Abstract
Companion cropping with potato onions (Allium cepa var. agrogatum Don.) can enhance the disease resistance of tomato plants (Solanum lycopersicum) to Verticillium dahliae infection by increasing the expressions of genes related to disease resistance. However, it is not clear how tomato plants physiologically respond to V. dahliae infection and what roles sulfur plays in the disease-resistance. Pot experiments were performed to examine changes in the physiology and sulfur metabolism of tomato roots infected by V. dahliae under the companion cropping (tomato/potato onion). The results showed that the companion cropping increased the content of total phenol, lignin and glutathione and increased the activities of peroxidase, polyphenol oxidase and phenylalanine ammonia lyase in the roots of tomato plants. RNA-seq analysis showed that the expressions of genes involved in sulfur uptake and assimilation, and the formation of sulfur-containing defense compounds (SDCs) were up-regulated in the V. dahlia-infected tomatoes in the companion cropping. In addition, the interactions among tomato, potato onion and V. dahliae induced the expression of the high- affinity sulfate transporter gene in the tomato roots. These results suggest that sulfur may play important roles in tomato disease resistance against V. dahliae.
Similar content being viewed by others
Introduction
Tomato Verticillium wilt, caused by Verticillium dahliae, is a severe soil-borne fungal disease that leads to severe economic losses in greenhouses and fields. Several management strategies have been developed for controlling this disease, such as cultivating the cultivars with Ve gene1, inducing plant resistance by using beneficial bacteria2,3, intercropping4,5,6, and enhancing plant defense against V. dahliae by using sulfur7,8. These strategies would lower production costs and reduce environmental pollution. Several studies have demonstrated that intercropping and companion cropping may alleviate soil-borne diseases by enhancing plant disease resistance to pathogens4,5,9,10. Previous studies have shown that companion cropping with potato onions alleviates tomato Verticillium Wilt by up-regulating the expression levels of genes related to disease resistance (encoding proteins such as pathogenesis-related proteins and those involved in lignin biosynthesis, phytohormone metabolism and signal transduction) in tomatoes infected with V. dahliae5, and by the inhibition effect of tomato root exudates on V. dahliae. However, the physiological responses of tomato to V. dahliae infection in the tomato/potato onion companion cropping system remain unknown.
Sulfur is an essential macronutrient for plants, and, importantly, it may enhance disease defenses in plants through the formation of sulfur-containing defense compounds (SDCs), such as glutathione, glutathione-S-transferase, phytochelatins and other sulfur-containing proteins11. Studies have shown that the accumulations of SDCs in pathogen-resistant cultivars are rapidly increased compared with that in susceptible ones. Notably, S0 is the only inorganic phytoalexin reported in the available literature12,13, and Cooper et al. have observed higher S0 accumulation in the resistant genotypes of Theobroma cacao infected with vascular-invading fungal pathogens than in susceptible T. cacao plants12,13. Moreover, the accumulation of S0 and glutathione rapidly increases in higher resistance tomato and pepper cultivars compared with lower resistance and susceptible plants14,15. These studies have demonstrated that sulfur plays important roles in plant disease resistance. However, additional studies are needed to determine whether sulfur is involved in the disease resistance of tomato plants against V. dahliae in the tomato/potato onion companion cropping system.
An increased sulfur supply increases the disease resistance of plants to pathogens and limits the spread of pathogens in plants, hence alleviating disease incidence and severity, whereas sulfur deficiency increases the susceptibility of plants to pathogens7,16,17,18. Thus, determining whether tomato/potato onion companion cropping may increase the availability of sulfur in the soil would be valuable for controlling tomato verticillium wilt. Interestingly, many studies have shown that intercropping increases soil phosphorus availability via the acidification resulting from root exudates and increases iron uptake by up-regulating the expression of genes encoding iron translocator (such as IRT gene family, AtNRAMP3, AtNRAMP4 and AtVIT1) mediated by roots interplay between different plant species19,20, thereby improving phosphorus and iron nutrition for crops. Thus, it is necessary to assess whether companion cropping may improve the availability of sulfur in the tomato rhizosphere, owing to the effects of root exudates from potato onions.
In this study, to elucidate the physiological responses of tomato plants to V. dahliae and determine the role of sulfur in tomato disease resistance against V. dahliae in the companion cropping, we examined 1) the physiological responses of tomato plant to V. dahliae infection, 2) the expression levels of genes related to sulfur uptake and assimilation and the formation of SDCs in tomato roots in the companion cropping by using RNA-seq, 3) changes in the expression level of the high-affinity sulfate transporter gene under different conditions by using qRT-PCR, 4) the accumulation of some SDCs in tomato roots, 5) the available sulfur content in the tomato rhizosphere, and 6) the effect of root exudates from potato onions on the available sulfur content in the soil.
Results
The disease symptoms
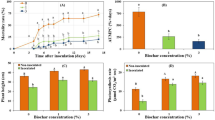
The disease symptoms of tomato verticillium wilt are presented as the disease incidence and disease index at 18 and 28 DAI (days after inoculation with Vd1). The results showed that at 18 DAI, the disease incidence was not significantly different between TM (tomatoes monoculture) and TC (tomatoes grown with potato onions) but was decreased in TC compared with that in TM (p <0.05) (Fig. 1A). In addition, the disease index was decreased in TC compared with that in TM at both 18 and 28 DAI (Fig. 1B,C).
Effects of companion cropping with potato onion on the disease incidence and index of tomato verticillium wilt.
TM indicates tomato monoculture; TC indicates tomato grown with potato onions. (A) Disease incidence. (B) Disease index. (C) Picture of disease symptom. The data are represented as the means ± SD, n = 3. The small letters above the column represent significant differences between two groups of mean values (p<0.05).
Changes in the content of total phenol, lignin, glutathione (GSH) and malonaldehyde (MDA) in tomato roots
Tomato roots were harvested at 0, 1, 3, 5, and 7 days after inoculation (DAI) with V. dahliae to measure the physiological responses of tomato roots to V. dahliae infection. The results showed that before inoculation with V. dahliae (0 DAI), the content of total phenol, lignin and GSH was not significantly changed between TC and TM (Fig. 2A–C), whereas the MDA content was lower in TC than in TM (p <0.05) (Fig. 2D). After inoculation with V. dahliae, the content of total phenol in TC significantly increased at 3, 5, and 7 DAI compared with TM, (Fig. 2A); the lignin content increased at 1 and 5 DAI (Fig. 2B); the GSH content increased at 1 and 5 DAI but decreased at 7 DAI (Fig. 2C); and the MDA content decreased at 1, 5 and 7 DAI (Fig. 2D).
Changes in content of total phenol, lignin, GSH and MDA in the tomato roots on different days after inoculation with V. dahliae.
(A) Total phenol content. (B) Lignin content. (C) GSH content. (D) MDA content. TM indicates tomato monoculture; TC indicates tomato grown with potato onions. The data are presented as the means ± SD, n = 3. The asterisk (*) indicates a significant difference between two groups of mean values (p <0.05).
Changes in the enzymatic activities of superoxide dismutase (SOD), peroxidase (POD), polyphenol oxidase (PPO) and phenylalanine ammonia lyase (PAL)
The activities of four enzymes in the tomato roots were determined at different days after inoculation with V. dahliae. The results showed no significant differences in the activities of the four enzymes between the two groups before inoculation with V. dahliae (0 DAI) (Fig. 3). At 1, 3, and 7 DAI, the SOD enzymatic activity in TC was lower than that in TM (p <0.05) (Fig. 3A). The POD activity was higher in TC at 1 and 3 DAI but was lower at 5 DAI than that in TM (Fig. 3B). The PPO activity was higher in TC at 3 and 5 DAI (Fig. 3C) than that in TM, and the trend in PAL activity was similar to that of POD (Fig. 3D).
Enzyme activities of SOD, POD, PPO and PAL in tomato roots on different days after inoculation with V. dahliae.
(A) SOD activity. (B) POD activity. (C) PPO activity. (D) PAL activity. TM indicates tomato monoculture; TC indicates tomato grown with potato onions. The data are presented as the means ± SD, n = 3. The asterisk (*) indicates a significant difference between two groups of mean values (p <0.05).
Expression levels of the genes related to sulfur uptake and assimilation and the formation of SDCs
The roots of tomatoes infected by V. dahliae for 3 days in tomato/potato onion companion cropping were used for RNA-seq analysis. The results showed that genes involved in sulfur uptake (high-affinity sulfate transporter 2) and assimilation (sulfate adenylyltransferase and sdenylyl-sulfate reductase) and the formation of SDCs (cystathionine gamma-lyase, cysteine synthase, glutathione-S-transferase-like protein, methionine sulfoxide reductase, S-adenosylmethionine decarboxylase proenzyme and S-adenosylmethionine-dependent methyltransferase) were all up-regulated in the tomato roots in TC compared with TM under the tomato/potato onion companion cropping (Table 1, Fig. 4). The expressions of genes encoding 1-aminocyclopropane-1-carboxylate synthase and 1-aminocyclopropane-1-carboxylate oxidase, which are key enzymes catalyzing methionine to ethylene, were also up-regulated (Table 1). Notably, the expression levels of the genes encoding high-affinity sulfate transporter 2 (Solyc09g082550.2.1), glutathione-S-transferase-like protein (Solyc09g011540.2.1, Solyc01g081250.2.1), 1-aminocyclopropane-1-carboxylate oxidase (Solyc07g026650.2.1) and 1-aminocyclopropane-1-carboxylate synthase (Solyc01g095080.2.1) in TC were 10 times higher than those in TM (Table 1).
Sulfur metabolism in plants.
The schematic flow diagram for sulfur metabolism, according to reviews by Thomas Rausch and Andreas Wachter11, and Kazu ki Saito21. ACC: 1-aminocyclopropane-1-carboxylate; APS: adenosine 5′- phosphosulfate; the arrow indicates induction (derepression); and the bar indicates inhibition (repression). The words in red indicate the genes encoding the enzymes (proteins) that were un-regulated in the roots of tomatoes infected by V. dahliae in tomato/potato onion companion cropping in the present study.
Expression level of the high-affinity sulfate transporter 2 gene (ST2 gene)
The expression levels of the ST2 gene were assessed under varying conditions. The results showed that after tomato seedlings were grown with potato onion plants for 10, 20 and 30 days, the relative expression of the ST2 gene in tomato roots was not different from that in monocultured tomatoes. However, after tomato plants were grown with potato onion plants for 30 days and simultaneously inoculated with V. dahliae (30d + Vd1), 10 days after inoculation the relative expression of the ST2 gene in TC was 4.31 times higher than that in TM (Fig. 5A). Compared with that of un-inoculated tomatoes, the relative expression of the ST2 gene in tomatoes inoculated with V. dahliae was not significantly different (Fig. 5B).
Relative mRNA expression of the ST2 gene in tomato roots infected by V. dahliae in tomato/potato onion companion cropping.
(A) Effect of companion cropping with potato onions on the relative mRNA expression of the ST2 gene. (B) Effect of V. dahliae inoculation on the relative mRNA expression of the ST2 gene. TM indicates tomato monoculture; TC means tomato grown with potato onions; and 10 d, 20 d and 30 d indicate companion cropping with potato onion for 10, 20 and 30 days, respectively. 30 d + Vd1 indicates companion cropping with potato onion for 30 days with simultaneous Vd1 inoculation starting at day 20. Vd1 indicates V. dahliae race 1. The data are presented as the means ± SD, n = 3. The asterisk (*) indicates a significant difference between two groups of mean values (p <0.05).
Changes in the content of total sulfur, GSH, methionine (Met) and S0 in tomato roots
The results showed that after inoculation with V. dahliae for 10 days, the content of total sulfur and GSH in the tomato roots in TC was higher than that in TM (p <0.05), whereas the Met content was lower (Table 2). S0 was not detected in either of the two groups.
Changes in the content of available sulfur in the soil
The results showed after the tomato plants were grown with potato onions for 10, 20 and 30 days without inoculation with V. dahliae, the available sulfur content in the tomato rhizosphere was not different from that in TM (Fig. 6A). After the tomato plants were grown with potato onions for 30 days and simultaneously inoculated with V. dahliae (30d + Vd1), 10 days after inoculation the available sulfur content in tomato rhizosphere was not different from that in TM (Fig. 6A). Compared with the un-inoculated tomatoes, tomatoes inoculated with V. dahliae did not show changes in the rhizosphere soil available sulfur content in either TM or TC (Fig. 6B). In addition, there was no significant difference in the available sulfur content in soil affected by the root exudates from potato onion plants (Fig. 6C).
Effects of companion cropping with potato onion and potato onion root exudates on soil available sulfur content.
(A) Effects of companion cropping with potato onion on the available sulfur content of tomato rhizosphere soil. (B) Effects of inoculation with V. dahliae on the available sulfur content of tomato rhizosphere soil. (C) Effects of potato onion root exudates on soil available sulfur content. The data are presented as the means ± SD, n = 3. The small letter “a” above the column represents no significance between the two groups of mean values (p > 0.05).
Discussion
The interspecific interactions of plants may enhance plant resistance to pathogens. For example, root interactions between maize and soybean increase the resistance of soybean to Cylindrocladium parasiticum10, and maize and pepper interactions enhance maize resistance to Bipolaris maydis9. Similarly, in a previous study, we have shown that tomato and potato onion interactions enhance tomato resistance to V. dahliae by up-regulating disease resistance-related genes under the tomato/potato onion companion cropping5. To explore the physiological defense responses of tomato to V. dahliae infection, we examined changes in the content of total phenol, lignin, GSH, and MDA, and the enzymatic activities of SOD, POD, PPO and PAL, all of which are key enzymes related to disease resistance1,21,22. The results showed that companion cropping with potato onions increased the contents of total phenol, lignin and GSH (Fig. 2A–C). Additional analyses showed that companion cropping with potato onions decreased the disease incidence and index (Fig. 1), confirming the results of the previous study5. These results indicated that companion cropping with potato onions increases the accumulation of substances related to resistance, thereby increasing the resistance of tomato plants against V. dahliae.
Furthermore, the activities of POD, PPO and PAL in the tomatoes grown with potato onion plants were higher than those in monocultured tomatoes at 3 days after inoculation, but POD and PAL activities in the tomatoes grown with potato onion plants were lower than those in monocultured tomatoes at 5 days after inoculation (Fig. 3B,D). These results suggested that companion cropping with potato onion plants rapidly increases enzymatic activity in the tomato roots, thereby enhancing disease resistance. In addition, POD and PAL are key enzymes in lignin biosynthesis23. The increased POD and PAL activities contribute to lignin accumulation, consistently with the increased lignin content in tomato roots (Fig. 2B). These results suggested that companion cropping with potato onions increases the levels and enzymatic activities of defensive substances, thereby decreasing the damage caused by V. dahliae. These results were confirmed by the decreased MDA content in the tomato roots (Fig. 2D), a result consistent with the decreased incidence and severity of tomato verticillium wilt (Fig. 1). Notably, the SOD activity was lower in tomatoes grown with potato onion plants than in monocultured tomatoes (Fig. 3A), probably because of enhanced pathogen defense and decreased V. dahliae attack5.
Sulfur enhances defenses against plant disease through the formation of sulfur-containing defense compounds (SDCs)11,13, and an increased sulfur supply increases the disease resistance of plants to pathogens7,16,17,18. Interestingly, the expression levels of genes related to sulfate uptake (high-affinity sulfate transporter) and assimilation and the formation of SDCs were all up-regulated in tomatoes grown with potato onion plants (Table 1), thus suggesting that sulfur-enhanced defense might play an important role in the enhanced resistance of tomatoes to V. dahliae, as described by Rausch and Wachter11. Furthermore, we examined the accumulation of SDCs, such as S0 and GSH, in the tomato roots. The results showed that after tomatoes were grown with potato onion plants and simultaneously inoculated with V. dahliae, the GSH content increased (Table 2), thus contributing to enhanced tomato resistance11. However, S0 was not detected in either of the two treatments (Table 2), possibly because S0 was not formed for 10 days after inoculation with V. dahliae. Indeed, Novo et al. have reported the accumulation of S0 at 10 days after the inoculation with V. dahliae in higher resistance pepper cultivars and at 15 days in lower resistance cultivars14. The tomato cultivar used in the present study was susceptible to V. dahliae, and these tomatoes were harvested at 10 days after inoculation. Nevertheless, the increased GSH content in the tomato roots (Fig. 2D, Table 2), coupled with the up-regulated expression of genes related to sulfur metabolism, such as those encoding glutathione-S-transferase, cysteine synthase and peptide methionine sulfoxide reductase, probably contribute to the enhancement of tomato resistance against V. dahliae.
Interestingly, RNA-seq analysis revealed that the expression level of the high-affinity sulfate transporter 2 gene (ST2), primarily responsible for sulfate uptake from the soil22, in tomatoes grown with potato onion plants was higher than that in monocultured tomatoes (Table 1). However, we were interested in the factors that induce the up-regulation of the ST2 gene. Therefore, we examined changes in the expression of the ST2 gene under different conditions. The results showed that after tomatoes were grown with potato onion plants without inoculation with V. dahliae, the expression of the ST2 gene in the intercropped tomatoes was not changed at each growth stage, as compared with that in monocultured tomatoes (Fig. 5A). However, after inoculation with V. dahliae, ST2 gene expression in tomatoes grown with potato onion plants was significantly higher than that in monocultured tomatoes (Fig. 5A). In addition, compared with un-inoculated tomatoes, tomatoes inoculated with V. dahliae did not show increased ST2 gene expression under monoculture (Fig. 5B), thus indicating that inoculation with V. dahliae did not induce the expression of the ST2 gene. This finding was not consistent with the result described by Howarth et al.24, in which infection with V. dahliae induced a high expression of LeST1-2 (one of the high-affinity sulfate transporters genes) in a resistant tomato cultivar. This discrepancy may be attributed to the susceptible tomato cultivar used in the present study. These results suggest that the up-regulated expression of the ST2 gene is induced by the interactions among all three factors—potato onion, tomato and V. dahliae—rather than interactions between two factors, i.e., potato onion and tomato, or tomato and V. dahliae.
There are two potential causes for the up-regulation of the ST2 gene in the tomato roots: sulfur-deficiency in tomato rhizosphere soils24,25 and pathogen infection11. Sulfur is taken up into plants as sulfate and subsequently assimilated into sulfur-containing amino acids, eventually forming SDCs11. Previous studies have shown that the genes encoding high-affinity sulfate transporters are up-regulated in tomato roots in response to sulfate deprivation24,25 and infection with V. dahliae in resistant tomato cultivars24. However, the results of the present study showed that inoculation with V. dahliae alone did not alter the expression level of the ST2 gene (Fig. 5B). In addition, we monitored the available sulfur content in the tomato rhizosphere, and the results showed that the available sulfur content in the rhizosphere was not significantly different between the groups, regardless of inoculation with V. dahliae (Fig. 6A,B). These results suggested that the up-regulated expression of the ST2 gene was not induced by either soil sulfate deficiency or V. dahliae infection alone.
In this case, we propose that a demand-driven mechanism might be responsible for the up-regulation of ST2 gene expression under infection conditions in tomato/potato onion companion cropping11,25. Plants positively respond to V. dahliae infection by forming more SDCs. The increased demand for SDCs requires an increase in the flux from cysteine to methionine (Met), GSH, S0 and other sulfur-rich proteins, and, therefore, leads to the up-regulation of the ST2 gene11,25. Feedback regulation was exhibited for the expression of the high-affinity sulfate transporter gene in response to the increased production of SDCs (Fig. 4). This mechanism was further supported by the results shown in Tables 1 and 2. Met is an intermediate in sulfur metabolism and a rate-limiting substance for sulfur-containing amino acid metabolism25,26. Met production is self-regulated through a feedback mechanism, i.e., if the Met content is sufficiently high, the production is decreased25. In this study, the Met content in tomatoes grown with potato onion plants was lower than that in monocultured tomatoes (Table 2). This result might reflect the high expression of genes encoding 1-aminocyclopropane-1-carboxylate oxidase and 1-aminocyclopropane-1-carboxylate synthase, which catalyze Met and form ethylene (Table 1, Fig. 4). The decreased Met level would lead to a temporary deficiency, thereby increasing cysteine consumption, which would lead to an increase in the expression of the high-affinity sulfate protein gene25.
Because the sulfur supply contributes to the disease resistance of plants7,8, the management of sulfur nutrition in the soil will be beneficial for plant defenses against pathogens. Several studies have demonstrated that the interactions between plants species may improve phosphorus and iron nutrition in plant rhizospheres through root exudates in intercropping systems19,27. Thus, it is important to evaluate whether the root exudates of potato onion plants may improve soil sulfur nutrition. In this study, we found that the root exudates of potato onion plants did not improve soil sulfur nutrition (Fig. 6C). However, the results suggested that companion cropping with potato onion plants increases sulfate uptake in tomato roots infected by V. dahliae, thereby increasing tomato resistance against V. dahliae. These findings might more effectively help farmers manage tomato Verticillium Wilt and reduce fungicide application and environmental pollution.
Conclusions
Companion cropping with potato onions increased defense substances (total phenol, lignin and GSH) and enzymatic activities (POD, PPO, and PAL) under V. dahliae stress. In addition, companion cropping with potato onions up-regulated the expression of genes related to sulfur metabolism and increased the total sulfur and GSH content in tomato roots, thereby indicating that sulfur might play important roles in tomato disease resistance to V. dahliae. Companion cropping with potato onion plants and potato onion root exudates did not alter soil sulfur availability. Thus, companion cropping might be the demand-driven mechanism by which potato onion plants up-regulate the expression of the high-affinity sulfate transporter gene. This discovery demonstrates an important step in the understanding of sulfur-enhanced defense, mediated by plant-plant-pathogen interactions in tomato/potato onion companion cropping.
Materials and Methods
Cultivation of plant materials and preparation of the fungal spore suspension
The tomato cultivar “Qiyanaifen” (V. dahliae-susceptible) and potato onion cultivar “Suihua” were used in this study. The fungal pathogen, V. dahliae race 1 (Vd1) was also used. Tomato cultivation and the preparation of the fungal spore suspension were performed according to a previous study5. Briefly, tomato seedlings were cultured in a greenhouse. The fungal spore suspension, at a concentration of 1.0 × 107 spores/mL, was prepared according to the methods described by Dobinson et al.28
Experiment design and sampling
The pot experiments were conducted from April to July 2015 in a greenhouse located in the Experimental Center of Northeast Agricultural University, Harbin, China. The experiments included the following two treatments: tomato/potato onion companion cropping (TC), in which one tomato seedling was grown with two potato onion plants at a distance of 10 cm between the tomato and potato onion plants, and tomato monoculture (TM), in which one tomato seedling was grown alone (without potato onion) in a single pot. Tomato seedlings with four true leaves were transplanted into pots (20 × 7.5 × 11.5 cm) filled with 2.5 kg autoclaved field soils per pot. Autoclaved underground water was applied throughout the experiment.
All the tomato seedlings were divided into three groups for different measurements (Table 3). In the first group, 20 days after transplantation, the tomato seedlings were inoculated with 20 mL of the Vd1 spore suspension (1 × 107 spores/mL) poured onto the rhizospheres of each tomato seedling. The tomato roots were harvested at 0, 1, 3, 5, and 7 days after inoculation (DAI) with V. dahliae, immediately frozen in liquid nitrogen after cleaning with autoclaved deionized water, subsequently stored at −80 °C for the determination of physiological changes. For each sampling, the roots from 3 tomato seedlings were collected and pooled together as one sample, with 3 samples per treatment (n = 3). For the remaining 60 tomato seedlings (10 tomato seedlings per treatment in one replication and there were 3 replications), the symptoms of Verticillium wilt disease were observed from 10 to 30 DAI at 3-day intervals. The disease symptoms were evaluated using a 0–5 scale according to a previous study5. The disease incidence was defined as the percentage of tomato seedlings with disease symptoms among all treated seedlings in each treatment per replication. The disease index (DI) was determined as DI = 100 × (∑Si × Ni)/(5 × Ni) according to the method described by Mercado-Blanco et al.29, where Si is the symptoms severity (disease scale) of an individual tomato plant, Ni is the number of plants with Si symptoms severity, and Nt is the total number of plants in each treatment per replication.
In the second group, 20 days after transplantation, tomato seedlings were inoculated with 20 mL of Vd1 spore suspension (1 × 107 spores/mL) poured onto the tomato seedling rhizospheres. Three days after inoculation with Vd1, the roots of 5 tomato seedlings in each treatment per replicate were harvested and pooled as one sample, with 3 samples per treatment (n = 3). The samples were instantly frozen in liquid nitrogen after cleaning with autoclaved deionized water and subsequently stored at −80 °C for RNA-seq library preparation.
In the third group, 10, 20 and 30 days after transplantation, the roots of tomato seedlings were harvested, immediately frozen in liquid nitrogen after being cleaned with autoclaved deionized water, and subsequently stored at −80 °C for determining the expression levels of high-affinity sulfate transporter gene. Additionally, 20 days after transplantation, parts of the tomato seedlings per treatment were inoculated with 20 mL of Vd1 spore suspension (1 × 107 spores/mL) poured onto the tomato seedling rhizospheres. 10 days after inoculation, the tomato seedlings roots and tomato rhizosphere soils were harvested and collected as above to determine the high-affinity sulfate transporter gene expression level and available sulfur content, respectively. The tomato rhizosphere soils were collected according to the method described by Wang et al.30. Briefly, the roots of tomato seedlings were carefully harvested and shaken gently to remove bulk soils. The soils adhering to the roots were treated as rhizosphere soil samples and collected using a brush. The rhizosphere soils were passed through a 2-mm mesh sieve and subsequently air-dried to measure the available sulfur content. Whole tomato plants from both treatments were harvested, cleaned, dried in an oven at 75 °C for 72 h, and subsequently mashed to determine the total sulfur content. For tomato roots and soil samples, 3 tomato seedlings and rhizosphere soil samples were pooled as single samples, in each treatment, with 3 samples per treatment in total (n = 3).
Experiments to determine the effects of potato onion root exudates on soil available sulfur content were performed according to the method described by Ae et al., with few modifications31. Briefly, 20 g of field soil was placed in a beaker, covered and autoclaved three times to eliminate soil microbes. Subsequently, 10 mL of sterile potato onion root exudates at a concentration of 1 g root/10 mL was added to the soil on a sterile operating platform at an interval of 24 h. Sterile deionized water instead of potato onion root exudates was added as a control treatment. There were 5 replicates of each treatment, and all beakers were stored in the dark at 25 °C. The soils were air-dried and passed through a 2-mm mesh sieve to determine the available sulfur content.
Measurement of the physiological parameters
A 0.1-g root sample of the fine powder material from each treatment and replication was used to examine the total phenol and lignin content, according to the methods described by Rodrigues et al.32. Total soluble phenolics were expressed as mg of catechol per kg of root fresh weight, and total lignin was expressed as mg/kg of root fresh weight by using lignin alkali as a standard. A 0.5-g frozen root sample from each treatment and replication was ground in 2 mL of cold extraction buffer (0.05 M phosphate buffer, pH 7.8), and the crude extract was used to determine SOD, POD, PPO, and PAL activities and the MDA content according to the methods described by Wang et al.33. POD activity was estimated using the guaiacol method34. PPO activity was spectrophotometrically measured on the basis of the increase in colored oxidation products within the first 3 min of the reaction35. PAL activity was assayed on the basis of the catalyzed rate of phenylalanine, as previously described33. A thiobarbituric acid reaction was used to determine the MDA content36. The levels of total glutathione content in tomato roots extracted in 2 mL of 0.1 M sodium phosphate buffer containing 5 mM EDTA (pH 7.5) were determined on the basis of the reaction of GSH with DTNB [5,50-dithio-bis (2-nitrobenzoic acid)], which produces the yellow derivative 50-thio-2-nitrobenzoic acid (TNB), according to the procedure described by Yu et al.37 The methionine level in tomato roots was determined using an Automatic Amino Acid Analyzer (Hitachi High-Technologies Corporation, Tokyo, Japan) after hydrolysis with HCl, and an external standard was used to calculate the concentration in tomato roots38,39. An Elementary Analysis System (Vario EL Cube, Germany) was used to determine the S0 level in tomato roots, according to the general procedure for elemental analyzers (JY/T017-1996, China). The total sulfur content in whole tomato plants was determined on the basis of HNO3-HClO4 digestion coupled with Barium Sulfate Turbidimetry40,41. The soil available sulfur content was determined through Ca(H2PO4)2-NaHCO3 extraction coupled with Barium Sulfate Turbidimetry42. All the measurements were performed in triplicate.
Screen of differentially expressed genes (DEGs) with RNA-seq
The tomato root samples were used to screen DEGs with RNA-seq 3 days after inoculation with Vd1 in tomato/potato onion companion cropping. Some of the results obtained in this study have been presented in a previous report (doi: 10.3389/fpls.2015.00726)5, in which we have summarized the DEGs related to disease resistance (such as pathogenesis-related protein genes, lignin biosynthesis related genes, plant hormones metabolism related genes), but the genes related to sulfur uptake and assimilation and the formation of SCDs were not listed. In this study, we presented the DEGs related to sulfur uptake and formation of SCDs from all DEGs.
Primer design and qRT-PCR assay for the high-affinity sulfate transporter (ST2) gene
The tomato actin gene was used as a reference gene43,44. The gene-specific primer pairs for the ST2 and actin genes were designed using Primer 5.0 software (Premier, Canada) and synthesized at the Sangon Biotech Company (Shanghai, China). The primer pair sequences for the ST2 and actin genes were F 5′-3′CAAAATTCTTCTGGATAAGTGCTA/ R 5′-3′CAAGGCGATGATACTGGTGAC, and F 5′-3′GAAATAGCATAAGATGGCAGACG/ R 5′-3′ATACCCACCATCACACCAGTAT, respectively. Total RNA from tomato roots was extracted using the TRIzol method described by Pattemore45. First-strand cDNA was synthesized using the TIANScript RT Kit (TIANGEN, China), according to the manufacturer’s instructions. The cDNA concentration was determined by using an Ultramicro ultraviolet spectrophotometer. The qRT-PCR reactions were performed with an iQ5 Multicolor Real-Time PCR Detection System (BIO-RAD, USA) with RealmasterMix (SYBR Green) (TIANGEN, China) according to the manufacturer’s instructions. The reaction mixture (20 μL) contained 0.6 μL of template cDNA, 0.3 μL of each primer oligonucleotide, 9 μL of fluorescent dyes and 9.8 μL of RNase-free ddH2O. Sterile water was used as a negative control, and all the samples were run in triplicate. The reaction mixture was denatured at 95 °C for 5 min, and this was followed by 35 cycles at 95 °C for 50 s, annealing at 57 °C for 30 s and extension at 72 °C for 40 s. At the end of 35 cycles, an additional final extension at 72 °C for 5 min was conducted to extend any premature DNA synthesis46. The mRNA expression levels of the target genes were normalized relative to the expression of the tomato actin gene and calculated using the 2−ΔΔCt method.
Statistical analysis
Analysis of variance (ANOVA) was performed using SPSS 16.0 analysis software (SPSS Inc., USA) and Tukey’s tests at the p = 0.05 significance level.
Additional Information
How to cite this article: Fu, X. et al. Physiological response and sulfur metabolism of the V. dahliae-infected tomato plants in tomato/potato onion companion cropping. Sci. Rep. 6, 36445; doi: 10.1038/srep36445 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Gayoso, C., Pomar, F., Novo-Uzal, E., Merino, F. & de Ilarduya, O. M. The Ve-mediated resistance response of the tomato to Verticillium dahliae involves H2O2, peroxidase and lignins and drives PAL gene expression. Bmc Plant Biology. 10, 232 (2010).
Shittu, H., Castroverde, D., Nazar, R. & Robb, J. Plant-endophyte interplay protects tomato against a virulent Verticillium. Planta. 229, 415–426 (2009).
Larena, I., Sabuquillo, P. P. & De, C. A. Biocontrol of fusarium and verticillium wilt of tomato by Penicillium oxalicum under greenhouse and field conditions. Journal of Phytopathology. 151, 507–512 (2003).
Xu, W., Wang, Z. & Wu, F. Companion cropping with wheat increases resistance to Fusarium wilt in watermelon and the roles of root exudates in watermelon root growth. Physiological & Molecular Plant Pathology. 90, 12–20 (2015).
Xuepeng Fu, X. W., Xingang, Zhou, Shouwei, Liu, Yanhui, Shen & Fengzhi, Wu . Companion cropping with potato onion enhances the disease resistance of tomato against Verticillium dahliae. Frontiers in Plant Science 6, 726 (2015).
Boudreau, M. A. Diseases in Intercropping Systems. Annual Review of Phytopathology. 51, 499–519 (2013).
Bollig, K., Specht, A., Myint, S. S., Zahn, M. & Horst, W. J. Sulphur supply impairs spread of Verticillium dahliae in tomato. European Journal of Plant Pathology. 135, 81–96, (2013).
Williams, J. S., Hall, S. A., Hawkesford, M. J., Beale, M. H. & Cooper, R. M. Elemental sulfur and thiol accumulation in tomato and defense against a fungal vascular pathogen. Plant Physiology. 128, 150–159 (2002).
Ding, X. et al. Priming maize resistance by its neighbours: activating 1,4-benzoxazine-3-ones synthesis and defence gene expression to alleviate leaf disease. Frontiers in Plant Science. 6, 1–8 (2015).
Gao, X. et al. Root Interactions in a Maize/Soybean Intercropping System Control Soybean Soil-Borne Disease, Red Crown Rot. PLoS ONE. 9, e95031–e95031 (2014).
Rausch, T. & Wachter, A. Sulfur metabolism: a versatile platform for launching defence operations. Trends in Plant Science. 10, 503–509 (2005).
Resende, M. L. V. et al. Novel phytoalexins including elemental sulphur in the resistance of cocoa (Theobroma cocoa L.) to Verticillium wilt (Verticillium dahliae Kleb.). Physiological & Molecular Plant Pathology. 48, 347–359 (1996).
Cooper, R. M. et al. Detection and cellular localization of elemental sulphur in disease-resistant genotypes of Theobroma cacao. Nature. 379, 159–162 (1996).
Novo, M. et al. Sulphur accumulation after Verticillium dahliae infection of two pepper cultivars differing in degree of resistance. Plant Pathology. 56, 998–1004 (2007).
Klug, K. et al. Spatial gene expression analysis in tomato hypocotyls suggests cysteine as key precursor of vascular sulfur accumulation implicated in Verticillium dahliae defense. Physiologia Plantarum. 153, 253–268 (2015).
Dubuis, P. H., Marazzi, C. E. & Mauch, F. Sulphur Deficiency Causes a Reduction in Antimicrobial Potential and Leads to Increased Disease Susceptibility of Oilseed Rape. Journal of Phytopathology. 153, 27–36 (2005).
Freitas, A. S. et al. Spatial distribution of Yellow Sigatoka Leaf Spot correlated with soil fertility and plant nutrition. Precision Agriculture 17, 1–15 (2015).
Klikocka, H. Influence of NPK fertilization enriched with S, Mg, and micronutrients contained in liquid fertilizer Insol 7 on potato tubers yield (Solanum tuberosum L.) and infestation of tubers with Streptomyces scabies and Rhizoctonia solani. Journal of Elementology. 14, 271–288 (2009).
Zuo, Y. & Zhang, F. Iron and zinc biofortification strategies in dicot plants by intercropping with gramineous species. A review. Agronomy for Sustainable Development 29, 63–71 (2009).
Long, L., Li, S., Zhou, L. & Zhang, H. Dive rsity Enhances Agricultural Productivity via Rhizosphere Phosphorus Facilitation on Phosphorus-deficient Soils. Proceedings of the National Academy of Sciences. 104, 11192–11196 (2007).
Shao, H. B., Chu, L. Y., Lu, Z. H. & Kang, C. M. Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. International Journal of Biological Sciences. 4, 8–14 (2008).
Bolter, C., Brammall, R. A., Cohen, R. & Lazarovits, G. Glutathione alterations in melon and tomato roots following treatment with chemicals which induce disease resistance to Fusarium wilt. Physiological & Molecular Plant Pathology. 42, 321–336 (1993).
Boerjan, W., Ralph, J. & Baucher, M. Lignin biosynthesis. Annual Review of Plant Biology. 54, 519–546 (2003).
Howarth, J. R., Pierre, F., Jean-Claude, D., Smith, F. W. & Hawkesford, M. J. Cloning of two contrasting high-affinity sulfate transporters from tomato induced by low sulfate and infection by the vascular pathogen Verticillium dahliae. Planta. 218, 58–64 (2003).
Saito, K. Regulation of sulfate transport and synthesis of sulfur-containing amino acids. Current Opinion in Plant Biology. 3, 188–195 (2000).
Wang, K. L., Li, H. & Ecker, J. R. Ethylene biosynthesis and signaling networks. Plant Cell. 14 Suppl, S131–S151 (2002).
Li, L. et al. Diversity enhances agricultural productivity via rhizosphere phosphorus facilitation on phosphorus-deficient soils. Proceedings of the National Academy of Sciences of the United States of America. 104, 11192–11196 (2007).
Dobinson, K. F., Tenuta, G. K. & Lazarovits, G. Occurrence of race 2 of Verticillium dahliae in processing tomato fields in southwestern Ontario. Canadian Journal of Plant Pathology. 18, 55–58 (1996).
Mercado-Blanco, J., Rodrı́Guez-Jurado, D. et al. Suppression of Verticillium wilt in olive planting stocks by root-associated fluorescent Pseudomonas spp. Biological Control. 30, 474–486 (2004).
Wang, G. et al. Effect of soil type and soybean genotype on fungal community in soybean rhizosphere during reproductive growth stages. Plant & Soil. 317, 135–144 (2009).
Ae, N. & Johansen, C. Phosphorus uptake by pigeon pea and its role in cropping systems of the Indian subcontinent. Science. 248, 477 (1990).
Rodrigues et al. Silicon influences cytological and molecular events in compatible and incompatible rice-Magnaporthe grisea interactions. Physiological & Molecular Plant Pathology. 66, 144–159 (2005).
Wang, M., Wu, C., Cheng, Z. & Meng, H. Growth and physiological changes in continuously cropped eggplant (Solanum melongena L.) upon relay intercropping with garlic (Allium sativum L.). Frontiers in Plant Science. 6, 262 (2015).
Bestwick, C. S. & Mansfield, J. W. Localized changes in peroxidase activity accompany hydrogen peroxide generation during the development of a nonhost hypersensitive reaction in lettuce. Acm Sigcomm Computer Communication Review. 118, 1067–1078 (1998).
Zheng, L. J. & Zhong, G. PPO Activity Determination and Anti-browning Measure for Amorphophallus albus. Journal of Southwest University. 29, 118–121 (2007).
Zhang, S. Z., Hua, B. Z. & Zhang, F. Induction of the activities of antioxidative enzymes and the levels of malondialdehyde in cucumber seedlings as a consequence of Bemisia tabaci (Hemiptera: Aleyrodidae) infestation. Arthropod-Plant Interactions. 2, 209–213 (2008).
Yu, G. B. et al. Glutathione biosynthesis and regeneration play an important role in the metabolism of chlorothalonil in tomato. Chemosphere. 90, 2563–2570 (2012).
Pazourek, K. Estimation of cystine, methionine and hydroxyproline in feed hydrolysates using the T 339 amino acid analyzer. Biologizace A Chemizace Živočišné Výroby Veterinaria. (1990).
Kim, S. Y. et al. Effect of Steaming, Blanching, and High Temperature/High Pressure Processing on the Amino Acid Contents of Commonly Consumed Korean Vegetables and Pulses. Preventive Nutrition & Food Science. 19, 220–226 (2014).
Soon, Y. K., Kalra, Y. P. & Abboud, S. A. Comparison of some methods for the determination of total sulfur in plant tissues. Communications in Soil Science & Plant Analysis. 27, 809–818 (1996).
Cong, J., Wang, C. Z., Yan, L. I. & Ke, W. U. Investigation of Barium Sulfate Turbidimetry on Determination the Sulfated Group of Sulfated Polysaccharides. Pharmaceutical Journal of Chinese Peoples Liberation Army. 19, 181–183 (2003).
Rukun, Lu. Analytical methods for soil agrochemistry. Agricultural science and technology press. (2000).
Løvdal, T. & Lillo, C. Reference gene selection for quantitative real-time PCR normalization in tomato subjected to nitrogen, cold, and light stress. Analytical Biochemistry. 387, 238–242 (2009).
Yang, Y. X. et al. RNA-seq analysis reveals the role of red light in resistance against Pseudomonas syringae pv. tomato DC3000 in tomato plants. Bmc Genomics. 16, 1–16 (2015).
Pattemore, J. A. RNA extraction from cereal vegetative tissue. Methods in Molecular Biology. 1099, 17–21 (2014).
Abdin, M. Z. et al. Constitutive expression of high-affinity sulfate transporter (HAST) gene in Indian mustard showed enhanced sulfur uptake and assimilation. Protoplasma. 248, 591–600 (2010).
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (project No. 31672200), National Staple Vegetable Industrial Technology Systems of China (CARS-25-08), and Northeast Agricultural University Innovation Foundation for Postgraduate (yjscx14074). The authors kindly thank those agencies for financing the research study.
Author information
Authors and Affiliations
Contributions
F.Z.W., X.G.Z. and X.P.F. designed the experiment; X.P.F. executed the experiments and drafted the manuscript; and C.X.L. assisted with the execution of the experiments. S.W.L. provided plant materials. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Fu, X., Li, C., Zhou, X. et al. Physiological response and sulfur metabolism of the V. dahliae-infected tomato plants in tomato/potato onion companion cropping. Sci Rep 6, 36445 (2016). https://doi.org/10.1038/srep36445
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36445
This article is cited by
-
Impact of Leifsonia xyli subsp. xyli titer on nutritional status, and metabolism of sugar cane
Plant and Soil (2023)
-
Chromosome-level genome assembly of bunching onion illuminates genome evolution and flavor formation in Allium crops
Nature Communications (2022)
-
Integrative transcriptomic and gene co-expression network analysis of host responses upon Verticillium dahliae infection in Gossypium hirsutum
Scientific Reports (2021)
-
Genomic and transcriptomic insights into Raffaelea lauricola pathogenesis
BMC Genomics (2020)
-
Machine learning approaches reveal genomic regions associated with sugarcane brown rust resistance
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.