Abstract
The morbidity and mortality of patients with ischemic cardiomyopathy resulted from ischemia/reperfusion injury are very high. The present study investigates whether our previously synthesized water-soluble phosphate prodrug of acacetin was cardioprotective against ischemia/reperfusion injury in an in vivo rat model. We found that intravenous administration of acacetin prodrug (10 mg/kg) decreased the ventricular arrhythmia score and duration, reduced ventricular fibrillation and infarct size, and improved the impaired heart function induced by myocardial ischemia/reperfusion injury in anesthetized rats. The cardioprotective effects were further confirmed with the parent compound acacetin in an ex vivo rat regional ischemia/reperfusion heart model. Molecular mechanism analysis revealed that acacetin prevented the ischemia/reperfusion-induced reduction of the anti-oxidative proteins SOD-2 and thioredoxin, suppressed the release of inflammation cytokines TLR4, IL-6 and TNFα, and decreased myocyte apoptosis induced by ischemia/reperfusion. Our results demonstrate the novel evidence that acacetin prodrug confer significant in vivo cardioprotective effect against ischemia/reperfusion injury by preventing the reduction of endogenous anti-oxidants and the release of inflammatory cytokines, thereby inhibiting cardiomyocytes apoptosis, which suggests that the water-soluble acacetin prodrug is likely useful in the future as a new drug candidate for treating patients with acute coronary syndrome.
Similar content being viewed by others
Introduction
Ischemic cardiomyopathy resulted from coronary artery disease is the most common cause of mortality worldwide1,2. Although timely myocardial reperfusion using either thrombolytic therapy or primary percutaneous coronary intervention (PPCI) is the effective therapy in limiting myocardial infarct (MI) size, preserving left-ventricular systolic function and reducing the onset of heart failure, the morbidity and mortality of patients with ischemic cardiomyopathy are significant concerns due to the myocardial reperfusion injury3,4,5. The blood reflow (reperfusion) of the ischemic myocardium induces oxidative stress and a series of intracellular responses, which may trigger life threatening ventricular fibrillation (VF), and may be accompanied with acute inflammatory responses, metabolic disorder, apoptosis and necrosis, and followed by cardiac remodelling and dysfunction6,7.
Earlier studies on ischemia/reperfusion injury focusing on preconditioning and post-conditioning cardioprotection3,8 have identified several intracellular molecular signals involved in ischemia/reperfusion injury (e.g. ROS, TNFα, polymorphonuclear leukocytes infiltration, apoptosis signals, etc.) and mobilization of endogenous cardioprotective mechanisms (e.g. adenosine, bradykinin, and opioid peptides, SOD-2, etc.)7,9. A number of pharmacological agents (e.g. adenosine, beta-blockers, nicorandil, cyclosporine, etc.) have been applied in patients with ischemic heart disorders before thrombolytic therapy or PPCI to reduce reperfusion injury10; however, the overall results in clinical trials with the pharmacological recruitment of cardioprotective signaling are not satisfactory, thus no ideal drug is yet available for preventing myocardial reperfusion injury yet4. New therapeutic interventions including effective drug therapy are therefore required to improve clinical outcomes5,11.
In the previous reports, we found that natural flavone acacetin from the Chinese medicinal herb Tianshanxuelian inhibits atrial IKur, IKACh, and Ito12,13,14 and prevents the induction of experimental atrial fibrillation in anesthetized canine12. We have recently synthesized a water-soluble phosphate prodrug of acacetin, and found that this acacetin prodrug can effectively terminate atrial fibrillation induced by vagal stimulation and burst pacing in beagle dogs after intravenous administration15. In the present study, we investigate whether this water-soluble acacetin prodrug is cardioprotective against ischemia/reperfusion injury. Our results demonstrated that intravenous administration of acacetin prodrug was very effective in reducing ventricular arrhythmias and myocardial infarct size induced by ischemia/reperfusion injury in anesthetized rats. Molecular mechanism analysis in ex vivo rat hearts revealed that acacetin prevented ischemia/reperfusion reduction of mitochondrial anti-oxidative kinases SOD-2 and thioredoxin as well as reduced inflammatory cytokines TLR4, IL-6 and TNFα, thereby inhibited myocyte apoptosis induced by ischemia/reperfusion.
Results
In vivo conversion of acacetin prodrug in rats
Our recent study has demonstrated that the custom-synthesized acacetin prodrug can be converted to the parent compound acacetin in in vivo beagle dogs15. The in vivo conversion was also confirmed in anesthetized rats. Figure 1 illustrates the representative high-performance liquid chromatography (HPLC) in plasma from an anesthetized rat with intravenous administration (250 μg/kg/min) of acacetin prodrug. The mobile phase containing 2 μg/mL acacetin showed the peak of acacetin on HPLC at 10–11 min. Acacetin was not detected in rat blood plasma before administration of acacetin prodrug (Fig. 1B), and significant plasma level of acacetin was observed at 5 min (Fig. 1C) and 40 min (Fig. 1D) with acacetin prodrug administration. These results indicate that acacetin prodrug is successfully converted in vivo into acacetin as reported previously in beagle dogs15.
HPLC result for in vivo conversion of the water-soluble acacetin prodrug to the parent compound acacetin in a rat after intravenous administration.
(A) Acacetin (2 μg/mL) in HPLC mobile phase. (B) HPLC with blank rat plasma (before administration of acacetin prodrug). (C) HPLC with rat plasma at 5 min after intravenous administration of acacetin prodrug (250 μg/kg/min). (D) HPLC with rat plasma at 40 min after intravenous administration of acacetin prodrug.
In vivo cardioprotection of acacetin prodrug against ischemia/reperfusion injury in rats
The effect of acacetin prodrug on ventricular arrhythmias induced by ischemia/reperfusion injury was determined in anesthetized rats. Ventricular arrhythmias including premature ventricular beats, ventricular tachycardia, and ventricular fibrillation were induced by ligating the left anterior descending (LAD) artery for 10 min, followed by a 10 min reperfusion as described previously16. Acacetin prodrug (5 mg, 10 mg and 20 mg/kg) or equivolume vehicle (5% glucose solution) was intravenously administered for 5 min before the artery ligation.
Ventricular fibrillation was frequently observed during the early reperfusion in control animals treated with intravenous vehicle (Fig. 2A), but not in animals treated intravenously with acacetin prodrug (Fig. 2B). Acacetin prodrug decreased the arrhythmia score (Fig. 2C) and arrhythmia duration (Fig. 2D) in a dose-dependent manner (n = 10–11, P < 0.05 or P < 0.01 vs. vehicle). Importantly, incidence of ventricular fibrillation was reduced by 26%, 67%, and 78% with bolus acacetin prodrug of 5, 10 and 20 mg/kg (Fig. 2E, P < 0.05 for 10 mg/kg or 20 mg/kg vs. vehicle).
Anti-arrhythmic effect of prodrug in anesthetized rats with ischemia/reperfusion.
(A) ECG in a rat treated with vehicle, showing ventricular fibrillation (VF) after reperfusion for 1 min. (B) ECG in a rat treated with bolus acacetin prodrug (10 mg/kg i.v. for 5 min) before ischemia, showing a brief ventricular tachycardia (VT) followed by normal rhythm after reperfusion. (C) Reduction of the ventricular arrhythmia score by acacetin prodrug. (D) Decrease of ventricular arrhythmia duration by acacetin prodrug. (E) Reduction of ventricular fibrillation (but not PVBs, premature ventricular beats) by acacetin prodrug in a concentration-dependent manner (n = 10–11 experiments, *P < 0.05, **P < 0.01 vs. vehicle).
The anti-infarct effect of acacetin prodrug was determined in anesthetized rats with 30 min LAD artery ligation, followed by 120 min reperfusion. Acacetin prodrug was administered by intravenous injection (10 mg/kg) before 10 min of the LAD artery ligation. Ventricular infarct size was remarkably reduced by 68.7% in rats treated with acacetin prodrug (Fig. 3A, n = 8 for each group, P < 0.001 vs. vehicle).
Intravenous administration of prodrug improves the impaired heart rate and ventricular contractile function induced by ischemia/reperfusion injury in anesthetized rats.
(A) Left ventricular slices with the TTC-staining, showing a reduced infarct area in animal treated with acacetin prodrug (10 mg/kg intravenous administration). (B) Acacetin prodrug antagonizes the reduction of mean femoral artery blood pressure by ischemia/reperfusion injury. (C) Acacetin prodrug slightly antagonizes the heart rate reduction by ischemia/reperfusion injury. (D) Acacetin prodrug improves the impaired systolic pressure of left ventricle (LVSP). (E) Acacetin prodrug improves the impaired developed pressure of left ventricle (LVDP). (F) Acacetin prodrug antagonizes the reduced left ventricular +dP/dT (n = 8 experiments, **P < 0.01, ***P < 0.001 vs. vehicle).
Moreover, the prodrug prevented the reduction of the mean femoral artery blood pressure (Fig. 3B) and the heart rate (Fig. 3C). It significantly improved the impaired ventricular contractile function, i.e. left ventricular systolic pressure (Fig. 3D, P < 0.01 or P < 0.001 vs. vehicle), developed pressure (Fig. 3E, P < 0.01 or P < 0.001 vs. vehicle), and +dP/dT (Fig. 3F, P < 0.01 or P < 0.001 vs. vehicle), induced by ischemia/reperfusion injury. These results show that acacetin prodrug is very effective in inhibiting ventricular arrhythmias, reducing infarct size, and preserving heart function in rat in vivo ischemia/reperfusion model. These results also indicate that acacetin prodrug is converted to the parent compound acacetin and exerts significant cardioprotective action against ischemia/reperfusion injury.
Ex vivo cardioprotection of acacetin against ischemia/reperfusion injury
To determine the potential molecular mechanisms underlying cardioprotection of acacetin prodrug, the parent compound acacetin was used to test the effect on myocardial ischemia/reperfusion injury in isolated rat hearts. The perfused hearts were subjected to LAD artery ligation for 30 min followed by 120 min reperfusion. The hearts were treated with different concentrations of acacetin before (10 min) and during the regional ischemia (Fig. 4A). The infarct area assessed by Evans blue/TTC staining in myocardial slices showed that a sizeable infarct area was present in the heart treated with vehicle, and the infarct size was remarkably reduced in the heart treated with 3 μM acacetin (Fig. 4B). The mean values (Fig. 4C) of the infarct area induced by ischemia/reperfusion injury was decreased by 36%, 54% and 73% respectively with 0.3, 1 and 3 μM acacetin (n = 10–11, P < 0.05 or P < 0.001 vs. vehicle). Significant reduction of the infarct size was observed at concentration as low as 0.3 μM acacetin. In addition, the heart function was also evaluated in isolated rat hearts subjected to ischemia/reperfusion injury (Fig. 5). LVP and +dP/dT was significantly reduced by ischemia/reperfusion. Acacetin at 3 μM acacetin significantly improved the impaired heart function by ischemia/reperfusion (P < 0.05 or P < 0.001 vs. vehicle).
Effects of acacetin on myocardial injury by ischemia/reperfusion in ex vivo rat hearts.
(A) The scheme of ischemia/reperfusion protocol in perfused ex vivo rat hearts with 30 min ischemia, followed by 2 h reperfusion. (B) Left ventricular slices of the heart with Evans blue/TCC-staining in a vehicle control heart and a heart treated with 3 μM acacetin. (C) Mean percentage of risk area and infarct area of left ventricle with ischemia/reperfusion injury, showing reduction of infarct area in hearts treated with 0.3, 1 or 3 μM in a concentration-dependent manner (n = 10–12 experiments, **P < 0.01, ***P < 0.001 vs. vehicle; #P < 0.05, ##P < 0.01 vs. 0.3 μM acacetin).
Effects of acacetin on heart rate and ventricular pressure in ex vivo rat hearts subjected to ischemia/reperfusion.
(A) Percentage values of heart rate during control, ischemia and reperfusion in isolated rat hearts treated with vehicle, 0.3, 1 or 3 μM acacetin. (B) Percentage values of left ventricular pressure (LVP) during control, ischemia and reperfusion in isolated rat hearts treated with vehicle, 0.3, 1 or 3 μM acacetin (n = 9–10 for each treatment, ##P < 0.01 vs control; *P < 0.05, **P < 0.01 vs. vehicle). (C) Percentage values of +dP/dT of left ventricular pressure during control, ischemia and reperfusion in isolated rat hearts treated with vehicle, 0.3, 1 or 3 μM acacetin (n = 9–10 for each treatment, ##P < 0.01 vs control; *P < 0.05, **P < 0.01 vs. vehicle).
It is well documented that myocardial ischemia/reperfusion injury induces intracellular oxidative stress, triggers inflammation, and initiates apoptosis signals to mediate the subsequent myocardial death6,7,17. Therefore, we determined whether the remarkable cardioprotection of acacetin involves anti-oxidation, anti-inflammation or anti-apoptosis.
Molecular mechanisms of cardioprotection of acacetin against myocardial ischemia/reperfusion injury
Previous studies demonstrated that the anti-oxidative proteins SOD (superoxide dismutase) and/or thioredoxin are decreased in myocardial ischemia/reperfusion injury or other oxidation stress18,19. Similarly, in isolated hearts with ischemia/reperfusion injury, we also found (Fig. 6A,B) that SOD-2 and thioredoxin were remarkably reduced in the left ventricle of rat hearts subjected to 30 min ischemia followed by 2 h reperfusion (n = 4, P < 0.01 vs. sham group). Interestingly, acacetin treatment prevented SOD-2 and thioredoxin reduction induced by ischemia/reperfusion injury (n = 4, P < 0.05 or P < 0.01 vs. vehicle), indicating that cardioprotective effects of acacetin involve the retention of endogenous anti-oxidants.
Effects of acacetin on endogenous anti-oxidant and inflammation response in ex vivo rat hearts with ischemia/reperfusion injury.
(A) Western blots of superoxide dismutase 2 (SOD-2) and thioredoxin (Trx). (B) Mean relative levels of SOD-2 and Trx. (C) Western blots of Toll like receptor-4 (TLR-4), interleukin-6 (IL-6), and tumour necrosis factor alpha (TNFα) in rat left ventricles with ischemia/reperfusion injury in the absence and the presence of 0.3, 1 and 3 μM acacetin. (D) Mean relative levels of TLR-4, IL-6 and TNFα, showing significant reduction of TLR-4, IL-6 and TNFα in hearts treated with acacetin (0.3, 1 and 3 μM). n = 4 experiments, #P < 0.05, ##P < 0.01 vs. Sham; *P < 0.05, **P < 0.01 vs. vehicle.
The release of inflammation mediators (TLR-4, IL-6, TNFα, etc.) has been found to play an important role in mediating and exacerbating myocardial ischemia/reperfusion injury7,17. Figure 6C,D show that TLR-4, IL-6 and TNFα were remarkably increased in ex vivo rat hearts subjected to ischemia/reperfusion injury (n = 4, P < 0.01 vs. sham group), and acacetin decreased the inflammatory responses in a concentration-dependent manner (n = 4 for each group, P < 0.05 or P < 0.01 vs. vehicle).
The potential involvement of molecular mechanism of anti-apoptosis by acacetin was further determined in ex vivo rat hearts subjected to ischemia/reperfusion insult. The pro-apoptotic proteins cleaved caspase-3 and Bax (Bcl-2-associated X), as well as the anti-apoptotic protein Bcl-2 (B-cell lymphoma 2) were examined in hearts treated with vehicle, 0.3, 1, or 3 μM acacetin. Figure 7A,B show that ischemia/reperfusion increased the pro-apoptotic cleaved caspase-3 and Bax and decreased the anti-apoptotic Bcl-2 (n = 4, P < 0.01 vs. sham). Acacetin significantly antagonized the decrease of anti-apoptotic molecule (Bcl-2) and the increase of pro-apoptotic molecules (Bax and caspase-3) induced by ischemia/reperfusion injury (n = 4, P < 0.05 or P < 0.01 vs. vehicle). The reduced Bcl-2/Bax ratio was reversed by acacetin treatment in a concentration-dependent manner (Fig. 7C, n = 4, P < 0.05 or P < 0.01 vs. vehicle). These results indicate that acacetin also has a secondary anti-apoptosis effect in addition to its anti-oxidation and anti-inflammation cardioprotective properties.
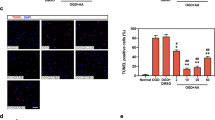
Effects of acacetin on myocardial apoptosis induced by ischemia/reperfusion in ex vivo rat hearts.
(A) Western blots for expression of the pro-apoptotic proteins cleaved caspase-3, Bax and the anti-apoptotic protein Bcl-2 in the absence and the presence of 0.3, 1 and 3 μM acacetin. (B) Mean relative protein levels. (C) Ratio of Bcl-2/Bax. n = 4 experiments, #P < 0.05, ##P < 0.01 vs. sham; *P < 0.05, **P < 0.01 vs. vehicle. (D) TUNEL-staining indicates significant myocyte apoptosis with dark-brown nuclei; acacetin decreases apoptosis. (E) Mean percentage of nuclei with TUNEL-staining in hearts treated with vehicle or 3 μM acacetin (n = 5 experiments, ##P < 0.01, vs. sham; **P < 0.01 vs. vehicle).
The TUNEL-staining for detecting apoptotic cells in left ventricular slices (Fig. 7D) shows that the increase of TUNEL-positive nuclei cells by ischemia/reperfusion injury was reduced in hearts treated with 3 μM acacetin (Fig. 7E). The TUNEL-positive nuclei was 51.1 ± 3.8% in vehicle control hearts, and was reduced to 20.2 ± 4.1% in acacetin-treated hearts (n = 5, P < 0.01 vs. vehicle), indicating that acacetin inhibits myocytes apoptosis thereby decreases myocardial infarct size in rat hearts subjected to ischemia/reperfusion injury. The histological damage induced by ischemia/reperfusion was also attenuated in hearts treated with 3 μM acacetin (Fig. S1).
Discussion
In the present study, we have demonstrated for the first time that the water-soluble prodrug of the natural flavone acacetin is converted into the parent compound acacetin and exerts a remarkable cardioprotective effect in anesthetized rats subjected to ischemia/reperfusion injury: reducing ventricular arrhythmia score and duration, ventricular fibrillation and myocardial infarct size and improving heart function. Molecular mechanisms of acacetin for cardioprotection determined in ex vivo rat hearts with ischemia/reperfusion injury reveals that acacetin prevents ischemia/reperfusion-induced reduction of anti-oxidants, decreases inflammation responses, and inhibits myocardial apoptosis.
Acacetin is a small molecule flavone (5,7-dihydroxy-4′-methoxyflavone) universally present in various plants and responsible for many of the colours in nature20. In addition to our previous reports of atrial-selective ion channels (IKur, IKACh, and Ito) blockade and anti-atrial fibrillation effect12,13,14, acacetin has been reported to have anti-cancer properties21, to reduce the airway hyper-responsiveness with dietary administration22, and to alleviate telomeric position effect in human cells23. The present study demonstrates that acacetin has the novel therapeutic cardioprotection against ischemia/reperfusion injury.
It has been well documented that myocardial ischemia/reperfusion (oxidative stress) stimulates the generation of reactive oxygen species (ROS) involved in myocardial injury3. These reactive oxygen species injure membrane lipids, protein, carbohydrates and DNA, and cause widespread inflammation associated with tissue damage24,25, which is an active process of cardiomyocyte apoptosis and/or necrosis26,27. Therefore, scavenging oxidative species or enhancing endogenous antioxidants could prevent the myocardial injury induced by oxidation. Cardioprotection relies on a complex signal transduction of triggers, mediators and effectors, which vary between different species28,29. It is generally believed that the main endogenous antioxidant is superoxide dismutase (SOD), which catalyses O2 reduction to produce H2O2. In patients and animals with myocardial ischemia/reperfusion, tumour necrosis factor-alpha (TNFα) and interleukin-6 (IL-6) were increased with other inflammatory cytokines30.
It should be noted that TNFα has an ambivalent role in myocardial ischemia/reperfusion injury. Both local ischemic conditioning and remote ischemic postconditioning are related to the decrease of the endogenous TNFα induced by myocardial ischemia/reperfusion in rodents31. On the other hand, evidence that ischemic preconditioning protection is lost in TNFα-KO mice and exogenous TNFα mimics the ischemia-preconditioning cardioprotection when administered prior to ischemia suggest that TNFα acts as a trigger of preconditioning32,33. Exogenous TNFα has been found to have biphasic effects: it is cardioprotective at low concentrations and acts as a signaling molecule, but it induces irreversible cell damage and increases infarct size at higher concentrations32,34,35.
It is well recognized that the overall preservation of cardiac structure and function is critical for cardioprotection against ischemia/reperfusion injury by inhibiting oxidative stress and inflammation and therefore decreasing apoptosis17,36,37. Earlier studies reported that preserving anti-oxidant systems using recombinant SOD38,39 or thioredoxin40,41,42 had significant cardioprotection against ischemia/reperfusion injury. Consistent with the previous reports3,7, the present study also showed that the endogenous antioxidant proteins SOD-2 and thioredoxin, and the anti-apoptotic Bcl-2 were remarkably reduced in rat hearts subjected to ischemia/reperfusion injury, while pro-inflammatory factors Toll-like receptor 4 (TLR-4)43, IL-6 and TNFα, the pro-apoptotic molecules cleaved caspase-3 and Bax were significantly increased by ischemia/reperfusion injury. It is interesting to note that acacetin treatment remarkably prevented the ischemia/reperfusion–induced reduction of SOD-2 and Bcl-2, preserved their activities, and thereby inhibited inflammation mediators (TLR-4, IL-6, and TNFα) and cardiomyocyte apoptosis and decreased the myocardial infarct size (Fig. 8). The preservation of the anti-oxidant systems (SOD and thioredoxin) may be the key effect of acacetin for cardioprotection against ischemia/reperfusion injury.
Schematic illustration shows the main cascade of molecules in myocardial ischemia/reperfusion injury.
Myocardial ischemia/reperfusion (oxidative stress) triggers ROS production and inflammation response (release of pro-inflammatory cytokines TLR-4, IL-6, TNFα, which induce ventricular arrhythmias) and decreases the endogenous anti-oxidants SOD and thioredoxin (Trx). Over-production of ROS and inflammation cytokines would initiate cardiomyocyte apoptosis cell death and cause myocardial infarct. Intravenous application of acacetin prodrug (bioconversion to acacetin) prevents the ischemia/reperfusion-induced reduction of the endogenous anti-oxidants and also reduces inflammation response, and thereby inhibits ventricular arrhythmias and myocardial apoptosis and significantly reduces infarct size.
It is generally recognized that cardiomyocytes suffer irreversible injury primarily during ischemia/reperfusion, the window time is important for clinical use of cardioprotective drugs to minimize additional cardiomyocyte loss during reperfusion. A large number of studies have recommended introducing protective drugs at the very beginning of reperfusion to reduce infarct size44. It is believed that the protective effect will be enhanced if cardioprotection is initiated before or during ischemia or as soon as possible after reperfusion, especially with longer duration of ischemia4,5. Although a number of clinical studies on cardioprotection were conducted with promising pharmaceutical agents, including the endogenous cytoprotective molecule adenosine, the mitochondrial permeability transition pore formation blocker cyclosporine A45, and the vasodilators nicorandil and nitrates46, beta-blockers4,5,11, the long term results were not satisfactory. Therefore, more effort is required to develop new cardioprotective strategies to improve the outcomes of acute myocardial infarction4,5,11. The present study demonstrated that acacetin prodrug has significant cardioprotection against ischemia/reperfusion injury by reducing ventricular fibrillation and infarct size and preserving ventricular contractile function.
Although the present study indicates that acacetin prodrug is likely a promising drug candidate to rescue the myocardial injury on reperfusion, the limitation is that all the results were obtained in rodents. Further experimental study is required in large animals (e.g. swine and/or canine) to gain the preclinical data which can be referred for clinical trials in the near future.
Conclusion
The present study demonstrates for the first time that acacetin prodrug confers significant cardioprotective effect against ischemia/reperfusion injury after converting into the parent compound acacetin by preventing ischemia/reperfusion-induced decrease of anti-oxidants, thereby reducing inflammatory response and inhibiting myocardial apoptosis and infarct size, which suggests that acacetin prodrug may improve the long term outcomes of patients undergoing thrombolytic therapy or PPCI.
Methods and Materials
Animal experiments
Sprague Dawley (SD) rats were employed in the present study following the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996), and the experimental procedure was approved by the Committee on the Use of Live Animals in Teaching and Research of the University of Hong Kong.
Blood sample collection for determining prodrug conversion in anesthetized rats with HPLC analysis
Adult male SD rats (250–300 g) was anesthetized with pentobarbital (50 mg/kg, i.p.), and the jugular vein and femoral vein were cannulated for drug administration and blood sample collection respectively. The water-soluble acacetin prodrug (Fig. S2) with purity of 99% (Fig. S3) was continuously administered for 40 min (250 μg/kg/min), and blood sample (~0.2 mL) was collected in heparinized tubes at 0 (pre-dose) and 5, 10, and 40 min during drug administration for HPLC analysis of acacetin prodrug conversion.
The simple liquid-liquid extraction with methanol was used for sample preparation. Pentamethylquercetin was selected as internal standard. The analysis was carried out on a Waters HPLC system (Milford, MA) equipped with an Alltech column (C18, 250 mm × 4.6 mm i.d., 5 μm, Grace, Deerfield, IL). The mobile phase consisted of methanol-water-phosphoric acid (70:30:0.15), and the flow rate was 1.0 mL/min. UV absorption was monitored at 260 nm.
Myocardial ischemia/reperfusion model in anesthetized rats
Ischemia-reperfusion model in anesthetized rats was established as described previously16. Adult male SD rats were anesthetized with pentobarbital (50 mg/kg, i.p.) and, supplemented during the experiment when needed. The animals were intubated and ventilated with room air. Body temperature was maintained at 37 °C with a temperature control system. The jugular vein was cannulated for drug administration and a PE-50 catheter was introduced into the left ventricle through right carotid artery to measure ventricular contractile function (e.g. left ventricular pressure). Femoral artery was cannulated to measure blood pressure. Lead II ECG was monitored during the experiment with a data acquisition system (RM6240, Chengdu Instrument Ltd, Chengdu, China). Regional ischemia was achieved by ligating LAD artery using a 5–0 silk suture with a section of silica gel tubing. Myocardial ischemia was confirmed by regional cyanosis and ST-segment elevation.
After 10-min stabilization, acacetin prodrug (5 mg, 10 mg and 20 mg/kg) or equivolume vehicle (5% glucose solution) was intravenously administered for 5 min before LAD artery was ligated for 10 min, followed by a 10-min reperfusion. Cardiac injury was characterized by a high incidence of ventricular arrhythmias during reperfusion in this model. Definitions of arrhythmias were based on the criteria of the Lambeth Conventions, including premature ventricular contractions (PVCs), ventricular tachycardia (VT), and ventricular fibrillation (VF)47. An arrhythmia score was calculated with scoring system as previously described: [(Log10PVCs) + 2 (Log10 episodes of VT) + 2 (Log10 episodes of VF) + (Log10 total duration of VF)]48,49.
To evaluate the ischemia/reperfusion-induced myocardial infarct size, rats were anesthetized and subjected to 30 min LAD artery ligation and followed by 120 min reperfusion. Acacetin prodrug (10 mg/kg, i.v.) was continuously administered for 10 min before LAD artery ligation and during 30 min ischemia. Lead II ECG, blood pressure, and left ventricular pressure were monitored and recorded on an IBM compatible PC computer using a data acquisition system (RM6240, Chengdu Instrument Ltd, Chengdu, China) for offline analysis.
Ex vivo rat myocardial ischemia/reperfusion model
Adult male SD rats were anesthetized, their hearts were isolated and retrogradely perfused on a Langendorff heart perfusion apparatus at 37 °C with modified Krebs-Henseleit solution (in mM): NaCl 118.5, KCl 4.7, KH2PO4 1.18, MgSO4 1.18, NaHCO3 25, glucose 11.1, CaCl2 2.5) gassed with 95% O2 and 5% CO2. Polyethylene tubing with a cling film balloon tip filled with water and connected to a pressure transducer was inserted into the left ventricle through a small incision in the left atrium to measure left ventricular pressure. ECG was recorded with two electrodes placed on heart surface and the physiological signals were saved on an IBM compatible PC computer using the data acquisition system (Chengdu Instrument Ltd).
Regional ischemia was achieved by ligating LAD artery using a 5–0 silk suture with a section of silica gel tubing. After 20-min equilibration, the hearts were treated by vehicle (DMSO) or different concentrations of acacetin (0.3, 1 or 3 μM) for 10 min, and during LAD artery ligation (30-min), followed by 2 hr reperfusion. Myocardial ischemia was confirmed by a decrease in left ventricular developed pressure (LVDP) and specific ST-segment elevation on ECG. The hearts were used to perform histological and molecular analysis at end of 2 hr reperfusion.
Data were discarded if one of the following criteria appeared: 1) LVDP was below 80 mmHg during equilibration, 2) lack of evidence of successful LAD occlusion, and 3) hearts with irreversible arrhythmia during equilibration or after ischemia.
Myocardial infarct size assessment
At end of reperfusion, LAD artery was re-occluded at the same location. 0.5% solution of Evans blue dye was infused via aorta to differentiate the area at risk from the non-ischemic zone. The heart was then rapidly excised and frozen at −20 °C for 2–3 hr, and cut into 2-mm transverse slices from apex to base. The slices were incubated in 1% triphenyltetrazolium chloride (TTC) in sodium phosphate buffer (pH 7.4) for 20 min at 37 °C and fixed in 10% formalin.
After Evans blue/TTC staining, myocardial ischemic risk area was identified as the myocardial tissue without blue staining and presented as a percentage of the total left ventricular area. Viable tissue in the risk area (deep red) was stained by TTC, while infarcted area was clearly visible as TTC-negative (pale) and expressed as a percentage of total myocardial risk area. Images of the heart slices were taken and size of risk area and infarct area was measured by Image J software.
Histopathological analysis
After the reperfusion, the heart was fixed with 10% buffered formalin, embedded in paraffin, and then sectioned into 5 μm slices. The tissue slices were stained with haematoxylin-eosin, and analysed by light microscopy.
Terminal deoxynucleotidyl transferase–mediated dUTP nick end labeling (TUNEL) assay
TUNEL staining of myocardial slices was performed to detect myocyte apoptosis in myocardium using an in situ cell death detection kit, POD (Roche Applied Sciences, Mannheim, Germany) following the manufacturer’s instruction. Samples were counterstained with haematoxylin prior to analysis by light microscope. Quantitative analysis of TUNEL-positive cells was performed in 10 random fields at 100× magnification on three different sections from the peri-infarction zone in each animal as reported previously50,51. Apoptotic myocytes were expressed as percentage of total cells in the field.
Western blotting analysis
The related proteins were determined with Western blotting analysis as described previously52. Myocardial tissue samples from the ischemic zone were homogenized with an ice cold modified RIPA buffer. Protein concentration was determined using a Bio-Rad protein assay (Bio-Rad Laboratories, Hercules, CA). Equal concentrations of proteins were mixed with SDS sample buffer and denatured at 95 °C for 5 min. The samples were resolved with 8% SDS–page gels which were then transferred onto nitrocellulose membranes. The membranes were blocked with 5% fat-free milk in 0.1% Tris-buffered saline with Tween (TTBS) for 2 hours and then probed with primary antibodies: anti-SOD-2, thiredoxin, TLR-4, TNFα, caspase-3, Bax and Bcl-2 antibodies (Santa Cruz Biotechnology, USA); anti-IL-6 antibody (Abcam, UK) at 4 °C overnight. After being washed for three times with TTBS, the membranes were incubated with secondary antibodies (Santa Cruz Biotechnology) in TTBS at room temperature for 2 hours. Membranes were washed again with TTBS three times and then visualized on X-ray films using a chemo-luminescence detection system (ECL, GE Healthcare). The housekeeping protein β-actin (Santa Cruz Biotechnology) was used to protein loading control. The relative band intensities were measured by image analysis software Gel-Pro Analyser.
Statistical analysis
Group data are expressed as mean ± S.E.M. The incidence of arrhythmias was analyzed using the Fisher exact test. Arrhythmia scores were analyzed using one-way ANOVA followed by a Tukey post hoc test. Quantitative data were analyzed using Student’s t-test for paired or unpaired observations to evaluate significant differences between two group means, and ANOVA for multiple groups. A value of P < 0.05 was taken to indicate statistical significance.
Additional Information
How to cite this article: Liu, H. et al. Water-soluble acacetin prodrug confers significant cardioprotection against ischemia/reperfusion injury. Sci. Rep. 6, 36435; doi: 10.1038/srep36435 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Kovacic, J. C. & Fuster, V. From treating complex coronary artery disease to promoting cardiovascular health: therapeutic transitions and challenges, 2010–2020. Clin Pharmacol Ther 90, 509–518 (2011).
Frohlich, G. M., Meier, P., White, S. K., Yellon, D. M. & Hausenloy, D. J. Myocardial reperfusion injury: looking beyond primary PCI. Eur Heart J 34, 1714–1722 (2013).
Frank, A. et al. Myocardial ischemia reperfusion injury: from basic science to clinical bedside. Semin Cardiothorac Vasc Anesth 16, 123–132 (2012).
Hausenloy, D. J. & Yellon, D. M. Myocardial ischemia-reperfusion injury: a neglected therapeutic target. J Clin Invest 123, 92–100 (2013).
Gerczuk, P. Z. & Kloner, R. A. An update on cardioprotection: a review of the latest adjunctive therapies to limit myocardial infarction size in clinical trials. J Am Coll Cardiol 59, 969–978 (2012).
Eltzschig, H. K. & Eckle, T. Ischemia and reperfusion-from mechanism to translation. Nat Med 17, 1391–1401 (2011).
Vilahur, G. et al. Molecular and cellular mechanisms involved in cardiac remodeling after acute myocardial infarction. J Mol Cell Cardiol 50, 522–533 (2011).
Sharma, V., Bell, R. M. & Yellon, D. M. Targeting reperfusion injury in acute myocardial infarction: a review of reperfusion injury pharmacotherapy. Expert Opin Pharmacother 13, 1153–1175 (2012).
Penna, C., Perrelli, M. G. & Pagliaro, P. Mitochondrial pathways, permeability transition pore, and redox signaling in cardioprotection: therapeutic implications. Antioxid Redox Signal 18, 556–599 (2013).
Sanz-Rosa, D., Garcia-Prieto, J. & Ibanez, B. The future: therapy of myocardial protection. Ann N Y Acad Sci 1254, 90–98 (2012).
Heusch, G. Cardioprotection: chances and challenges of its translation to the clinic. Lancet 381, 166–175 (2013).
Li, G. R. et al. Acacetin, a natural flavone, selectively inhibits human atrial repolarization potassium currents and prevents atrial fibrillation in dogs. Circulation 117, 2449–2457 (2008).
Wu, H. J. et al. Acacetin causes a frequency- and use-dependent blockade of hKv1.5 channels by binding to the S6 domain. J Mol Cell Cardiol 51, 966–973 (2011).
Wu, H. J. et al. Properties and molecular determinants of the natural flavone acacetin for blocking hKv4.3 channels. PLoS One 8, e57864 (2013).
Liu, H. et al. Synthesis of a highly water-soluble acacetin prodrug for treating experimental atrial fibrillation in beagle dogs. Sci Rep 6, 25743 (2016).
Feng, M. et al. Activation of epidermal growth factor receptor mediates reperfusion arrhythmias in anaesthetized rats. Cardiovasc Res 93, 60–68 (2012).
Eefting, F. et al. Role of apoptosis in reperfusion injury. Cardiovasc Res 61, 414–426 (2004).
Yu, Q. J. & Yang, Y. Function of SOD1, SOD2, and PI3K/AKT signaling pathways in the protection of propofol on spinal cord ischemic reperfusion injury in a rabbit model. Life Sci 148, 86–92 (2016).
D’Annunzio, V., Perez, V., Boveris, A., Gelpi, R. J. & Poderoso, J. J. Role of thioredoxin-1 in ischemic preconditioning, postconditioning and aged ischemic hearts. Pharmacol Res, doi: 10.1016/j.phrs.2016.03.009 (2016).
Cody, V. Crystal and molecular structures of flavonoids. Prog Clin Biol Res 280, 29–44 (1988).
Jung, S. K. et al. The P110 subunit of PI3-K is a therapeutic target of acacetin in skin cancer. Carcinogenesis 35, 123–130 (2014).
Huang, W. C. & Liou, C. J. Dietary acacetin reduces airway hyperresponsiveness and eosinophil infiltration by modulating eotaxin-1 and th2 cytokines in a mouse model of asthma. Evid Based Complement Alternat Med 2012, 910520 (2012).
Boussouar, A. et al. Acacetin and chrysin, two polyphenolic compounds, alleviate telomeric position effect in human cells. Mol Ther Nucleic Acids 2, e116 (2013).
Lefer, D. J. & Granger, D. N. Oxidative stress and cardiac disease. Am J Med 109, 315–323 (2000).
Oyama, J. et al. Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 109, 784–789 (2004).
Zong, W. X. & Thompson, C. B. Necrotic death as a cell fate. Genes Dev 20, 1–15 (2006).
Golstein, P. & Kroemer, G. Cell death by necrosis: towards a molecular definition. Trends Biochem Sci 32, 37–43 (2007).
Heusch, G. Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116, 674–699 (2015).
Skyschally, A. et al. Across-Species Transfer of protection by remote ischemic preconditioning with species-specific myocardial signal transduction by reperfusion injury salvage kinase and survival activating factor enhancement pathways. Circ Res 117, 279–288 (2015).
Dewald, O. et al. Of mice and dogs: species-specific differences in the inflammatory response following myocardial infarction. Am J Pathol 164, 665–677 (2004).
Zhang, J. Q. et al. Ischemic preconditioning produces more powerful anti-inflammatory and cardioprotective effects than limb remote ischemic postconditioning in rats with myocardial ischemia-reperfusion injury. Chin Med J 126, 3949–3955 (2013).
Lecour, S., Rochette, L. & Opie, L. Free radicals trigger TNF alpha-induced cardioprotection. Cardiovasc Res 65, 239–243 (2005).
Smith, R. M., Suleman, N., McCarthy, J. & Sack, M. N. Classic ischemic but not pharmacologic preconditioning is abrogated following genetic ablation of the TNFalpha gene. Cardiovasc Res 55, 553–560 (2002).
Kleinbongard, P., Schulz, R. & Heusch, G. TNFalpha in myocardial ischemia/reperfusion, remodeling and heart failure. Heart Fail Rev 16, 49–69 (2011).
Lecour, S. et al. Identification of a novel role for sphingolipid signaling in TNF alpha and ischemic preconditioning mediated cardioprotection. J Mol Cell Cardiol 34, 509–518 (2002).
Bialik, S. et al. Myocyte apoptosis during acute myocardial infarction in the mouse localizes to hypoxic regions but occurs independently of p53. J Clin Invest 100, 1363–1372 (1997).
Colucci, W. S. Apoptosis in the heart. N Engl J Med 335, 1224–1226 (1996).
Hatori, N., Sjoquist, P. O., Marklund, S. L., Pehrsson, S. K. & Ryden, L. Effects of recombinant human extracellular-superoxide dismutase type C on myocardial reperfusion injury in isolated cold-arrested rat hearts. Free Radic Biol Med 13, 137–142 (1992).
Liu, J. et al. Recombinant PTD-Cu/Zn SOD attenuates hypoxia-reoxygenation injury in cardiomyocytes. Free Radic Res 47, 386–393 (2013).
Wu, X. W. et al. Human thioredoxin exerts cardioprotective effect and attenuates reperfusion injury in rats partially via inhibiting apoptosis. Chin Med J 121, 819–826 (2008).
Turoczi, T. et al. Thioredoxin redox signaling in the ischemic heart: an insight with transgenic mice overexpressing Trx1. J Mol Cell Cardiol 35, 695–704 (2003).
Matsushima, S., Zablocki, D. & Sadoshima, J. Application of recombinant thioredoxin1 for treatment of heart disease. J Mol Cell Cardiol 51, 570–573 (2011).
Yang, J. et al. Sequential expression of TLR4 and its effects on the myocardium of rats with myocardial ischemia-reperfusion injury. Inflammation 31, 304–312 (2008).
Hausenloy, D. J., Mocanu, M. M. & Yellon, D. M. Cross-talk between the survival kinases during early reperfusion: its contribution to ischemic preconditioning. Cardiovasc Res 63, 305–312 (2004).
Hausenloy, D. J., Duchen, M. R. & Yellon, D. M. Inhibiting mitochondrial permeability transition pore opening at reperfusion protects against ischaemia-reperfusion injury. Cardiovasc Res 60, 617–625 (2003).
Kitakaze, M. et al. Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction (J-WIND): two randomised trials. Lancet 370, 1483–1493 (2007).
Walker, M. J. et al. The Lambeth Conventions: guidelines for the study of arrhythmias in ischaemia infarction, and reperfusion. Cardiovasc Res 22, 447–455 (1988).
Curtis, M. J. & Walker, M. J. Quantification of arrhythmias using scoring systems: an examination of seven scores in an in vivo model of regional myocardial ischaemia. Cardiovasc Res 22, 656–665 (1988).
Miller, L. E., Hosick, P. A., Wrieden, J., Hoyt, E. & Quindry, J. C. Evaluation of arrhythmia scoring systems and exercise-induced cardioprotection. Med Sci Sports Exerc 44, 435–441 (2012).
Singla, D. K., Lyons, G. E. & Kamp, T. J. Transplanted embryonic stem cells following mouse myocardial infarction inhibit apoptosis and cardiac remodeling. Am J Physiol Heart Circ Physiol 293, H1308–H1314 (2007).
Liao, S. Y. et al. Proarrhythmic risk of embryonic stem cell-derived cardiomyocyte transplantation in infarcted myocardium. Heart Rhythm 7, 1852–1859 (2010).
Zhang, Y. H. et al. Functional transient receptor potential canonical type 1 channels in human atrial myocytes. Pflugers Archiv 465, 1439–1449 (2013).
Acknowledgements
This work was supported in part by a grant (ITS/339/09) from Innovation and Technology Commission of the Hong Kong SAR Government, China, and a Seeding Fund from the University of Hong Kong.
Author information
Authors and Affiliations
Contributions
H.L., L.Y., H.J.W., G.S.X., Y.W. and G.R.L. conceived and designed the project, H.L., L.Y., H.J.W., K.H.C., F.L., G.L. and H.Y.S. conducted the experiments. H.L., L.Y., H.Y.S. and G.R.L. analyzed the data. H.L. and G.R.L. prepared the manuscript. All authors read and approved the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, H., Yang, L., Wu, HJ. et al. Water-soluble acacetin prodrug confers significant cardioprotection against ischemia/reperfusion injury. Sci Rep 6, 36435 (2016). https://doi.org/10.1038/srep36435
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep36435
This article is cited by
-
Acacetin inhibits myocardial mitochondrial dysfunction by activating PI3K/AKT in SHR rats fed with fructose
Journal of Natural Medicines (2023)
-
Acacetin attenuates mice endotoxin-induced acute lung injury via augmentation of heme oxygenase-1 activity
Inflammopharmacology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.