Abstract
To investigate the applicability of compound specific stable carbon isotope ratios (δ13C) of organics in assessment of their photochemical aging in the atmosphere, batch UV irradiation experiments were conducted on two ambient (anthropogenic and biogenic) aerosol samples in aqueous phase for 0.5–120 h. The irradiated samples were analyzed for δ13C of diacids, glyoxylic acid (ωC2) and glyoxal. δ13C of diacids and related compounds became larger with irradiation time (i.e., aging), except for few cases. In general, δ13C of C2-C4 diacids showed an increasing trend with decreasing chain length. Based on δ13C of diacids and related compounds and their relations to their concentrations, we found that C2 and C3 are enriched with 13C during the photochemical decomposition and production from their higher homologues and oxoacids. Photochemical breakdown of higher (≥C3) to lower diacids is also important in the enrichment of 13C in C3-C9 diacids whereas their production from primary precursors causes depletion of 13C. In case of ωC2 and glyoxal, their photochemical production and further oxidation to highly oxygenated compounds both cause the enrichment of 13C. This study reveals that δ13C of diacids and related compounds can be used as a proxy to trace the aging of organic aerosols during long-range atmospheric transport.
Similar content being viewed by others
Introduction
Organic aerosols (OA), which represent a large fraction of fine particles1, play an important role in atmospheric processes and have serious impacts on the Earth’s climate system and human health2,3,4,5. OA are emitted directly into the atmosphere from various primary sources and secondarily produced in the atmosphere by oxidation of volatile organic compounds (VOCs) followed by condensation on pre-existing particles and/or nucleation1,6. The oxidation products are highly water-soluble and typically contain several types of functional groups including carboxylic acids, carbonyls, and hydroxyl groups as well as peroxides7,8. In the presence of moisture (i.e., clouds and fog), water-soluble compounds can partition into the aqueous phase and react with dissolved oxidants or undergo direct photolysis by actinic radiation9,10, thus OA are subjected to photochemical processing (aging) for several days until they are removed from the atmosphere through wet and/or dry deposition11,12,13. It is well known that aqueous phase oxidation processes can be very different from those in gas phase. Most of the recent studies have been focused on the ability of aqueous phase reactivity of selected organic compounds and other studies on the effects of aging of complex organic mixtures in aqueous aerosols9,10,11,12,13,14,15,16,17,18,19. However, identification of proxies, except for some molecular tracers9, for the assessment of aging of OA during long-range atmospheric transport remains unclear yet.
Low molecular weight dicarboxylic acids and related polar compounds are known as ubiquitous in atmospheric aerosols and atmospheric waters20,21,22,23,24. Although diacids, oxoacids and α−dicarbonyls can be emitted directly into the atmosphere from primary sources such as incomplete combustion of fossil fuels25 and biomass burning26, they are mainly produced by secondary processes of VOCs of both anthropogenic and biogenic origin14,16,19,27,28,29. They are further subjected to significant photochemical oxidation in the atmosphere during long-range transport; e.g., carbonyls to carboxylic acids30 and breakdown of higher to lower diacids16,31. The molecular distributions of diacids have been considered to understand aging of organic aerosols in the atmosphere21.
Stable carbon isotopic composition (δ13C) of specific organic compound can provide highly valuable information on its aging in the atmosphere32,33,34,35, because the isotopic fractionation of carbon occurs upon chemical reactions or phase transfer35. Unidirectional reactions show a preferential enrichment of 12C in reaction products with the remaining reactants being isotopically heavier36. On the other hand, chemical processing within the particle (or aqueous) phase causes the enrichment of 13C in the reactant retained in the same phase, if some of the reaction products are volatile that are isotopically lighter37. It was found that remaining aliphatic/aromatic hydrocarbons including isoprene become more enriched with 13C after photochemical oxidation with hydroxyl radical (HO•) in laboratory experiments and field measurements32,33. It was also found that 13C is enriched in the remaining oxalic acid (C2) during the photolysis catalyzed by Fe3+ (and Fe2+) in aqueous phase in a laboratory experiment34. Therefore, it is very likely that δ13C of diacids and related compounds could be used as a proxy for assessing the aging of organic aerosols in the atmosphere.
Wang and Kawamura38 found a decrease in the concentrations and an increase in δ13C of saturated diacids, especially C2-C4 diacids, in marine aerosols from midlatitudes toward the equator over the western Pacific. In addition, they reported an increasing trend in the relative abundance of C2 in total diacids (C2%), a measure of aging21 and interpreted the enrichment of 13C in diacids toward the equator due to the kinetic isotope effects (KIE) caused by photochemical degradation. Pavuluri et al.39 also reported higher δ13C of C2-C4 diacids in the tropical Indian aerosols with an increase in δ13C from C4 to C2, which are considered to be aged during long-range transport from distant source regions. They interpreted an enrichment of 13C in C2-C4 diacids as a result of photochemical breakdown of higher to lower diacids and C2 diacid to CO2 during long-range atmospheric transport, despite a linear relationship between concentrations of C2 and C2%. However, there are no laboratory studies on the changes in δ13C of organic compounds with aging under atmospherically relevant conditions.
In this study, we conducted batch UV irradiation experiments using two types of ambient aerosol samples, which represent anthropogenic (AA) and biogenic aerosols (BA), collected from Chennai, India in the presence of moisture for different time periods from 0.5 to 120 h to better understand the applicability of δ13C of diacids, oxoacids and α-dicarbonyls in assessing the atmospheric aging of OA. Here, we report the changes in δ13C of diacids, glyoxylic acid and glyoxal as a function of irradiation time. Based on the observed results together with the changes in their concentrations, we discuss possible photochemical processes responsible for an enrichment or depletion of 13C in diacids and related compounds with the aging of aqueous aerosols, thus infer the use of δ13C of diacids and related compounds as a proxy for photochemical aging.
Results
Table 1 presents δ13C of normal saturated α,ω-diacids (C2-C6 and C9) and aromatic diacid (phthalic acid, Ph), glyoxylic acid (ωC2) and glyoxal (Gly) determined in non-irradiated AA and BA samples as well as their average and median values in the irradiated (0.5 to 120 h) AA and BA samples. On average, δ13C of C2, C3, Ph, ωC2 and Gly in irradiated AA and BA are higher than those of non-irradiated AA and BA samples (Table 1). Further, δ13C of C2 is highest followed by C3 and C4 in both non-irradiated and irradiated (on average) AA and BA (Table 1). In contrast, average δ13C of C4 and C5 diacids in irradiated AA and BA samples are lower than those of non-irradiated samples. C6 diacid shows an enrichment of 13C in irradiated samples whereas C9 presents a depletion of 13C in irradiated samples, compared to that in non-irradiated samples (Table 1).
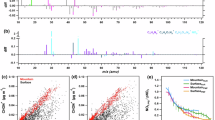
Figure 1 shows changes in δ13C of diacids and related compounds together with changes in their concentrations as a function of UV irradiation time. We found that δ13C of C2 diacid increase with irradiation time, except for few points, in both AA and BA, although its concentrations declined (Fig. 1a,j). δ13C of C3 diacid also increased, being opposite to its concentrations in both samples, although the trend is not clear during early stages of irradiation in the case of BA (Fig. 1b,k). Interestingly, δ13C of C4 showed a general decrease with irradiation up to 6 h followed by an increase until the end of in both AA and BA experiments, except for few cases, despite a significant increase in its concentrations with irradiation up to 72 h in BA and 96 h in AA (Fig. 1c,l). However, the enrichment of 13C in C4 diacid was small in both samples, except for later stages of irradiation (72 to 120 h) in BA when the concentration of C4 decreased drastically (Fig. 1c,l).
Changes in δ13C of diacids (C2-C6, C9 and Ph), glyoxylic (ωC2) acid and glyoxal (Gly) together with the changes in their concentrations with UV irradiation time in AA and BA.
Concentrations data is obtained from Pavuluri et al.19. Bars show the experimental errors in δ13C of diacids and related compounds.
Similarly, δ13C of C5 diacid decreased with irradiation time up to 18 h in BA and 24 h in AA, except for few cases, and then increased until the end of AA experiment and up to 48 h in BA experiment followed by a decrease thereafter (Fig. 1d,m). δ13C of C6 diacid in AA also decreased with time up to 36 h and then increased, except for few points, until the end of experiment (Fig. 1e). In case of BA, C6 was depleted with 13C in 1.5 h and enriched with 13C in 12 h compared to non-irradiated sample (Fig. 1n). Such trends of δ13C of C5 and C6 are opposite to those of their concentrations, except for few points (Fig. 1d,e,m,n). Being consistence with other normal diacids, C9 diacid in AA showed an enrichment of 13C with irradiation, except for 12 h (Fig. 1f). On the contrary, δ13C of C9 in BA showed a decrease with irradiation, despite a decrease in its concentration (Fig. 1o). δ13C of Ph also showed an increase with time in both AA and BA samples (Fig. 1g,p). δ13C of ωC2 and Gly in AA showed a general increase with time, although their concentrations increased during early stages of irradiation (i.e., up to 24 h) and decreased gradually during later stages of irradiation (Fig. 1h,i). They did not show clear trends in the case of BA (Fig. 1q,r).
Discussion
We generally found an inverse relation between δ13C and concentrations of individual diacids and related compounds, although correlations are weak in most cases (Fig. 1, Table 2). This trend is consistent with that reported in remote marine aerosols from midlatitudes toward the equator over the western Pacific38. The decrease in C2 and C3 diacid concentrations with irradiation time (i.e., aging) has been attributed to the overwhelming photolysis catalyzed by water-soluble iron19, because C2 and C3 diacids tend to form a complex with Fe3+ (and C2 even with Fe2+) by acting as ligands in aqueous phase and then both the diacids photolyze upon the absorption of UV light to result in Fe2+ and CO240,41,42,43. Fe2+-oxalato complex undergoes a charge transfer from the Fe2+ to the surrounding solvent molecule, upon the absorption of UV light, forming Fe3+-oxalato complex, and then results in Fe2+ and CO241.
In fact, Fe2+ and Fe3+ species are abundant in both AA (20.5 ng m−3 and 36.6 ng m−3, respectively) and BA (30.0 ng m−3 and 48.4 ng m−3, respectively)19, which could promote the photolysis of C2 and C3 diacids upon irradiation. The photolysis of C2 (and C3) in the presence of iron species could cause an isotopic fractionation and enrichment of 13C in the remaining C2 (and C3) diacids, because unidirectional reaction causes an enrichment of 13C in the remaining reactant34,36. Further the formation of Fe-oxalato (and malonato) complex and subsequent photolysis mainly depends on concentration ratios of Fe to oxalic (and malonic) acid rather than the UV light intensity34,40,44. Pavuluri and Kawamura34 reported an enrichment of 13C in authentic C2 diacid upon its photolysis in the presence of Fe3+ (and Fe2+) in aqueous phase in a laboratory experiment. Therefore, we interpret that an enrichment of 13C in C2 and C3 diacids with aging in AA and BA should have been mainly driven by their enhanced photochemical decomposition to CO2 (Fig. 2).
It is well established that C2 and C3 diacids are produced by the photochemical breakdown of higher homologous diacids preferably via ketomalonic acid (kC3) and hydroxysuccinic acid (hC4), respectively. In addition, C2 is secondarily produced by the photochemical oxidation of unsaturated short-chain aliphatic, and aromatic hydrocarbons via ωC2 acid whereas C3 may be produced from unsaturated fatty acids and/or cyclic olefins via oxopropionoic (ωC3) or oxoheptanoic (ωC7) acids (Fig. 2)19,21,24. Hence, the trends of mass ratios of diacid species to their immediate precursor compounds could provide insights on the formation pathways and subsequent changes in δ13C of diacids. We found a linear relation between δ13C of C2 diacid and mass ratios of C2 to kC3 in both AA and BA (Fig. 3a) and also between δ13C of C2 and C2/ωC2 in BA but not in AA (Fig. 3b). On the other hand, although δ13C of C3 diacid showed no relation with mass ratios of C3 to hC4 in both AA and BA samples and with ωC3 and ωC7 acids in AA, it did show linear relations with C3/ωC3 and C3/ωC7 in BA (Fig. 3c–e). Such positive relations infer that enrichment of 13C in C2 and C3 is partly caused by the production from higher homologues and/or other precursors by sequential decarboxylation reactions (Fig. 2) because the substrate (i.e., reaction products retained in the same phase; particle or aqueous) are enriched with 13C, if some of the reaction products are volatile that are isotopically lighter37. This process is supported by the enhanced enrichment of 13C in C2 followed by C3 and C4 (Table 1).
Scatter plots between mass ratios of selected diacids (C2-C6 and oxoacid (ωC2) to their immediate precursor compounds and δ13C of diacids (C2-C6) and ωC2.
Please see text for abbreviations. Mass ratios data is obtained from Pavuluri et al.19. The outliers (1.75 × whisker length) that are not included in regression coefficient (r2) estimation are shown in brackets. Negligible r2 values (≤0.10) are not shown here.
On the contrary, a depletion of 13C in C2 diacid observed in few samples and a decrease in δ13C of C3 diacid during early stages of irradiation (Fig. 1a,b,j,k) can be attributed to an enhanced photochemical production of C2 and C3, whose δ13C are isotopically lighter than their precursor compounds such as oxoacids32,33,36 that are freshly produced from the primary precursors available in the original samples (Fig. 2). The reason for the observed inverse relation between δ13C of C2 and C2/ωC2 in AA is not clear, but we presume that photochemical degradation of C2 and its production from higher homologues could have played more important role in controlling δ13C than oxoacids in AA (Fig. 2). It is likely because the major precursors of ωC2; Gly and methylglyoxal (mGly), are mainly derived from biogenic emissions rather than anthropogenic sources45 and the formation pathway of ωC2 from mGly involves the decarboxylation reactions (Fig. 2), which causes an enrichment of 13C in the substrate (ωC2). Moreover, the ωC2 derived from aromatic hydrocarbons in AA might be enriched with 12C, rather than 13C, due to several unidirectional reactions.
Similarly, the production of C3 from C4 diacid via hC3 may be minor in both the samples as well as from oxoacids (ωC3 and ωC7) in AA. It is likely because the photochemical oxidation of longer-chain diacids results in either the immediate lower homologue or different short-chain oxoacid and diacid species46 whereas ωC3 and ωC7 are mainly derived from fatty acids that originate from biogenic emissions21. On the other hand, δ13C of C3 could have more influenced by its photochemical degradation and transformations rather than the production processes (Fig. 2), because the removal of CO2 in the former two cases leads to a significant enrichment of 13C in the remaining C3 diacid37. In the case of BA, production of C3 diacid via ωC3 and ωC7 acids (Fig. 2) should be significant because the abundances of fatty acids are higher (total concentration of C8-C34 fatty acids is 297 ng m−3) in BA than in AA (167 ng m−3)19.
The depletion and enrichment of 13C in C4-C6 diacids during early (up to 24~36 h) and later stages of irradiation, respectively, except for few cases, (Fig. 1c–e,l–n; Table 1) can also be attributable to the enhanced photochemical production from the first generation products, which are originally present in the samples and subsequent degradation to lower homologues (Fig. 2). However, plots between δ13C of C4 and mass ratios of C4 to C5 and oxooctanoic (ωC8) acid in AA, and oxobutanoic (ωC4) acid, and those between δ13C of C5 and C5/C6 and δ13C of C6 and C6/C7 in both the AA and BA samples are highly scattered (Fig. 3f–j), suggesting that production of C4-C6 diacids from their immediate higher homologues (and other precursors such as long-chain oxoacids), except for C4 in BA, may be insignificant in controlling δ13C of C4-C6.
Further, a significant enrichment of 13C in C4 and C6 in BA and AA, respectively, during the later stages of irradiation (Fig. 1) can be attributable to the photochemical breakdown to lower homologues (Fig. 2), because their concentrations were significantly low in those samples (Fig. 1l,e). The differences in δ13C trends of C4-C6 diacids between AA and BA (Fig. 1c–e,l–n) should have been driven by a significant difference in the abundances of their precursors in the original samples. For example, fatty acids are abundant in BA (297 ng m−3) than in AA (167 ng m−3)19. It is also important to note that depletion of 13C in C4-C6 in irradiated samples compared to that in non-irradiated samples (Fig. 1c–e,l–n; Table 1) should be a result of the enhanced production from the primary precursors that are originally present in the samples rather than the transformations of C4-C6 to their lower homologues (Fig. 2).
Although δ13C of C9 diacid were measured in few samples during early stages of irradiation, an enrichment of 13C was found in early stage of aging of AA (Fig. 1f), which can be attributed to photochemical degradation of C9 to lower homologues. In contrast, a depletion of 13C in C9 was found with the aging of BA as well as in 12 h irradiated sample of AA (Fig. 1f,o), which might be involved with simultaneous photochemical production of C9 diacid from its primary and first generation precursors such as fatty acids and ωC9 acid21. We found an enrichment of 13C in Ph with aging in both AA and BA (Fig. 1g,p), which can be explained by the enhanced photochemical degradation.
We also found an enrichment of 13C in ωC2 acid, except for few samples in BA, and Gly with aging (Fig. 1h,i,q,r). This increase may be caused by fragmentation reactions of their precursors (e.g., isoprene oxidation products, Fig. 2) that results in an enrichment of 13C in the remaining ωC2 and Gly in the same phase (i.e., particle/aqueous)37. Moreover, the breakdown of ωC2 and Gly to other oxygenated species (Fig. 2) on prolonged irradiation could also cause an enrichment of 13C in the remaining ωC2 and Gly36. It is noteworthy that the relations between δ13C of ωC2 and mass ratios of ωC2 to its precursors (Gly and pyruvic acid) are not linear but rather scattered (Fig. 3k,l). Simultaneous production of ωC2 from various precursors and further oxidation to C2 may cause a complicated behavior of the intermediate species (ωC2) in terms of stable isotopic composition (Fig. 2). As discussed in the cases of diacids, the differences in the abundances of precursor compounds in AA and BA might cause the variations in trends of δ13C of ωC2 and Gly between AA and BA samples. In fact, concentrations of ωC2 and Gly were significantly higher in BA than in AA during the early stages of irradiation (Fig. 1h,i,q,r), suggesting that their formation is more prominent than degradation in BA compared to that in AA because the original BA sample contains a large amount of organics (9820 ng m−3 of organic carbon) than AA (6400 ng m−3)19.
Thus the observed changes in δ13C of diacids, ωC2 and Gly. with aging in aqueous AA and BA were similar, except for few cases, which infer that the photochemical formation and degradation pathways of diacids and related compounds are almost same irrespective of their precursors origin and the δ13C of diacids and related compounds can be used as a proxy for aging. However, the differences in δ13C between AA and BA for some cases should have been driven by the differences in the abundances of diacids and related compounds as well as their precursor compounds in the non-irradiated AA and BA samples, which cause the differences in rates of their formation and/or degradation. In fact, diacids and related compounds are more abundant in non-irradiated AA than in BA whereas, organic carbon content that contains several precursors of those compounds is higher in BA than AA19. It is also of important to note that a significant formation of oligomers of carboxyl and carbonyl compounds in our experiment is unlikely because photolysis of oligomers should be more prominent during the prolonged aging of organics for several hours to days11,13.
Finally, it is noteworthy that a general trend of increased δ13C of C2 to C4 diacids with aging in both AA and BA samples are consistent with those reported in ambient aerosols from the western Pacific38, Sapporo, northern Japan47, and Chennai, India39, which are considered to be aged during long-range transport. Such consistency further supports that δ13C of diacids and related compounds can be used as a new proxy to trace aging of organic aerosols in the atmosphere. However, due to a lack of quantitative evaluation of oxidants (e.g., HO•) and complexity of photochemical processes of diacids and related compounds during the experiment, it is difficult to derive the KIE factors from this study, which is a subject of future research.
Materials and Methods
Batch UV irradiation experiments were conducted using two ambient aerosol (PM10) samples that were collected in winter on 28 January (IND104) and summer on 25 May (IND178) 2007 during daytime (06:00~18:00 h local time) from Chennai (13.03°N; 80.17°E), India. The procedure flow chart and schematic of the irradiation experimental setup are shown in Fig. 4. Details of sampling, chemical characteristics (source assessment) of aerosols and irradiation experiment are provided elsewhere19,22. Briefly, PM10 samples were collected using a high volume air sampler and pre-combusted (450 °C, 4 h) quartz fiber filters. The sample filter was placed in a pre-combusted glass jar with a Teflon-lined screw cap and stored in a dark freezer room at −20 °C prior to the experiment22.
As detailed by Pavuluri et al.19, backward air mass trajectories showed that air masses for IND104 originated from the north Indian subcontinent passing over the Bay of Bengal, where emissions from fossil fuel combustion and forest fires are significant. In contrast, the air masses for IND178 originated from the Arabian Sea passing over the south Indian subcontinent, where the emissions from combustion of biofuels and livestock are important. The concentrations of elemental carbon, organic carbon, hopanes (C27-C35, tracer for fossil fuel combustion), and fatty acids (C8-C34) and fatty alcohols (C14-C34) (biomarkers for biogenic emissions) were found to be 4810, 6400, 11.8, 167 and 93.3 ng m−3, respectively, in IND104 sample and 1810, 9820, 3.9, 297 and 178 ng m−3, respectively, in IND178 sample19. In addition, the trace metals that mainly originate from fossil fuel combustion (Cr, Pb and V) are higher (5.33, 133 and 9.60 ng m−3, respectively) by up to several times in IND104 than in IND178 (0.00, 39.9 and 0.00 ng m−3, respectively)19. Such chemical signatures indicate that IND104 sample is enriched with anthropogenic emissions whereas IND178 sample is with biogenic emissions. Hence, we considered that IND104 represents AA whereas IND178 represents BA.
Irradiation experiments of each sample were conducted for 0.5, 1.5, 3.0, 6.0, 12, 18, 24, 36, 48, 72, 96 and 120 h using a separate filter cut for every experiment. In each experiment, an aliquot (~12 cm2) of sample filter was placed vertically in a cleaned quartz reaction vessel (cylinder, 100 ml) with the sample surface facing to UV light and wetted with ~0.4 ml of ultra-pure organic free distilled water and then sealed with Teflon-lined screw cap under the ambient pressure. The filter samples were then irradiated with a low-pressure mercury lamp (Ushio, UL0-6DQ) that emits a UV light wavelength primarily at 254 nm and a minor peak at 185 nm in the presence of moisture in the reaction vessel.
The main objective of UV irradiation of aqueous aerosols (wetted sample filter) at 254 nm, rather than a solar spectrum, was to produce significant amount of hydroxyl radicals (HO•) via different pathways. Irradiation of aqueous aerosol at 254 nm induces the formation of O3 from the dissolved O2, followed by the generation of H2O2; the photolysis of H2O, NO3−, NO2−, H2O2, Fe(OH)2+ and certain organic compounds; and Fenton’s reaction of photochemically formed Fe2+ and H2O219. Such HO• sources are similar to those of atmospheric waters48 and the produced HO• should be sufficient enough to act as the main oxidant in our experimental system because both the samples contain a large amount of Fe (2070 ng m−3 in AA and 553 ng m−3 in BA, including water-soluble iron)19 that could promote Fenton’s reaction upon UV irradiation. Moreover, O3, H2O2, HOO• and NO2 formed in aqueous phase reactions may be partitioned into gas phase and generate the gaseous HO• in the reaction cylinder that should be re-partitioned into aqueous phase48. However, we could not approximate the actual concentrations of HO• in experiments, because we did not add any chemicals (e.g., a standard compound whose kinetics are known) into our experimental system in order to keep it as realistic as possible. We believe that the concentration of HO• is almost constant in all experiments irrespective of the duration of each experiment, because the consumption and production of the HO• could occur simultaneously during the experiment. Bateman et al.11 found no significant change in the concentration of H2O2, a major source of HO•, during aging of secondary OA of d-limonene in laboratory, which further supports our presumption.
Although we do not preclude a minor photolysis of some organic compounds present in aerosol samples by irradiation at 254 nm, it has been reported that targeted compounds of this study; low molecular weight diacids, oxoacids including pyruvic acid, and α-dicarbonyls such as mGly, have negligible absorbance at 254 nm and exhibit minimal photolysis, particularly when HO• reactions of organics are significant14,18,31. The photolysis of organics by the absorbance of 185 nm light should also be insignificant during the experiment because the intensity of 185 nm peak is about 100 times lower than that of 254 nm and such small intensity of 185 nm light is mostly absorbed by water due to its high absorption coefficient (1.8 cm−1 at 25 °C)49. In addition, radiation at 254 nm has been reported to impose only a marginal photolysis of most inorganic species, except for nitrate, which is one of the HO• sources31. The temperature of experimental system was maintained at 25 ± 1 °C to avoid any potential temperature effect on chemical reactions during the experiment.
Immediately after the irradiation on filter sample, diacids and related compounds in each sample were extracted and δ13C relative to Pee Dee Belemnite were determined using the method developed by Kawamura and Watanabe50. Briefly, irradiated filter sample was extracted with ultra-pure organic free distilled water (10 mL × 3) under ultra-sonication for 10 min. The extracts were concentrated to near dryness using a rotary evaporator under vacuum, and then diacids and related compounds were derivatized with 14% BF3/n-butanol at 100 °C to butyl esters and/or butoxy acetals. The derivatized samples were extracted with n-hexane and then determined for their peak identification and quantification using a capillary GC (HP 6890) and GC-MS (Thermo Trace MS)20,22,51, prior to the stable carbon isotope measurements.
After an appropriate amount of internal standard (n-C13 alkane) was spiked to a fraction of the derivatized sample, δ13C of the derivatives were determined using GC (HP6890) coupled to isotope ratio mass spectrometry (Finnigan MAT Delta Plus). δ13C of free diacids and related compounds in the sample were then calculated using an isotopic mass balance equation based on the measured δ13C of the derivatives and derivatizing agent (1-butanol)49. Each sample was analyzed in duplicate and an average is reported here. Difference in δ13C of free acids in duplicate analysis was generally below 1‰. However, the uncertainties for minor species were sometimes up to 1.5‰ and occasionally over 2‰. The experimental errors, including analytical errors, from replicate experiments (n = 3) conducted for 18 h irradiation of AA were found to be within 2‰, except for C3 diacid (3.2‰). No peaks were detected in the procedural blank of irradiation experiments (1.5 h and 6.0 h), except for a small peak for C2 and Ph acids, using a clean quartz filter.
Additional Information
How to cite this article: Pavuluri, C. M. and Kawamura, K. Enrichment of 13C in diacids and related compounds during photochemical processing of aqueous aerosols: New proxy for organic aerosols aging. Sci. Rep. 6, 36467; doi: 10.1038/srep36467 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Kanakidou, M. et al. Organic aerosol and global climate modelling: a review. Atmos Chem. Phys. 5, 1053–1123 (2005).
Andreae, M. O. & Crutzen, P. J. Atmospheric Aerosols: Biogeochemical Sources and Role in Atmospheric Chemistry. Science 276, 1052–1058 (1997).
Ramanathan, V., Crutzen, P. J., Kiehl, J. T. & Rosenfeld, D. Aerosols, climate, and the hydrological cycle. Science 294, 2119–2124 (2001).
Asa-Awuku, A. et al. Airborne cloud condensation nuclei measurements during the 2006 Texas air quality study. J. Geophys. Res 116, D11201 (2011).
Baltensperger, U. et al. Combined determination of the chemical composition and of health effects of secondary organic aerosols: The POYSOA project. J. Aerosol Med. Pulm. Drug Delivery 21, 145–154 (2008).
Kroll, J. H. & Seinfeld, J. H. Chemistry of secondary organic aerosol: Formation and evolution of low-volatility organics in the atmosphere. Atmos. Environ. 42, 3593–3624 (2008).
Saxena, P. & Hildemann, L. M. Water-soluble organics in atmospheric particles: A critical review of the literature and application of thermodynamics to identify candidate compounds. J. Atmos. Chem. 24, 57–109 (1996).
Hallquist, M. et al. The formation, properties and impact of secondary organic aerosol: current and emerging issues. Atmos. Chem. Phys. 9, 5155–5236 (2009).
Ervens, B., Turpin, B. J. & Weber, R. J. Secondary organic aerosol formation in cloud droplets and aqueous particles (aqSOA): a review of laboratory, field and model studies. Atmos. Chem. Phys. 11, 11069–11102 (2011).
Epstein, S. A., Tàpavicza, E., Furche, F. & Nizkorodov, S. A. Dircet photolysis of carbonyl compounds dissolved in cloud and fog droplets. Atmos. Chem. Phys. 13, 9461–9477 (2013).
Bateman, A. P., Nizkorodov, S. A., Laskin, J. & Laskin, A. Photolytic processing of secondary organic aerosols dissolved in cloud droplets. Phy. Chem. Chem. Phys. 13, 12199–12212 (2011).
Liu, Y. et al. Aqueous phase processing of secondary organic aerosol from isoprene photooxidation. Atmos. Chem. Phys. 12, 5879–5895 (2012).
Renard, P. et al. Aqueous phase oligomerization of methyl vinyl ketone through photooxidation – Part 1: Aging processes of oligomers. Atmos. Chem. Phys. 15, 51–35 (2015).
Carlton, A. G., Turpin, B. J., Lim, H. J., Altieri, K. E. & Seitzinger, S. Link between isoprene and secondary organic aerosol (SOA): Pyruvic acid oxidation yields low volatility organic acids in clouds. Geophys. Res. Lett. 33, L06822 (2006).
Chen, Z. M. et al. Aqueous phase ozonolysis of methacrolein and methyl vinyl ketone: a potentially important source of atmospheric aqueous oxidants. Atmos. Chem. Phys. 8, 2255–2265 (2008).
Charbouillot, T. et al. Mechanism of carboxylic acid photooxidation in atmospheric aqueous phase: Formation, fate and reactivity. Atmos. Environ. 56, 1–8 (2012).
Tan, Y., Carlton, A. G., Seitzinger, S. P. & Turpin, B. J. SOA from methylglyoxal in clouds and wet aerosols: Measurement and prediction of key products. Atmos. Environ. 44, 5218–5226 (2010).
Tan, Y., Lim, Y. B., Altieri, K. E., Seitzinger, S. P. & Turpin, B. J. Mechanisms leading to oligomers and SOA through aqueous photooxidation: insights from OH radical oxidation of acetic acid and methylglyoxal. Atmos. Chem. Phys. 12, 801–813 (2012).
Pavuluri, C. M., Kawamura, K., Mihalopoulos, N. & Swaminathan, T. Laboratory photochemical processing of aqueous aerosols: formation and degradation of dicarboxylic acids, oxocarboxylic acids and α-dicarbonyls. Atmos. Chem. Phys. 15, 7999–8012 (2015).
Kawamura, K. & Ikushima, K. Seasonal changes in the distribution of dicarboxylic acids in the urban atmosphere. Environ. Sci. Technol. 27, 2227–2235 (1993).
Kawamura, K. & Sakaguchi, F. Molecular distributions of water soluble dicarboxylic acids in marine aerosols over the Pacific Ocean including tropics. J. Geophys. Res. 104, D3 3501 (1999).
Pavuluri, C. M., Kawamura, K. & Swaminathan, T. Water-soluble organic carbon, dicarboxylic acids, ketoacids, and α-dicarbonyls in the tropical Indian aerosols. J. Geophys. Res.-Atmos. 115, D11302 (2010).
Sorooshian, A., Wang, Z., Coggon, M. M., Jonsson, H. H. & Ervens, B. Observations of Sharp Oxalate Reductions in Stratocumulus Clouds at Variable Altitudes: Organic Acid and Metal Measurements During the 2011 E-PEACE Campaign. Environ. Sci. Technol. 47, 7747–7756 (2013).
Kawamura, K. & Bikkina, S. A review of dicarboxylic acids and related compounds in atmospheric aerosols: Molecular distributions, sources and transformation. Atmos. Res. 170, 140–160 (2016).
Kawamura, K. & Kaplan, I. R. Motor exhaust emissions as a primary source for dicarboxylic acids in Los-Angeles ambient air. Environ. Sci. Technol. 21, 105–110 (1987).
Narukawa, M., Kawamura, K., Takeuchi, N. & Nakajima, T. Distribution of dicarboxylic acids and carbon isotopic compositions in aerosols from 1997 Indonesian forest fires. Geophys. Res. Lett. 26, 3101–3104 (1999).
Kawamura, K., Kasukabe, H. & Barrie, L. A. Source and reaction pathways of dicarboxylic acids, ketoacids and dicarbonyls in arctic aerosols: One year of observations. Atmos. Environ. 30, 1709–1722 (1996).
Lim, H. J., Carlton, A. G. & Turpin, B. J. Isoprene forms secondary organic aerosol through cloud processing: model simulations. Environ. Sci. Technol. 39, 4441–4446 (2005).
Warneck, P. In-cloud chemistry opens pathway to the formation of oxalic acid in the marine atmosphere. Atmos. Environ. 37, 2423–2427 (2003).
Tilgner, A. & Herrmann, H. Radical-driven carbonyl-to-acid conversion and acid degradation in tropospheric aqueous systems studied by CAPRAM. Atmos. Environ. 44, 5415–5422 (2010).
Yang, L. M., Ray, M. B. & Yu, L. E. Photooxidation of dicarboxylic acids- Part 1: effects of inorganic ions on degradation of azelaic acid. Atmos. Environ. 42, 856–867 (2008).
Rudolph, J., Czuba, E. & Huang, L. The stable carbon isotope fractionation for reactions of selected hydrocarbons with OH-radicals and its relevance for atmospheric chemistry. J. Geophys. Res.-Atmos. 105, 29329–29346 (2000).
Rudolph, J. et al. The stable carbon isotope ratio of biogenic emissions of isoprene and the potential use of stable isotope ratio measurements to study photochemical processing of isoprene in the atmosphere. J. Atmos. Chem. 44, 39–55 (2003).
Pavuluri, C. M. & Kawamura, K. Evidence for 13-carbon enrichment in oxalic acid via iron catalyzed photolysis in aqueous phase. Geophys. Res. Lett. 39, L03802 (2012).
Ren, L. et al. Molecular distributions and compound-specific stable carbon isotopic compositions of lipids in wintertime aerosols from Beijing. Sci. Rep. 6, 27481 (2016).
Hoefs, J. Stable Isotope Geochemistry, 6th ed., Springer, New York (2009).
Turekian, V. C., Macko, S. A. & Keene, W. C. Concentrations, isotopic compositions, and sources of size-resolved, particulate organic carbon and oxalate in near-surface marine air at Bermuda during spring. J. Geophys. Res.-Atmos. 108, D5 4157 (2003).
Wang, H. B. & Kawamura, K. Stable carbon isotopic composition of low-molecular-weight dicarboxylic acids and ketoacids in remote marine aerosols. J. Geophys. Res.-Atmos. 111, D07304 (2006).
Pavuluri, C. M., Kawamura, K., Swaminathan, T. & Tachibana, E. Stable carbon isotopic compositions of total carbon, dicarboxylic acids and glyoxylic acid in the tropical Indian aerosols: Implications for sources and photochemical processing of organic aerosols. J. Geophys. Res.-Atmos. 116, D18307 (2011).
Zuo, Y. G. & Hoigne, J. Photochemical decomposition of oxalic, glyoxalic and pyruvic acid catalyzed by iron in atmospheric waters. Atmos. Environ. 28, 1231–1239 (1994).
Zuo Y. & Deng, Y. Iron(II)-catalyzed photochemical decomposition of oxalic acid and generation of H2O2 in atmospheric liquid phases. Chemosphere 35, 2015–2018 (1997).
Wang, Z. H. et al. Photochemical cycling of iron mediated by dicarboxylates: special effect of malonate. Environ. Sci. Technol. 44, 263–268 (2010).
Weller, C., Tilgner, A., Bräuer, P. & Herrmann, H. Modeling the impact of iron-carboxylate photochemistry on radical budget and carboxylate degradation in cloud droplets and particles. Environ. Sci. Technol. 48, 5652–5659 (2014).
Weller, C., Horn, S. & Herrmann, H. Effects of Fe(III)-concentration, speciation, excitation-wavelength and light intensity on the quantum yield of iron(III)-oxalato complex photolysis. J. Photochem. Photobiol. A: Chem. 255, 41–49 (2013).
Fu, T. M. et al. Global budgets of atmospheric glyoxal and methylglyoxal, and implications for formation of secondary organic aerosols. J. Geophys. Res.-Atmos. 113, D15303 (2008).
Yang, L. M., Ray, M. B. & Yu, L. E. Photooxidation of dicarboxylic acids–Part II: Kinetics, intermediates and field observations. Atmos. Environ. 42, 868–880 (2008).
Aggarwal, S. G. & Kawamura, K. Molecular distributions and stable carbon isotopic compositions of dicarboxylic acids and related compounds in aerosols from Sapporo, Japan: Implications for photochemical aging during long-range atmospheric transport. J. Geophys. Res.-Atmos. 113, D14301 (2008).
Arakaki, T. & Faust, B. C. Sources, sinks, and mechanisms of hydroxyl radical (•OH) photoproduction and consumption in authentic acidic continental cloud waters from Whiteface Mountain, New York: The role of the Fe(r) (r = II, III) photochemical cycle. J. Geophys. Res.-Atmos. 103, 3487–3504 (1998).
Weeks, J. L., Meaburn, G. M. & Gordon, S. Absorption coefficients of liquid water and aqueous solutions in the far ultraviolet. Radiat. Res. 19, 559–567 (1963).
Kawamura, K. & Watanabe, T. Determination of stable carbon isotopic compositions of low molecular weight dicarboxylic acids and ketocarboxylic acids in atmospheric aerosol and snow samples. Anal. Chem. 76, 5762–5768 (2004).
Kawamura, K. Identification of C2-C10 ω-oxocarboxylic acids, pyruvic acid, and C2-C3 α-dicarbonyls in wet precipitation and aerosol samples by capillary GC and GC/MS. Anal. Chem. 65, 3505–3511 (1993).
Acknowledgements
This study was in part supported by Japan Society for the Promotion of Science (JSPS) (Grant-in-aid Nos 19204055 and 24221001), Japan, and 985 Project of National Key Universities, Tianjin University, China. We thank T. Swaminathan for his support in aerosol sampling and E. Tachibana for the help in isotope measurements.
Author information
Authors and Affiliations
Contributions
C.M.P. and K.K. designed the research and C.M.P. performed the experiments at Hokkaido University. The data interpretation and writing were done by both the authors.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Pavuluri, C., Kawamura, K. Enrichment of 13C in diacids and related compounds during photochemical processing of aqueous aerosols: New proxy for organic aerosols aging. Sci Rep 6, 36467 (2016). https://doi.org/10.1038/srep36467
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36467
This article is cited by
-
Combined influences of sources and atmospheric bleaching on light absorption of water-soluble brown carbon aerosols
npj Climate and Atmospheric Science (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.