populations 298, 312 (1971).
Marquis, R. J., Diniz, I. R. & Morais, H. C. Patterns and correlates of interspecific variation in foliar insect herbivory and pathogen attack in Brazilian cerrado. Journal of Tropical Ecology 17, 127–148 (2001).
Keane, P. How pathogens attack plants. Microbiology Australia 33, 26–28 (2012).
Bayandala, Fukasawa, Y. & Seiwa, K. Roles of pathogens on replacement of tree seedlings in heterogeneous light environments in a temperate forest: a reciprocal seed sowing experiment. Journal of Ecology 104, 765–772 (2016).
Butenschoen, O. & Scheu, S. Climate change triggers effects of fungal pathogens and insect herbivores on litter decomposition. Acta Oecologica 60, 49–56 (2014).
García-Guzmán, G. & Benítez-Malvido, J. Effect of litter on the incidence of leaf-fungal pathogens and herbivory in seedlings of the tropical tree Nectandra ambigens. Journal of Tropical Ecology 19, 171–177 (2003).
Condit, R., Hubbell, S. P. & Foster, R. B. Recruitment near conspecific adults and the maintenance of tree and shrub diversity in a neotropical forest. American Naturalist 140, 261–286 (1992).
Comita, L. S. & Hubbell, S. P. Local neighborhood and species’ shade tolerance influence survival in a diverse seedling bank. Ecology 90, 328–334 (2009).
Kobe, R. K. & Vriesendorp, C. F. Conspecific density dependence in seedlings varies with species shade tolerance in a wet tropical forest. Ecology letters 14, 503–510 (2011).
Bai, X. et al. Effects of local biotic neighbors and habitat heterogeneity on tree and shrub seedling survival in an old-growth temperate forest. Oecologia 170, 755–765 (2012).
Lin, L., Comita, L. S., Zheng, Z. & Cao, M. Seasonal differentiation in density‐dependent seedling survival in a tropical rain forest. Journal of Ecology 100, 905–914 (2012).
Lin, F. et al. The contribution of understory light availability and biotic neighborhood to seedling survival in secondary versus old-growth temperate forest. Plant Ecology 215, 795–807 (2014).
Jonard, M., Andre, F. & Ponette, Q. Modeling leaf dispersal in mixed hardwood forests using a ballistic approach. Ecology 87, 2306–2318 (2006).
Zhao, Q., Liu, H., Jonard, M., Wang, Z. & Wang, X. Applicability analysis of spatially explicit model of leaf litter in evergreen broad-leaved forests Chinese Journal of Applied Ecology 25, 3117–3124 (2014).
Stone, D. M. Leaf dispersal in a pole-size maple stand. Canadian Journal of Forest Research 7, 189–192 (1977).
Ferguson, D. K. The origin of leaf-assemblages—new light on an old problem. Review of Palaeobotany and Palynology 46, 117–188 (1985).
Staelens, J., Nachtergale, L., Luyssaert, S. & Lust, N. A model of wind-influenced leaf litterfall in a mixed hardwood forest. Canadian Journal of Forest Research 33, 201–209 (2003).
Xu, T. & Ge, Q. Fungous Diseases on Shade Trees in Hangzhou. Journal of Zhejiang Forestry College 2, 45–52 (1985).
Yuan, Z. Q. Fungi and associated tree diseases in Melville Island, Northern territory, Australia. Australian Systematic Botany 9, 337–360 (1996).
Délye, C., Laigret, F. & Corio-Costet, M. F. A mutation in the 14 alpha-demethylase gene of Uncinula necator that correlates with resistance to a sterol biosynthesis inhibitor. Applied & Environmental Microbiology 63, 2966–2970 (1997).
Abras, S. et al. Phytosanitary monitoring of woody species from the banks of watercourses in Wallonia. Phytoparasitica 6, 69–80 (2005).
Wang, Q. Volatile organic compounds analysis of 22 garden plants and analytic hirerarchy process evaluation Master thesis, Zhejiang A&F University (2014).
Frost, C. J. et al. Priming defense genes and metabolites in hybrid poplar by the green leaf volatile cis-3-hexenyl acetate. New Phytologist 180, 722–734 (2008).
De Moraes, C. M., Lewis, W. J., Pare, P. W., Alborn, H. T. & Tumlinson, J. H. Herbivore-infested plants selectively attract parasitoids. Nature 393, 570–573 (1998).
Xiong, S. & Nilsson, C. The effects of plant litter on vegetation: a meta-analysis. Journal of Ecology 87, 984–994 (1999).
Hart, N. et al. Alkaloids of several Litsea species from New Guinea. Australian Journal of Chemistry 22, 2259–2262 (1969).
Cheng, W. et al. Chemical constituents of the bark of Machilus wangchiana and their biological activities. Journal of natural products 72, 2145–2152 (2009).
Putnam, A. R. Allelochemicals from Plants as Herbicides. Weed Technology 2, 510–518 (1988).
Canham, C. D., LePage, P. T. & Coates, K. D. A neighborhood analysis of canopy tree competition: effects of shading versus crowding. Canadian Journal of Forest Research 34, 778–787 (2004).
Song, Y. & Wang, X. Vegetation and flora of Tiantong national forest park, Zhejiang province (Shanghai Scientific Documentary Press, 1995).
Yang, Q. et al. Community structure and species composition of an evergreen broad-leaved forest in Tiantong’s 20 ha dynamic plot, Zhejiang Province, eastern China Biodiversity Science 19, 215–223 (2011).
Xu, T., Chen, Y., de Jong, P. C., Oterdoom, H. J. & Chang, C. Flora of China - Aceraceae. Vol. 11 529 (Science Press, 2008).
Zhang, Z., Zhang, H. & Peter, K. E. Flora of China - Hamamelidaceae. Vol. 9 29 (Science Press, 2003).
Li, X. et al. Flora of China - Lauraceae. Vol. 7 140–141&214 (Science Press, 2008).
Packer, A. & Clay, K. Soil pathogens and spatial patterns of seedling mortality in a temperate tree. Nature 404, 278–281 (2000).
Liu, X. et al. Experimental evidence for a phylogenetic Janzen–Connell effect in a subtropical forest. Ecology letters 15, 111–118 (2012).
McCarthy-Neumann, S. & Ibáñez, I. Plant–soil feedback links negative distance dependence and light gradient partitioning during seedling establishment. Ecology 94, 780–786 (2013).
Gunton, R. M. & Kunin, W. E. Density‐dependence at multiple scales in experimental and natural plant populations. Journal of ecology 97, 567–580 (2009).
Uriarte, M., Bruna, E. M., Rubim, P., Anciães, M. & Jonckheere, I. Effects of forest fragmentation on the seedling recruitment of a tropical herb: assessing seed vs. safe-site limitation. Ecology 91, 1317–1328 (2010).
Chanthorn, W., Caughlin, T., Dechkla, S. & Brockelman, W. Y. The relative importance of fungal infection, conspecific density and environmental heterogeneity for seedling survival in a dominant tropical tree. Biotropica 45, 587–593 (2013).
Zuur, A., Ieno, E. N., Walker, N., Saveliev, A. A. & Smith, G. M. Mixed effects models and extensions in ecology with R. (Springer Science & Business Media, 2009).
Chen, L. et al. Community‐level consequences of density dependence and habitat association in a subtropical broad‐leaved forest. Ecology Letters 13, 695–704 (2010).
Comita, L. S. et al. Abiotic and biotic drivers of seedling survival in a hurricane‐impacted tropical forest. Journal of Ecology 97, 1346–1359 (2009).
Zhu, Y., Comita, L. S., Hubbell, S. P. & Ma, K. Conspecific and phylogenetic density-dependent survival differs across life stages in a tropical forest. Journal of Ecology 103, 957–966 (2015).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-Theoretical Approach 2nd edn. (Springer, 2002).
Bates, D., Maechler, M., Bolker, B. & Walker, S. lme4: Linear mixed-effects models using Eigen and S4. R package version 3.2.1. http://lme4.r-forge.r-project.org/ (2013).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31210103920 & 31470487). We thank Sean Maurice McMahon and Sabrina Ermatina Russo for their great critical comments to improve this manuscript. We are also grateful to Zhanghua Wang, Qingqing Zhao, Xiao Lei, Jiuzhou Xing, Xiyang Fei, Mingjiao Yuan, Shu Dong and Yi Zong for their help in the field survey.
Author information
Authors and Affiliations
Contributions
H.M.L. X.H.W. Z.P.M. and X.F.F. conceived and designed the experiments. H.M.L. X.H.W. Z.P.M. Q.S.Y. and X.F.F. and performed the experiments. H.M.L. G.C.S. and X.H.W. analyzed the data. H.M.L. G.C.S. J.Y.X. and X.F.F. contributed reagents/materials/analysis tools. H.M.L. G.C.S. X.H.W. J.Y.X. wrote the paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, H., Shen, G., Ma, Z. et al. Conspecific Leaf Litter-Mediated Effect of Conspecific Adult Neighborhood on Early-Stage Seedling Survival in A Subtropical Forest. Sci Rep 6, 37830 (2016). https://doi.org/10.1038/srep37830
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37830
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.