Abstract
Various per- and poly-fluorinated compounds (PFCs) were first systematically investigated in North Canal Basin, Beijing, China. A total of 68 surface water samples were collected from North Canal Basin, Beijing, at high spatial resolution. The seasonal disparity was compared and associated with source variation. PFCs concentrations in low-water period ranged from 26 to 207 ng/L, and significantly declined levels were found in high-water period. The individual component proportions among different sites varied less in high-water period, when runoff played a role in mixing and diluting PFCs. A methodology combined with principal component analysis (PCA), heat map-hierarchical cluster analysis (HM-HCA), and correlation analysis were introduced to discriminate sources of PFCs in surface water. The statistical results agreed with each other, and daily domestic consumption, fire-fighting products and related industries were identified as sources of PFCs in this region. In addition, two composition ratios were proposed through the methodology to distinguish the impact of nonpoint source, and the outcome demonstrates that great disparities exist in compositional profiles between nonpoint source and others. Overall, the results showed that this comprehensive analysis method has great potential for source apportionment in surface water and other environmental compartments.
Similar content being viewed by others
Introduction
Per- and poly-fluorinated compounds (PFCs), including perfluorinated carboxylates (PFCAs), sulfonates (PFSAs) and various precursors, have been applied to industrial and household products for decades1,2. PFCs have been widely used in industry as surfactants, lubricants, fire retardants, paper and textile treatment3,4,5. They are water-soluble, bioaccumulative and persistent in the environment6. PFCs have been ubiquitously detected in abiotic and biotic media, including surface and ground water, sediments, aquatic organisms, human blood and breast milk7,8,9,10. In addition, some PFCs have been found to have acute and subchronic toxicity as peroxisome proliferators, gap junction and intercellular communication inhibitor3,11,12,13,14. Thus, long time exposure to PFCs would pose potential risk to human being and ecosystem15,16.
In recent years, much attention has been paid to PFCs in aqueous environments. PFCs pollution in surface water has been reported globally17,18,19,20,21. Generally, perfluorooctanoic acid (PFOA) and Perfluorooctane sulfonate (PFOS) were the most detected and concentrated perfluorinated compounds in surface water. The concentration of each chemical could reach the level of several hundred ng/L in river basin5,22,23,24,25. The contamination level often exceeds the recommended standards of PFCs for drinking water proposed by state of New Jersey (0.04 μg/L for PFOA26) and the German Drinking Water Commission (0.1 μg/L for PFOA plus PFOS27). Past studies showed that occurrence of PFCs was strongly correlated with population density and highly influenced by urban activities28,29,30. It is also interesting to note that higher PFCs loads in surface water have been observed during wet-weather31,32.
Generally, PFCs input into the aqueous environment occurs via four routes: (1) discharge from wastewater treatment plants (WWTP); (2) influx of runoff contaminated by nonpoint source; (3) atmospheric deposition of oxidized precursors; (4) seepage and illegal discharge. Significant variances were observed between the profiles of different sources33,34,35. Although quantification of PFCs in water environment has been successfully explored and applied, source identification can still be a challenging task, because PFCs were widely used and they were dispersed and underwent complex transformation. In some cases, it was handled by analyzing specific composition of all possible sources29,33. In other complicated environment system, the sources of PFCs were able to be traced by studying their spatial and temporal distribution and the ratios of discrete components. Xu et al.9 employed principal component analysis (PCA) to investigate the possible sources for sediments. Nguyen et al.27 used loading ratio method to evaluate the source of non-/point sources. Cluster analysis can help to differentiate contributions of each sources30. However, exploited separately, these models are not enough to provide accurate and comprehensive information of source apportionment.
Beijing is a very typical monsoon region. The maximum and minimum rainfall occurred in July and December respectively, and the former is about 100 times as much as the later. The contamination status and seasonal variation of various PFCs in Beijing, one of the most densely populated regions in the world, has not been properly studied. This study is aimed to fill in the data gap of PFCs contamination status of North Canal basin in both high-water period (HWP) and low-water period (LWP), to statistically analyze the spatial distribution pattern and seasonal variation, and to develop a methodology in order to identify sources. The method is based on multivariate data analysis, including PCA, heat map-hierarchical cluster analysis (HM-HCA), and correlation analysis, which make it easier to analyze raw data of environmental monitoring, and to track down the sources of PFCs. Besides, widely application of chlorinated polyfluorinated ether sulfonate (locally called F-53B, C8ClF16O4SK), a Chinese PFOS alternative, has been encouraged by Chinese government, as an important part of implementing Stockholm Convention. To the best of our knowledge, this is the first systematic investigation of F-53B together with various PFCs in a regional environment, after Wang et al.36 first reported its usage in China.
Results and Discussion
Concentration and composition profile of PFCs in the surface water
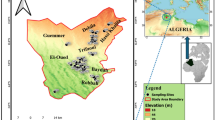
The concentrations of ∑PFCs (the sum of 9 measured PFCs concentrations) in North Canal and its tributaries ranged from 4.49 to 40.12 ng·L−1 in the HWP, and 26.11 to 207.56 ng·L−1 in LWP. The highest ∑PFCs concentration of LWP was observed in W8, where several tributaries join the main stream. The lowest total concentration was found in A1 for both HWP and LWP. According to Figs 1 and 2 and Table S1, in both two samplings, the contamination status of Xiaozhong River (A1, A2), Yunchaojian River, (C1, C2) are lower than the North Canal, which can be explained by running through the less developed areas. Nevertheless, flowing past the highly populated and industrialized areas may account for the high ∑PFCs loadings of Qing River (Q1 to Q4) and Liangshui River (L1 to L5)37.
Sampling sites in North Canal and its tributaries, northeastern Beijing.
(We created this figure using ArcMap 10.2, http://www.esrichina.com.cn/).
The average concentrations of individual PFCs were exhibited in Tables S4 and 5, PFOA showed the highest average concentration (2.89 ng·L−1) in the HWP, followed by PFBS (2.61 ng·L−1), PFOS (1.94 ng·L−1), while PFOS, PFOA and PFBS predominated in the LWP, with the average concentrations of 32.17 ng·L−1, 16.82 ng·L−1, and 12.57 ng·L−1, respectively. Together the three compounds took up 16.98–80.41% (median of 64.34%) and 40.95–92.56% (78.85%) of total PFCs concentration in HWP and LWP, respectively. Most of the compounds had been identified and peaked in Liangshui River (the biggest municipal drainage channel of Beijing, Fig. 1). The consequence is verified by a previous report that seven discharge outlets along the Liangshui River discharged 60,000 tons of untreated wastewater into the river per day38.
In LWP, the compounds were spotty distributed (Figs 2 and 3), suggesting the influence of point discharges30,39, while in HWP, the distribution was less fluctuated (Supplementary Tables S4 and 5, Fig. S2), whereas an overall concentration decline was clearly observed. Such circumstance might result from the mixing and diluting effects of large amount runoff during continuous wet weather27,31,32,35. Particularly, in HWP, the percentage of PFOS was significantly decreased, while the share of several other PFCs (PFBA, PFPeA, PFHxA, PFHpA, PFOA and F-53B, Fig. 2) were clearly elevated. The comparison suggested that the surface runoff source might have merged multiple point sources27. It is interesting to note that the PFOS decreased in both concentration and proportion could be explained by its almost absence from precipitation and runoff water.
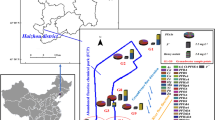
PCA and HM-HCA results of PFCs in two water periods.
(A) HWP Component Plot in Rotated Space; (B) LWP Component Plot in Rotated Space; (C) two-dimention HM-HCA of HWP, for 31 sites (right of the map) and 9 PFCs (bottom of the map); (D) HM-HCA for LWP, for 34 sites and 9 PFCs. In Heat map, the color gradient represents the concentration intensity from the lowest (−3) to the highest (3). (More interpretation of the references to HM-HCA and PCA models were listed in the supplementary information).
Few previous studies have reported the occurrence of F-53B. This compound was mainly used as mist suppressant by chrome plating industry and it is also demanded as a PFOS alternative in China36. In this study, F-53B was detected in all sampling sites. It was unevenly distributed; Q-3 and W(M)-8 were found to be the most polluted sites in HWP and LWP, with concentrations of 29.42 (ng/L) and 8.14 (ng/L), respectively. This might be associated with nearby point source (Fig. 1). During LWP, Liangshui River was the most contaminated tributaries, whereas, in the HWP, there was no evident disparity in contamination status. Moreover, compared with PFOS, the observed concentrations of F-53B also dropped dramatically in HWP, whereas, the proportion remained at the same level, suggesting the PFCs compositional differences in runoff. (More information concerning F-53B was listed in the Supplementary Information).
Source apportionment
In this part, three statistical approaches were employed to analyze the contributions of possible sources to PFC pollution in North Canal Catchment.
Source Categories
PCA and HM-HCA were applied to aggregating the distributing pattern of PFCs in order to investigate the possible sources categories. These methods have been proved effective and widely used in source apportionment of environmental pollutants9,30,40.
The results of PCA (Fig. 3(A,B)) states that in both two samplings, concentrations of PFCAs (PFBA, PFPeA, PFHxA, PFHpA and PFNA (the clustered group)) were correlated, while PFOS, PFBS or F-53B were independent from other compounds. According to the reported source marker4,23, this environmental occurrence pattern should be associated with the source categories. The clustered compounds were widely used in industrial processes, however, due to the low concentration, and extensive and similar distribution patterns, they were more likely derived from nonpoint inputs. This is consisted with the previous studies that consumer products (e.g. textile, paper and food/pharmaceutical package) were the major PFCAs sources4,29,30. Thus, PC1 in both periods could be identified as the household emission source. PFOS and F-53B containing chemicals were used in galvanizing, nickel, chromium plating and anodizing industry36,41,42, therefore, PC 2 can be pinpointed as relevant industry9. This was proved by previous researches that highly concentrated PFOS and other PFSAs were observed in the effluents of metal industries impacted WWTP4,30,43. Moreover, although PC 3 in both periods was of minor importance, the distribution pattern of indicative compounds reflected the influence of certain sources. PFBS was used in aqueous film-forming foam (AFFF) to replace PFOS41 and it was also observed in the impacted surface water29,44,45, besides, PFBS in this study also presented in similar pattern (e.g. W5–7, downstream of the Airport). Accordingly, PC 3 can be identified as AFFF using units (e.g. airport and fire station). It is interesting to note that PFOA was not in the same group in HWP and LWP. This indicates that PFOA had sources other than domestic input, which were most likely to be food-packaging and paper grease-proofing treatments (Fig. 1)30.
Geographic distribution of Sources
In cluster analysis, surface water contamination with similar source categories would be included in one group and the major pollutants would be displayed on the heat map9,30. As shown in Fig. 3(C,D), in both periods, the midstream of North Canal was clustered with Qinghe River and PFBS was the main chemical in the region, which was consisted with the geographic distribution of an airport and several fire station with large AFFF consumption (Fig. 1). In HWP (Fig. 3(C)), downstream of North Canal was grouped with all the tributaries but Qinghe River, which was otherwise clustered with the mid- and upstream of North canal. Besides, Compared with situations in LWP, a wider extension and jointed area was clustered as groups, and on the heat map, red color was less concentrated. This is another evidence to prove the mixing and diluting effect of runoff. In LWP (Fig. 3(D)), PFOS contamination was scattered and it was highlighted on the heat map. As expected, the result conformed to the geographic distribution of relevant industries, indicating surface water was heavily influenced by point emissions.
According to Beijing Statistical Yearbook46, 150,713 ton wastewater was generated in 2014 and entered natural surface water with or without proper treatment, and over 90% of which was discharged into North Canal. Besides, there were 3, 686 industrial enterprises above designed size (the main business income >RMB 20,000,000), among which over 40% were PFCs related industries and together they account for 17% total industrial output value (Supplementary Table S8).
Impact of nonpoint source
In several previous studies, runoff water has been regarded as an important source of PFCs in surface water, and it was reported that precipitation contributed over 50% of the overall loads in some areas33,47. Nevertheless, these studies only considered the first flush of precipitation, which probably only caused minor runoff with very high pollutant loadings, but the effects of consecutive rainfall and large runoff were not taken into consideration. North Canal catchment is the largest drainage receptor of Beijing, therefore, its contamination status is heavily influenced by precipitation. In this study, we sampled the surface water right after consecutive storms. Figure 2 clearly reveals that PFCs concentrations in LWP were higher than those in HWP in almost all sampling sites. This circumstance indicates that the continuous rainfalls will bring rainwater with lower pollutant loading into the rivers, compared with the reported highly contaminated first flush, and hence, they played a dilution role27.
In order to discriminate the impact of runoff, the correlation among each compound was studied. Pearson correlation coefficients between individual PFCs are displayed in Fig. 4. PFBA, PFPeA, PFHxA, PFHpA and PFNA are more related to each other in HWP (Fig. 4(a)) than in LWP (Fig. 4(b)). The results are consistent with the outcomes of HM-HCA and PCA. These compounds are often found in the domestic discharges4,23,30, and also highly influenced by traffic34 and deposition of atmospherically transformed telomer alcohols48, and therefore, they are usually concentrated in runoff49,50. Therefore, in this case, these compounds are probably brought into surface water by runoff, and the distribution pattern variation between two periods may also be explained by elevated runoff input4.
Concentration ratios can be employed to identify sources of pollutants, because of their different composition and behaviors in various environments27,51,52,53. In this case, precipitation runoff is considered as an extra source in HWP, leading to strong dilution of PFOS. Therefore, two indicators are proposed as tracers to discriminate the impact of runoff on HWP’s water environments: F-53B/PFOS concentration ratio and (PFBA + PFPeA + PFHxA + PFHpA)/PFOS concentration ratio. Firstly, F-53B and PFOS have the same number of fluorinated carbons and they are derived from similar point sources35,36. Also, Fig. 4 revealed that F-53B have a very close correlation with PFOS (P < 0.01) in LWP samples, rather than in HWP. Such contrast indicates source change, and consequently, the concentrations ratio can possibly be used as a source tracer. Secondly, PFBA, PFPeA, PFHxA and PFHpA share close statistical correlations with each other in all the samples (Fig. 4). Besides, as mentioned before, they were found highly loaded in nonpoint source, which varied much from the emission regularity of the point-source compound (i.e. PFOS). Therefore, another PFCs source indicator can be drew from (PFBA + PFPeA + PFHxA + PFHpA)/PFOS concentration ratio.
F-53B/PFOS ratio in LWP ranged from 0.05 to 0.10 (25% percentile to 75% percentile, the same for other), and from 0.15 to 0.41 in HWP (Fig. 5(C)). (PFBA + PFPeA + PFHxA + PFHpA)/PFOS ratio ranged from 0.21 to 1.13 and 1.41 to 2.61, respectively (Fig. 5(B)). Given the disparity of pollution status and lack of enough validation, these measure might not be specific enough to absolutely identify nonpoint source. Nevertheless, clear difference has been shown and these ratios are potentially useful for the evaluation of the contributions from point source and non-point source to PFCs in multiple water environments. Thus further investigations of the various sources are needed to validate the proposed indicators.
PFOA/PFOS was reported typically greater than 1.1 in the surface water27. Nevertheless, this ratio is not suitable for the studied area, especially in dry weather (median: 0.53 for LWP), probably because China is the only one where PFOS is still being manufactured. Conversely, it is interesting to note that this ratio was significantly elevated to 1.70 (median) in HWP (Fig. 5(A)), and the rational explanation is the input from runoff. This theory is supported by several previous studies. The ratio in runoff is reported around 9, which could make contribution to the increment of this ratio in HWP35; a study conducted in Tianjin, a metropolis adjacent to Beijing, states that the frequency and content of PFOS is much less than most PFCAs, throughout all the seasons54.
Besides runoff, previous studies have also mentioned several other weighty nonpoint sources for surface water6,27,35,49,55. The ratio of PFHpA/PFOA (Fig. 5(D)) is regarded as an effective tracer of atmospheric deposition9,41,56. In this study, this ratio ranged from 0.03 to 0.34 with little variance between two sampling samplings, indicating the distribution of PFCs was not much affected by atmospheric deposition. Also, this ratio is used to measure the nearness to urbanized and industrialized areas33, which is consisted with the case in this study. PFCs precursors existed in wastewater, and they can be degraded/biodegraded into PFCs as they enter surface water30,35,56. PFCs loads had even been found increasing during wastewater treatment process43. It is reported that biodegradation of the same precursors produced mainly PFOA and secondarily PFNA35. In the present study, the ratio of PFNA/PFOA ranged from 0 to 0.18 for both two samplings. PFNA and PFOA had close correlations in HWP (p < 0.01) and LWP (p < 0.05), indicating the possibility that precursors degradation is another source.
Conclusion
This study is the first large-scale surface water investigation of PFCs and alternative F-53B occurrence in Beijing, the capital of China. It provides important data on the occurrence and distribution patterns of PFCs in different water periods. In North Canal basin, Liangshui River is the most polluted tributary in both high-water period and low-water period. The rivers flowing through the central city tends to show higher loadings than those running past rural area. The PFCs contamination of surface water is found to be dominated by PFOS, PFOA and PFBS all the time, which are considered to come mainly from relevant industrial discharges. In HWP, large amount of runoff mixes and dilutes PFCs concentration in surface water and it is identified as an input, because it obviously changes the PFCs compositional profile between different water periods. Moreover, source apportionment was achieved by investigating multivariate statistical techniques to distinguish the traits of pollution and determine the sources. The consequences are geographically consisted with known sources. Two new composition ratios are proposed as indicators to discriminate the influence nonpoint source. F-53B/PFOS <0.07 and (PFBA + PFPeA + PFHxA + PFHpA)/PFOS <0.62 indicate the impact of point source. But nonpoint source, specially runoff, comes to make a difference when the ratios >0.28 and >1.87, respectively. The application of the methodology combined with PCA, HM-HCA, and correlation analysis have greatly reduced the complexity of the data, allowing to build up meaningful relation between sources and pollution status. This technique is believed to make a contribution to optimizing river/environment monitoring system.
To the best of our knowledge, this is the first systematic report of F-53B together with various PFCs in a regional environment in China, which was found heavily used in this region and its occurrence and distribution patterns varied much along seasonal change. Its occurrence is similar to the level of PFPeA, PFHxA, PFHpA and PFNA. In the LWP, F-53B have the same source category with PFOS; whereas, it can be brought into surface water by runoff in HWP, which is not considered as a major source for PFOS. F-53B was first synthesized in China and had been produced for more than 30 years; nevertheless, few studies had attached importance to this compound. Recently, as a part of the performance of Stockholm Convention, Chinese ministry of environmental protection have been encouraging the application of PFOS’s alternatives (including F-53B)57, so the larger usage and output of F-53B can be expected. Besides, owing to the lacking knowledge of its toxicity, fate and occurrence in various environments, sustained attention should be paid to the environmental impacts of its large output and usage.
Materials and Methods
Standards, Reagents and other materials
In this study, 11 PFCs were selected for target compounds, including 7 PFCAs (4A to 8A) and 4 PFSAs (4S, 6S, 8S and F-53B) (detailed in Supplementary Information). 13C4-PFBA, 13C5-PFPeA, 13C2-PFHxA, 13C4-PFHpA, 13C4-PFOA, 13C5-PFNA, 13C2-PFDA, 13C4-PFBS, 13C3-PFHxS(Na), 13C4-PFOS(Na) and 13C8-PFOS(Na) were purchased from Wellington Laboratories Inc. (ON, Canada) with purities of >98% as internal standards. F-53B was obtained from Shanghai Synica Co., Ltd. (Shanghai, China). Solvents for cleaning and extraction purposes were supplied by J.T. Baker (NJ, USA) and ultrapure water was used (18 MΩ·cm, Millipore, MA).
Sampling
Water samples were collected from North Canal Basin, which is located in the northeast Beijing with a catchment area of 4,293 km2. This is one of the most urbanized and industrialized area in the world, resided by 70% population and receiving 90% city drainage of Beijing58,59. As shown in Fig. 1, all the major WWTPs (Supplementary Table S9) in Beijing were located near the tributaries of North Canal, which bring in more than 3 million tons of wastewater per day59. Due to the insufficiency of wastewater collection and treatment system, the catchment was considered as the most polluted water environment in Beijing (Supplementary Table S1).
Total 34 Samples were collected along main stream (13 sites) and 6 tributaries (21 sites) in Beijing section of North Canal within one day (July and December of 2015 respectively, Fig. 1 and Table S1). The high-water period (HWP) in Beijing usually lasts from June to September, and the low-water period (LWP) lasts from October to March. According to Beijing water authority60, before the first sampling, several intense storms (detailed in Supplementary Information) hit Beijing city and caused major runoff. While during the second sampling, there was very little rain in Beijing. Therefore, runoff amount and flow rate were dramatically different between two samplings.
Water samples were taken 0.5 m under the surface and stored in 500 mL polypropylene (PP) bottles with screw caps (Vitlab, Grossostheim, Germany). PP is the most suitable material for anionic PFCs61. All samples were carried back to laboratory within 12 hours when they were stored in a refrigerator before further actions.
Sample pretreatment
Samples were filtered under vacuum through glass fiber filters (GF/F. 47 mm, Whatman, Kent, UK) to remove the suspended particulate matters (SPM). After filtration, PFCs samples were extracted and fractionated respectively. 25 ng internal standard was added and aged for 30 minutes. The PWAX-SPE cartridge (150 mg, 6 ml, AGELA Technologies, Tianjin, China) was conditioned by passing 4 mL 0.5% ammonia methanol solution, 4 mL methanol and 4 mL high purity water (HPW), respectively. The analytical sample was loaded onto the cartridge, and the aqueous eluate was collected at a rate of 3–5 mL/min. The SPE cartridge was then washed by 4 mL 25 mM sodium acetate solution, and dehydrated by pump air at 10 bar pressure. The analytes were eluted with 4 mL methanol and 4 mL 0.5% ammonia methanol solution successively. The elutes were concentrated by a stream of purified N2 to 250 μL. Prior to instrumental analysis, 250 μL ultrapure water and 25 ng injection standard, was added to each sample. Samples were finally filtered by 0.22 um microfiltration membrane (Pall Inc., Shanghai, China) and prepared to be analyzed.
HPLC-MS/MS analysis
Instrumental analysis was performed by a high-performance liquid chromatograph interfaced with a tandem mass spectrometer (HPLC-MS/MS) operated in electrospray negative ionization mode. Target compounds were separated on a Xbrige BEH C18 column (3.5 μm, 3.0 × 150 mm, Waters lnc., Shanghai, China) using an UltiMate 3000 HPLC (Dionex by Thermo Fisher Scientific Inc., Massachusetts, USA) and detected by an API 3200 triple quadrupole mass spectrometer (AB SCIEX, ON, Canada). The mobile phase flowed at a rate of 0.3 mL/min, consisting of 2 mM ammonium acetate (A) and methanol (B). The elution program was started with 40% B, and increased to 100% B from 1 min to 7 min. After another 3.5 min, it returned to 40% B and kept for 3 min. Target compounds were measured by MS/MS in multiple reaction monitoring (MRM) mode. Ion transitions monitored were, m/z 498.8 > 98.9(80) for PFOS, m/z 412.8 > 369.0 for PFOA, 502.7 > 98.9 for 13C4-PFOS and 416.7 > 269.9 for 13C4-PFOA (other substances were discussed in Supplementary Information).
Statistical analysis and data processing
In this study, statistical analyses were carried out by using IBM SPSS Statistics 21 (IBM Lnc., New York, USA), Origin 8 (OriginLab Corp., Massachusetts, USA) and R (the R foundation., Vienna, Austria). Values below detection limits were assigned as one-half of the limits, prior to statistical analysis. Two compounds (PFHxS, PFDA) were not quantifiable at all the sites. Therefore, they were not taken into further discussion or the following sections. More details were given in the Supplementary Information.
QA/QC
A strict quality control regime was operated during the experiment. Field blanks, transport blanks and solvent blanks were conducted with every sample set. PFC quantification was based on the calibration curves of which the correlation coefficients were all higher than 0.99. The limit of detection (LOD) and limit of quantification (LOQ) were defined as the peak of analyte that yield a signal-to-noise (S/N) ratio of 3:1 and 10:1, respectively. The procedure recoveries ranged from 83% to 133% at moderate concentration (5 ng standard added), and 62% to 126% at high concentration (20 ng). Detailed QA/QC measurements of PFCs in water samples were given in Supplementary Information.
Additional Information
How to cite this article: Zhang, Y.-Z. et al. Occurrence and source apportionment of Per- and poly-fluorinated compounds (PFCs) in North Canal Basin, Beijing. Sci. Rep. 6, 36683; doi: 10.1038/srep36683 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
OECD. Hazard assessment of perfluorooctane sulfonate (PFOS) and its salts: ENV/JM/RD (2002) 17/FINAL. Organisation for Economic Co-operation and Development Network http://www.oecd.org/chemicalsafety/risk-assessment/2382880.pdf. (Accessed: 27th December 2015) (2002).
Olsen, G. W. et al. Historical comparison of perfluorooctanesulfonate, perfluorooctanoate, and other fluorochemicals in human blood. Environ Health Persp. 113, 539–545 (2005).
Lau, C., Butenhoff, J. L. & Rogers, J. M. The developmental toxicity of perfluoroalkyl acids and their derivatives. Toxicol Appl Pharm. 198, 231–241 (2004).
Clara, M., Scheffknecht, C., Scharf, S., Weiss, S. & Gans, O. Emissions of perfluorinated alkylated substances (PFAS) from point sources-identification of relevant branches. Water Science & Technology 58, 59–66 (2008).
Castiglioni, S. et al. Sources and fate of perfluorinated compounds in the aqueous environment and in drinking water of a highly urbanized and industrialized area in Italy. J Hazard Mater 282, 51–60 (2015).
Björklund, J. A., Thuresson, K. & De Wit, C. A. Perfluoroalkyl compounds (PFCs) in indoor dust: Concentrations, human exposure estimates, and sources. Environ Sci Technol. 43, 2276–2281 (2009).
Yamashita, N. et al. A global survey of perfluorinated acids in oceans. Mar Pollut Bull. 51, 658–668 (2005).
Kärrman, A. et al. Exposure of perfluorinated chemicals through lactation: Levels of matched human milk and serum and a temporal trend, 1996–2004, in sweden. Environ Health Persp. 115, 226–230 (2007).
Xu, J. et al. Source apportionment of perfluorinated compounds (PFCs) in sediments: Using three multivariate factor analysis receptor models. J Hazard Mater 260, 483–488 (2013).
Midasch, O., Schettgen, T. & Angerer, J. Pilot study on the perfluorooctanesulfonate and perfluorooctanoate exposure of the German general population. Int J Hyg Envir Heal. 209, 489–496 (2006).
Berthiaume, J. & Wallace, K. B. Perfluorooctanoate, perflourooctanesulfonate, and N -ethyl perfluorooctanesulfonamido ethanol; Peroxisome proliferation and mitochondrial biogenesis. Toxicol Lett. 129, 23–32 (2002).
Lau, C. et al. Perfluoroalkyl acids: A review of monitoring and toxicological findings. Toxicol Sci. 99, 366–394 (2007).
Upham, B. L., Deocampo, N. D., Wurl, B. & Trosko, J. E. Inhibition of gap junctional intercellular communication by perfluorinated fatty acids is dependent on the chain length of the fluorinated tail. Int J Cancer 78, 491–495 (1998).
Yan, S. et al. Perfluorooctanoic acid exposure for 28 days affects glucose homeostasis and induces insulin hypersensitivity in mice. Sci. Rep. 5 (2015).
Egeghy, P. P. & Lorber, M. An assessment of the exposure of Americans to perfluorooctane sulfonate: A comparison of estimated intake with values inferred from NHANES data. Journal of Exposure Science & Environmental Epidemiology. 21, 150–168 (2011).
Stein, C. R., Mcgovern, K. J., Pajak, A. M., Maglione, P. J. & Wolff, M. S. Perfluoroalkyl and Polyfluoroalkyl Substances and Indicators of Immune Function in Children Aged 12–19 years: National Health and Nutrition Examination Survey. Pediatr Res. 183, 147–162 (2015).
Taniyasu, S., Kannan, K., Horii, Y., Hanari, N. & Yamashita, N. A survey of perfluorooctane sulfonate and related perfluorinated organic compounds in water, fish, birds, and humans from Japan. Environ Sci Technol. 37, 2634–2639 (2003).
Boulanger, B., Vargo, J., Schnoor, J. L. & Hornbuckle, K. C. Detection of perfluorooctane surfactants in Great Lakes water. Environ Sci Technol. 38, 4064–4070 (2004).
So, M. K. et al. Perfluorinated compounds in coastal waters of Hong Kong, South China, and Korea. Environ Sci Technol. 38, 4056–4063 (2004).
Mclachlan, M. S., Holmström, K. E., Margot Reth, A. & Berger, U. Riverine Discharge of Perfluorinated Carboxylates from the European Continent. Environ Sci Technol. 41, 7260–7265 (2007).
Xie, Z. et al. Neutral Poly-/perfluoroalkyl Substances in Air and Snow from the Arctic. Sci. Rep. 5 (2015).
Pan, C. G. et al. Contamination profiles of perfluoroalkyl substances in five typical rivers of the Pearl River Delta region, South China. Chemosphere. 114, 16–25 (2014).
Wang, P. et al. Occurrence and transport of 17 perfluoroalkyl acids in 12 coastal rivers in south Bohai coastal region of China with concentrated fluoropolymer facilities. Environ Pollut. 190, 115–122 (2014).
Niisoe, T. et al. Perfluorinated carboxylic acids discharged from the Yodo River Basin, Japan. Chemosphere. 138, 81–88 (2015).
Ahrens, L. et al. Distribution of polyfluoroalkyl compounds in water, suspended particulate matter and sediment from Tokyo Bay, Japan. Chemosphere. 79, 266–272 (2010).
Post, G. Guidance for PFOA in Drinking Water at Pennsgrove Water Supply Company. Pennsgrove Water Supply Company Network http://www.nj.gov/dep/watersupply/pdf/pfoa_dwguidance.pdf. (Accessed: 28th December 2015) (2013).
Nguyen, V. T., Reinhard, M. & Karina, G. Y. Occurrence and source characterization of perfluorochemicals in an urban watershed. Chemosphere. 82, 1277–1285 (2011).
Murakami, M. et al. Occurrence and sources of perfluorinated surfactants in rivers in Japan. Environ Sci Technol. 42, 6566–6572 (2008).
Müller, C. E., Gerecke, A. C., Alder, A. C., Scheringer, M. & Hungerbühler, K. Identification of perfluoroalkyl acid sources in Swiss surface waters with the help of the artificial sweetener acesulfame. Environ Pollut. 159, 1419–1426 (2011).
Xiao, F., Halbach, T. R., Simcik, M. F. & Gulliver, J. S. Input characterization of perfluoroalkyl substances in wastewater treatment plants: Source discrimination by exploratory data analysis. Water Res. 46, 3101–3109 (2012).
Lin, Y. C., Panchangam, S. C. & Lo, C. C. The impact of semiconductor, electronics and optoelectronic industries on downstream perfluorinated chemical contamination in Taiwanese rivers. Environ Pollut. 157, 1365–1372 (2009).
Labadie, P. & Chevreuil, M. Biogeochemical dynamics of perfluorinated alkyl acids and sulfonates in the River Seine (Paris, France) under contrasting hydrological conditions. Environ Pollut. 159, 3634–3639 (2011).
Kim, S. K. & Kannan, K. Perfluorinated acids in air, rain, snow, surface runoff, and lakes: Relative importance of pathways to contamination of urban lakes. Environ Sci Technol. 41, 8328–8334 (2007).
Murakami, M. & Takada, H. Perfluorinated surfactants (PFSs) in size-fractionated street dust in Tokyo. Chemosphere. 73, 1172–1177 (2008).
Murakami, M., Shinohara, H. & Takada, H. Evaluation of wastewater and street runoff as sources of perfluorinated surfactants (PFSs). Chemosphere. 74, 487–493 (2009).
Wang, S. et al. First report of a Chinese PFOS alternative overlooked for 30 years: Its toxicity, persistence, and presence in the environment. Environ Sci Technol. 47, 10163–10170 (2013).
So, M. K. et al. Perfluorinated compounds in the pearl river and yangtze river of china. Chemosphere. 68, 2085–2095 (2007).
Wang, B. et al. Linking the environmental loads to the fate of PPCPs in Beijing: Considering both the treated and untreated wastewater sources. Environ Pollut. 202, 153–159 (2015).
Ahrens, L., Felizeter, S., Sturm, R., Xie, Z. & Ebinghaus, R. Polyfluorinated compounds in waste water treatment plant effluents and surface waters along the River Elbe, Germany. Mar Pollut Bull. 58, 1326–1333 (2009).
Qi, Y. et al. Spatial distribution and source apportionment of PFASs in surface sediments from five lake regions, China. Sci. Rep. 6, (2016).
Wang, Z., Cousins, I. T., Scheringer, M. & Hungerbühler, K. Fluorinated alternatives to long-chain perfluoroalkyl carboxylic acids (PFCAs), perfluoroalkane sulfonic acids (PFSAs) and their potential precursors. Environ Int. 60, 242–248 (2013).
Xie, S. et al. Industrial source identification and emission estimation of perfluorooctane sulfonate in China. Environ Int. 52, 1–8 (2013).
Kim, S. K. et al. Wastewater treatment plants (WWTPs)-derived national discharge loads of perfluorinated compounds (PFCs). J Hazard Mater. 201–202, 82–91 (2012).
Anderson, R. H., Long, G. C., Porter, R. C. & Anderson, J. K. Occurrence of select perfluoroalkyl substances at U.S. Air Force aqueous film-forming foam release sites other than fire-training areas: Field-validation of critical fate and transport properties. Chemosphere. 150, 678–685 (2016).
Houtz, E. F., Sutton, R., Park, J. S. & Sedlak, M. Poly- and perfluoroalkyl substances in wastewater: Significance of unknown precursors, manufacturing shifts, and likely AFFF impacts. Water Res. 95, 142–149 (2016).
Beijing water authority. Rainfall regime statistics. Beijing Water Authority Network http://hdsw.bjhd.gov.cn/zxfw/bmcxfw/hdqyqcx/index_2.htm. (Accessed: 18th December 2015) (2015).
Scott, B. F. et al. Analysis for perfluorocarboxylic acids/anions in surface waters and precipitation using GC-MS and analysis of PFOA from large-volume samples. Environ Sci Technol. 40, 6405–6410 (2006).
Dinglasan-panlilio, M. J. & Mabury, S. A. Significant residual fluorinated alcohols present in various fluorinated materials. Environ Sci Technol. 40, 1447–1453 (2006).
Müller, C. E., Spiess, N., Gerecke, A. C., Scheringer, M. & Hungerbühler, K. Quantifying diffuse and point inputs of perfluoroalkyl acids in a nonindustrial river catchment. Environ Sci Technol. 45, 9901–9909 (2011).
Zushi, Y., Takeda, T. & Masunaga, S. Existence of nonpoint source of perfluorinated compounds and their loads in the Tsurumi River basin, Japan. Chemosphere. 71, 1566–1573 (2008).
Meyers, P. A. Preservation of elemental and isotopic source identification of sedimentary organic matter. Chem Geol. 114, 289–302 (1994).
Yunker, M. B. et al. PAHs in the Fraser River basin: A critical appraisal of PAH ratios as indicators of PAH source and composition. Org Geochem. 33, 489–515 (2002).
Armitage, J. M., Macleod, M. & Cousins, I. T. Comparative assessment of the global fate and transport pathways of long-chain perfluorocarboxylic acids (PFCAs) and perfluorocarboxylates (PFCs) emitted from direct sources. Environ Sci Technol. 43, 5830–5836 (2009).
Zhao, L., Zhou, M., Zhang, T. & Sun, H. Polyfluorinated and perfluorinated chemicals in precipitation and runoff from cities across eastern and central china. Archives of Environmental Contamination & Toxicology. 64, 198–207 (2013).
Clara, M. et al. Perfluorinated alkylated substances in the aquatic environment: An Austrian case study. Water Res. 43, 4760–4768 (2009).
Simcik, M. F. & Dorweiler, K. J. Ratio of perfluorochemical concentrations as a tracer of atmospheric deposition to surface waters. Environ Sci Technol. 39, 8678–8683 (2005).
Foreign Economic Cooperation Office. The project of the performance of Stockholm convention in China-the bidding announcement of testing the environmental characteristics of PFOS’s alternatives. Ministry of Environmental Protection Network http://www.mepfeco.org.cn/dtxx/cgzb/201605/t20160525_65806.html. (Assessed: 15th June 2016) (2016).
Jiang, N., Feng, S. Y., Zheng, Y. X., Meng, Q. Y. & Tang, Z. J. An analysis of problems and countermeasures for the surface water environment pollution of the north canal watershed in beijing. China Rural Water & Hydropower (2010).
Guo, J., Jing, H. W., Li, J. X. & Li, L. J. Surface water quality of Beiyun Rivers basin and the analysis of acting factors for the recent ten years. Environmental Science. 33, 1511–1518 (2012).
Beijing water authority. Rainfall regime statistics. Beijing Water Authority Network http://hdsw.bjhd.gov.cn/zxfw/bmcxfw/hdqyqcx/index_2.htm. (Accessed: 18th December 2015) (2015).
Dekleva, L. A. Adsorption/desorption of ammonium perfluorooctanoate to soil. OECD. 106, 13–17 (2003).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21577075), Tsinghua University Initiative Scientific Research Program (20131089193), and Program for Changjiang Scholars and Innovative Research Team in University (IRT1261).
Author information
Authors and Affiliations
Contributions
Z.-Y.Z. and B.W. designed the experiments; Z.-Y.Z., B.W., W.W. and W.-C.L. performed the experiments; Z.-Y.Z. and B.W. analyzed the data and wrote the manuscript. J.H., S.-B.D. and Y.-J.W. contributed to and edited the manuscript. G.Y. supervised the project. All the authors have reviewed this manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, YZ., Wang, B., Wang, W. et al. Occurrence and source apportionment of Per- and poly-fluorinated compounds (PFCs) in North Canal Basin, Beijing. Sci Rep 6, 36683 (2016). https://doi.org/10.1038/srep36683
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36683
This article is cited by
-
A study on the effectiveness of sodium selenite in treating cadmium and perfluoro octane sulfonic (PFOS) poisoned zebrafish (Danio rerio)
Biological Trace Element Research (2024)
-
The relationship between maternal perfluoroalkylated substances exposure and low birth weight of offspring: a systematic review and meta-analysis
Environmental Science and Pollution Research (2021)
-
The association between maternal perfluoroalkyl substances exposure and early attention deficit hyperactivity disorder in children: a systematic review and meta-analysis
Environmental Science and Pollution Research (2021)
-
Distribution of perfluorinated compounds in drinking water treatment plant and reductive degradation by UV/SO3 2− process
Environmental Science and Pollution Research (2018)
-
Mechanochemical mineralization of “very persistent” fluorocarbon surfactants ‒ 6:2 fluorotelomer sulfonate (6:2FTS) as an example
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.