Abstract
TBN, a novel tetramethylpyrazine derivative armed with a powerful free radical-scavenging nitrone moiety, has been reported to reduce cerebral infarction in rats through multi-functional mechanisms of action. Here we study the therapeutic effects of TBN on non-human primate model of stroke. Thirty male Cynomolgus macaques were subjected to stroke with 4 hours ischemia and then reperfusion. TBN were injected intravenously at 3 or 6 hours after the onset of ischemia. Cerebral infarction was examined by magnetic resonance imaging at 1 and 4 weeks post ischemia. Neurological severity scores were evaluated during 4 weeks observation. At the end of experiment, protein markers associated with the stroke injury and TBN treatment were screened by quantitative proteomics. We found that TBN readily penetrated the blood brain barrier and reached effective therapeutic concentration after intravenous administration. It significantly reduced brain infarction and modestly preserved the neurological function of stroke-affected arm. TBN suppressed over-expression of neuroinflammatory marker vimentin and decreased the numbers of GFAP-positive cells, while reversed down-regulation of myelination-associated protein 2′, 3′-cyclic-nucleotide 3′-phosphodiesterase and increased the numbers of NeuN-positive cells in the ipsilateral peri-infarct area. TBN may serve as a promising new clinical candidate for the treatment of ischemic stroke.
Similar content being viewed by others
Introduction
Stroke is one of the most devastating diseases affecting the health and life of human beings. Despite the remarkable progresses achieved in the last two decades in understanding the pathophysiology of stroke, tissue-type plasminogen activator (tPA) remains the sole therapeutics approved by the US FDA for the treatment of acute ischemic stroke. However, tPA has a narrow therapeutic window of 3–4.5 h within the occurrence of a stroke, limiting its clinical use. Besides, tPA has no neuroprotective effect. Although many pharmacological agents for stroke have shown considerable promise in rodent models, none have successfully been translated for use in humans1. Nevertheless, finding an effective neuroprotective agent remains a strongly sought after goal2,3.
One of the most extensively studied classes of neuroprotective agents is free radical scavengers, as free radical over-production is a major contributor to brain injury after stroke. NXY-059, a free radical-trapping nitrone, showed positive results when evaluated in various animal stroke models but failed its second phase III clinical trial4,5. The cause of this failure is multifactorial, including poorly designed preclinical and clinical studies6 and its poor ability in penetrating the blood-brain barrier (BBB)7,8. Edaravone, also a free radical scavenger, has been approved for treatment of patients with ischemic stroke in Japan9 and China. However, it is not approved for use in Western countries due to limited efficacy and side effects, such as acute renal failure, liver dysfunction and acute allergic reactions10.
Tetramethylpyrazine (TMP), a main active ingredient of Ligusticum wallichii Franchat (Chuanxiong), has been widely used as a treatment of ischemic stroke for a long time in China11. However, until now, there has not ever been a methodologically sound clinical trial demonstrating its benefit. Although the exact mechanisms of action of TMP have not been completely understood, a variety of mechanisms may be attributed to its beneficial effects in stroke patients, including inhibiting platelet aggregation, lysing blood clots, blocking intracellular calcium entry and scavenging free radicals12. Nitrone is originally developed as free radical-trapping agents in free radical chemistry, potently scavenges free radicals and protects biological systems from oxidative stress8. Nitrones have been tested as therapeutic agents for neural and systemic dysfunctions including atherosclerosis, septicemia, stroke, and Alzheimer’s disease13,14. Taking advantage of the beneficial effects of both TMP and nitrone, therefore, we designed and synthesized (2-[[(1,1-dimethylethyl)oxidoimino]-methyl]-3,5,6-trimethylpyrazine, TBN), a TMP derivative armed with a powerful free radical-scavenging nitrone moiety. TBN was desired to possess the pharmacological properties of both TMP and nitrone.
We previously demonstrated that TBN protected rats from ischemic stroke damage15,16. A multi-functional mechanisms possibly contributed to TBN’s beneficial effects in stroke models, including directly scavenging various free radicals, blockade of calcium entry and inhibition of platelet aggregation16. To bridge the biological gap between the rodent animal experiments and clinical trials, in the present study, we investigated the therapeutic efficacy of TBN in non-human primate (NHP) model of ischemic stroke, as well as its pharmacokinetics, fulfilling the requirements for initiating investigational new drug application.
Results
TBN reduces cerebral infarction and improves neurological functions in stroke monkeys
We evaluated TBN in M. fascicularis (cynomolgus macaque) subjected to t-MCAo. At one week post-surgery, TBN treatment administered at 3 h post-ischemia caused a reduction in overall infarct size in a dose-dependent manner as measured by T2-weighted MRI scans (Fig. 1A,B). Since only a small percentage (~3%) of stroke patients receive therapeutic intervention within 3 h after onset of ischemic stroke17, it was important to establish whether TBN is effective even under delayed treatment scenarios. TBN was found to be effective even when administered 6 h post-ischemia, indicating the potential for an expanded therapeutic window (Fig. 1A,B). At four weeks, similar dose-dependent trends were observed, with all treatments resulting in significant reductions in infarct size (Fig. 1A,C).
TBN attenuates brain infarct volume and improves neurological function in monkeys subjected to t-MCAo.
(A) Representative images of T2-weighted MRI scan. (B,C) Brain infarct size of ipsilateral hemisphere. Data were expressed as dot plots with means in the left panel and mean ± s.e.m. in the right panel (n = 6 for each group). (D) Success rate of food catching of the contralateral, stroke-affected hand. Data were expressed as mean ± s.e.m. (n = 6 for each group). ‘@3 h’ and ‘@6 h’ mean TBN treatment initiating at 3 h and 6 h post-ischemia.
Functional neurological assessments were performed throughout the 28-day observation period, which ascertained that TBN treatment ameliorated the long-term neurological impairment produced by t-MCAo. Monkeys treated with TBN trended to have lower neurological deficit scores at 1 and 4 weeks post-ischemia (Table 1). Scores included assessments of consciousness, sensory and motor system function, and skeletal muscle coordination. Impressively, at 4 weeks post-ischemia, both the 30 and 90 mg kg−1 TBN treatment regiments produced a ~50% recovery of arm function, as demonstrated by the restored ability of the contralateral, stroke-affected arm to acquire food (Fig. 1D and see Supplementary Movie 1 and 2).
TBN readily penetrates the blood-brain barrier and reaches therapeutic concentration
One reason that NXY-059 failed in clinical trials was its poor penetration of the BBB, with only 1% penetration in rats8. To investigate its ability to penetrate the BBB, TBN’s concentration in the monkey plasma and cerebrospinal fluid were examined. After intravenous (i.v.) administration of 30 and 90 mg kg−1 in monkeys, the plasma Cmax of TBN reached 370 and 549 μM, respectively (Fig. 2A). In the CSF, the concentrations for the 30 and 90 mg kg−1 TBN treatment regiments were 176 μM at 15 min and 379 μM at 45 min, respectively (Fig. 2B). At 2 h after administration, TBN was still present at concentrations of 58 and 148 μM, respectively. TBN concentrations achieved in vivo were at similar levels to those (30–300 μM) that protected cortical neurons against glutamate-induced excitotoxicity effectively (Fig. 2C), and attenuated oxygen and glucose deprivation (OGD)-induced release of lactate dehydrogenase (LDH) in cortical neurons (Fig. 2D).
TBN readily penetrates the blood-brain barrier and reaches neuroprotective concentrations.
(A,B) Blood and brain cerebrospinal fluid (CSF) concentrations vs. time profile after drug administration. (C) TBN protects primary cortical neurons against glutamate-induced excitotoxicity. #P < 0.05 versus control (Ctrl); **P < 0.01 versus glutamate treatment alone. Data were from three independent experiments. (D) TBN attenuates OGD-induced LDH release in primary cortical neurons. #P < 0.05 versus control (Ctrl); *P < 0.05 and **P < 0.01 versus OGD alone. Data were from three independent experiments.
Characterization of molecular events involved in neuroprotection by iTRAQ-based quantitative proteomic analyses
To gain insight into the molecular mechanisms underlying TBN’s neuroprotection in vivo, the cerebral tissues of the untreated and 30 mg kg−1 TBN-treated monkeys were subjected to iTRAQ-based quantitative proteomic analyses (see Supplementary Fig. S1). A total of 2935 non-redundant proteins were confidently identified (local FDR <5%) (see Supplementary Dataset 1), with the differential expression profiles of 1497 quantified proteins compared in the model peri-infarct tissue versus the contralateral normal tissue and the TBN-treated peri-infarct tissue versus the contralateral normal tissue (see Supplementary Dataset 2). Seven and 37 proteins were observed to be dysregulated exclusively in the untreated and TBN-treated cerebral tissues, respectively, with 2 proteins showing reciprocal expression and 5 proteins being upregulated in both sample groups (see Supplementary Table 1 and Supplementary Dataset 3). Thirty-three of these 51 dysregulated proteins were functionally annotated to cellular processes associated with the chronic phase of stroke, including angiogenesis, neurogenesis, astrocytosis and inflammation18,19,20,21,22 (Fig. 3). These results revealed the major cellular processes associated with the observed proteomic changes. Interestingly, 8 of the 18 dysregulated proteins that were not annotated to the aforementioned processes are associated with various aspects of cellular energy metabolism and mitochondria. All were downregulated after TBN treatment (see Supplementary Table 2).
Dysregulated proteins in quantitative proteomic analyses functionally annotate to cellular processes associated with the chronic phase of stroke.
(A) Hierarchical cluster analysis of dysregulated proteins for the stroke peri-infarct tissue versus the contralateral normal tissue and the TBN-treated peri-infarct tissue versus the contralateral normal tissue. (B) Dysregulated proteins functionally annotate to cellular processes of angiogenesis, neurogenesis, astrogenesis and inflammation.
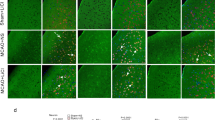
Two of the significantly dysregulated proteins, namely vimentin and 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNP) were chosen for further validation using Western blotting, as changes of both vimentin and CNP have been previously reported in rodent models of ischemic stroke23,24,25. Vimentin expression was markedly up-regulated in the ipsilateral (Ipsi) region of the brain compared to that of the contralateral part. TBN treatment notably suppressed the up-regulation of vimentin in the ipsilateral region (Fig. 4A,B and see Supplementary Fig. S2). Vimentin is a component of the intermediate filament system and a prominent marker of microglia and reactive astrocytes23. Up-regulation of vimentin demonstrates that microglia and reactive astrocytes are activated and/or are proliferating in response to the presence of injured neural tissues (Fig. 4A,B and see Supplementary Fig. S2). Microglia and astrocytes activation and proliferation were further confirmed by the increased presence of GFAP+ cells by immunohistochemistry (Fig. 4C,D). Unlike vimentin expresson, the expression level of CNP was dramatically decreased in the stroke peri-infarct tissue in comparison to that in the contralateral normal tissue (Fig. 4A,B and see Supplementary Fig. S2). CNP is a highly expressed enzyme in the myelin sheath and is believed to play an essential role in axon myelination24 (Fig. 4A,B and see Supplementary Fig. S2). The decreased CNP expression implies neuronal damage and demyelination in stroke24,26. TBN treatment caused a reversal in the expression of CNP (Fig. 4A,B and see Supplementary Fig. S2), suggesting possible neuronal repair or regeneration by TBN. In accordance with this finding, TBN treatment notably increased the number of NeuN+ neurons with respect to the stroke model group (Fig. 4D,E).
Molecular and cellular events involved in the protection of TBN against stroke injury.
(A) Representative immunoblots of monkey brain tissue. (B) Densitometric quantification of the immunoblots as shown in Supplementary Fig. S1. #P < 0.05 verses contralateral (Cont); *P < 0.05 and **P < 0.01 verses ipsilateral (Ipsi) without TBN treatment. (C) Representative immunohistochemistry images of cortical sections. Scale bars, 50 μm. (D) Quantification of the number of NeuN and GFAP positive cells. Data were expressed as percentage of sham group (n = 5 for Contralateral; n = 4 Ipsilateral without TBN and n = 6 for Ipsilateral with TBN). ##P < 0.01 verses sham group; *P < 0.05 and **P < 0.01 verses model group.
Discussion
The high-order brain of NHP is anatomically and functionally similar to that of humans and is amenable to quantitative assessment of the ischemic damage via MRI17. In addition, comprehensive functional evaluations using standardized tests are available due to the monkey’s extensive behavioral repertoire17. Researchers hope that monkey data will advance the field of stroke therapy with neuroprotective agents. NA-1(also termed Tat-NR2B9c), a 20-mer peptide functioned as a PSD-95 inhibitor, has been assessed in stroke model macaques monkey and now advances into Phase 3 clinical trial2. In reporting NA-1, Cook and his colleagues found that monkeys treated with NA-1 exhibited a significant reduction in infarct volume compared with placebo (38.7% reduction by T2-weighted MRI scans at 30-day post-stroke), NA-1 was effective even when administrated up to 3 h after the onset of stroke2. In our present study, TBN effectively reduced brain infarction by 43.84–56.08% reduction measured by T2-weighted MRI scans at 28-day post-stroke. More importantly, TBN was still effective administrated up to 6 h after the onset of stroke, suggesting that more patients can be treated. In addition to significantly reduced brain infarction, TBN treatment also trended to recover the food-catching function of stroke-affected arm. Severe contralateral hemiparesis is a long-lasting and typical clinical presentation after ischemic stroke, with functional recovery of the hemiplegic limb considered as a hallmark indicator for evaluation of the efficacy and prognosis of stroke therapeutics27. Our result suggests that TBN possibly improves the major clinical manifestations of the stroke subjects.
One of the principle features of an effective stroke neuroprotective agent is its ability to penetrate the BBB to reach the site of action. NXY-059 has two negatively charged sodium sulfonate moieties, and it is well known that negatively charged compounds hardly penetrating BBB6,8. To effectively deliver the free radical-scavenging nitrone moiety into brain tissues, TBN was designed using TMP as a carrier. TMP’s superior ability to enter the brain was implied as early as 500 years ago in the Compendium of Materia Medica (Bencao Gangmu). Using a microdialysis method, Tsai et al. demonstrated that TMP effectively penetrated the BBB, yielding a progressively higher brain/blood ratio from 10 to 120 min following intravenous administration28. In our present study, pharmacokinetic analysis in monkeys demonstrated that TBN readily penetrated the BBB and reached therapeutic concentrations in targeted cerebral tissues. This feature of TBN is pivotally important accounting for its high therapeutic efficacy.
In addition to taking advantage of TMP’s superior capability to penetrating BBB to deliver the nitrone into brain, we also take TMP’s another property, namely its ability to inhibit Ca2+ influx into cells. Thus, TBN can effectively enter the injured brain tissues to both inhibit Ca2+ influx and scavenge free radicals, two major causes of ischemic neuronal damage29. Previously, we have demonstrated that a multi-functional mechanism, including direct scavenging free radicals, blockade of intracellular Ca2+ entry, as well as inhibition of platelet aggregation, possibly contributed to TBN’s neuroprotective effects in cellular and in rodent stroke models16.
Tetramethylpyrazine (TMP) has been widely used as a treatment of ischemic stroke for a long time in China. A Chinese meta-analysis evaluated the safety of TMP injection plus conventional medicine for acute cerebral ischemic stroke. Compared to conventional medicine treatment alone, TMP injection (80–300 mg daily for 14 or 21 days) plus conventional medicine showed no significant side effects30. In the preclinical study, we compared the acute toxicity of TBN (LD50: 858 mg/kg in mice by i.v.) and TMP (LD50: 351 mg/kg in mice by i.v.), TBN was two times less toxic than TMP. In addition, because TMP has activities of inhibiting platelet aggregation and lysing blood clots, we concern if TBN causes hemorrhage when the drug will presumably be used in conjunction with thrombolytic, such as tPA, and anti-platelet agent, such as aspirin. In the preclinical study, we evaluated the effect of TBN on anti-platelet aggregation, and demonstrated that TBN had marginal effect on platelet aggregation and there was no synergism with aspirin (data not shown). Based on the safety of TMP in clinical use and the preclinical safety evaluation of TBN, we speculate that TBN would be safe and has few side effects in human.
Here we used iTRAQ-based quantitative proteomic analyses to elucidate the molecular events involved in the long-term benefit effect of TBN. The major cellular processes observed in our proteomic changes were associated with the chronic phase of stroke, including angiogenesis, neurogenesis, astrocytosis and inflammation, revealing that TBN possibly regulates the brain tissue repair and recovery from stroke injury. In addition, several proteins related to various aspects of cellular energy metabolism and mitochondria were downregulated after TBN treatment. This suggests that one of the mechanisms involved in neuroprotection of TBN may be similar to one involved in hypothermia and ischemic preconditioning, which have been shown to exhibit remarkable neuroprotection in laboratory studies and correlated with the downregulation of tissue and cellular metabolism31,32.
CNP is a highly expressed enzyme in the myelin sheath and is believed to play an essential role in axon myelination24, which is essential for the proper function of nervous system33. The decreased CNP expression implies neuronal damage and demyelination in stroke24,26. Demyelination, loss of myelin sheath around an axon, causes impaired sensation, movement, cognition, or other function aspects depending on which nerves are involved34. Remyelination in the peri-infarct area has been observed in the central nerve system after focal ischemia35. In both proteomic analysis and Western blotting validation, TBN significantly upregulated the decreased CNP expression level in the stroke peri-infarct tissue. Thus, TBN may promote the axon remyelination.
Neurogenesis is an important step in neural development, which plays a vital role in neuronal repair after damage during disease processes36,37,38. After stroke injury, the brain attempts to repair neural cell damage through initiation of neurogenesis. It has been reported that TMP could promote the proliferation and differentiation of neuronal precursor cells39,40, enhance migration toward the ischemic region in the MCAo rat model41. Adult brain has the capacity for self-repair after stroke insults via neuronal replacement from endogenous precursors42 or a latent neurogenic program in the striatal astrocytes43. Our recent unpublished data suggest that TBN notably promotes polarization of cortical neurons in vitro, and increases the production of newly born (BrdU-positive) neurons in the peri-infarct area of rat MCAo models.
In the present study, all animal experiments were randomized and blinded by an independent researcher. Researchers remained blinded throughout behavioural, imaging, histological assessments. Groups were decoded at the end of each experiment upon statistical analysis. The design of this study imitated the clinical randomized and blinded trials to test the preclinical drug efficacy, which reduces the confounding effects of bias. Our approach may help to advance the translational roadblock, in which promising preclinical candidates fail to be translated to clinically effective treatments44,45. Recently, the clinical trial of TBN for ischemic stroke therapy has been approved by the China Food and Drug Administration.
In conclusion, TBN reduced cerebral infarction and improved neurological functions with unique multifunctional mechanisms of action in an ischemic stroke model of non-human primates. Finally, the therapeutic efficacy of neuroprotective TBN for ischemic stroke in preclinical findings should be further tested in clinical trials.
Methods
Monkey transient middle cerebral artery (MCA) occlusion (t-MCAo) model
Thirty adult male Cynomolgus macaques were used. Animals were 4–5 years of age and 3.0–4.0 kg of body weight at the start of the experiment. The animals were housed individually in stainless steel cages with 63 cm wide by 76 cm high by 76 cm deep (confirmation to NIH requirement) and maintained on a 12 h light-dark cycle (light on 8:00 to 20:00), had good visual and auditory interaction with other monkeys. All animals received water ad libitum and were provided monkey chow, supplemented with milk, fruit and seeds throughout the day. Primate-specific toys and audiovisual media were provided during light hours. Animals were fasted for 12 h before administration of anesthesia or behavioral testing.
The details of stroke surgical procedure were described in Supplementary Method. Two monkeys with failure of ischemic and/or reperfusion were excluded from further experiments. Three monkeys died within 3 h post-surgery and before saline or drug administration as a result of surgical/anesthetic complications. No animals died because of ischemic stroke or drug treatment during the experimental period, and no animals were excluded from data analysis at the end of the experiment.
Body weights were monitored and not significantly affected by surgery and drug treatment during the experimental period (see Supplementary Fig. S3B). The monkeys’ behaviour was measured for 28 days. Brain infarct size was inspected using magnetic resonance imaging (MRI) on days 7 and 28 after surgery. The representative images of Tof-3D-multi-slab scan at 7 days post stroke showed that the M1 segment of the left middle cerebral artery in all monkeys were open (see Supplementary Fig. S4), suggesting that surgical process of MCA reperfusion succeeded, no artery injury and reperfusion good.
Surgery, behavioural testing, MRI inspection and histological analysis were all done blinded to those who performed the experiments.
All procedures were performed in accordance with testing facility Standards of Practice (SOP) and regulations of Kunming Biomedical International (KBI Ltd., Yunnan Province, China). Animal experimental protocols were approved by the institutional animal care and use committee (IACUC) of KBI Ltd., which ensured humane and proper care of research animals. All efforts were made to minimize the numbers of animals used and ensure minimal suffering.
Experimental groups
Thirty monkeys with successful t-MCAo surgery were block-randomly divided into five groups with 6 per group: (1) Model, stroke animals treated with saline; (2) 10 mg kg−1 TBN; (3) 30 mg kg−1; (4) 90 mg kg−1 TBN; (5) 90 mg kg−1 TBN delayed treatment. On the day of surgery, saline or drugs (dissolved in saline) were given (i.v. bolus, 1 mL kg−1) at 3 and 6 h (groups 1–4) or at 6 and 9 h (group 5) after t-MCAo. Drugs were then given twice daily for a total of 7 days post-surgery. TBN (Lot. No. 20101102, purity 99.3%) used in this study was synthesized by Shanghai Medicilon Inc. (Shanghai, China).
Assessment of neurological behavior
The neurological severity score (NSS), based on that described by Kito et al.46, included assessments of consciousness, sensory and motor system function, and skeletal muscle coordination (see Supplementary Table 3). In addition to the NSS, we also tested the functional recovery of the contralateral limb using food catching. The monkeys were measured on their ability to freely catch food (apple pieces of 1 cm3 cubic shape). Before the stroke surgery, all monkeys were trained to successfully catch 15 pieces of apple within 5 min using their both limbs. When food catching task was performed, monkey was restrained in a special monkey chair (as shown in Supplementary Movie 1 and 2), their left limbs could only reach the left foot container, and their right limbs could only reach the right foot container. Then, the numbers of successful catching and total catching (times of touching the food container, including successful and failed catching) using both limbs were recorded. The success rate of food catching was calculated as: times of successful catching/times of total catching×100%.
Evaluation of brain infarct size by MRI
For the imaging study, after the animals had been anesthetized by intramuscular administration of ketamine hydrochloride (10 mg kg−1) containing atropine sulfate (0.05 mg kg−1), MRI imaging was performed from the frontal to the posterior brain regions employing an MRI scanner (Siemens-MR-Verio 3.0, Siemens, Germany) with Tof-3D-multi-slab, T1-f12d-tra and T2-tse-tra scan. The sequence of Tof-3D-multi-slab was the cerebrovascular dynamic scanning for determining cerebral blood vessel integrity and patency. The sequence of T1-f12d-tra scan determined the gross anatomy map of animal brain tissue. The sequence of T2-tse-tra scan detected the infarct area of animal brain tissue, in which the infarct area showed high signal and normal brain tissue showed low signal. Transverse images with a 4-mm slice thickness were obtained from each T2-MRI sequence. The infarct size on T2-MRI imaging of the ischemic side was defined as the white portions and was compared to those on the contralateral side using Adobe Photoshop 7.0 software (San Jose, CA, USA).
Tissue preparation
At termination of the experiment, monkeys were euthanized with a sodium pentobarbital overdose (150 mg kg−1, i.v.). The brain was removed and 4 mm brain slices (total 12 pieces from each brain) were sectioned coronally on ice with the brain immersed in cold saline (0.9%) for less than 15 min. For every brain slice within the areas of visible infarct, the normal cerebral cortex of the contralateral cerebral hemisphere and the peri-infarct area (adjacent to infarcted tissue) of the cerebral cortex from the ipsilateral hemisphere were dissociated for protein extraction and proteomics analysis. The tissues were immediately frozen in liquid nitrogen, and stored at −80 °C. The other brain slices were immersed in 4% paraformaldehyde to fix for 48 h. The tissues were then processed for histopathologic evaluation.
Pharmacokinetics
Six Cynomolgus macaque monkeys were used for pharmacokinetic study. TBN were given intravenously at doses of 30 and 90 mg kg−1, 3 monkeys for each dose. Blood samples were collected through the hind limb vein using 5 mL heparinized syringes at 0, 2.5, 5, 10, 20, 40, 80 and 160 min post drug administration. Blood samples were centrifuged at 3,200 g for 10 min at 4 °C. Following centrifugation, plasma was collected and frozen until further analysis.
After the animals were anesthetized by intramuscular administration of ketamine hydrochloride (10 mg kg−1) containing atropine sulfate (0.05 mg kg−1), cerebrospinal fluid samples were collected from the cisterna magna at 15, 45, 60 and 120 min post drug administration. Drug concentrations were determined by HPLC analysis.
Primary cortical neurons culture and neuroprotective assays
We prepared cortical cultures from E18 rat embryos (Experimental Animal Center of Sun Yat-Sen University) as previously described47. Cortical neurons were grown at 37 °C in a humidified incubator with 5% CO2/95% air atmosphere. After 4–6 h, unattached cells and debris were removed by replacing the initial medium with fresh neurobasal medium containing B27 supplements. Subsequent partial medium replacement was carried out twice a week and cultures at 11 days were used for the experiments. TBN at indicated concentrations were applied 2 h before exposure to glutamate (200 μM), and the cells were cultured for another 24 h. Cell viability was assessed by 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay15.
The neuroprotective effect of TBN was further assessed by attenuation of LDH release on OGD-insulted cortical neurons. TBN at indicated concentrations were applied 2 h before exposure to OGD for 4 h, and the cells were cultured for another 24 h. LDH release assay was conducted according to our previous report48.
Isobaric tags for relative and absolute quantitation (iTRAQ)-based quantitative proteomic analyses
The details of iTRAQ-based quantitative proteomic analyses were described in Supplementary Methods, including proteomics sample preparation, iTRAQ labelling, liquid chromatography, mass spectrometry and data analysis, and iTRAQ ratio calculations and criteria for the selection of dysregulated proteins.
Western blotting
For protein extraction, tissues were extracted with RIPA lysis buffer containing 1% PMSF and 1% protease inhibitor cocktail on ice for 10 min. The lysates were centrifuged (12,500 × g, 20 min, 4 °C) and the supernatants were then collected and stored at –80 °C until use. Protein contents were assayed using a BCA protein quantification kit (Pierce, Rockford, IL, USA). Protein samples (30 μg) were resolved using SDS-PAGE and transferred to PVDF membranes. The immunoblots were analyzed with the appropriate primary antibodies (1:1000). Horseradish peroxidase-conjugated secondary antibodies (1:2500) were used to detect the proteins of interest through enhanced chemiluminescence.
Immunohistochemistry
The fixed brain slices were dehydrated with graded alcohol, embedded in paraffin wax, and coronally sectioned at 6 μM. Immunohistochemistry of sections was performed using antibodies against glial fibrillary acidic protein (GFAP, Cat.no. MAB360, Millipore) and neuronal nuclei (NeuN, Cat.no. MAB 377, Millipore) to evaluate the infiltration of inflammatory cells and viability of neurons. The positive cells were randomly counted in 5 areas per section and 3 sections of each monkey from 6 monkeys per group, and then averaged the data.
Data and statistical analyses
We carried out statistical analysis with Prism 6.0 software (GraphPad Software). Unless otherwise specified, all data were expressed as mean ± s.e.m, and multiple comparisons were assessed by one-way ANOVA followed by the Tukey’s post hoc test for significance. The significance level was set as P < 0.05.
Additional Information
How to cite this article: Zhang, Z. et al. Tetramethylpyrazine nitrone, a multifunctional neuroprotective agent for ischemic stroke therapy. Sci. Rep. 6, 37148; doi: 10.1038/srep37148 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
O’Collins, V. E. et al. 1,026 experimental treatments in acute stroke. Ann Neurol 59, 467–477 (2006).
Cook, D. J., Teves, L. & Tymianski, M. Treatment of stroke with a PSD-95 inhibitor in the gyrencephalic primate brain. Nature 483, 213–217 (2012).
Papadakis, M. et al. Tsc1 (hamartin) confers neuroprotection against ischemia by inducing autophagy. Nat Med 19, 351–357 (2013).
Lees, K. R. et al. NXY-059 for acute ischemic stroke. N Engl J Med 354, 588–600 (2006).
Shuaib, A. et al. NXY-059 for the treatment of acute ischemic stroke. N Engl J Med 357, 562–571(2007).
Feuerstein, G. Z. et al. Missing steps in the STAIR case: a Translational Medicine perspective on the development of NXY-059 for treatment of acute ischemic stroke. J Cereb Blood Flow Metab 28, 217–219 (2008).
Ginsberg, M. D. Life after cerovive: a personal perspective on ischemic neuroprotection in the post-NXY-059 era. Stroke 38, 1967–1972 (2007).
Kuroda, S., Tsuchidate, R., Smith, M. L., Maples, K. R. & Siesjo, B. K. Neuroprotective effects of a novel nitrone, NXY-059, after transient focal cerebral ischemia in the rat. J Cereb Blood Flow Metab 19, 778–787 (1999).
Effect of a novel free radical scavenger, edaravone (MCI-186), on acute brain infarction. Randomized, placebo-controlled, double-blind study at multicenters. Cerebrovasc Dis 15, 222–229 (2003).
Higashi, Y., Jitsuiki, D., Chayama, K. & Yoshizumi, M. Edaravone (3-methyl-1-phenyl-2-pyrazolin-5-one), a novel free radical scavenger, for treatment of cardiovascular diseases. Recent Pat Cardiovasc Drug Discov 1, 85–93 (2006).
Chen, K. J. & Chen, K. Ischemic stroke treated with Ligusticum chuanxiong. Chin Med J (Engl) 105, 870–873 (1992).
Gao, H. J. et al. Ligustrazine monomer against cerebral ischemia/reperfusion injury. Neural Regen Res 10, 832–840 (2015).
Floyd, R. A. Nitrones as therapeutics in age-related diseases. Aging Cell 5, 51–57 (2006).
Hensley, K. et al. Nitrone-based free radical traps as neuroprotective agents in cerebral ischaemia and other pathologies. Int Rev Neurobiol 40, 299–317 (1997).
Sun, Y. et al. Antioxidative and thrombolytic TMP nitrone for treatment of ischemic stroke. Bioorg Med Chem 16, 8868–8874 (2008).
Sun, Y. et al. Therapeutic effects of tetramethylpyrazine nitrone in rat ischemic stroke models. J Neurosci Res 90, 1662–1669 (2012).
Roitberg, B. et al. Chronic ischemic stroke model in cynomolgus monkeys: behavioral, neuroimaging and anatomical study. Neurol Res 25, 68–78 (2003).
Borgens, R. B. & Liu-Snyder, P. Understanding secondary injury. Q Rev Biol 87, 89–127 (2012).
Dirnagl, U., Iadecola, C. & Moskowitz, M. A. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22, 391–397 (1999).
Font, M. A., Arboix, A. & Krupinski, J. Angiogenesis, neurogenesis and neuroplasticity in ischemic stroke. Curr Cardiol Rev 6, 238–244 (2010).
Tobin, M. K. et al. Neurogenesis and inflammation after ischemic stroke: what is known and where we go from here. J Cereb Blood Flow Metab 34, 1573–1584 (2014).
Xiong, Y., Mahmood, A. & Chopp, M. Angiogenesis, neurogenesis and brain recovery of function following injury. Curr Opin Investig Drugs 11, 298–308 (2010).
Jiang, S. X., Slinn, J., Aylsworth, A. & Hou, S. T. Vimentin participates in microglia activation and neurotoxicity in cerebral ischemia. J Neurochem 122, 764–774 (2012).
Jing, L., He, Q., Zhang, J. Z. & Li, P. A. Temporal profile of astrocytes and changes of oligodendrocyte-based myelin following middle cerebral artery occlusion in diabetic and non-diabetic rats. Int J Biol Sci 9, 190–199 (2013).
Vogel, U. S. & Thompson, R. J. Molecular structure, localization, and possible functions of the myelin-associated enzyme 2′,3′-cyclic nucleotide 3′-phosphodiesterase. J Neurochem 50, 1667–1677 (1988).
Inuzuka, T. et al. Changes in the concentrations of cerebral proteins following occlusion of the middle cerebral artery in rats. Stroke 21, 917–922 (1990).
da Silva, P. B., Antunes, F. N., Graef, P., Cechetti, F. & Pagnussat Ade, S. Strength training associated with task-oriented training to enhance upper-limb motor function in elderly patients with mild impairment after stroke: a randomized controlled trial. Am J Phys Med Rehabil 94, 11–19 (2015).
Tsai, T. H. & Liang, C. Pharmacokinetics of tetramethylpyrazine in rat blood and brain using microdialysis. Int J Pharm 216, 61–66 (2001).
Kostandy, B. B. The role of glutamate in neuronal ischemic injury: the role of spark in fire. Neurol Sci 33, 223–237 (2012).
Ni, X. J., Liu, S. N. & Guo, X. F. Meta analysis on efficacy and safety of ligustrazine injection for cerebral stroke. Chinese Journal of Experimental Traditional Medical Formulae 19, 325–332 (2013).
Scornavacca, G. et al. Proteomic analysis of mouse brain cortex identifies metabolic down-regulation as a general feature of ischemic pre-conditioning. J Neurochem 122, 1219–1229 (2012).
Yenari, M., Kitagawa, K., Lyden, P. & Perez-Pinzon, M. Metabolic downregulation: a key to successful neuroprotection? Stroke 39, 2910–2917(2008).
Nave, K. A. Myelination and the trophic support of long axons. Nat Rev Neurosci 11, 275–283 (2010).
Konopaske, G. T. et al. Effect of chronic antipsychotic exposure on astrocyte and oligodendrocyte numbers in macaque monkeys. Biol Psychiatry 63, 759–765 (2008).
Ishibashi, T. et al. Astrocytes promote myelination in response to electrical impulses. Neuron 49, 823–832 (2006).
Gage, F. H. Mammalian neural stem cells. Science 287, 1433–1438 (2000).
van Praag, H. et al. Functional neurogenesis in the adult hippocampus. Nature 415, 1030–1034 (2002).
Zhao, C., Deng, W. & Gage, F. H. Mechanisms and functional implications of adult neurogenesis. Cell 132, 645–660 (2008).
Tian, Y. et al. Tetramethylpyrazine promotes proliferation and differentiation of neural stem cells from rat brain in hypoxic condition via mitogen-activated protein kinases pathway in vitro. Neurosci Lett 474, 26–31(2010).
Xiao, X. et al. Neuroprotection and enhanced neurogenesis by tetramethylpyrazine in adult rat brain after focal ischemia. Neurol Res 32, 547–555 (2010).
Kong, X. et al. Tetramethylpyrazine Promotes Migration of Neural Precursor Cells via Activating the Phosphatidylinositol 3-Kinase Pathway. Mol Neurobiol, doi: 10.1007/s12035-015-9551-1 (2015).
Arvidsson, A., Collin, T., Kirik, D., Kokaia, Z. & Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat Med 8, 963–970 (2002).
Magnusson, J. P. et al. A latent neurogenic program in astrocytes regulated by Notch signaling in the mouse. Science 346, 237–241(2014).
Bornstein, S. R. & Licinio, J. Improving the efficacy of translational medicine by optimally integrating health care, academia and industry. Nat Med 17, 1567–1569 (2011).
Endres, M. et al. Improving outcome after stroke: overcoming the translational roadblock. Cerebrovasc Dis 25, 268–278 (2008).
Kito, G. et al. Experimental thromboembolic stroke in cynomolgus monkey. J Neurosci Methods 105, 45–53 (2001).
Vogiatzi, T., Xilouri, M., Vekrellis, K. & Stefanis, L. Wild type alpha-synuclein is degraded by chaperone-mediated autophagy and macroautophagy in neuronal cells. J Biol Chem 283, 23542–23556 (2008).
Zhou, X. et al. Protective Effect of Edaravone in Primary Cerebellar Granule Neurons against Iodoacetic Acid-Induced Cell Injury. Oxid Med Cell Longev 2015, 606981 (2015).
Acknowledgements
This work was partially supported by grants from the National Innovative Drug Program of China (2009ZX09103-031; 2012ZX09103-055), Scientific Projects of Guangdong Province (2013A022100030; 2014A030310174; 2015A020211019; 2015B020232011) and the National Science Foundation of China (30973618), as well as the Science and Technology Program of Guangzhou for Key Laboratory (151800010).
Author information
Authors and Affiliations
Contributions
Y.Q.W. designed and supervised the project. I.Q.C. supervised the proteomics experiments. Z.J.Z., G.X.Z. and E.S. performed the monkey stroke experiments, collected and analyzed the data. S.S.W.S., H.C.H.L., Q.Q., G.H.L., M.K.W.S., S.M.Y.L. and I.K.C. performed the proteomics experiments, collected and analyzed the data. Y.W.S. and P.Y. performed the cell culture, pharmacokinetics and toxicological experiments, collected and analyzed the data. Z.J.Z. and S.S.W.S. drafted the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, Z., Zhang, G., Sun, Y. et al. Tetramethylpyrazine nitrone, a multifunctional neuroprotective agent for ischemic stroke therapy. Sci Rep 6, 37148 (2016). https://doi.org/10.1038/srep37148
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37148
This article is cited by
-
Protective Mechanism and Treatment of Neurogenesis in Cerebral Ischemia
Neurochemical Research (2020)
-
Proteomic-Based Approaches for the Study of Ischemic Stroke
Translational Stroke Research (2019)
-
JM-20 Treatment After MCAO Reduced Astrocyte Reactivity and Neuronal Death on Peri-infarct Regions of the Rat Brain
Molecular Neurobiology (2019)
-
Tetramethylpyrazine Nitrone Reduces Oxidative Stress to Alleviate Cerebral Vasospasm in Experimental Subarachnoid Hemorrhage Models
NeuroMolecular Medicine (2019)
-
Neuroprotective Effect and Mechanism of Action of Tetramethylpyrazine Nitrone for Ischemic Stroke Therapy
NeuroMolecular Medicine (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.