Abstract
Raphanobrassica is an allopolyploid species derived from inter-generic hybridization that combines the R genome from R. sativus and the C genome from B. oleracea var. alboglabra. In the present study, we used a high-throughput sequencing method to identify the mRNA and miRNA profiles in Raphanobrassica and its parents. A total of 33,561 mRNAs and 283 miRNAs were detected, 9,209 mRNAs and 134 miRNAs were differentially expressed respectively, 7,633 mRNAs and 39 miRNAs showed ELD expression, 5,219 mRNAs and 57 miRNAs were non-additively expressed in Raphanobrassica. Remarkably, differentially expressed genes (DEGs) were up-regulated and maternal bias was detected in Raphanobrassica. In addition, a miRNA-mRNA interaction network was constructed based on reverse regulated miRNA-mRNAs, which included 75 miRNAs and 178 mRNAs, 31 miRNAs were non-additively expressed target by 13 miRNAs. The related target genes were significantly enriched in the GO term ‘metabolic processes’. Non-additive related target genes regulation is involved in a range of biological pathways, like providing a driving force for variation and adaption in this allopolyploid. The integrative analysis of mRNA and miRNA profiling provides more information to elucidate gene expression mechanism and may supply a comprehensive and corresponding method to study genetic and transcription variation of allopolyploid.
Similar content being viewed by others
Introduction
Polyploidization, an ancient and ongoing evolutionary process, is a pervasive phenomenon in all angiosperm and played an important role in plant evolution through genomic merging and doubling1,2,3,4. Polyploids are typically classified as autopolyploids (duplication of the genome of a single species) and allopolyploids (duplicated combination of divergent genomes through hybridization). In fact, allopolyploid plays an important role in plant diversification and remains a significant speciation process today3,5, and many important agricultural plants; including wheat (Triticum aestivum), cotton (Gossypium hirsutum) and rapeseed (Brassica napus), have shown their hallmarks of allopolyploidy. Allopolyploid plants often show enhanced adaptability and arresting traits compared with their progenitors, which may contribute to crop domestication and natural selection6. Studies had reported a common occurrence in newly formed allopolyploids, which might cause genetic, epigenetic and transcriptional changes3,5, such as gene conversion events7,8 and deletions4,9,10, epigenetic changes, including DNA methylation11,12, and small RNA regulation13,14. Altered expression patterns14,15 and ELD (expression level dominate)4 have been reported as prominent phenomena in allopolyploids. Furthermore, recent studies on allopolyploidy have focused on ‘transcriptomic shock’: a rapid change in gene expression, reflecting non-additive gene expression from the predicted null hypothesis16, which has become a widespread phenomenon in newly formed allopolyploids. The ‘transcriptomic shock’ resulting from non-additive expression has recently been recognized as a crucial immediate outcome of allopolyploidy17,18,19,20,21,22.
Allopolyploidization is intriguing because it involves two processes: hybridization and whole genome doubling (WGD). Previous studies on allopolyploids have revealed that hybridization alters ancestral expression patterns immediately, massively, and irregulary23. Notably, hybridization between species is the driving force of genome evolution and new species formation. In fact, hybridization and WGD are often jointly involved in natural settings, resulting in growth vigour and phenotypic variation24. To study the effects of these two indispensable processes in allopolyploids, various results have been obtained from different plant taxa. Hybridization markedly affects Arabidopisis, but few variations have been associated with WGD25. Some reports have demonstrated that transcriptomic shock generates a modified pattern of WGD in Senecio; however, this effect cannot be explained by transcriptional changes26. Moreover, a semblable effect of both hybridization and WGD was observed in cotton27, while an oppositional effect of hybridization and WGD also has been reported in Tragopogon18. In addition, Xu showed that WGD has a distinct effect on gene expression and DNA methylation changes in synthesized B. napus28. The forgoing studies involved in hybridization and WGD coaction in interspecies hybrid allopolyploids have supplied valuable information. However, contrasting models have explained the respective effect of hybridization and WGD upon genomic and trancriptomic changes in inter-generic hybrid allopolyploids. Further investigations in inter-generic hybrid allopolyploids could increase the current understanding of the transcriptomic alterations that depend on hybridization and WGD.
Small RNAs (sRNAs) are endogenous single-stranded non-coding RNAs of approximately 21–25 nt in length including microRNAs (miRNAs) and small interfering RNAs (siRNAs), which regulate gene expression at the post-transcriptional level through complementary base pairing29,30 and epigenetic modifications31. MiRNAs often negatively regulate gene expression by promoting the degradation or suppressing the translation of target mRNAs. For example, miRNA regulation has been oberved in pathogens32, in response to stress, such as drought33,34 and extreme temperatures35,36. Wang suggested that miRNAs preferentially repress A. thaliana homologous loci when using the former as the maternal parent37. These observations demonstrate that miRNAs may serve as buffers against genomic shock and regulate DNA methylation. Despite these efforts, little attention has been paid to understanding how miRNA and mRNA expression is integrated into a dynamic and complex regulatory network, which might alter gene expression regulation and contribute to the enhancement of adaptation in inter-generic hybrid allopolyploids.
Studies on artificially synthesized polyploids and their progenitors are needed to understand the effects of polyploidization. The genus Brassica is a useful and importent model for agriculture and human nutrition38. Raphanus sativus (2n = 18, RR) is a valuable vegetable species that has contributed to human health and nutrition for hundreds of years and comprises multiple distinct cultivar terms with diverse morphological and phytochemical attributes. Brassica oleracea var. alboglabra (2n = 18, CC) is native to southern China, as a nutritious vegetable in Brassica plants. The name of this amphidiploid, Raphanobrassica (2n = 36, RRCC), reflects a combination of Raphanus and Brassica39. The tetraploid Raphanobrassica plant might be an ideal model to study gene expression dynamics and the corresponding genetic and epigenetic regulation in allopolyploids of inter-generic origins.
In this context, it is important to examine the regulation of gene expression in polyploids. Insights into miRNA-mRNA networks are necessary to understand the modulation of gene expression at the post-transcriptional level. However, the modulations that occur in the artificially synthesized tetraploid Raphanobrassica at the gene expression level and the interactions between miRNAs and mRNAs for the regulation of gene expression remain unknown. High-throughput sequencing technology offers unparalleled opportunities to address such issues and contributes to the current understanding of the molecular mechanisms that may contribute to adaption in the tetraploid Raphanobrassica. Thus, we executed mRNA and miRNA analyses of the leaves of R. sativus, B. oleracea var. olablagra and Raphanobrassica using RNA-Seq. The abundance data generated from gene expression and miRNAs analysis might suggest a RNA-mediated regulatory mechanism for homologous expression that may be essential for the tetraploid Raphanobrassica and its parents.
Results
Analysis of mRNA sequencing data
After employing the RNA-seq (modified) method, an average of 20 million reads were detected in each library, and 84.03%, 86.85%, and 86.22% clean reads could be individually mapped to reference genomes in the R. sativus, B. oleracea var. alboglabra, and Raphanobrassica libraries, respectively (Table 1). In the present study, a total of 33,561 genes were detected in Raphanobrassica and its parents, and analysis of the gene coverage distribution showed that 14,875, 18,454 and 18,628 genes had more than 90% coverage in R. sativus, B. oleracea var. alboglabra and Raphanobrassica, respectively (Fig. S1), indicating that a significant number of genes are satisfactorily matched, and the recent Abecedarian data are usable for further studies. Venn diagram shows visible gene expression in R. sativus, B. oleracea var. alboglabra, and the tetraploid Raphanobrassica (Fig. 1). A total of 26,531 genes were co-expressed in the tetraploid Raphanobrassica and its parents. Furthermore, 929 genes (R. sativus), 1,206 genes (B. oleracea var. alboglabra), and 1,240 genes (tetraploid Raphanobrassica) genes were specifically expressed.
Gene ontology (GO) enrichment annotation analysis of all detected genes
In the present study, 28,314 genes (84.36%) among the 33,560 detected genes had at least one GO annotation. All matched relevant genes were classified into 3 functional categories: molecular functions, biological processes and cellular components, including 44 terms (Fig. S2). These genes were classified into 23 terms in the biological process category, while 12 terms were matched in the molecular function category, and 9 terms were matched in cellular component category. The terms ‘cell’ and ‘organelle’, which included 19,096, and 12,526 genes, were dominant in cellular component category. In the molecular function category, the terms ‘binding’ and ‘catalytic activities’ were most significantly enriched, with 13,249 and 11,547 genes, respectively. There was also enrichment for the terms in response to ‘biological process’, ‘categories’, ‘cellular process’ and ‘metabolic process’, indicating that Raphanobrassica and its parents underwent exquisite metabolic activities, suggesting further exploration of additional mechanisms that relevant to metabolic activities in polyploids.
Differentially expressed genes (DEGs) between Raphanobrassica and its parents with GO functional categories analysis
A total of 9,209 genes were differentially expressed (log2Ratio ≥ 1, p ≤ 0.05) between tetraploid Raphanobrassica and its parents (Fig. 2), and 6,042 genes were significantly differentially expressed between tetraploid Raphanobrassica and R. sativus, among these, 4,679 genes were up-regulated and 1,363 genes were down-regulated in the tetraploid Raphanobrassica. Moreover, 4,741 genes were differentially expressed between Raphanobrassica and B. oleracea var. alboglabra, with 2,754 up-regulated and 1,987 down-regulated genes in Raphanobrassica. These data suggested that differentially expressed genes are up-regulated in Raphanobrassica compared with its parents and there were more differentially expressed genes between Raphanobrassica and its maternal parent R. sativus.
To examine the gene expression differences between Raphanobrassica and its parents, we performed a GO enrichment analysis and categorized the DEGs to the secondary GO terms classification. A total of 9 terms belonged to the cellular component category, 12 functional terms belonged to the molecular function category and 22 functional terms belonged to the biological process category (Fig. 3). In the cellular component category, the terms ‘cell’, ‘cell part’, ‘organelle’ ‘organelle part’ and ‘macromolecular complex’ were significantly enriched, and the term ‘organelle part’ was more significantly expressed in contrast with the other three terms in the Raphanobrassica/B. oleracea var. alboglabra comparison for the cellular component category. The terms ‘binding’, ‘catalytic’ and ‘transporter’ were dominant in the molecular function category, with ‘binding’ biased to the comparison Raphanobrassica/R. sativus; however, the terms ‘catalytic’ and ‘transporter’ were biased to paternal parent B. oleracea var. alboglabra. ‘Cellular process’, ‘metabolic process’ and ‘response to stimulus’ were the highest three GO terms significantly enriched in the biological process category. Interestingly, the terms ‘developmental process’, ‘establishment of localization’, ‘biological regulation’ and ‘localization’ were slightly lower but also significantly enriched in the biological process category. In this category, ‘biological regulation’, ‘cellular process’, ‘developmental process’ and ‘metabolic process’ were biased to the maternal parent R. sativus, and the terms ‘establishment of localization’ and ‘response to stimulus’ were biased to the paternal parent B. oleracea var. alboglabra. These results imply that tetraploid Raphanobrassica was distinct reflecting a complex process of expression.
KEGG analysis of DEGs between Raphanobrassica and its parent
After mapping to the reference canonical pathways, the 9,209 DEGs (Q value ≤ 0.05) detected from Raphanobrassica and its parents were assigned to 127 pathways. In the comparison R. sativus/Raphanobrassica, ‘biosynthesis of secondary metabolites’, ‘metabolic pathway’, ‘plant-pathogen interaction’ and ‘plant hormone signal transduction’ were significantly enriched. In the comparison of B. oleracea var. alboglabra/Raphanobrassica, the genes were significantly enriched in the ‘metabolic’, ‘plant-pathogen interaction’ and ‘plant signal transduction’ pathways. The top 10 (Table S1) pathways showed that the high gene enrichment was sorted in order of gene numbers in Raphanobrassica compared with its parents, with the terms ‘metabolic pathways’, ‘biosynthesis of secondary metabolites’, ‘plant-pathogen interaction’ and ‘plant hormone signal transduction’. The metabolic pathway contained the largest gene numbers (1,716) among all detected pathways, while the pathway ‘ribosome’, containing 180 genes, showed the highest genes expression levels among all detected KEGG pathways. Thus, some remarkable gene expression changes might occur for one or more of these genes.
Analysis of differentially expressed genes among putative transcription factor genes
Using the rape transcription factor database regarding the identified DEGs, 3,387 genes were associated with 63 transcription factor families, and the expressions of the genes in most of these families was down regulated. The highest gene expression family was ‘bHLH’, with an average RPKM value of 1,870 (total amount divided by the number of genes expressed), and the second highest gene expression family was ‘MYB’, with an average expression quantity of 1,705. ‘AP2-EREBP’ and ‘C3H’ were the third and fourth highest gene expression families, possessing 1,525 and 1,225 average expression quantities, respectively. The last gene expression family, ‘bZIP’ had a gene expression quantity of 1,150 (Fig. S3). The highest expressed family in the present study, ‘bHLH’, has a reported function of transcriptional activation of gene expression under drought stress in Arabidopsis, and growth-promoting bHLH degradation by inhibition shade avoidance based on a phytochrome-mediated process. This result suggests that the family ‘bHLH’ might also be a nonnegligible force, played an important role for variation and adaptation in polyploidy.
Identification of non-additively expressed genes in the tetraploid Raphanobrassica relative to its parents
To determine non-additively expressed genes, we compared gene expression levels in Raphanobrassica with the average genes expression values in the parents (MPVs). If the value of a gene in Raphanobrassica showed at least a two-fold change and P < 0.05 relative to the MPV, then the gene was regarded as non-additive expression37. A total of 5,219 genes, accounting for 15.55% of the total detected genes, were non-additively expressed in the tetraploid Raphanobrassica. Among these non-additive genes, 2,280 genes were up-regulated and 2,932 genes were down-regulated in the tetraploid Raphanobrassica compared to its parents. We observed additive gene expression for a majority of the expressed genes in Raphanobrassica, whereas non-additively expressed genes represented only a small portion of the expressed genes. To further explore the difference in the functional category distribution between additively and non-additively expressed genes, we assigned secondary classification GO terms. The genes belonged to 9, 12 and 24 functional terms in cellular component, molecular function and biological process, respectively. In the cellular component category, the number of additive and non-additive gene significantly differed for the functional terms ‘membrane-enclosed lumen’, ‘organelle part’, ‘extracellular region’, ‘macromolecular complex’ and ‘envelope’ and showed significant enrichment in additive genes compared with non-additive genes except ‘extracellular region’. In the molecular function category, the term ‘structural molecule activity’ and ‘translation regulator’ showed significant differences in gene numbers between additive and non-additive gene, and the terms ‘antioxidant’ and ‘transporter’ were significantly enriched with non-additive genes predominant. In the biological process category, four functional terms, ‘death’, ‘immune system process’, ‘multi/-organism processes’ and ‘response to stimulus’ had significantly more non-additive than additive genes. The non-additive genes were belonged to 9, 11 and 21 terms in cellular component, molecular function, and biological process, respectively (Fig. 4). More than 40% of the non-additively expressed genes were enriched in ‘cell’, ‘cell part’ and ‘organelle’ in the cellular component category, while ‘catalytic activity’, ‘binding’ and ‘transporter’ were significantly enriched in the molecular function category, and the terms ‘metabolic process’, ‘response to stimulus’ and ‘cellular process’ showed significant enrichment in the biological process category.
Parental gene expression level dominance (ELD) in allotetraploid Raphanobrassica
To accurately analyse the gene expression patterns between Raphanobrassica and its parents, we further classified all of the detected genes into 12 categories according to the methods of Rapp22 and Grover40. Among these, the differentially expressed genes in only 4 categories were detected between the parental lines and exhibited statistically similar levels of expression in Raphanobrassica to those in the parents, indicating parental expression level dominance (ELD) (Fig. 5). In the 4 categories, we designated genes with expression levels in Raphanobrassica that were statistically similar to those in R. sativus as ELD-a genes and those similar to B. albaglabra as ELD-b genes.
In Raphanobrassica, 7,633 genes showed parental ELD. The down-regulated genes (884) in ELD-a patterns showed relatively lower expression in Raphanobrassica compared with the up-regulated genes (3,583), and the genes in ELD-b did not show significant discrepancy ratios between up-regulated (1,905) and down-regulated (1,261) patterns. Generally, Raphanobrassica had relatively more ELD-a genes than ELD-b genes (4,467/3,166). Thus, the gene expression in Raphanobrassica displayed ELD bias towards the maternal progenitor R. sativus.
We further pursued the potential functions of the genes showing parental ELD in Raphanobrassica. The GO analysis of ELD-a genes (4,467) revealed the enrichment of genes for the biological processes ‘establishment of localization’, ‘metabolic process’, ‘localization’ and ‘cellular process’, and among the ELD-b genes in biological processes, three terms, ‘response to stimulus’, ‘metabolic process’, and ‘cellular process’, were enriched among the ELD-b genes in the biological process category (Fig. S4). In addition, the terms ‘establishment of localization’ and ‘localization’ were specifically enriched in ELD-a genes, and the ELD-b genes were significantly enriched for ‘response to stimulus’ after removing the two coexisting terms. In the molecular function category, the terms ‘catalytic activity’ and ‘binding’ coexisted in both ELD-a and ELD-b genes with the term ‘transporter activity’ and were specifically enriched in ELD-a genes.
Identification of miRNAs and their target genes in Raphanobrassica and its parents
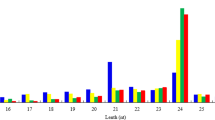
A total of 85,593,286 small RNA sequencing reads were generated from three libraries. After removing adapter and structural RNA sequences, we identified 27,776,413, 27,290,301, and 30,093,748 clean reads within a range of 21–25 nucleotides from R. sativus, B. oleracea var. alboglabra, and Raphanobrassica, respectively. The reads distributed between 21–25 nt were consistent with the common features of sRNAs (Fig. 6), and 24 nt was the most abundantly enriched, accounting for 39.57% of clean reads in R. sativus, 45.07% in B. oleracea var. alboglabra and 52.75% in Raphanobrassica. In addition, the second most abundant read was 21 nt, accounting for 26.60% of clean reads in R. sativus, 19.54% in B. alboglabra, and 24.92% in Raphanobrassica.
Compared with the miRBase database, we generated 281 miRNAs in total, with 277 in R. sativus, 274 in B. oleracea var. alboglabra and 271 in Raphanobrassica (Fig. S5). Furthermore, 267 miRNAs were conserved in all three libraries, accounting for 95.01% of the 281 miRNAs. Subsequently, 3 miRNAs were co-expressed in R. sativus and B. oleracea var. alboglabra, and 7 miRNAs were co-expressed in Raphanobrassica and R. sativus, while 4 miRNAs were co-expressed in Raphanobrassica and B. oleracea var. alboglabra. Because a deep-sequencing approach was used to recover small RNA reads, the relative expression level of a specific miRNA can be measured from the frequency of its read count. Based on the abundance of miRNAs, we observed that the miRNAs expression levels were highly variable, from 1 to 784,100 sequence reads among the three libraries (Table 2). Specifically, 43.53% of miRNAs was observed for expression of less than 100 reads, while 20.64% of miRNA expression levels ranged from 100 to 500 reads, and 16.13% of those were between 1,000 and 5,000 reads. The percentage of miRNAs whose expression levels were more than 10,000 reads was 7.59%. For example, one of the most abundant (>10,000 reads) miRNAs in the three libraries was miR172, with 495,478 reads in R. sativus, 474,293 reads in B. oleracea var. alboglabra, and 632,699 reads in Raphanobrassica. Interestingly, one of the targets of miR172 is AP2 gene, which is essential for regulation of leaf conversion from young to mature41. MiR166, miR167, miR172, and miR158 had more than 10,000 reads in all three libraries, with similar expression profiles. These data suggest that the abundant miRNAs in Raphanobrassica and its parents might involve in a number of key biological processes.
Subsequently, compared with R. sativus, 134 significantly differentially expressed miRNAs (log2Ratio ≥ 1, p ≤ 0.05) were detected, with 64 up-regulated and 70 down-regulated miRNAs in Raphanobrassica (Fig. 7). In 120 differentially expressed miRNAs between Raphanobrassica and B. oleracea var. alboglabra, 50 up-regulated and 70 down-regulated miRNAs were detected in Raphanobrassica.
To identify the potential roles of target genes of differentially expressed miRNAs in Raphanobrassica/R. sativus and Raphanobrassica/B. oleracea var. alboglabra, a total of 182 differentially expressed miRNAs and 3,303 target genes were detected (Table S2), with 182, 179 and 182 miRNAs targeting 1,103, 1,098 and 1,102 genes in R. sativus, B. oleracea var. alboglabra and Raphanobrassica, respectively. In addition, 85 and 74 miRNAs with 394 and 478 target genes existed in Raphanobrassica/R. sativus and Raphanobrassica/B. oleracea var. alboglabra, respectively. Moreover, differentially expressed miRNA targets for Raphanobrassica/R. sativus and Raphanobrassica/B. oleracea var. alboglabra were placed in secondary GO terms classification to compare the difference of Raphanobrassica from its two parents (Fig. S6). In the high frequency of occurrence terms ‘organelle’ and ‘organelle part’, Raphanobrassica showed a significant difference from its paternal parent B. oleracea var. alboglabra than R. sativus, while in the conversed terms ‘cell’, ‘cell part’, ‘catalytic, ‘cellular process’ and ‘localization’, Raphanobrassica showed a consistent difference from its maternal parent R. sativus. The difference in the miRNA target expression between the progeny and its parents might reflect the different relationship types.
ELD-miRNAs between Raphphanobrassica and its parents
We further divided all miRNAs into 12 patterns (Fig. 8) and observed that 39 miRNAs in Raphanobrassica were ELD-expressed compared with the MPVs, 23 miRNAs (pattern II and XI) exhibited ELD-a, and all ELD-a miRNAs were biased to up-regulated (pattern II). Interestingly, differentially expressed miRNAs in ELD-b were average, with 8 ELD-b miRNAs in pattern IV showing biased towards up-regulated patterns, and the other 8 ELD-b miRNAs showing down-regulated pattern IX. Furthermore, 120 differentially expressed miRNAs were transgressively down-regulated, while 114 differentially expressed miRNAs were transgressive up-regulated. Among the transgressive miRNAs, we identified 2 miRNAs with more deflection than the paternal parent among the transgressive down-regulated miRNAs in pattern X, but pattern III (biased to the maternal parent) conceived a comparatively high proportion (31 of 224) of transgressive down-regulated miRNAs. This result may illustrate the existence of maternal-bias miRNAs in Raphanobrassica. Overall, miRNAs displayed preferential pattern III in transgressive down-regulated miRNAs, likely reflecting the regulation of their target gene expression in Raphanobrassica.
Comprehension of non-additive miRNAs and their target genes
Compared with MPV, 57 non-additively expressed miRNAs and 167 target genes were detected. To further explore the functional categories distribution of the non-additively expressed miRNAs target genes in Raphanobrassica, we assigned these genes to secondary classification GO terms (Fig. S7). In the cellular component category, cell and organelle were significantly enriched. The terms that include ‘binding’, ‘catalytic’, and ‘transporter’ were significantly enriched in molecular function category. ‘Biological regulation’, ‘cellular process’, ‘developmental process’, ‘localization’, ‘metabolic process’, ‘multicellular organismal process’, ‘pigmentation’ and ‘response to stimulus’ were significantly enriched in the coherent target genes of non-additive miRNAs.
Correlation analysis of related target genes based on the mRNA-miRNA interaction network
We defined a predicted miRNA target gene when its expression pattern was in contrast with that of the miRNA, reflecting the fact that mRNA expression was negatively correlated with miRNA expression. Based on this rule, we identified a total of 178 related target genes in the two comparisons between the polyploidy and the two parents (Fig. 9), with 32 down-regulated and 62 up-regulated genes in Raphanobrassica/R. sativus, and 30 down-regulated and 82 up-regulated genes in Raphanobrassica/B. oleracea var. alboglabra retro-regulation by miRNAs. Among the related target genes, only 31 genes were non-additively expressed, targeting 14 miRNAs. Obviously, miRNA5658 contains a considerable number of related target genes, and 15.00% (12/80) related target genes targeted by miRNA5658 were non-additively expressed. Moreover, both miRNA156a and miRNA8005a have 14 related target genes, achieving the second and third most highly correlated miRNAs. Among the 14 genes targeted by miRNA156a, only bra014212 was non-additively expressed. In miRNA8005a, two genes, bra015748 and bra032246, were non-additively expressed. To screen these differentially expressed miRNAs target genes, we performed a functional enrichment analysis based on GO functional annotations. Interestingly, the term ‘metabolic process’ and ‘response to stimulus’ were significantly enriched in the relevant target genes of Raphanobrassica/B. oleracea var. alboglabra compared with Raphanobrassica/R. sativus (Fig. 10). As a result of frequently restrained miRNAs, the genes related to ‘metabolic process’ and ‘response to stimulus’ in Raphanobrassica were up-regulated compared with its parents. Based on the information contained in GO enrichment of mRNAs and miRNAs, the GO term ‘metabolic process’ was significantly enriched, no matter in differentially expressed mRNAs/miRNAs (target genes) or non-additively expressed mRNAs/miRNAs (target genes) in Raphanobrassica compared with its parents. MiRNA5658a, which contained 44.94% of miRNA-target pairs in correlation network in the present study, was also significantly enriched in the terms ‘protein catabolic process’, ‘response to stress’ and ‘response to biotic and abiotic stress’ in Ocimum basilicum42. The 31 related target genes showing non-additive expression were also significantly enriched in ‘response to stress’ and response to biotic’ in the present study, but the non-additively expressed gene targets of miRNA5658a were significantly enriched in the ‘metabolic process’ category (Fig. S8).
qRT-PCR validation of significant differentially expressed mRNA-genes and miRNAs
To confirm the expression profiles obtained from high-throughput sequencing, stem-loop real-time quantification RT-RCR was performed to study the ten mRNAs and eight miRNAs (Fig. 11 and Table 3). The expression patterns of the selected miRNAs and miRNAs were consistent with those determined using high-throughput sequencing, indicating that the sequencing data produced in the present study were reliable and could be subjected to further analysis.
Discussion
Until recently, many studies have examined the mechanism gene expression in polyploidy4,14,15,20. For example, Zhao15 suggested that high ploidy levels in the Brassica hexaploid might reflect increased genome and transcriptome changes. In Arabidopsis allopolyploids, small RNAs act as a buffer against genomic shock by analysing miRNA expression and siRNA distribution35. Raphanobrassica was artificially synthesized from modern cultivars of R. sativus and B. oleracea var. alboglabra without obvious reproductive barriers, indicating almost instant genetic stabilization. The advent of high-through sequencing technology provided unprecedented opportunities to examine this inter-generic hybridization allopolyploid with the data obtained from two pivotal components: mRNA and microRNA.
Insight into gene expression changes in polyploids derived from inter-generic and interspecific hybridization
Non-additively expressed mRNAs and miRNAs in polyploidy have been detected in allotetraploid Arabidopsis, hexaploid wheat and citrus allopolyploids4,29,42. In the present study, the RNA-seq technique were used to compare mRNA and miRNA expression profiles between Raphanobrassica and its parents, and a total of 9,209 (27.43%) genes showed expression divergence, while 5,219 (15.55%) genes were non-additively expressed in the tetraploid Raphanobrassica. Interestingly, compared with the interspecific polyploids Brassica hexaploid in the study of Zhao15, 17.44% genes were differentially expressed, and only 7.79% genes were non-additively expressed in the allohexaploid. The frequency of changes in non-additive gene expression increased in the inter-generic tetraploid, compared with the Brassica hexaploid, likely reflecting higher gene expression divergence between the inter-generic parents. Among the miRNAs, 41.79% of miRNAs were non-additively expressed and differentially expressed between tetraploid Raphanobrassica and its parents, while 69.11% of miRNAs were non-additively expressed in the interspecific hybrid Brassica hexaploid14, which was obviously higher than that in the inter-generic tetraploid Raphanobrassica. Subsequently, a previous study reported that the volume of production was not increased following the ploidy level in R. sativus × B. alboglabra hybrids, with the tetraploid showed highest output among diploid, tetraploid, hexaploid and octaploid species43. The higher output in this tetraploid compared with diploid and other polyploid species may also reflect the increased frequency of changes in non-additive gene expression. Thus, inter-generic tetraploid Raphanobrassica may have great genomic and transcriptomic changes in mRNA expression.
The phenomenon of genes showing parent-biased has been frequently observed in many speices4,6,37. For example, Li4 showed that the parent-biased expressed genes were likely major contributors to key adaptation traits, such as stress tolerance and flexibility under flowering conditions. Del44 reported that some species show a maternal bias in mRNA transcripts and gene activity in early embryogenesis. However, a few studies also suggested that the contribution of maternal genomes had no visible distinction from the paternal genomes. Not surprisingly, a marked number of genes also showed parental-bias expression in inter-generic Raphanobrassica. We used ELD-genes to explain this phenomenon, and among the 9,209 differential expression genes between progeny and progenitors, a total of 4,467 (48.50%) genes belonged to ELD-a, while 3,166 (34.37%) genes belonged to ELD-b in Raphanobrassica, indicating that a relatively larger portion of genes showed biased expression to the maternal parent R. sativus in Raphanobrassica. In addition, model II (gene up-regulated in Raphanobrassica than in R. sativus, and relatively unaltered compared with B. oleracea var. alboglabra) was significantly higher than model XI (Fig. 5), suggesting that the genes in ELD-a exhibit increased rather than decreased expression level in the progeny, indicating that interesting variations in the gene expression might have occurred in this region. Moreover, all of the ELD-a miRNAs belonged to model II (miRNA up-regulated in Raphanobrassica than in R. sativus, and relatively unaltered compared with B. oleracea var. alboglabra), but miRNAs in the ELD-b showed equal distribution, indicating that if the progeny miRNAs showed no significant changes compared with the other parents, then the miRNAs that the maternal parent R. sativus transmitted to Raphanobrassica were down-regulated. However, the miRNA in Raphanobrassica transmitted from the paternal parent B. oleracea var. alboglabra were impartially changed in an up and down regulated mode. This result may suggest a comparatively specific difference between Raphanobrassica and its maternal parent R. sativus. These data, combined with the gene and miRNA expression analysis, showed that the directivity between Raphanobrassica and R. sativus was larger than the contrast between Raphanobrassica and B. oleracea var. alboglabra. The maternal biased was consistent with the previous conclusions of Zhao15 and Shen14 concerning the Brassica hexaploid.
Correlation analysis of miRNAs and mRNAs are significant to study polyploidy
Recent studies have reported that the target genes repressed by miRNAs are typically involved in salt, cold or drought response treatments4,44, and 178 related target genes were detected in Raphanobrassica, and most of these target genes were significantly enriched in down-regulated compared with up-regulated related target genes, except the terms ‘macromolecular complex’, ‘molecular transducer’ and ‘response to stimulus’ (Fig. S9), suggesting that the related target genes exhibited an increased influence in most of the biological pathways in the progeny. In the interspecific hybrid allopolyploid Brassica hexaploid, only the term ‘response to stimulus’ was significantly enriched in the down-regulated miRNA target genes14. These results demonstrate that the related target genes generated from inter-generic hybridization might play an important role in the growth and development changes observed in the inter-generic Raphanobrassica.
Some studies have suggested that the miRNAs-mRNAs regulation pattern includes both the coherent and incoherent relationship15,45 and that one miRNAs may have diverse target genes. For example, a single miRNA ‘miR167a’ in this Raphanobrassica can target multiple mRNAs, including Bra004125, Bra010776, Bra028110, and Bra035727. Among these, some targets show up-regulated expression compared with miRNAs, while other targets show down-regulated expression compared with miRNAs. Indeed, miRNAs may have a certain specific target mRNA and contribute to the regulation of miRNA (for example, target Bra028668 can be regulated by miR162a in Raphanobrassica). However, target genes may be regulated by several up- and down-regulated miRNAs. Bra004674 can be passively regulated by up-regulated miR156a in Raphanobrassica/B. oleracea var. alboglabra and down-regulated miR156a in tetraploid Raphanobrassica/R. sativus. This consequence indicated the target genes that did not respond to monotone miRNA expression in Raphanobrassica are complicated. Moreover, even miRNAs in a same miRNA family can display different regulation patterns. For example, miR156a was down-regulated in Raphanobrassica, but miR156b-3p was up-regulated, suggesting a difference in the regulation of expression levels between miRNAs and their target mRNAs. The miRNA expression mechanism is analogous to that in cold-stressed populus34 and the peach46. A previous study showed the indirect miRNAs modulatory effects of mRNAs45, and miRNA was also demonstrated to indirectly interact with mRNAs transport factor genes47. Furthermore, among the 178 related target genes, 31(17.41%) genes were non-additively expressed and significantly enriched in the terms ‘metabolic process’, ‘response to abiotic stimulus’ and ‘response to stress’ in Raphanobrassica. miRNA5658, which has the largest related non-additive genes in the present study, was reported to be significantly enriched in the terms ‘response to stress’ and ‘response to biotic and abiotic stress’ in Ocimum basilicum42 and significantly enriched in the term ‘metabolic process’ in Raphanobrassica. miRNA5658 might not only play particular roles in response to drought in plant regulating mechanism but also make a non-negligible elemental contribution to the regulation of biological metabolism. Thus, the mRNAs-miRNAs regulation pattern is a potential key regulator that is critical for the fitness and development of the tetraploid Raphanobrassica.
Regulation of gene expression in inter-generic allopolyploidy may increase the potential of a defence response
A marketable study demonstrated that polyploidy enhances stress responses compared with diploidy14,16,27. Analysis of Arabidopsis gene expression suggests that the stress-related genes are conserved during polyploidization48. Chen25 observed that the genes SCHLAFMUTZE and SCHNARCHZAPFEN in the AP2 TF family were inhibited by miR172 to regulate flowering time in Arabidopsis. Because overexpressing miR172 induced early flowering and organ mutations49, moderate inhibition could promote growth50. In the Brassica hexaploid15, the genes involved in the biological processes of ‘response to stimulus’ and ‘metabolic pathway’ were significantly up-regulated compared with its parents. In the present study, the related target genes involved in ‘response to stimulus’ and ‘metabolic pathway’ were differentially expressed in Raphanobrassica compared with its parents. For example, miR169, which is down-regulated under drought conditions, and its target gene NFYA5 were up-regulated and contributed to strong drought resistance in Arabidopsis51, and these genes were up-regulated in Raphanobrassica compared with its parents. Bra015496, encoding a salt tolerance-related protein targeted by miR5256, was also up-regulated in Raphanobrassica and its parents.
The metabolic pathway plays an unneglectable role in the plant defence response. Under hyperthermia, ARR5 gene family shows a negative correlation with the plant defence mechanism52. In the present study, the transcription factor gene bra001641 of the homologous gene family ARR5-B was detected and implicated in the metabolic pathway. Remarkably, this gene is down-regulated in Raphanobrassica compared with its parent, reflecting a negative correlation; thus, an efficient defence mechanism may exist. The differentially expressed gene bra001641 was also non-additively expressed in the present study and showed maternal bias. These findings suggested that the regulation of non-additively expressed genes in Raphanobrassica might increase the defence response.
Conclusion
The present study is the first to explore the data of mRNAs and miRNAs to identify genes and miRNA expression levels in the inter-generic hybrid tetraploid Raphanobrassica and its parents. Several mRNAs and miRNAs from Raphanobrassica and its parents that are involved in diverse pathways were detected. Non-additively expressed genes in the inter-generic hybrid tetraploid Raphanobrassica revealed some key pathways (‘antioxidant’, ‘auxiliary transport protein’ and ‘multi/-organism process’), which were different from non-additively expressed genes in the interspecific Brassica hexaploid15, providing informative results to detect the difference between inter-generic and interspecific polyploids. Interestingly, the ‘metabolic process’ pathway was significantly enriched both in non-additively expressed mRNAs/miRNAs (target genes) or related target genes from the miRNA-mRNA network in Raphanobrassica compared with its parents, and this pathway might play an important role in Raphanobrassica. The relationship between miRNAs and mRNAs is complex and dynamic, and further studies on the mRNA-mRNA network are required to deeply analyse the superiority of allopolyploids.
Methods
Plant materials
Plant materials included Brassica oleracea var. alboglabra (CC 2n = 18), Raphanus sativus (RR 2n = 18) and Raphanobrassica (RRCC 2n = 36). The amphidiploid Raphanobrassica (RRCC 2n = 36) was generated by crossing paternal cabbage mustard (B. oleracea var. alboglabra) with maternal radish (R. sativus) followed by chromosome doubling, and the plants were grown and self-pollinated in the field of Huazhong Agricultural University, China, in the sixth generation39. Immature leaves of Raphanobrassica and its parents were cut and immediately frozen in liquid nitrogen, then store them in ultra-low temperature freezers for later use.
Library construction and high-throughput sequencing
The sequencing libraries were prepared using the Illumina RNA-seq kit as described by Xu et al.28. Total RNA from Raphanobrassica and its parents was extracted using TRIzol reagent (Invitrogen, Burlington, ON, Canada) and then RNase-free DNase I was used to avoid genomic DNA contamination. Dynabeads oligo (dT) (Dynal, Invitrogen) was performed to extract mRNA. The double-stranded cDNAs were synthesized using the reversed transcriptase (Superscript Double Stranded cDNA synthesized kit; Invitrogen) and random hexamer primers (Illumina, San Diego, CA, USA). Then the RNA-seq libraries were constructed by standard Illumna protocol. RNA quality was corrected on Agilent 2100 NanoDrop bioanalyzer. Only samples containing suitable standards (m ≥ 10 μg, c ≥ 200 ng/μL, OD260/280 ≥ 1.8) were accepted for further analysis16.
mRNA sequencing data analysis and gene identification
To study the effect of ployploidization on gene expression in inter-generic synthetic tetraploid Raphanobrassica, we used the method of RNA-seq (modified) to compare the difference of gene expression between Raphanobrassica and its parents. After discarding the low quality raw reads, a total of 61,396,492 sequence reads (average length of 140 bp) were generated from cDNA libraries driven from R. sativus, B. oleracea var. alboglabra, and Raphanobrassica.
Functional analysis and characterization of unigenes and DEGs
Gene function was annotated based on the following databases: Nr (NCBI non-redundant protein sequences); Nt (NCBI non-redundant nucleotide sequences); GO (Gene Ontology), KO (KEGG Ontholog database). Plant Transcription Factor Database (http://brassicadb.org/brad/index.php) was used for identification and classification of transcriptional factors. For further study of the significance variation of the detected genes in biological processes/pathway, GO functional classes (http://www.geneontology.org/) and BLAST2GO (version 2.3.5) (http://www.blast2go.org/) were used to precisely define relevance genes. GO enrichment analysis was identified with the BinGO plugin Cytoscape. Kyoto Encyclopedia of Genes and Genomes (KEGG) is a database linking genomic information with higher order functional information by mapping them to reference canonical pathways (http://www.genome.ad.jp/kegg/). KEGG pathway enrichment analysis was done using KOBAS software (KOBAS, Surrey, UK).
Identification of non-additive expressed genes
To determine non-additively expressed genes, we compared gene expression levels in Raphanobrassica with the average genes expression values in the parents (MPVs). If the value of a gene in Raphanobrassica showed at least a two-fold change and p < 0.05 relative to the MPV, then the gene was regarded as non-additive expression37.
Analysis of differential expressed gene
Differential expressed genes were performed using the R package DEGseq53. Genes with log2Ratio ≥ 1, 0.05 were defined differentially expressed genes (DEG).
Quantitative RT-PCR validation for mRNAs
Total RNA was fetched from young leaves of R. sativus, B. oleracea var. alboglabra and tetraploid Raphanobrassica. Quantitative PCR was used to quantify the significantly differentially expressed mRNAs, ten mRNAs were obtained from high through-put sequencing, and qPCR assays were carried out using SYBR kit under the LightCycler Real-Time PCR System with the parameters: 30 s at 95 °C, 40 cycles at 95 °C for 5 s, 30 s at 60 °C. Triplicates of each reaction were performed as the mean ± SD. CT method were used to calculate comparative the relative fold change values. The detail primers used in all quantitative qRT-PCR are given in Table 3.
Small RNA library preparation
For sRNA library construction, young leaves of R. sativus, B. oleracea var. olablagra and Raphanobrassica were separately extracted using Trizol reagent (Invitrogen, USA). Then use RNase-free DNase I (Fermentas, Canada) to remove the residual DNA. Sequencing libraries were generated using Illumina high-throughput sequencing technology at Beijing Genomics Institute (BGI).
Analysis of small RNA sequencing data
Illumina high-throughput sequencing technology was employed to study the variation of small RNAs and their potential roles in gene regulation Raphanobrassica and its parents. After cluster generation, the library preparations were sequenced on Illumina Hiseq 2000 platform and based on BGI small RNA pipeline, and the adaptor sequences and lower quality reads were removed. Then the 18–30 nt RNAs were aligned to the B. rapa genome using SOAP software34 to identify the repeat-associated sRNAs and to assess the expression of sRNAs. The cellular structural RNAs which include tRNA, scRNA, snoRNA or snRNA, were removed comparison with the NCBI nucleotide database and Rfam RNA family database. The miRNAs were indentified using miRBase. In order to realize the differential expression of miRNAs, the abundance of miRNAs in three libraries was normalized as values to compare between each two libraries and were calculated in the form of fold changes (fold change = log2). In addition, p-value was used to be a previously described formula54. The gene function includes biological process, cellular component and molecular function which were analyzed for the predicted target genes.
Differential expression analysis of miRNAs
Differentially expressd miRNAs between Raphanobrassica and its parents were performed using the DEGseq software55. P-value was adjusted-using q-value. Q-value < 0.01 and log2-fold change >1 was set as the threshold significantly differential expression by default.
Identification of ELD-miRNAs between Raphphanobrassica and its parents
In order to understand how hybridization and polyploidy regulate miRNAs expression, we compared miRNAs expression in Raphanobrassica with the mid-parent value (MPV) from parents species to deem the ELD-miRNAs (If the value of a miRNAs showed at least a twofold change and 0.05 compared with MPV), the others were considered to be additive miRNAs.
MiRNA target gene prediction
Prediction of conserved miRNAs target genes was performed by psRobot_tarscripts in psRobot56.
Quantitative RT-PCR validation for miRNAs
Total RNA was fetched from young leaves of R. sativus, B. oleracea var. alboglabra and tetraploid Raphanobrassica. Stem-loop RT-PCR was used to validate miRNA expression, and the miRNA-specific stem-loop primers were assigned as Chen39. qRT-PCR was performed in triplicate combine with the SYBR kit, and the samples were amplified using the LightCycler Real-Time PCR System under the process: 30 s at 95 °C, 40 cycles at 95 °C for 10 s, 30 s at 56 °C, 15 s at 72 °C. Fold changes in miRNAs of different samples were calculated by CT method. The primers were detail listed in Table 3.
Correlation analysis of miRNA and mRNA
In order to build the miRNA-mRNA interaction network, including positive and negative relationships between mRNA and miRNA, we used an in-house method to construct miRNA-mRNA network. We integrated the expression data with mRNAs, and the main concern was negatively correlated miRNAs and mRNAs. We defined a predicted miRNAs target gene as related target gene if its expression pattern was contrary with the miRNAs, for example, of all miRNAs up-regulated in a Raphanobrassica, the down-regulated expression target gene we call relevance target genes, the others we called non-related target genes; contrapose down-regulated miRNAs in Raphanobrassica, the target genes which up-regulated we call related target genes, the others called non-related target genes.
Additional Information
How to cite this article: Ye, B. et al. Correlation analysis of the mRNA and miRNA expression profiles in the nascent synthetic allotetraploid Raphanobrassica. Sci. Rep. 6, 37416; doi: 10.1038/srep37416 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Wendel, J. F. Genome evolution in polyploids. Plant Mol. Biol. 42, 225–249 (2000).
Leitch, A. R. & Leitch, I. J. Genomic plasticity and the diversity of ployploid plants. Science 320, 481–483 (2008).
Soltis, D. E. et al. Polyploidy and angiosperm diversification. Am. J. Bot. 96, 336–348 (2009).
Li, A. et al. mRNA and small RNA transcriptomes reveal insights into dynamic homoeolog regulation of allopolyploid heterosis in nascent hexaploid wheat. Plant Cell 26, 1878–1900 (2014).
Abbott, R. et al. Hybridization and speciation. J. Evol. Biol. 26, 229–246 (2013).
Ng, D. W. et al. Proteomic divergence in Arabidopsis autopolyploids and allopolyploids and their progenitors. Heredity 108, 419–430 (2012).
Kovarik, A. et al. Concerted evolution of 18-5.8-26S rDNA repeats in Nicotiana allotetraploids. Biol. J. Linn. Soc. 82, 615–625 (2004).
Kovarik, A. et al. Rapid concerted evolution of nuclear ribosomal DNA in two Tragopogon allopolyploids of recent and recurrent origin. Genetics 169, 931–944 (2005).
Ma, X. F. & Gustafson, J. P. Timing and rate of genome variation in triticale following allopolyploidization. Genome 49, 950–958 (2006).
Tate, J. A. et al. Evolution and expression of homeologous loci in Tragopogon miscellus (Asteraceae), a recent and reciprocally formed allopolyploid. Genetics 173, 1599–1611 (2006).
Lukens, L. N. et al. Patterns of sequence loss and cytosine methylation within a population of newly resynthesized Brassica napus allopolyploids. Plant Physiol. 140, 336–348 (2006).
Xu, Y., Zhong, L., Wu, X., Fang, X. & Wang, J. Rapid alterations of gene expression and cytosine methylation in newly synthesized Brassica napus allopolyploids. Planta 229, 471–483 (2009).
Hegarty, M. J. et al. Nonadditive changes to cytosine methylation as a consequence of hybridization and genome duplication in Senecio (Asteraceae). Mol. Ecol. 20, 105–113 (2011).
Shen, Y. et al. Characterization and expression patterns of small RNAs in synthesized Brassica hexaploids. Plant Mol. Biol. 85, 287–299 (2014).
Zhao, Q., Zou, J., Meng, J., Mei, S. & Wang, J. Tracing the transcriptomic changes in synthetic trigenomic allohexaploids of Brassica using an RNA-seq approach. PLoS One 8, e68883 (2013).
Dina, G., harsh, D., Torsten, G. & Olga, S. Alteration of mRNA and microRNA expression profiles in rat muscular type vasculature in early postnatal development. Sci. Rep. 5, 11106 (2015).
Chagué, V. et al. Genome wide gene expression changes in genetically stable synthetic and natural wheat allohexaploids. New Phytol. 187, 1181–1194 (2010).
Buggs, R. et al. Transcriptomic shock generates evolutionary novelty in a newly formed, natural allopolyploid plant. Curr. Biol. 21, 551–556 (2011).
Bottley, A., Xia, G. M. & Koebner, R. M. D. Homoeologous gene silencing in hexaploid wheat. Plant J. 47, 897–906 (2006).
Chao, D. Y. et al. Polyploids exhibit higher potassium uptake and salinity tolerance in Arabidopsis. Science 341, 658–659 (2013).
Chelaifa, H. et al. Prevalence of gene expression additively in genetically stable wheat allohexaploids. New Phytol. 197, 730–736 (2013).
Rapp, R. A., Udall, J. A. & Wendel, J. F. Genomic expression dominance in allopolyploids. BMC Biol. 7, 18 (2009).
Chaudhary, B. et al. Reciprocal silencing, transcriptional bias and functional divergence of homeologs in polyploid cotton (Gossypium). Genetics 182, 503–517 (2009).
Ni, Z. et al. Altered circadian rhythms regulate growth vigor in hybrids and allopolyploids. Nature 457, 327–331 (2009).
Chen, Z. et al. Pseudomonas syringae type III effector AvrRpt2 alters Arabidopsis thaliana auxin physiology. Proc. Natl. Acad. Sci. USA 104, 20131–20136 (2007).
Hegarty, M. J. et al. Transcriptome shock after interspecific hybridization in Senecio is ameliorated by genome duplication. Curr. Biol. 16, 1652–1659 (2006).
Flagel, L., Udall, J., Nettleton, D. & Wendel, J. Duplicate gene expression in allopolyploid Gossypium reveals two temporally distinct phases of expression evolution. BMC Biol. 6, 16 (2008).
Xu, Y., Xu, H., Wu, X., Fang, X. & Wang, J. Genetic changes following hybridization and genome doubling in synthetic Brassica napus. Biochem. Genet. 50, 616–624 (2012).
He, L. & Hannon, G. J. MicroRNAs: small RNAs with a big role in gene regulation. Nat. Rev. Genet. 5, 522–531 (2004).
Bartel, D. P. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297 (2004).
Vaucheret, H. Post-transcriptional small RNA pathways in plants: mechanisms and regulations. Gene Dev. 20, 759–771 (2006).
Li, C. et al. RNA-seq analysis of mucosal immune responses reveals signatures of intestinal barrier disruption and pathogen entry following Edwardsiella ictaluri infection in channel catfish, Ictalurus punctatus. Fish. Shellfish. Immun. 32, 816–827 (2012).
Li, B., Qin, Y., Duan, H., Yin, W. & Xia, X. Genome-wide characterization of new and drought stress responsive microRNAs in Populus euphratica. J. Exp. Bot. 62, 3765–3779 (2011).
Liu, H. H., Tian, X., Li, Y. J., Wu, C. A. & Zheng, C. C. Microarray-based analysis of stress-regulated microRNAs in Arabidopsis thaliana. RNA 14, 836–843 (2008).
Yang, X., Wang, L., Yuan, D., Lindsey, K. & Zhang, X. Small RNA and degradome sequencing reveal complex miRNA regulation during cotton somatic embryogenesis. J. Exp. Bot. 64, 1521–1536 (2013).
Guan, Q. et al. A DEAD box RNA helicase is critical for pre-mRNA splicing, cold-responsive gene regulation, and cold tolerance in Arabidopsis. Plant Cell 25, 342–356 (2013).
Wang, J. et al. Genome wide non-additive gene regulation in Arabidopsis allotetraploids. Genetics 172, 507–517 (2006).
Gaeta, R. T., Pires, J. C., Iniguez-Luy, F., Leon, E. & Osborn, T. C. Genomic changes in resynthesized Brassica napus and their effect on gene expression and phenotype. Plant Cell 19, 3403–3417 (2007).
Chen, H., Wu, J. & Cheng, Y. Synthesis and GISH analysis of R. sativus-B. oleracea var. alboglabra allotetraploid. J. Agron. Crop. Sci. 32, 1117–1120 (2006).
Grover, C. E. et al. Homoeolog expression bias and expression level dominance in allopolyploids. New Phytol. 196, 966–971 (2012).
Jiang, Y., Tian, E., Li, R., Chen, L. & Meng, J. Genetic diversity of Brassica carinata with emphasis on the interspecific crossability with B. rapa. Plant Breeding 126, 487–491 (2007).
Singh, N. & Sharma, A. In-silico identification of miRNAs and their regulating target functions in Ocimum basilicum. Gene 552, 277–282 (2014).
Niimi, H. et al. Amiprophosmethyl-induced efficient in vitro production of polyploids in Raphanobrassica with the aid of aminoethoxy vinyl glycine (AVG) in the culture medium. Breeding Sci. 65, 396 (2015).
Khraiwesh, B., Zhu, J. K. & Zhu, J. Role of miRNAs and siRNAs in biotic and abiotic stress responses of plants. BBA-Gene. Regul. Mech. 1819, 137–148 (2012).
Jeon, J. et al. A subset of cytokinin two-component signaling system plays a role in cold temperature stress response in Arabidopsis. J. Biol. Chem. 285, 23371–23386 (2010).
Barakat, A. et al. Genome wide identification of chilling responsive microRNAs in Prunus persica. BMC Genomics 13, 481 (2012).
Le, H. S. & Bar-Joseph, Z. Integrating sequence, expression and interaction data to determine condition-specific miRNA regulation. Bioinformatics 29, i89–i97 (2013).
Casneuf, T., Bodt, S., Raes, J., Maere, S. & Van, D. Y. Non-random divergence of gene expression following gene and genome duplications in the owering plant Arabidopsis thaliana. Genome Biol. 7, R13 (2006).
Aukerman, M. J. & Sakai, H. Regulation of flowering time and floral organ identity by a miRNA and its APETALA2-like target genes. Plant Cell 15, 2730–2741 (2003).
Chen, M., Meng, Y. J., Gu, H. B. & Chen, D. J. Functional characterization of plant small RNAs based on next-generation sequencing data. Comput. Biol. Chem. 34, 308–312 (2010).
Li, W. X. et al. The Arabidopsis NFYA5 transcription factor is regulated transcriptionally and post-transcriptionally to promote drought resistance. Plant Cell 20, 2238–2251 (2008).
Danilova, M. N. et al. Molecular and physiological responses of Arabidopsis thaliana plants deficient in the genes responsible for ABA and cytokinin reception and metabolism to heat shock. Russ. J. Plant Physiol. 3, 308–318 (2016).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B. 57, 289–300 (1995).
Gao, Z. H. et al. High-throuput sequencing of small RNAs and analysis of differentially expressed microRNAs associated with pistil development in Japanese apricot. BMC Genomics 13, 371–384 (2012).
Anders, S. & Huber, W. Differential expression analyses for sequence count data. Genome Biol. 11, R106 (2010).
Wu, H. et al. PsRobot: a web-based plant small RNA meta-analysis toolbox. Nucleic. Acids. Res. 40, W22–W28 (2012).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31370258, 31070204).
Author information
Authors and Affiliations
Contributions
J.W. and B.Y. designed the research. B.Y. performed the laboratory experiments and result analysis, and wrote the main part of the manuscript. J.W. and R.W. revised the manuscript. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ye, B., Wang, R. & Wang, J. Correlation analysis of the mRNA and miRNA expression profiles in the nascent synthetic allotetraploid Raphanobrassica. Sci Rep 6, 37416 (2016). https://doi.org/10.1038/srep37416
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37416
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.