Abstract
Antennae are among the most elaborate sensory organs in adult flies, and they provide rich information for phylogenic studies. The antennae of five out of eight species of Gasterophilus Leach (G. haemorrhoidalis (Linnaeus), G. intestinalis (De Geer), G. nasalis (Linnaeus), G. nigricornis (Loew) and G. pecorum (Fabricius)), were examined using scanning electron microscopy. The general morphology, including distribution, type, size, and ultrastructure of antennal sensilla were presented, and the definition of auriculate sensilla and sensory pits were updated and clarified. Eighteen antennal characters were selected to construct the first species-level phylogeny of this genus. The monophyly of Gasterophilus was supported by the presence of coeloconic sensilla III on the antennal arista. The species-level cladogram showed G. pecorum branching off at the base, and the remaining species forming the topology (G. intestinalis+ (G. haemorrhoidalis+ (G. nasalis+ G. nigricornis))). Our research shows the importance of the antennal ultrastructure as a reliable source for phylogenetic analysis.
Similar content being viewed by others
Introduction
Morphology is an indispensable and near-unlimited source for the study of dipteran systematics and evolution. While molecular techniques have gained much popularity in phylogenetic analyses, morphologists continue to apply novel morphological characters to reconstruct phylogenetic relationships of Diptera1,2,3. This is especially evident for small and complex structures that are difficult to investigate by traditional methods such as light microscopy4. Morphology undoubtedly has the potential for providing a large amount of information for systematics from the numerous character systems that still remain unexplored5.
Our morphological investigation focusses on the antennae, one of the most elaborate and morphologically diverse sensory organs. Antennae perform crucial functions in the life-cycle of most Diptera6,7,8,9,10,11,12,13,14,15,16, with their sensillar equipment fine-tuned by strong selection17. Diptera antennae offer an abundance of structures that provide excellent taxonomic and phylogenetic characters. Antennal ultrastructure has been used in classification and phylogenetic reconstruction at least since the beginning of last century18, yet new information continues to emerge19,20,21. There are numerous studies of antennal ultrastructure in Calyptratae, but they mainly focus on morphology, physiology and function speculation. The antennae of calyptrate flies offer a wealth of information about their evolutionary history, as well as insights into the ecology of each species, which has a valuable potential in phylogenetic studies16,21.
Gasterophilus Leach (horse stomach bot flies) is a relatively small genus that contains eight species worldwide22,23. These species are obligate intestinal parasites of equids (including horses, donkeys and zebras), sharing similar behaviour and life-cycles23,24,25. The unique morphology, biology and epidemiology of these flies have been documented extensively23,26,27,28,29,30,31.
In this study, we conducted a comprehensive morphological comparison of five species of Gasterophilus, including the distribution, types, size and ultrastructure of antennal sensilla. We used these data to construct the first (partial) species-level phylogeny for Gasterophilus.
Materials and Methods
Morphological study
Adult male and female specimens of G. haemorrhoidalis (Linnaeus), G. intestinalis (De Geer), G. nasalis (Linnaeus), G. nigricornis (Loew) and G. pecorum (Fabricius) were captured during 2009–2011 in Kalamaili Ungulate Nature Reserve, Xinjiang, northwest China. The specimens were pinned and air-dried on site before being deposited in Beijing Forestry University. Material of the Palaearctic species G. inermis (Brauer), as well as of the two Afrotropical species G. meridionalis (Pillers & Evans) and G. ternicinctus Gedoelst, were not available for the semi-destructive sampling necessary for the study of antennal sensilla. For general morphology, antennae were studied using an Olympus SZX16 stereoscopic microscope. Series of photographs were taken using a Canon 500D digital camera (Canon, Inc., Tokyo, Japan) mounted on the stereoscopic microscope and superimposed using Helicon Focus (Helicon Soft Ltd, Kharkov, Ukraine) on a Windows 7 platform. All micrographs were processed in Adobe Photoshop CS5 (Adobe Systems, Inc., San Jose, CA, USA).
To prepare samples for scanning electron microscopy (SEM) documentation, the heads of exemplar specimens were excised and rinsed in PBS buffer (pH 7.0) to remove surface debris. For further cleaning, the antennal funiculus (including arista) was dissected from the remaining antenna, cleaned with detergent solution in an ultrasonic cleaner (50 °C, 15 min) and dehydrated in a graded ethanol series. The prepared samples were air-dried and mounted on aluminium stubs with double-sided adhesive tape and coated with gold before observing in a HITACHI S34Q scanning electron microscope (Hitachi Corp., Tokyo, Japan) at the Microscopy Core Facility, Biological Technology Center, Beijing Forestry University (Beijing, China). The length, basal diameter, distal dilation diameter, and distribution of antennal sensilla were measured.
As shown by Zhang et al.12, the types of antennal sensilla in male G. nigricornis are highly similar to those of the female, so for this species only the distribution of sensilla on the female antenna is given in this paper. The terminology follows Zhang et al.12,13,14,15,16.
Phylogenetic analysis
Eighteen characters (13 binary, 5 multistate) were included in the analysis, which covered characters from antennal scape, pedicel, funiculus (including arista) and sensilla (Appendix 1). The matrix (Appendix 2) was compiled using MESQUITE 1.0532. No data are available on the antennal ultrastructure for the genus Gyrostigma Brauer, which is the most probable sister-group to Gasterophilus33, and no specimens are available for the semi-destructive sampling required for proper SEM-documentation. We chose Hypoderma lineatum (Villers) (Hypodermatinae) and Rhinoestrus purpureus (Brauer) (Oestrinae) as outgroup representatives, as the antennal ultrastructure of these two species has been extensively documented by Liu et al.9 and Li et al.10, respectively. For proper rooting of the cladogram, we used Lucilia sericata (Meigen) (Calliphoridae) as a more distant outgroup whose Antennal ultramorphology was reported by Zhang et al.15.
Phylogenetic analyses were performed under the parsimony criterion with the program TNT 1.134. All multistate characters were considered to have their states forming linear transformation series as indicated under the relevant character following the description of character states. They were treated as either additive (=ordered) or non-additive as given in the discussion. Exhaustive searches using implicit enumeration were conducted under equal weights (=unweighted) and with implied weighting35 under a range of k-values as given in the discussion. Branches were collapsed if the maximum branch length was zero.
Branch support for individual nodes was estimated by Bremer’s decay index35,36. Suboptimal trees allowing for five extra steps were produced in TNT to calculate absolute Bremer support values.
Results and Discussion
General description of the antennal morphology in G. haemorrhoidalis, G. intestinalis, G. nasalis, G. nigricornis and G. pecorum
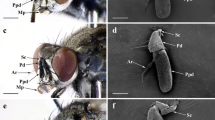
Like other schizophoran Diptera, the antennae of these five species are a pair of segmented sensory appendages arising from membranous sockets between the compound eyes and below the lunule (Figs 1A–C, 2A–C, 3A,B and 4A–C). Each antenna consists of a proximal scape (Sc), a pedicel (Pd) and a distal flagellum composed of a pyriform funiculus (Fn) and a slender, bristle-like arista (Ar) (Figs 1A–C,E, 2A–D,F, 3A–C,E and 4A–C).
(A) Antennae located centrally between compound eyes. (B) Anterodorsal surface, dorsolateral margin and posteroventral surface of antenna in resting position. (C) Antennal scape and pedicel. (D) Base and (inset) middle parts of setae on antennal pedicel. (E) Flagellum. (F–H) Base, middle and tip of arista. Scale bars: A = 1 mm, B = 500 μm, C = 200 μm, D = 100 μm, 5 μm in inset, E = 250 μm, F = 50 μm, G, H = 10 μm. Abbrevations: Ad, anterodorsal surface; Ar, arista; Br, bristle; Dl, dorsolateral margin; Fn, funiculus; Pd, pedicel; Pv, posteroventral surface; Sc, scape.
(A) Antennae located centrally between compound eyes. (B) Antenna in resting position, showing anterodorsal surface, dorsolateral margin in front and posteroventral surface. (C) Antennal scape. (D) Antennal pedicel. (E) Bristle (mechanoreceptor) on antennal pedicel. (F) Flagellum. Scale bars: A = 1 mm, B, F = 250 μm, C = 200 μm, D = 150 μm, E = 50 μm. Abbrevations: Ad, anterodorsal surface; Ar, arista; Br, bristle; Dl, dorsolateral margin; Fn, funiculus; Pd, pedicel; Pv, posteroventral surface; Sc, scape.
(A) Antennae located centrally between compound eyes. (B) Antenna in resting position, showing anterodorsal surface, dorsolateral margin in front and posteroventral surface. (C) Antennal scape and pedicel. (D) Bristle (mechanoreceptor) on antennal pedicel. (E) Anterodorsal surface of antennal funiculus and (inset) aristal tip. Scale bars: A, B = 1mm, C = 200 μm, D = 25 μm, E = 250 μm, 50 μm in inset. Abbrevations: Ad, anterodorsal surface; Ar, arista; Br, bristle; Dl, dorsolateral margin; Fn, funiculus; Pd, pedicel; Pv, posteroventral surface; Sc, scape. SP, sensory pit.
(A) Antennae located centrally between compound eyes. (B) Antenna in resting position, showing anterodorsal surface, dorsolateral margin in front and posteroventral surface. (C) Antennal scape and pedicel. (D) Bristle (mechanoreceptor) on antennal pedicel. Scale bars: A = 1 mm, B = 0.5 mm, C = 100 μm, D = 50 μm, 20 μm in inset. Abbrevations: Ad, anterodorsal surface; AR, annular ridge; Ar, arista; Br, bristle; C, cone; Dl, dorsolateral margin; Fn, funiculus; PB, pedicellar button; Pd, pedicel; Pv, posteroventral surface; Sc, scape.
The scape, the first and shortest segment of the antenna, has a smooth cuticular surface without microtrichia. The pedicel (Figs 1A–C, 2A–C, 3A–C and 4A–C), the second segment, is flattened and disc-like and is covered with microtrichia on the surface. In G. pecorum, the pedicel is elongated and is the longest segment (Fig. 1A–C). Mechanoreceptors (Figs 1C,D, 2C–E, 3C,D and 4C,D) are distributed both on the scape and pedicel. These are short, straight setae or bristles of varying length and with the usual longitudinal grooves on the cuticular surface. The pedicellar button (PB) (Fig. 5) is a button-like structure situated on the articular surface near the pedicellar cleft, and it consists of a circular dome centrally and a slightly convex (G. intestinalis and G. pecorum) or distinctly ridgy (G. haemorrhoidalis, G. nasalis, G. nigricornis) ring peripherally (Fig. 5). Gasterophilus pecorum has two buttons on the pedicel (Fig. 5A), whereas there is only one in the other four species.
The funiculus is the most conspicuous segment of the antenna, housing a variety of sensilla. It is roughly pyriform and can be divided into three aspects: the anterodorsal surface (Ad), the dorsolateral surface (with the arista) (Dl), and the posteroventral surface (Pv) (Figs 1B, 2B, 3B and 4B). A total of five types of antennal sensilla are found, including trichoid sensilla (Figs 6A–D and 7A–D), basiconic sensilla (Figs 6B,C,E,F and 7B,C,E,F), coeloconic sensilla (Figs 8 and 9), clavate sensilla (Fig. 10) and auriculate sensilla (Fig. 11A–C). The length, basal diameter, and distal dilation diameter of these sensilla for both males and females are summarized in Tables 1, 2, 3 and 4, and their distribution is shown in Figs 12, 13, 14, 15 and 16. The arista consists of one or two short basal segments and one long distal segment that tapers gradually (Figs 1A,B,E–H, 2B,F, 3B,E and 4B). The short basal segment(s) are without sensilla, while the distal aristal segment is equipped with coeloconic sensilla III.
(A) Trichoid sensilla in G. pecorum. B Basiconic sensilla I in G. pecorum. C Basiconic sensilla II in G. pecorum. D Trichoid sensilla in G. nasalis. E Basiconic sensilla I in G. nasalis. F Basiconic sensilla II in G. nasalis. Scale bars: A, B, E, F = 5 μm, C, D = 10 μm. Abbreviations: Ba, basiconic sensilla; Ba I, basiconic sensilla I; Ba II, basiconic sensilla II; Mt, microtrichia; Tr, trichoid sensilla.
(A) Trichoid sensilla in G. intestinalis. (B) Basiconic sensilla I in G. intestinalis. (C) Basiconic sensilla II in a sensory pit in G. intestinalis. (D) Trichoid sensilla in G. haemorrhoidalis. E Basiconic sensilla I in G. haemorrhoidalis. F Basiconic sensilla II in G. haemorrhoidalis. Scale bars: A, B, C, D, E = 5 μm, F = 2.5 μm. Abbreviations: Ba, basiconic sensilla; Ba I, basiconic sensilla I; Ba II, basiconic sensilla II; Tr, trichoid sensilla.
(A) Coeloconic sensilla I in G. pecorum. (B) Coeloconic sensilla II in G. pecorum. (C) Coeloconic sensilla III on basal part of arista in G. pecorum. (D) Magnification of coeloconic sensilla III in C. (E) Coeloconic sensilla III in depression on antennal funiculus in G. nasalis. (F) Coeloconic sensilla III on basal part of arista in G. nasalis. (G) Coeloconic sensilla III in F. Scale bars: A, B, E = 2.5 μm, C = 30 μm, D, G = 5 μm, F = 25 μm. Abbreviations: Co I, coeloconic sensilla I; Co II, coeloconic sensilla II; Co III, coeloconic sensilla III.
(A) Coeloconic sensilla I within a sensory pit in G. intestinalis. (B) Coeloconic sensilla III on antennal arista in G. intestinalis. (C) Coeloconic sensilla III on basal part of arista in G. intestinalis. (D) An coeloconic sensillum I in G. haemorrhoidalis. (E) Coelocronic sensilla III on antennal arista in G. haemorrhoidalis. (F) Coeloconic sensilla III on basal part of arista in G. haemorrhoidalis. Scale bars: A, B, D = 5 μm, C = 20 μm, E, F = 10 μm. Abbreviations: Co I, coeloconic sensilla I; Co III, coeloconic sensilla III.
(A) Clavate sensillum in shallow depression in G. nasalis. (B) Clavate sensilla within a sensory pit in G. nasalis. (C) Clavate sensillum in shallow depression in G. intestinalis. (D) Clavate sensillum in shallow depression in G. haemorrhoidalis. Scale bars: A, C = 2.5 μm, B, D = 5 μm. Abbreviation: Cl, clavate sensilla.
(A) Auriculate sensillum in Gasterophilus nasalis. (B) Auriculate sensillum in G. intestinalis. (C) Auriculate sensilla clustered in a sensory pit in G. haemorrhoidalis. (D) Microtrichiae on antennal funiculus in G. pecorum. (E) Microtrichiae on antennal funiculus in G. nasalis. (F) Microtrichiae on antennal funiculus in G. intestinalis. (G) Microtrichiae on antennal funiculus in G. haemorrhoidalis. Scale bars: A, C = 5 μm, B = 2.5 μm, D = 20 μm, E, F, G = 10 μm, F = 10 μm, G = 10 μm. Abbreviations: Au, auriculate sensilla; Mt, microtrichia.
General description of the funicular sensilla
Trichoid sensilla
Trichoid sensilla (Tr) are the longest sensilla on the antennal funiculus (Figs 6A–D and 7A–D; Tables 1, 2, 3 and 4). They are slender, tapering and blunt-tipped structures extending above the microtrichia at various lengths. The trichoid sensilla are concentrated on the anterodorsal surface and show a conspicuous density gradient with the number increasing from base to tip of the funiculus (Figs 12, 13, 14, 15 and 16).
Basiconic sensilla
Basiconic sensilla (Ba) are shorter than trichoid sensilla and less widespread on the funicular surface (Figs 6B,C,E,F, 7B,C,E,F, 12, 13, 14, 15 and 16, Tables 1, 2, 3 and 4). Two subtypes (Ba I, II) of basiconic sensilla can be distinguished by their size in each species. Ba I (Figs 6B,E and 7B,E) and Ba II (Figs 6C,F and 7C,F) are both digitiform with a cylindrical shaft, and have abruptly blunt tips, but Ba I is 1.04 (G. intestinalis) to 2.60 (G. haemorrhoidalis) times longer and 1.40 (G. haemorrhoidalis) to 1.71 (G. nasalis) times thicker at base than Ba II (Tables 1, 2, 3 and 4).
Coeloconic sensilla
Coeloconic sensilla (Co) are the shortest of the sensilla, and are located in sunken cavities (Figs 8 and 9; Tables 1, 2, 3 and 4). Three subtypes (Co I, II, III) of Co can be distinguished: Co I and Co II are distributed on the mid-proximal region of the anterodorsal and the posteroventral surface of the funiculus, whereas Co III is found only on the arista. Co I (Figs 8A,E and 9A,D) is a short peg with deep, longitudinal grooves over the distal 1/3–1/4 of the sensillum, and it is found in all five species studied. Co II (Fig. 8B) is a relatively long peg with an irregular pattern on the terminal part. This type is found only in G. pecorum. Co III is usually short and smooth (Figs 8C,D,F,G and 9B,C,E,F), except in G. nasalis, in which it is approximately triangular in shape and with grooves (Fig. 8E–G), and found exclusively at the base of the distal aristal segment. In G. intestinalis and G. pecorum, Co III are clustered in shallow depressions, while in G. haemorrhoidalis, G. nasalis, and G. nigricornis, they are distributed singly (Figs 8 and 9).
Clavate sensilla
The clavate sensilla (Cl) are characterised by a subapical dilation or swelling, giving them a club-like or spatulate appearance. This type of sensillum is seated in a superficial cavity (Fig. 10) and can only be observed on the most proximal region of the funiculus (Figs 12, 13, 14, 15 and 16). In G. nasalis (Fig. 10B) and G. nigricornis, each clavate sensillum has a short, tapering tip, while in the other species of Gasterophilus, the Cl is more abruptly tapered with the tip broadly rounded (Fig. 10A) or angulated (Fig. 10C,D). Additionally, clavate sensilla are distributed either singularly on the surface (Fig. 10A) or clustered in pits (Fig. 10B).
Auriculate sensilla
In Gasterophilus spp., the auriculate sensilla (Au) are gradually tapered from the base, with a length-width ratio <3 (Tables 1, 2, 3 and 4), somewhat resembling the ear of a rabbit (Fig. 11A–C). Au are distributed either singly on the surface or clustered in sensory pits.
Auriculate sensilla were first discovered by Setzu et al.37 in Protophormia terraenovae (Robineau-Desvoidy) (Calliphoridae) and described as ear- or spoon-like formations presenting a concavely indented or sunken surface distally. In this study, we observed this type of sensilla in four species (G. haemorrhoidalis, G. intestinalis, G. nasalis and G. nigricornis). It should be noted that distinguishing antennal sensilla solely by their cuticular surface concavity can be misleading, since several sensilla that we observed (e.g., clavate sensilla) had partly sunken cuticular surfaces. They were situated amongst morphologically similar sensilla without any sign of concavity. Shrinkage and deflation happen occasionally during the dehydration process for scanning electron microscopy, which may explain this inconsistency38,39.
Sensory pit
The sensory pit is a cave-like depression in the funicular surface that contains several sensilla of the same type (Figs 3E, 7C, 9A, 10B, 11C, 12, 13, 14, 15, 16 and 17). All Gasterophilus spp. have the funiculus equipped with numerous sensory pits.
(A) Overview of sensory pits on funiculus in Gasterophilus haemorrhoidalis. (B) Dorsal view of a sensory pit with a fringe of microtrichiae in G. pecorum. (C) Dorsal view of a sensory pit with a fringe of microtrichiae in G. nasalis. (D) Lateral view of a sensory pit with broken microtrichiae. Scale bars: A = 50 μm, B–D = 10 μm.
The concept of sensory (or ‘olfactory’) pits needs clarification, as any depression on the cuticle in connection with sensilla are sometimes referred to as a sensory pit37,40. McAlpine19 differentiated between “simple pits” as opposed to the “deep, sac-like invagination of the cuticle of segment 3 containing several trichoid sensilla and opening to the exterior by a relatively small pore”, which Lowne41 termed the ‘sacculus’. Similarly, Zhang et al.14 separated the ‘sensory pit’ as a single-chambered invagination containing a cluster of sensilla from the ‘sacculus’, which was defined as a multi-chambered invagination stretching into the cavity of the antennal funiculus, and often with a complement of different types of sensilla. We propose that the term ‘sensory pit’ should be used to describe a cluster of sensilla located inside a saucer- or bowl-like depression of the cuticular surface with at most one third of their length emerging above the surrounding surface. A fringe of microtrichia is usually found around the edge of a sensory pit. Pezzi et al.42 used the term ‘olfactory pit’, but as antennal sensilla may have other functions, e.g., hygro- or thermoreception43, we prefer the broader term ‘sensory pit’.
Phylogeny
Analysing the matrix under equal weights and with all characters treated as non-additive (i.e., unordered) yielded two most parsimonious trees. The strict consensus of the two trees gave a sister-group relationship between the clade Hypodermatinae + Oestrinae and a largely unresolved Gasterophilus (Fig. 18). Analysing the data as non-additive under implied weights resulted in a fully resolved tree for all values of k ≥1, which differs from the equally weighted analysis in the topology of Gasterophilus, with G. pecorum as sister taxon to all other species of Gasterophilus (Fig. 19). An identical tree is obtained in analyses with the multistate characters treated as additive, both in the equally weighted analysis and in analyses with implied weighting and for all k-values (i.e., k ≥1). Bremer supports for every node of the minimum-length cladogram are given in Fig. 19.
Numbers refer to characters (above branches) and states (below branches) as discussed in the text and in Appendix 1, black circles represent unique character state changes, white circles represent homoplasious character state changes. Underlined numbers are Bremer support values obtained with characters treated as additive using the implicit enumeration analysis.
The genus Gasterophilus is supported as a monophyletic group (Fig. 19) based on two synapomorphies: both flat and hair-like microtrichia on antennal funiculus (C8: 1) and the arista with coeloconic sensilla III (C11: 0) (Figs 8C,D,F,G and 9B,C,E,F). A great diversity of antennal sensilla types and locations are evident within this small genus, compared with 3–6 types of antennal sensilla arranged mostly on the funicular surface in other flies8,14,15,43,44,45,46. All species of Gasterophilus have seven types of sensilla, except for G. pecorum having only six types. These cover nearly all types of sensilla (e.g., plaques are not detected) described in cyclorrhaphan flies. Some sensilla can be divided into several subtypes, making a strict calculation of the total number of sensillar types in this genus potentially misleading. Besides, large numbers of sensilla are located in numerous sensory pits and so are more difficult to study. There is increasing evidence that specific types of antennal sensilla have specific functions47,48. The diverse sensilla and large number of sensory pits in Gasterophilus spp., may increase sensitivity to specific odours, while simultaneously maximizing protection of the fragile sensilla from damage8,9,10,13,15,49.
Within Gasterophilus, G. pecorum branches off at the base, emerging with four autapomorphies: the microtrichia are extensive on the outer side but absent on the inner side of the pedicel (C0: 2) (Fig. 1C), length-width ratio of antennal pedicel more than 0.8 (C1: 2), two antennal pedicel buttons (C3: 2) (13A), and two subtypes of coeloconic sensilla on funiculus (C10: 1) (Fig. 8A,B), and with four homoplasious character states: the pedicel partly enveloping the funiculus (C2: 1), the slim mechanoreceptor (C7: 0) with twisting grooves (C6: 1) but no socket (C5: 1) (Fig. 1D).
Gasterophilus pecorum has one more subtype of Co than all the other species included in the present analysis (C10: 1). Co are sensitive to many olfactory cues48,50,51,52,53,54 instead of being only hygro- or thermo-sensitive as speculated previously55,56,57. Further behavioural and electrophysiological studies are required to understand why G. pecorum has acquired more types of antennal coeloconic sensilla than other bot flies, but it is noteworthy that G. pecorum will deposit eggs at a distance from its host rather than directly on the host23, which is probably a derived behaviour that may require additional sensory input to monitor host location.
The second clade in Gasterophilus is supported by three synapomorphies: length-width ratio of antennal pedicel less than 0.5 (C1: 0), only flat and grooved microtrichia on antennal funiculus (C8: 0), the presence of auriculate sensilla on antennal funiculus (C14: 0) (Fig. 11A–C). The third clade is supported by two synapomorphies: the pedicel button with distinctly ridgy ring (C4: 1) and coeloconic sensilla III distributed singly on the arista (C12: 0). In this clade, a sister-group relationship of G. nigricornis and G. nasalis is supported, based on six homoplasious character states: the strong mechanoreceptor (C7: 1) with straight grooves (C6: 0) and socket (C5: 0) (Figs 2E, 3D and 4D), clavate sensillum with a tapering tip (C13: 0), arista with two segments (C16: 0) and distal aristomere with sparse microtrichia (C17: 1).
No prior study has specifically investigated a species-level phylogeny for Gasterophilus. Otranto et al.58 analysed partial sequence data from the mitochondrial COI gene and the ribosomal genes 16S and 28S in a study of the “differentiation and phylogenesis” of five species of Gasterophilus. Unrooted Maximum Likelihood “phylograms” were given based on each gene, but no explicit phylogeny was presented. Rooting their three phylograms (Fig. 20) will generate phylogenies strongly conflicting with the present study (Fig. 19), which may be caused by the sparcity of molecular data. Otranto et al.58 also suggested that G. haemorrhoidalis and G. intestinalis could be morphotypes of the same species based on high genetic similarity, in particular of the 28S gene (Fig. 20A). They even considered the main site of development of the third instar larva as sufficiently similar to support conspecificity in spite of known differences31. This view did not consider significant evidence that G. haemorrhoidalis and G. intestinalis are biologically and morphologically quite distinct in several features like oviposition site, oviposition behaviour, details of first instar larva (especially the cephaloskeleton) and adult male and female morphology (for an exquisite treatment see Grunin31, which incidentally was not cited by Otranto et al.58). In the present study, we found significant differences in the antennal morphology of these two species (Figs 14 and 15), such as a distinctly ridgy ring of the pedicellar button and singly distributed aristal Co III in G. haemorrhoidalis, while G. intestinalis has the alternative state for both these characters (see the results and list of characters in Appendix 1). Actually, the existing similarities between the two species may relate entirely to their shared ancestry, i.e., be symplesiomorphic, because G. intestinalis is cladistically subordinate to G. haemorrhoidalis, with the latter being more closely related to G. nigricornis and G. nasalis.
Three rooted cladograms constructed using maximum likelihood analysis of 28S rDNA sequences (A), COI sequences (B) and 16S rRNA sequences (C) (modified from Otranto et al.58).
Additional Information
How to cite this article: Zhang, D. et al. The antenna of horse stomach bot flies: morphology and phylogenetic implications (Oestridae, Gasterophilinae: Gasterophilus Leach). Sci. Rep. 6, 34409; doi: 10.1038/srep34409 (2016).
References
Giroux, M., Pape, T. & Wheeler, T. A. Towards a phylogeny of the flesh flies (Diptera: Sarcophagidae): morphology and phylogenetic implications of the acrophallus in the subfamily Sarcophaginae. Zool J Linn Soc-Lond 158, 740–778 (2010).
Lambkin, C. L. et al. The phylogenetic relationships among infraorders and superfamilies of Diptera based on morphological evidence. Syst Entomol 38, 164–179 (2012).
Yeates, D. K. & Wiegmann, B. M. Phylogeny and evolution of Diptera: recent insights and new perspectives. In: The evolutionary biology of flies (eds Yeates, D. K. & Wiegmann, B. M. ) 14–44 (Columbia University Press, 2005).
Araujo, D. P., Tuan, M. J. M., Yew, J. Y. & Meier, R. Analysing small insect glands with UV-LDI MS: high-resolution spatial analysis reveals the chemical composition and use of the osmeterium secretion in Themira superba (Sepsidae: Diptera). J Evolution Biol 27, 1744–1750 (2014).
Wipfler, B., Courtney, G. W., Craig, D. A. & Beutel, R. G. First μ-CT-based 3D reconstruction of a dipteran larva—the head morphology of Protanyderus (Tanyderidae) and its phylogenetic implications. J Morphol 273, 968–980 (2012).
Amer, A. & Mehlhorn, H. The sensilla of Aedes and Anopheles mosquitoes and their importance in repellency. Parasitol Res 99, 491–499 (2006).
Hallem, E. A., Dahanukar, A. & Carlson, J. R. Insect odor and taste receptors. Annu Rev Entomol 51, 113–135 (2006).
Hunter, F. F. & Adserballe, C. F. Cuticular structures on the antennae of Hypoderma bovis De Geer (Diptera: Oestridae) females. Int J Insect Morphol Embryol 25, 173–181 (1996).
Li, X. Y., Liu, X. H., Ge, Y. Q. & Zhang, D. Scanning electron microscopy of antennal sensory organs of the cattle grub, Hypoderma lineatum (Diptera: Oestridae). Parasitol Res 114, 3865–3871 (2015).
Liu, X. H., Li, X. Y., Li, K. & Zhang, D. Ultrastructure of antennal sensory organs of horse nasal-myiasis fly, Rhinoestrus purpureus (Diptera: Oestridae). Parasitol Res 114, 2527–2533 (2015).
Wang, Q. K., Zhang, M., Li, K. & Zhang, D. Olfactory sensilla on antennae and maxillary palps of Fannia hirticeps (Stein, 1892) (Diptera: Fanniidae). Microsc Res Techniq 75, 1313–1320 (2012).
Zhang, D., Wang, Q. K., Hu, D. F. & Li, K. Sensilla on the antennal funiculus of the stomach bot fly, Gasterophilus nigricornis (Diptera: Oestridae). Med Vet Entomol 26, 314–322 (2012a).
Zhang, D., Wang, Q. K., Hu, D. F. & Li, K. Cuticular structures on antennae of the bot fly, Portschinskia magnifica (Diptera: Oestridae). Parasitol Res 111, 1651–1659 (2012b).
Zhang, D., Wang, Q. K., Yang, Y. Z., Chen, Y. O. & Li, K. Sensory organs of the antenna of two Fannia species (Diptera: Fanniidae). Parasitol Res 112, 2177–2185 (2013a).
Zhang, D., Liu, X. H., Li, X. Y., Zhang, M. & Li, K. Antennal sensilla of the green bottle fly, Lucilia sericata (Meigen) (Diptera: Calliphoridae). Parasitol Res 112, 3843–3850 (2013b).
Zhang, D. et al. Ultrastructural investigation of antennae in three cutaneous myiasis flies: Melophagus ovinus, Hippobosca equina, and Hippobosca longipennis (Diptera: Hippoboscidae). Parasitol Res 114, 1887–1896 (2015).
Hallberg, E. & Hansson, B. S. Arthropod sensilla: morphology and phylogenetic considerations. Microsc Res Techniq 47, 428–439 (1999).
Williston, S. W. The antennae of Diptera: a study in phylogeny. Biol Bull-US 13, 324–332 (1907).
McAlpine, D. K. Observation on antennal morphology in Diptera with particular reference to the articular surfaces between segments 2 and 3 in the Cyclorrhapha. Rec Aust Mus 63, 113–166 (2011).
Urbaneka, A., Piotrowicz, M., Szadziewski, R. & Giłka, W. Sensilla coeloconica ringed by microtrichia in host-seeking biting midges. Med Vet Entomol 28, 355–363 (2014).
Wang, Q., Liu, X., Lu, P. & Zhang, D. Ultrastructure of antennal sensilla in Hydrotaea armipes (Fallén) (Diptera: Muscidae): New evidence for taxonomy of the genus Hydrotaea . Zootaxa 3790, 577–586 (2014).
Cogley, T. Status of the stomach bot fly Gasterophilus lativentris (Brauer) 1858 (Diptera: Gasterophilidae). Syst Entomol 16, 135–136 (1991a).
Zumpt, F. Myiasis in man and animals of the old world. (Butterworths, 1965).
Colwell, D. D., Hall, M. J. R. & Scholl, P. J. The oestrid flies: biology, host-parasite relationships, impact and management. (CABI Publishing, 2006).
Bezdekova, B., Jahn, P. & Vyskocil, M. Pathomorphological study on gastroduodenal ulceration in horses: localization of lesions. Acta Vet Hung 55, 241–249 (2007).
Cogley, T., Anderson, J. & Cogley, L. Migration of Gasterophilus intestinalis larvae (Diptera: Gasterophilidae) in the equine oral cavity. Int J Parasitol 12, 473–480 (1982).
Cogley, T. Key to the eggs of the equid stomach bot flies Gasterophilus Leach 1817 (Diptera: Gasterophilidae) utilizing scanning electron microscopy. Syst Entomol 16, 125–133 (1991b).
Cogley, T. & Cogley, M. C. Inter-relationship between Gasterophilus larvae and the horse’s gastric and duodenal wall with special reference to penetration. Vet Parasitol 8, 127–142 (1999).
Cogley, T. & Cogley, M. C. Field observations of the host–parasite relationship associated with the common horse bot fly, Gasterophilus intestinalis. Vet Parasitol 88, 93–105 (2000).
Colwell, D. D., Otranto, D. & Horak, I. G. Comparative scanning electron microscopy of Gasterophilus third instars. Med Vet Entomol 21, 255–264 (2007).
Grunin, K. J. Die Fliegen der Palaearktischen Region. (Schweizerbart’sche, 1969).
Maddison, W. P. & Maddison, D. R. Mesquite: a modular system for evolutionary analysis. Version 2.73. http://mesquiteproject.org (2010).
Pape, T. Phylogeny of Oestridae (Insecta: Diptera). Syst Entomol 26, 133–171 (2001).
Goloboff, P., Farris, S. & Nixon, K. T. N. T. : tree analysis using new technology. Version 1.1. Program and documentation available from the authors. http://www.zmuc.dk/public/phylogeny (2003).
Goloboff, P. Estimating character weights during tree search. Cladistics 9, 83–91 (1993).
Bremer, K. Branch support and tree stability. Cladistics 10, 295–304 (1994).
Setzu, M. D., Poddighe, S. & Angioy, A. M. Sensilla on the antennal funiculus of the blow fly, Protophormia terraenovae (Diptera: Calliphoridae). Micron 42, 471–477 (2011).
Boyde, A. & Maconnachie, E. Morphological correlations with dimensional change during SEM specimen preparation. Scanning Electron Microscopy 4, 27–34 (1981).
Jones, C. G. Scanning electron microscopy: preparation and imaging for SEM. Methods in Molecular Biology 915, 1–20 (2012).
Fernandes, F. F., Linardi, P. M. & Chiarini-Garcia, H. Morphology of the antenna of Dermatobia hominis (Diptera: Cuterebridae) based on scanning electron microscopy. J Med Entomol 39, 36–43 (2002).
Lowne, B. T. The anatomy, physiology, morphology, and development of the blow-fly (Calliphora erythrocephala) in: A study in the comparative anatomy and morphology of insects; with plates and illustrations executed directly from the drawings of the author, Vol. 2 (ed. Porter, R. H. ) 22–52, 351–778 (Rarebooksclub, 1895).
Pezzi, M. et al. Ultrastructural morphology of the antenna and maxillary palp of Sarcophaga tibialis (Diptera: Sarcophagidae). J Med Entomol 10.1093/jme/tjw061 (2016).
Shanbhag, S. R., Singh, K. & Singh, R. N. Fine structure and primary sensory projections of sensilla located in the sacculus of the antenna of Drosophila melanogaster . Cell Tissue Res 282, 237–249 (1995).
Liu, X. H., Zhang, M., Shi, J. N., Li, K. & Zhang, D. Ultrastructure of antennal sensilla of a parasitoid fly, Pales pavida Meigen (Diptera: Tachinidae). Micron 54–55, 36–42 (2013).
Shanbhag, S. R., Müller, B. & Steinbrecht, R. A. Atlas of olfactory organs of Drosophila melanogaster 2. Internal organization and cellular architecture of olfactory sensilla. Arthropod Struct Dev 29, 211–229 (2000).
Smallegange, R. C., Kelling, R. J. & Den Otter, C. J. Types and numbers of sensilla on antennae and maxillary palps of small and large houseflies, Musca domestica (Diptera, Muscidae). Microsc Res Tech 71, 880–886 (2008).
Chapman, R. F. Chemoreception: the significance of receptor numbers. Adv Insect Physiol 16, 247–356 (1982).
Couto, A., Alenius, M. & Dickson, B. J. Molecular, anatomical, and functional organization of the Drosophila olfactory system. Curr Biol 15, 1535–1547 (2005).
Bruyne, M., Foster, K. & Carlson, J. R. Odor coding in the Drosophila antenna. Neuron 30, 537–552 (2001).
Mei, G. et al. Variant ionotropic receptors are expressed in olfactory sensory neurons of coeloconic sensilla on the antenna of the desert locust (Schistocerca gregaria). Int J Biol Sci 10, 1–14 (2013).
Schneider, D. & Steinbrecht, R. A. Checklist of insect olfactory sensilla. Symp. Zool Soc Lond 23, 279–297 (1968).
Shanbhag, S. R., Muller, B. & Steinbrecht, R. A. Atlas of olfactory organs of Drosophila melanogaster 1. types, external organization, innervation and distribution of olfactory sensilla. Int J Insect Morphol Embryol 28, 377–397 (1999).
Steinbrecht, R. A. The fine structure of thermo-/hygrosensitive sensilla in the silkmoth Bombyx mori: receptor membrane structure and sensory cell contacts. Cell Tissue Res 255, 49–57 (1989).
Yao, C. A., Ignell, R. & Carlson, J. R. Chemosensory coding by neurons in the coeloconic sensilla of the Drosophila antenna. J Neurosci 25, 8359–8367 (2005).
Altner, H., Schaller-Selzer, L., Stetter, H. & Wohlrab, I. Poreless sensilla with inflexible sockets; a comparative study of a fundamental type of insect sensilla probably comprising thermo- and hygroreceptors. Cell Tissue Res 234, 279–307 (1983).
Zacharuk, R. Y. Antennal sensilla. In: Comparative Insect Physiology, Biochemistry and Pharmacology, vol. 6 (eds Kerkut, G. A. & Gilbert, L. I. ) 1–69 (Pergamon Press, 1985).
Ochieng, S. A., Park, K. C., Zhu, J. W. & Baker, T. C. Functional morphology of antennal chemoreceptors of the parasitoid Microplitis croceipes (Hymenoptera: Braconidae). Arthropod Struct Dev 29, 231–240 (2000).
Otranto, D., Traversa, T., Milillo, P., de Luca, F. & Stevens, J. Utility of mitochondrial and ribosomal genes for differentiation and phylogenesis of species of gastrointestinal bot flies. J Econ Entomol 98, 2235–2245 (2005).
Acknowledgements
The authors are grateful to Prof. Mark Elgar (University of Melbourne) for the thorough linguistic revision. This study was supported by the Fundamental Research Funds for the Central Universities (No. JC2015-04), National Science Foundation of China (No. 31572305), Program for New Century Excellent Talents in University (No. NCET-12-0783), Beijing Higher Education Young Elite Teacher Project (No. YETP0771), and the State Scholarship Fund of China Scholarship Council (No. 201306515009).
Author information
Authors and Affiliations
Contributions
D.Z., T.P. and X.Y.L. conceived and designed the study. X.H.L. and Q.K.W. performed the LM and SEM documentation. X.H.L. draw the distribution of sensilla. D.Z., X.Y.L. and T.P. produced the matrix and analysed the phylogenetic data. D.Z., X.Y.L., X.H.L., Q.K.W. and T.P. wrote the manuscript and made contributions to the discussion. D.Z., X.Y.L., Q.K.W. and T.P. revised the manuscript, and all authors approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, D., Li, X., Liu, X. et al. The antenna of horse stomach bot flies: morphology and phylogenetic implications (Oestridae, Gasterophilinae: Gasterophilus Leach). Sci Rep 6, 34409 (2016). https://doi.org/10.1038/srep34409
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep34409
This article is cited by
-
Antennal and palpal sensilla of three predatory Lispe species (Diptera: Muscidae): an ultrastructural investigation
Scientific Reports (2021)
-
Ultrastructure of adult Gasterophilus intestinalis (Diptera: Gasterophilidae) and its puparium
International Journal of Tropical Insect Science (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.