Abstract
Understanding the movement of threatened species is important if we are to optimize management and conservation actions. Here, we describe the age and sex specific spatial and temporal ranging patterns of 19 bearded vultures Gypaetus barbatus tracked with GPS technology. Our findings suggest that spatial asymmetries are a consequence of breeding status and age-classes. Territorial individuals exploited home ranges of about 50 km2, while non-territorial birds used areas of around 10 000 km2 (with no seasonal differences). Mean daily movements differed between territorial (23.8 km) and non-territorial birds (46.1 km), and differences were also found between sexes in non-territorial birds. Daily maximum distances travelled per day also differed between territorial (8.2 km) and non-territorial individuals (26.5 km). Territorial females moved greater distances (12 km) than males (6.6 km). Taking into account high-use core areas (K20), Supplementary Feeding Sites (SFS) do not seem to play an important role in the use of space by bearded vultures. For non-territorial and territorial individuals, 54% and 46% of their home ranges (K90), respectively, were outside protected areas. Our findings will help develop guidelines for establishing priority areas based on spatial use, and also optimize management and conservation actions for this threatened species.
Similar content being viewed by others
Introduction
Movement ecology is an emerging discipline that can play a critical role in the design and improvement of management and conservation measures that target threatened taxa. Thus, the link between movement ecology and biodiversity is a key factor in understanding ecological and evolutionary processes and in improving the efficacy of conservation efforts1,2. Several recent examples show the importance of movement ecology studies in topics such as age-related aspects of migration3, the energetics and timing of migration4, the conservation of migratory flyways5, the selection of stopover, wintering and breeding areas6,7,8,9, the influence of environmental conditions10, transboundary management policies11 and foraging behaviour12.
Age classes of most large territorial vertebrates segregate spatially as a consequence of their breeding status and the different ways in which they exploit food resources. For example, the foraging movements of pre-adult individuals during dispersal periods are typically aimed at finding food resources13 and so, due to this non-territorial lifestyle, patches with higher food availability are preferably selected by non-breeding individuals. This is a very important and sensitive period from a conservation perspective because pre-breeding dispersal in large vertebrates with deferred maturity usually lasts for several years, during which time individuals exploit large areas; their success in finding food will thus greatly affect their survival prospects14,15,16. Consequently, estimation of the use of space during these pre-settlement movements is a key element in the design of any adaptive management program designed to improve geographical expansion and restore viable metapopulations17,18.
By contrast, when individuals reach adulthood, settle and occupy breeding territories, their central-place foraging behaviour restricts their movements to the nest surroundings. Foraging movements of breeding individuals are linked to obtaining food resources but also include aspects related to habitat choice, functional responses and the trade-offs between food-seeking and safety19,20. Breeding status obliges individuals to pursue a cost-benefit equilibrium in their foraging strategy that takes into account the distance they need to cover and the benefits (i.e. food) they can obtain. This is more obvious for species such as avian scavengers that depend on spatially and temporally unpredictable food resources21,22. In these species, days may elapse before food is obtained and so individuals try to optimize their energetic gain at each feeding event at carcasses23,24.
The bearded vulture Gypaetus barbatus is a threatened species in the Western Palearctic, where it is restricted to mountain biomes such as the Pyrenees (n = 130 pairs, 61% of the European population in 2015) (A. Margalida, unpubl. data). Nevertheless, several ongoing reintroduction projects are currently being performed in certain subpopulations (i.e. Alps, Andalusia). The largest natural European population of bearded vultures is confined to the Pyrenees and knowledge of the movement of this species will help improve management actions aimed at increasing the distribution of this species and minimizing the risk of metapopulation extinction. The identification of the core areas this vulture uses is an important step in the conservation of the species and will improve management strategies. The difficulties inherent in the long-term monitoring of long-lived species greatly hinder the collection of reliable information on this subject. To date, information on the ranging behaviour of bearded vultures in the Pyrenees is limited to nine non-breeding individuals tracked using ARGOS satellite telemetry, which yielded 427 locations25, and a further nine non-breeding individuals monitored with GPS satellite telemetry17. Another study has been carried out on the subspecies Gypaetus barbatus meridionalis in South Africa, in which 18 individuals were monitored26. The current study aims to provide the first comprehensive understanding of the ranging behaviour of this vulture’s threatened Pyrenean population via the use of a larger sample size of all age-classes, tracked over a number of years/seasons with highly accurate GPS satellite technology.
Through an analysis of existing information on the pre-breeding dispersal and territorial movements of GPS-tracked bearded vultures in the Pyrenees, in this study we offer insights into the use of space by this endangered species. We analyzed the movements of 19 bearded vultures in the Pyrenees (Spain, France and Andorra) between 2006 and 2014 and were thus able to identify home range sizes and daily movements in terms of age, sex and period (breeding vs. non-breeding seasons). The nesting areas occupied by breeding individuals are commonly used as surrogates of critical areas for conservation and buffer areas are established around nesting sites on this basis27. However, from a conservation perspective, high-quality areas in which individuals obtain most resources should similarly be considered as vital in the design and implementation of conservation priorities. Accordingly, we predicted that, apart from inherent individual age-, sex- and breeding-status-related differences28, no substantial asymmetries would appear between the vultures’ life periods. Specifically, we predicted that movements in the non-breeding and breeding periods would be similar because the poor weather in winter is compensated for by a network of supplementary feeding sites (also known as vulture restaurants) that function between November and April29. On the other hand, the summer (non-breeding) period offers greater food availability during the transhumance season30. To date, the aim behind the management of the Pyrenean population of bearded vultures has primarily been to enhance the region’s environmental carrying capacity by installing a network of feeding sites, which have been found to play an important role in the observed movement patterns of the species17. Based on the data gathered, we here develop guidelines for the establishment of priority areas in which management and conservation actions targeting this threatened species should be concentrated.
Results
We obtained a total of 66 467 GPS locations of 19 bearded vultures from 29 November 2006 to 31 December 2014 (Table 1). The mean monitoring period per bird was 1104.0 ± 789.3 days (range 29–2058), while the mean number of locations per bird was 3509.1 ± 2542.8 (range 57–7886). The distribution of locations was as follows: by sex 36.1% females and 63.9% males, and by age class 80.6% adults, 15.3% subadults, 3.3% immatures and 0.8% juveniles. During this period, six individuals died (two poisoned, two intoxicated by lead and two after collisions with power lines) and the transmitter was lost or failed in nine birds (Table 1).
Daily movements
Non-territorial adults recorded the highest mean daily movements at 46.1 ± 41.4 km, the maximum distance travelled by a bearded vulture in one day being 259.5 km. Territorial adults moved a mean of 23.8 ± 20.9 km per day but with an average maximum distance reached from the initial point per day of 8.2 ± 12 km (see Table 2 for complete results; see also Supplementary Information Table S1). The maximum distance travelled by a tracked bearded vulture in a single day was 176.2 km from the initial point. Juveniles moved shorter distances, 8.4 ± 7.3 km, and their maximum distance travelled per day was 5.7 ± 5.2 km. Subadults and non-territorial adults moved similar average hourly distances of 4.5 ± 7.3 km per hour. Territorial adults moved an average hourly distance of 2.5 ± 3.9 km per hour; juveniles were the slowest age group with an hourly distance of 1.1 ± 2.1 km per hour (Table 2). We found significant differences between the mean daily distances travelled by territorial and by non-territorial birds (Wilcoxon test z = 10.4, p < 0.001) and likewise found differences between the maximum distances travelled per day by territorial and by non-territorial birds (z = 19.3, p < 0.001). Non–territorial birds moved to more distant areas than territorial birds (26.5 ± 27.5 km vs. 8.2 ± 12.0 km).
Differences between sexes were not detected in the mean daily distances travelled by territorial birds (z = 0.92, p = 0.35) but were found between sexes in non-territorial birds (z = −3.81 p < 0.001), as females travelled shorter distances (37.4 ± 34.0 km) than males (47.2 ± 44 km; Table 2). No sex-related differences in maximum distances travelled per day were found in non-territorial birds (z = −1.60, p = 0.11); territorial females moved further then than males (12.0 ± 18.2 km vs. 6.6 ± 7.6 km; z = 3.37, p < 0.001).
Home range
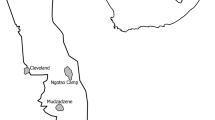
The total area used by all tracked bearded vultures was 48 738.6 km2 (Minimum Convex Polygon, MCP; Fig. 1), which encompassed the whole of the current breeding range of the species in the Pyrenees. Annual bearded vulture home ranges showed important differences between age classes (see Table 3 for complete data, Figs 2 and 3, and Supplementary Information Table S2). These differences were statistically significant in the core area (Wilcoxon test z = 6.41, p < 0.001), home range (z = 6.36, p < 0.001) and MCP (z = 5.97, p < 0.001). Territorial individuals exploited home range areas of about 50 km2, while non-territorials birds used areas of around 10,000 km2. When assessing home range differences between non-territorial age classes, we only found significant differences between juveniles and non-territorial adults in all home range estimators (K50 z = −1.94, p = 0.05; K90 z = −2.04, p = 0.041; MCP z = −1.99, p = 0.045; Fig. 3). Differences between sexes in home range size were found in territorial adults but not in non-territorial birds (K50 z = −0.49, p = 0.63; K90 z = −1.14, p = 0.26; MCP z = −1.22, p = 0.22). Territorial males used smaller core areas than territorial females (z = 2.01, p = 0.043) (see Table 3). No sex-related differences were found in MCP or home range in territorial adults (K90 z = 1.57, p = 0.12; MCP z = 0.74, p = 0.45).
Minimum convex polygon used by 19 GPS-tracked bearded vultures Gypaetus barbatus in the Pyrenees from 2006 to 2014.
Maps were created in Rstudio 0.99 (https://www.rstudio.com) and R 3.0.3 (http://www.R-project.org) in combination with ArcGIS 9.1 (http://www.esri.com/ see Material and Methods).
Bearded vulture Gypaetus barbatus ranging behaviour tracked in the Pyrenees from 2006 to 2014.
We show utilization density areas UD kernel 90% (thick line) and UD kernel 50% (thin line) for (a) territorial adult in the breeding season (yellow) and non-breeding season (violet) and (b) we show yearly UD areas for a non-territorial adult (blue) versus a territorial adult (red). Maps were created in Rstudio 0.99 (https://www.rstudio.com) and R 3.0.3 (http://www.R-project.org) in combination with ArcGIS 9.1 (http://www.esri.com/ see Material and Methods).
No seasonal differences were detected in the home range size of non-territorial birds (K50 z = −0.50, p = 0.62; K90 z = −0.29, p = 0.77; MCP z = 0.50, p = 0.61) or territorial adults (K50 z = −0.45, p = 0.65; K90 z = −0.05, p = 0.95), with the exception of the MCP (MCP z = 1.96, p = 0.049; Fig. 4). Territorial adult females used larger MCP areas during the breeding season (z = 2.01, p = 0.044) compared to the non-breeding season. By contrast, no differences were found in home range (z = 0.18, p = 0.85) or core area (z = 0.18, p = 0.85). Territorial males showed no differences in any home range estimator (K50 z = −0.55, p = 0.58; K90 z = −0.04, p = 0.96; MCP, z = 1.14, p = 0.25).
Factors related to home range size
At core-area scale, territorial status, age, interaction between sex, territorial status and the full model including all the variables performed better than the null model. At home-range scale, all of these factors were included, together with two other factors (season and the interaction between season and territorial status). For territorial birds at core-area scale, season was the only factor that improved the null model (see Supplementary Information Table S3). At home-range scale, no factors improved the null model. For non-territorial individuals, age was the principal factor related to the size of their home and core ranges. In relation to the pairwise comparison between age classes in the core area, juvenile birds showed significant differences to the other age classes (see Table 4).
Effects of supplementary feeding sites
Up to 75 SFS (70.1% of the total SFS established in the Pyrenees and surrounding regions) were located within the MCP of all tracked bearded vultures (Fig. 5). In all, 39.9%, 4.8% and 2.4% of locations were situated less than 5, 1 and 0.5 km, respectively, from a SFS. Although no differences relating to territorial status were found at smaller buffer scales (0.5 km z = −0.40, p = 0.69; 1 km z = 1.49, p = 0.14), at 5 km (z = 6.08, p < 0.001), territorial individuals had a significant higher percentage of locations near SFS than non-territorials (67.6% ± 36.8% vs. 30.0% ± 17.0%).
Example of bearded vulture Gypaetus barbatus ranging behaviour tracked in the Pyrenees in relation to location of Supplementary Feeding Sites (Suppl. Feed. Sites).
We show utilization density areas UD kernel 90% (thick line), 50% (medium line) and 20% (thin line) for territorial bird (red) and non-territorial bird (blue) and Suppl. Feed. Sites (green stars). Maps were created in Rstudio 0.99 (https://www.rstudio.com) and R 3.0.3 (http://www.R-project.org) in combination with ArcGIS 9.1 (http://www.esri.com/ see Material and Methods).
Territorial bearded vultures included significantly fewer SFS within their core areas and home ranges than non-territorial birds (Table 5; K50 z = 5.93, p < 0.001; K90 z = 6.08, p < 0.001). In the high-use core areas (K20) of territorial birds, no SFS was found, a contrast to the 2.8 ± 2.3 SFS that were found in non-territorial birds (z = 5.16, p < 0.001).
In the case of territorial individuals, we found no differences between the sexes in either the number of SFS inside the kernels (K50 z = 1.29, p = 0.197; K90 z = 0.96, p = −0.34) or the frequency of locations at different distances from the SFS (0.5 km z = −0.89, p = 0.37; 1 km z = −0.75, p = 0.46; 5 km z = −0.37, p = 0.71). For non-territorial individuals, we found no differences in the number of SFS inside the kernels (K20 z = −0.75, p = 0.45; K50 z = −3.42, p = 0.73; K90 z = −1.53, p = −0.13) but did find differences in the frequency of locations at 5 km from the SFS (0.5 km z = 0.54, p = 0.59; 1 km z = 0.59, p = 0.56; 5 km z = −2.16, p = 0.03), where male locations were more frequent (34.2 ± 18.7% vs. 23.4 ± 12.6%).
Overlapping foraging range with protected areas
In all, 43.8% of the home range of non-territorial birds (K90 22 723.6 km2) was within protected areas. A similar overlap percentage, 44.5%, was found for the core area (K50 6669.9 km2). Overlap with protected areas was higher in territorial birds, with 54.3% of the home range (K90 838.7 km2) and 65.7% of the core area (K50 158.9 km2) lying within protected areas.
Discussion
This study provides the first description and comparison of spatial and temporal ranging patterns of territorial and non-territorial Pyrenean bearded vultures tracked with GPS technology (see also refs 17 and 25). Such high-quality information will help optimize management and conservation actions for this threatened species in the context of the European metapopulation. It may also assist in assessing food availability in the area used by the species, as well the functionality of the SFS designed to enhance survival, breeding success and geographical expansion31,32,33. Thus, actions aimed at 1) increasing the range of the species; 2) establishing SFS; and 3) minimizing the detrimental effects of territory shrinkage should be based on available data on range movements since almost no geographical expansion of this population has occurred in the last few decades31.
The bearded vulture, an obligate avian scavenger characterized by energy-efficient foraging flight, exploits large areas in search of unpredictable carcasses34,35,36,37,38. Individual breeding status and the temporally variable food requirements (more meat is required during the chick-rearing period39) could explain the differences in the use of space. Our findings show asymmetries in space use between territorial and non-territorial individuals. Breeding individuals exploit smaller areas (K90 63 ± 59.5 km2) in contrast to non-territorial individuals, which cover areas of 1818–11 616 km2 depending on age-class (Table 4). In a previous study25 based on nine pre-adult individuals in the Pyrenees tracked with satellite telemetry (but not GPS technology), the MCP obtained ranges of 945–19 691 km2. A similar pattern26 was observed in the subspecies Gypaetus barbatus meridionalis in South Africa in which breeding individuals covered on average 95 ± 19 km2, whilst non-territorial individuals covered areas of 10 540–25 985 km2. The differences are also significant in MCP, with 940.8 ± 1524.4 km2 for breeding individuals and 1566–13 270 km2 for non-breeding individuals. The South African subspecies had significantly larger MCP home range estimates, with home ranges for breeding adults around 5220 km2 and for non-breeding individuals at 21 151–40 961 km2. This marked difference between subspecies may result from factors such as conspecific attraction, the abundance of food resources and the modification of habitat quality by SFS, all of which may influence the movement ecology of Pyrenean bearded vultures17. It is generally assumed that, for vertebrates, foraging range size is inversely related to resource abundance and spatio-temporal predictability40. In other large-bodied raptors, home ranges vary according to prey density and individual reproductive status, with habitat quality acting as the main regulatory mechanism of space use41,42. This is the case of the bearded vulture in the Pyrenees, an area characterized by abundant natural food resources (i.e. wild and domestic ungulates32,33) that also possesses a network of SFS providing additional food. These predictable sites attract concentrations of dozens of mainly pre-adult individuals29. These differences may have important conservation implications given the threatened status of the species in Europe and the need to connect subpopulations so as to reduce the extinction risk. In this sense, movements of the non-territorial population of Pyrenean bearded vultures differ from those in other reintroduced subpopulations (Andalusia and Alps), in which there are greater range movements17. Several non-mutually exclusive factors could explain the differences found between wild and reintroduced individuals17. Firstly, captivity (individuals from Alps and Andalusia) can relax selective pressures, change the direction of selection or impose completely novel pressures, thereby provoking noticeable behavioural changes. In addition, the sharp decline of the European bearded vulture population may have had serious genetic consequences and the genetic differences found in the Pyrenean population could be linked to individual dispersal propensity. Finally, human activities can alter selective environments and disassociate certain behavioural and life history decisions from the outcomes that are normally expected of them, thereby creating evolutionary traps and thus rapid environmental changes that result in shifted behavioural decisions17. The small home ranges of Pyrenean bearded vultures limit the geographical expansion of the species. For example, over the last 20 years, only a small expansion of the distribution area has occurred in the Pyrenees (40 km westwards)31 despite its population growth (from 53 territories in 1991 to 130 in 2015). Habitat quality and conspecific attraction regulated by SFS may be responsible for the shrinkage of territories, the density-dependent effects on breeding output and the lack of geographical expansion. Thus, actions designed to extend the predictable food sources provided by the network of SFS have been proposed as the main actions to be employed to facilitate the expansion process17,31.
Our results show sexual and age-class asymmetries in both the daily mean movements and maximum distance travelled. Non-territorial females travelled less far than males but territorial females moved to more distant places than males. In addition, territorial females exploited larger areas (MCP and K50) than males. The potential drivers of this pattern are difficult to understand from ecological and evolutionary points of view since the division of parental roles in breeding bearded vultures is equitable43 and so a priori we would expect similar foraging distances in the sexes in the search for food resources. The shorter distances travelled by non-territorial females could be explained by their greater dependence on predictable food sources or patches, which might reduce their movements. However, this pattern will also be applicable to breeding individuals visiting distant SFS, in which foraging movements are compensated for by the predictability (spatial and temporal) of the available food resource. In fact, females of Egyptian vultures (Neophron percnopterus majorensis) have been reported to visit supplementary feeding stations more frequently than males44. In other species such as the California Condor (Gymnogyps californianus) monthly home ranges of adults were significantly larger than those of immatures; however, males and females had monthly home ranges of similar sizes throughout the year and breeding adults did not differ from non-breeding adults in their average monthly home range size45. On the contrary, in Eurasian griffon vultures (Gyps fulvus) no differences were found in home ranges between adults and immatures22. This differentiated foraging behaviour in obligate avian scavengers suggests that selective foraging habitat varies between species and age classes. Although predictable resources, lack of experience and reproductive constraints could partially explain such differences, our findings show that non-territorial bearded vultures included more SFS in their home ranges than territorial individuals, although the intensity of use (frequency of nearby locations) of the SFS was similar. Inexperienced individuals included more SFS in their movements because they act as predictable food supplies and as a site for interaction between conspecifics. Territorial birds used these sites less often, probably because they are able to exploit unpredictable but higher-quality prey more efficiently. This pattern could explain the territory-related differences in the use of SFS by bearded vultures. We found no differences between the sexes in territorial individuals in any of the buffers considered. The only significant result was found in non-territorial birds, in which male locations appeared more frequently in the 5-km buffers. However, these results could also be influenced by other factors such as the home range sizes of each individual, habitat quality (density of wild and domestic ungulates as the main food resource) and the spatial configuration of the SFS network. Indeed, the SFS network is not arranged in a spatially random way but is located mainly in high-quality breeding areas or near occupied breeding territories. Thus, used as a simple measure, the distance of locations from the SFS does not seem to be an objective proxy for determining their use. This could be due to the fact that the territorial birds in our study did not include SFS within their core areas but, by contrast, did have a higher frequency of locations near the SFS. Thus, if we take into account the smaller home range sizes of territorial bearded vultures, the use of large buffers (i.e. 5 km) as a surrogate measure for SFS use would seem to be unsuitable25. As a result, new methods and measures for studying the use of SFS by bearded vultures and the effects they have on vulture movements still need to be explored further.
With respect to the overlap with protected areas, our data suggest that around 44% of the home range of Pyrenean non-territorial bearded vultures lies within the network of protected areas, a figure that increases to 54% (K90) and 66% (K50) in territorial individuals. This implies that bearded vultures spend a significant amount of time foraging outside protected areas, thereby increasing the inherent risks of non-natural mortality due to anthropogenic factors46. Biodiversity conservation strategies are increasingly focused on regions outside protected areas, where animals face numerous anthropogenic threats and have to coexist with human settlements, livestock and agriculture47. As a result, an increase in conservation actions outside protected areas is a priority in any attempt to reduce the risk of non-natural mortality46. Of non-natural mortality factors, the risk of ingesting poison bait is one of the most serious factors affecting bearded vultures36 and other avian scavengers48,49,50,51,52 around the world. Other factors such as wind farms, lead poisoning and collisions with power lines have also been identified as drivers of non-natural mortality36,38,53. In addition, the authorization of the veterinary product Diclofenac in Spain in recent years is considered to be a potential threat to scavenger species. Carrion controls are needed to detect residues of veterinary pharmaceuticals that are potentially toxic to scavengers54,55.
Future decisions regarding the implementation of specific protected areas and the management of SFS should take advantage of the emerging discipline of movement ecology and be based on the robustness of data obtained via telemetry. For example, in addition to breeding sites as surrogates of priority protected areas, the data herein provided will aid the identification of important feeding areas (at a spatial and temporal scale) and thus help the conservation of specific species. This information will also allow us to improve the design and spatial distribution of the SFS network by evaluating the natural biomass provided by the habitat in relation to birds’ energetic requirements. Managers and policy-makers should consider these tools when seeking to implement evidence-based conservation actions56.
Material and Methods
Ethics Statement
All the work was conducted in accordance with relevant national and international guidelines, and conforms to all legal requirements. Captures and blood sample collection were carried out in compliance with the Ethical Principles in Animal Research. Thus, protocols, amendments and other resources were conducted in accordance to the guidelines approved by the Catalan Autonomous Government (Generalitat de Catalunya) following the R.D.1201/2005 (10 October 2005, BOE 21 October 2005) of the Ministry of Presidency of Spain. All experimental protocols were approved by the Catalan Autonomous Government and MAGRAMA (References 15546 and 25.306).
Study species
The bearded vulture is a long-lived, threatened species that nests on mountain cliffs in Africa, Europe and Asia57. Declines in bearded vulture populations have been documented throughout its range, with habitat loss, direct persecution (poisoning and hunting) and reduced food availability being the main aggravating factors58,59. In Europe the species is restricted to the Pyrenees (170 breeding pairs), Alps (31 pairs), Crete (6 pairs), Corsica (5 pairs) and Andalusia (1 pair) (A. Margalida, unpubl. data). The primary risks to this population are the illegal use of poisons for predator control, lead intoxication, collisions with energy infrastructures and food shortages58,60.
Capture and tracking
A total of 19 bearded vultures were fitted with satellite transmitters in the period 2006–2013. Transmitters were fitted to birds captured using radio-controlled bow-nets at feeding stations (n = 16), to fledglings in the nest (n = 1) or to individuals released after recovery at official rescue centres (n = 2).
Birds were assigned to one of four age classes according to previous demographic studies61: juveniles (birds in their first year of life), immatures (2–3 years), sub-adults (4–5 years) and adults (≥6 years). Individuals were sexed by molecular analysis of blood samples. Their territoriality, breeding status and annual breeding success were assessed through field observations.
We used solar-powered 70 g (GPS/PTT) satellite transmitters (Microwave Telemetry, Inc. Columbia, MD) to track movements. We attached transmitters using a backpack harness made from 0.64 cm Teflon ribbon (Bally Ribbon Mills, Bally, PA). We programmed transmitters to collect GPS locations (manufacturer’s estimated error: 18 metres) each hour from 6:00 to 21:00 UTC. Units on two birds (Dulantz and Revilla) were programmed to transmit a fix every two hours.
Movement modelling
To study temporal variation in bearded vulture movement ecology, we divided the year into two seasons according to the breeding cycle: the breeding period (Br) from 1 January to 31 July and the non-breeding period (Non-br) from 1 August to 31 December.
Different measures were calculated to investigate daily movement patterns. The daily movement (km) was calculated summing the straight-line distances between successive locations on the same day. The maximum distance travelled per day (km) was calculated as the maximum distance from the initial daily location to any location reached on the same day. The average hourly distance between locations (km) was calculated as the straight-line distance between two consecutive locations and standardized in relation to the hours spent between locations. We limited the hourly frequency between locations to a maximum of two hours (i.e. seven or more locations per day). The distance between locations was calculated using basic trigonometry.
We used dynamic kernel models in the adehabitatHR package to calculate utilization distributions (UD)62. This package was run in Rstudio 0.99 and R version 3.0.363 in combination with ArcGIS 9.1 (ESRI 2003) to build UD surfaces. Annual and seasonal UDs were estimated per bird using the ad hoc method as a smoothing parameter for comparison with previous studies26. Resolution of UD surfaces was established at 1 ha. We calculated the 90% (K90) and 50% kernel (K50) density contours. K90 represents the home range and K50 the core area of activity. Birds with fewer than 50 locations per season or 15 per month were removed from the analyses. Additionally, we calculated the Minimum Convex Polygon (MCP), which is traditionally used as a measure of the maximum area of activity. All values are presented as mean ± standard deviation (sd).
Effects of supplementary feeding stations
A total of 107 SFS were located in the range of bearded vultures in Spain and France. We evaluated the effects of SFS presence on bearded vulture movements by calculating (i) the percentage of locations inside buffers of 0.5 km, 1 km and 5 km around each SFS25 and (ii) the number of SFS located inside the annual UD surfaces of tracked bearded vultures. We used the core area (K50) and home range (K90), and calculated the 20% kernel (K20), which allowed us to determine which areas were used most frequently by each bearded vulture.
Overlap of foraging movements with protected areas
We calculated the core area (K50) and home range (K90) of non-territorial and territorial birds separately. Protected area cover for France, Andorra and Spain was obtained from government and institutional online sources (https://inpn.mnhn.fr, http://www.iea.ad and http://www.idee.es, respectively). For this study, we only took into account the following protected area categories: National and Natural/Regional Parks and Special Protected Area (SPAs, Birds Directive 2009/147/CE) due their importance for bird protection.
Statistical performance
We analyzed age- and sex-related differences in distance travelled per day, maximum distance per day, mean hourly distance and home-range size estimators using a Wilcoxon rank sum test. We analyzed each relationship independently.
After assessing the combined effect, we evaluated the effect of four factors: age, sex, territorial status (territorial vs. non-territorial) and season (breeding season vs. non-breeding season) on home range size using generalized linear mixed models (GLMM)64. We built nested models that included each factor separately (i.e. age), factors combined with other factors (i.e. age + season), and the interactions between factors (i.e. age*season). We included a null model (1 + random factor) to evaluate the performance of the models. To avoid pseudoreplication, we included as a random factor individual identity and year. GLMMs were fitted by maximum likelihood (ML) using the function “glmer” in “lme4” R package65 with a Gaussian error distribution and log link. Akaike’s information criterion (AIC) was used to compare the nested models with null models; differences in goodness-of-fit between models were evaluated by a log-likelihood ratio test calculated using the R function anova66. Additionally, to detect differences in factor categories, pairwise comparisons between age classes and breeding season were performed using the “multcomp” package67. P values were adjusted using the Tukey method, the default for pairwise comparisons between adjusted means.
We used the paired Wilcoxon rank sum test to study differences in territorial status in the percentage of locations at 0.5 km, 1 km and 5 km from a SFS and the number of SFS within K20, K50 and K90. For these analyses, juvenile birds were excluded from the non-territorial class due to their strong dependence on the parental territory during the first months after fledgling. All statistical procedures were performed in R version 3.0.363.
Additional Information
How to cite this article: Margalida, A. et al. Spatial and temporal movements in Pyrenean bearded vultures (Gypaetus barbatus): Integrating movement ecology into conservation practice. Sci. Rep. 6, 35746; doi: 10.1038/srep35746 (2016).
References
Wikelski, M. et al. Going wild: what a global small-animal tracking system could do for experimental biologists. J. Exp. Biol. 210, 181–186 (2007).
Jeltsch, F. et al. Integrating movement ecology with biodiversity research-exploring new avenues to address spatiotemporal biodiversity dynamics. Mov. Ecol. 1, 6 (2013).
Sergio, F. et al. Individual improvements and selective mortality shape lifelong migratory performance. Nature 515, 410–413 (2014).
Flack, A. et al. Costs of migratory decisions: A comparison across eight white stork populations. Sci. Adv. 2, e1500931 (2016).
Kirby, J. S. et al. Key conservation issues for migratory land-and waterbird species on the world’s major flyways. Bird Conserv. Int. 18, S49 (2008).
Robinson, W. D. et al. Integrating concepts and technologies to advance the study of bird migration. Front. Ecol. Environ. 8, 354–361 (2009).
Cano, L. S. & Tellería, J. L. Migration and winter distribution of Iberian and central European black storks Ciconia nigra moving to Africa across the Strait of Gibraltar: a comparative study. J. Avian Biol. 44, 189–197 (2013).
Silva, M. A., Prieto, R., Jonsen, I., Baumgartner, M. F. & Santos, R. S. North Atlantic Blue and Fin Whales suspend their spring migration to forage in middle latitudes: building up energy reserves for the journey? PLoS ONE 8, e76507 (2013).
Trierweiler, C. et al. Migratory connectivity and population-specific migration routes in a long-distance migratory bird. Proc. R. Soc. Lond. B 281, 20132897 (2014).
Dodge, S. et al. Environmental drivers of variability in the movement ecology of turkey vultures (Cathartes aura) in North and South America. Phil. Trans. R. Soc. Lond. B 369, 1471–2970 (2014).
Lambertucci, S. et al. Apex scavenger movements call for transnational conservation policies. Biol. Conserv. 170, 145–150 (2014).
Wakefield, E. D. et al. Space partitioning without territoriality in Gannets. Science 341, 68–70 (2013).
Moleón, M., Bautista, J., Sánchez-Zapata, J. A. & Gil-Sánchez, J. M. Diet of non-breeding Bonelli’s Eagles Hieraaetus fasciatus at settlement areas of southern Spain. Bird Study 56, 142–146 (2009).
Weimerskirch, H., Åkesson, S. & Pinaud, D. Postnatal dispersal of wandering albatrosses Diomedea exulans: implications for the conservation of the species. J. Avian Biol. 37, 23–28 (2006).
Grande, J. M. et al. Survival in a long-lived territorial migrant: effects of life-history traits and ecological conditions in wintering and breeding areas. Oikos 118, 580–590 (2009).
Ortega, E. et al. A demographic description of the recovery of the Vulnerable Spanish imperial eagle Aquila adalberti. Oryx 43, 113–121 (2009).
Margalida, A. et al. Uneven large-scale movement patterns in wild and reintroduced pre-adult bearded vultures: conservation implications. PLoS ONE 8, e65857 (2013).
Chandler, R. B. et al. Spatial occupancy models for predicting metapopulation dynamics and viability following reintroduction. J. Appl. Ecol. 52, 1325–1333 (2015).
Stephens, D. W. & Krebs, J. R. Foraging theory. Princeton: Princeton University Press (1986).
Stephens, D. W., Brown, J. S. & Ydenberg, R. C. Foraging: behavior and ecology. Chicago: University of Chicago Press (2007).
Carrete, M. & Donázar, J. A. Application of central-place foraging theory shows the importance of Mediterranean dehesas for the conservation of the cinereous vulture, Aegypius monachus. Biol. Conserv. 126, 582–590 (2005).
Monsarrat, S. et al. How predictability of feeding patches affects home range and foraging habitat selection in avian social scavengers? PLoS ONE 8, e53077 (2013).
Dermody, B. J., Tanner, C. J. & Jackson, A. L. The evolutionary pathway to obligate scavenging in Gyps vultures. PLoS ONE 6, e24635 (2011).
Moreno-Opo, R., Trujillano, A. & Margalida, A. Behavioral coexistence and feeding efficiency drive niche partitioning in European avian scavengers. Behav. Ecol. 27, 1041–1052 (2016).
Gil, J. A. et al. Home ranges and movements of non-breeding bearded vultures tracked by satellite telemetry in the Pyrenees. Ardeola 61, 379–387 (2014).
Krüger, S., Reid, T. & Amar, A. Differential range use between age classes of Southern African Bearded Vultures Gypaetus barbatus. PLOS ONE 9, e114920 (2014).
Arroyo, B. & Razin, M. Effect of human activities on bearded vulture behaviour and breeding success in the French Pyrenees. Biol. Conserv. 128, 276–284 (2006).
Johnson, M. L. & Gaines, M. S. Evolution of dispersal: theoretical models and empirical tests using birds and mammals. Annu. Rev. Ecol. Syst. 21, 449–480 (1990).
Margalida, A., Oro, D., Cortés-Avizanda, A., Heredia, R. & Donázar, J. A. Misleading population estimates: biases and consistency of visual surveys and matrix modelling in the endangered bearded vulture. PLoS ONE 6, e26784 (2011).
Olea, P. P. & Mateo-Tomás, P. The role of traditional farming practices in ecosystem conservation: the case of transhumance and vultures. Biol. Conserv. 142, 1844–1853 (2009).
Margalida, A., Donázar, J. A., Bustamante, J., Hernández, F. J. & Romero-Pujante, M. Application of a predictive model to detect long-term changes in nest-site selection in the Bearded Vulture Gypaetus barbatus: conservation in relation to territory shrinkage. Ibis 150, 242–249 (2008).
Margalida, A., Colomer, M. A. & Sanuy, D. Can wild ungulate carcasses provide enough biomass to maintain avian scavenger populations? An empirical assessment using a bio-inspired computational model. PLoS ONE 6, e20248 (2011).
Margalida, A. & Colomer, M. A. Modelling the effects of sanitary policies on European vulture conservation. Sci. Rep. 2, 753 (2012).
Kane, A., Jackson, A. L., Monadjem, A., Colomer, M. A. & Margalida, A. Carrion ecology modeling for vulture conservation: are vulture restaurants needed to sustain the densest breeding population of the African White-backed Vulture? Anim. Conserv. 18, 279–286 (2015).
Pennycuick, C. J. Soaring behaviour and performance of some East African birds, observed from a motor-glider. Ibis 114, 178–218 (1972).
Pennycuick, C. Modelling the flying bird. Elsevier (2008).
Phipps, W. L., Willis, S. G., Wolter, K. & Naidoo, V. Foraging ranges of immature African White-Backed Vultures (Gyps africanus) and their use of protected areas in Southern Africa. PLoS ONE 8, e52813 (2013).
Rivers, J. W. et al. An analysis of monthly home range size in the critically endangered California Condor Gymnogyps californianus. Bird Conserv. Int. 24, 492–504 (2014).
Margalida, A., Bertran, J. & Heredia, R. Diet and food preferences of the endangered Bearded Vulture Gypaetus barbatus: a basis for their conservation. Ibis 151, 235–243 (2009).
Maher, C. R. & Lott, D. F. A review of ecological determinants of territoriality within vertebrate species. Amer. Midl. Nat. 143, 1–29 (2000).
Fernández, M., Oria, J., Sánchez, R., González, L. M. & Margalida, A. Space use of adult Spanish Imperial Eagles (Aquila adalberti). Acta Ornithol. 44, 17–26 (2009).
Pérez-García, J. M. et al. Interannual home range variation, territoriality and overlap in breeding Bonelli’s eagles (Aquila fasciata) tracked by GPS satellite telemetry. J. Orn. 154, 63–71 (2013).
Margalida, A. & Bertran, J. Breeding behaviour of the Bearded Vulture (Gypaetus barbatus): minimal sexual differences in parental activities. Ibis 142, 225–234 (2000).
García-Heras, M. S., Cortés-Avizanda, A. & Donázar, J. A. Who are we feeding? Asymmetric individual use of surplus food resources in an insular population of the endangered Egyptian Vulture Neophron percnopterus. PLoS ONE 8, e80523 (2013).
Rivers, J. W. et al. An analysis of monthly home range size in the critically endangered California Condor Gymnogyps californianus. Bird Conserv. Int. 24, 492–504 (2014).
Cortés-Avizanda, A., Colomer, M. A., Margalida, A., Ceballos, O. & Donázar, J. A. Modeling consequences of the demise and potential recovery of a keystone species: wild rabbits and avian scavengers in Mediterranean landscapes. Sci. Rep. 5, 17033 (2015).
Ahlering, M. A. et al. Conservation outside protected areas and the effect of human-dominated landscapes on stress hormones in savannah elephants. Conserv. Biol. 27, 569–575 (2013).
Hernández, M. & Margalida, A. Poison-related mortality effects in the endangered Egyptian Vulture (Neophron percnopterus) population in Spain: conservation measures. Eur. J. Wildl. Res. 55, 415–423 (2009).
Hernández, M. & Margalida, A. Pesticide abuse in Europe: effects on the Cinereous vulture (Aegypius monachus) population in Spain. Ecotoxicology 17, 264–272 (2008).
Margalida, A. Baits, budget cuts: a deadly mix. Science 338, 192 (2012).
Mateo-Tomás, P., Olea, P. P., Sánchez-Barbudo, I. S. & Mateo, R. Alleviating human–wildlife conflicts: identifying the causes and mapping the risk of illegal poisoning of wild fauna. J. Appl. Ecol. 49, 376–385 (2012).
Sanz-Aguilar, A. et al. Action on multiple fronts, illegal poisoning and wind farm planning, is required to reverse the decline of the Egyptian vulture in Southern Spain. Biol. Conserv. 187, 10–18 (2015).
Carrete, M. et al. Habitat, human pressure, and social behaviour: partialling out factors affecting large-scale territory extinction in an endangered vulture. Biol. Conserv. 136, 143–154 (2007).
Margalida, A. et al. One health approach to use of pharmaceuticals. Science 346, 1296–1298 (2014).
Green, R. E., Donázar, J. A., Sánchez-Zapata, J. A. & Margalida, A. The threat to Griffon Vultures Gyps fulvus in Spain from veterinary use of the drug diclofenac. J. Appl. Ecol. 53, 993–1003 (2016).
Arlettaz, R. et al. From publications to public actions: when conservation biologists bridge the gap between research and implementation. BioScience 60, 835–842 (2010).
Del Hoyo, J., Elliott, A. & Sargatal, J. Handbook of the birds of the World. Vol. 4. Barcelona: Lynx Edicions (1994).
Margalida, A., Heredia, R., Razin, M. & Hernández, M. Sources of variation in mortality of the Bearded Vulture Gypaetus barbatus in Europe. Bird Conserv. Int. 18, 1–10 (2008).
Krüger, S. C., Simmons, R. E. & Amar, A. Anthropogenic activities influence the abandonment of Bearded Vulture (Gypaetus barbatus) territories in southern Africa. The Condor 117, 94–107 (2015).
Hernández, M. & Margalida, A. Assessing the risk of lead exposure for the conservation of the endangered Pyrenean bearded vulture (Gypaetus barbatus) population. Environ. Res. 109, 837–842 (2009).
Margalida, A., Colomer, M. A. & Oro, D. Man-induced activities modify demographic parameters in a long-lived species: effects of poisoning and health policies. Ecol. Appl. 24, 436–444 (2014).
Worton, B. J. Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70, 164–168 (1989).
R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org (2014).
Zuur, A., Ieno, E. N. & Smith, G. M. Analysing ecological data. Springer Science & Business Media (2007).
Bates, D. & Martin, M. Lme4: linear mixed-effects models using S4 classes. R package version 0.999375-32, http://CRAN.R-project.org/package=lme4 (2009).
Bolker, B. M. et al. Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol. Evol. 24, 127–13 (2009).
Hothorn, T., Bretz, F., Westfall, P. & Heiberger, R. M. multcomp: Simultaneous inference for general linear hypotheses. R package version 0.993-1, http://CRAN.R-project.org/package=multcomp (2008).
Acknowledgements
V. García-Matarranz, J. Canut, D. García, R. Heredia, E. Vega, S. Pinedo and the forestry rangers of Pallars Jussà, Alta Ribagorça and Pallars Sobirà helped with the captures. D. García (Generalitat of Catalunya), M. Alcántara and J.M. Martínez (Government of Aragón), Martine Razin (LPO), P. Muñoz, J.M. Vadillo, D. Campión and A. Llamas (Government of Navarra) and R. Heredia provided data about supplementary feeding sites and data movement. The comments of T. Katzner and two anonymous reviewers improved a previous version of this manuscript. M. Lockwood reviewed the English. A.M. was supported by a Ramón y Cajal research contract by the Ministry of Economy and Competitiveness (RYC-2012-11867). J.M.P.G. was supported by a Juan de la Cierva research contract by the Ministry of Economy and Competitiveness (FJCI-2015-25632). This study was supported by MAGRAMA, Generalitat de Catalunya, Government of Navarra and the project CGL2015-66966-C2-2-R.
Author information
Authors and Affiliations
Contributions
A.M. conceived the idea. A.M., J.M.P.-G. and I.A. collected and analysed the data. A.M. and J.M.P.-G. wrote the paper. A.M., J.M.P.-G., I.A. and R.M.-O. discussed the results and commented on the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Margalida, A., Pérez-García, J., Afonso, I. et al. Spatial and temporal movements in Pyrenean bearded vultures (Gypaetus barbatus): Integrating movement ecology into conservation practice. Sci Rep 6, 35746 (2016). https://doi.org/10.1038/srep35746
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35746
This article is cited by
-
Daily movements of the Common Crane (Grus grus) during the premigration period
Journal of Ornithology (2023)
-
Factors modulating home range and resource use: a case study with Canarian houbara bustards
Movement Ecology (2022)
-
Influence of individual biological traits on GPS fix-loss errors in wild bird tracking
Scientific Reports (2020)
-
Landscape anthropization shapes the survival of a top avian scavenger
Biodiversity and Conservation (2020)
-
Evidence of niche differentiation for two sympatric vulture species in the Southeastern United States
Movement Ecology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.