Abstract
Carbon storage is affected by photosynthesis (Pn) and soil respiration (Rs), which have been studied extensively in natural and agricultural systems. However, the effects of Pn and Rs on carbon storages in the presence of arbuscular mycorrhizal fungi (AMF) in coalfields remain unclear. A field experiment was established in 2014 in Shendong coal mining subsidence area. The treatments comprised two inoculation levels (inoculated with or without 100 g AMF inoculums per seedlings) and four plant species [wild cherry (Prunus discadenia Koebne L.), cerasus humilis (Prunus dictyneura Diels L.), shiny leaf Yellow horn (Xanthoceras sorbifolium Bunge L.) and apricot (Armeniaca sibirica L.)]. AMF increased Pn of four species ranging from 15.3% to 33.1% and carbon storage, averaged by 17.2% compared to controls. Soil organic carbon (OC), easily extractable glomalin-relation soil protein (EE-GRSP) and total glomalin-relation soil protein (T-GRSP) were significantly increased by AMF treatment. The effect of AMF on the sensitivity of Rs depended on soil temperature. The results highlighted the exponential models to explain the responses of Rs to soil temperature and for the first time quantified AMF caused carbon sequestration and Rs. Thus, to our knowledge, AMF is beneficial to ecosystems through facilitating carbon conservation in coalfield soils.
Similar content being viewed by others
Introduction
Carbon storage depends on the balance between carbon sequestration by plant Pn and carbon release to atmosphere through Rs1,2. Therefore, if plant Pn or Rs is altered, carbon balance will be affected. Rs, especially seasonal variations, are significantly affected by soil temperature and moisure3,4. However, the impact of soil microbes on Rs or carbon storage remains unclear. So far, quantified contribution of soil microbes, especially AMF has not been reported yet. AMF are obligate plant symbionts that associates with the roots of more than 80% vascular land plants, which significantly enhance long-term success of mine site reclamation5. Studies showed that AMF are helpful for building up a productive, healthy and sustainable post-mine land ecosystem with vegetation cover. The positive impact of AMF on reclaimed soil fertility and plant communities succession has been well documented6,7,8. However, few studies focused on the effects of AMF on Rs in post-mine land. A recent study conducted in Germany grassland communities reported that AMF stimulated Rs on pasture soil, leading to elevated CO2 level and temperature, with most carbon sequestered in belowground parts, making Rs an important component of carbon balance9,10. Factors such as soil moisture, soil temperature, CO2 enrichment and precipitation changes were all affected Rs. However, relevant studies remain scare and the complexity of various interactions that affect Rs (such as soil temperature vs moisture, soil temperature vs microbes, moisture vs microbes interactions) are still poorly understood.
Rs includes intergated CO2 flux of root respiration, mycorrhizal respiration (considered as part of autotrophic respiration) and heterotrophic respiration, which are controlled by carbon supply and temperature9,10. Carbon supply is an important Rs impact factor, which primarily depends on plant productivity and generally responds positively to CO2 enrichment with the increased Pn11. Numerous studies confirmed that carbon storage depended on plant Pn and Rs via AMF in greenhouse or field experiments7,9,10,11.
AMF usually form symbiotic associations with trees in diverse forests. The fungi rely on host plant to obtain carbon supply and utilize 5–20% of the net photosynthate of the symbiotic system11. Global forest soil releases 24 Pg Carbon per year into atmosphere via CO2 efflux and generates CO2 from a widely variety of belowground organisms, with AMF as the dominant carbon source12. AMF as an important carbon and soil CO2 efflux source have been well studied under various temperature and moisture conditions12. A pulse-labeling experiment showed that in temperate prairie, about 4–6% photoassimilates were facilitated via AMF10. Simultaneously, AMF enhanced 16% soil CO2 efflux when Lolium pernne roots were colonized and 8% carbon supply was derived from AMF in a barley field13. To date, no studies have been carried out on the contribution of AMF on carbon storage in coal mining soil.
Respiratory quotient (Q10) is defined as the temperature sensitivity of Rs which is derived from substrate availability, which is the respiration variation ratio when soil temperature rises 10 °C. Q10 is largely affected by a series of environmental factors, such as soil physicochemical properties and soil moisture, etc. However, few studies have quantified the effects of AMF on apparent Q10 from different tree species. Thus, knowledge about the function of AMF on Rs, Pn and carbon storage is insufficient14.
Notably, CO2 concentration indirectly influences Rs and carbon allocation from host species to fungus, suggesting that root participates in carbon fixation15,16,17. Most colonized plants enhanced productivity or nutritional status of the plant symbioses system when exposed to higher CO2 concentration18. In addition, mycorrhizal hyphae assisted carbon re-distribution from aboveground parts to roots via utilizing the photosynthate and provides multiple adherent agents such as extracellular polymer and amino acid, etc than non-colonized plants19. Briefly, AMF enhance soil carbon proportion in higher CO2 environment20,21,22. However, lacking a better understanding of these responses in coal mining soil limited to prediction soil carbon storage when AMF existed in current scenarios.
Thus, the experiment planted wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot in coal mining soil and half seedlings were inoculated with AMF inoculums. Based on the experimental results, two hypotheses was proposed that: (i) plant species preferentially facilitate their photosynthetic ability and increase Rs rates, leading to different carbon storage amount; and (ii) soil temperature rather than soil moisture is sensitive to Rs due to the extreme drought. The future goal is to investigate the effect of AMF on carbon sequestration in coal mining fields.
Results
Pn and Rs temporal dynamics
Pn was significantly greater with AMF inoculation in all the four species from July to September (Fig. 1). Specifically, AMF treatment increased Pn in wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot by 21.2%, 15.0%, 53.1% and 7.4%, respectively. Meanwhile, Pn in all four tree species increased from July to August and then strikingly decreased from August to September (Fig. 1).
In all tree species, Rs was significantly decreased from July to September, with slight decrease from July to August and reduced sudden reduction of 21.3–26.9% in September (Fig. 2).
Cumulative carbon measures
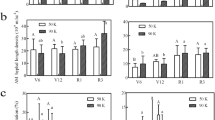
Annual cumulative carbon in AMF treated wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot were about 1706, 1784, 2049 and 2065 g C.m−2.yr−1 (Fig. 3a–d). Compared to the corresponding controls, the annual cumulative carbon levels in the four tree species were increased by 28.3%, 44.6%, 31.8% and 33.5%, respectively (Fig. 3a–d). In growing season, AMF increased the carbon storage by 29.4%, 45.7%, 32.4% and 34.4% (P < 0.0001) (Fig. 3a–d). AMF also significantly increased the annual cumulative Rs carbon in the four tree species by 32.4%, 36.6%, 25.2% and 29.9%, when compared to the corresponding controls (P < 0.0001) (Fig. 4a–d).
Carbon storage in growing and non-growing season.
(a–d) Indicated wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot, respectively. Bar represented means ± SE. −M and +M denoted without and with AMF inoculation. Different lowercase letters indicated that −M and +M were significantly different at 5% level by LSD.
Soil carbon releases via Rs in growing and non-growing seasons.
(a–d) Showed that wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot, respectively. Bar represented means ± SE. −M and +M denoted without and with AMF inoculation, respectively. Different lowercase letters indicated that −M and +M were significantly different at 5% level by LSD.
Soil temperature sensitivity to Rs
Rs was calculated using exponential function (Eq. 1), temperature dynamic showed over 70% Rs variation in AMF treated groups and the correlation of Rs showed only 40% variation in control groups (Table 1). Apparent Q10 and the coefficient b were decreased significantly in control apricot and wild cherry (P < 0.05) (Table 1; Fig. 5a,d). However, AMF decreased previous two parameters in cerasus humilis and shiny leaf Yellowh orn (Fig. 5b,c). Apparent Q10 responded differently to temperature (the precipitation in 2014 was higher than 10 year’s average precipitation) in August from that in July or September (Fig. 6a–d).
Exponential models of Rs based on soil temperature.
Soil temperature was plotted against Rs for plant species with or without AMF inoculation (n = 36). (a–d) Indicated that wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot, respectively. Solid and hollow circles represented data from without and with AMF inoculation. Solid and dash lines represented the fitted curve of Rs to soil temperature without and with AMF inoculation, respectively.
Apparent Q10 values represents the sensitivity of soil respiration and soil temperature when rises 10 °C.
Monthly apparent Q10 values variation of four tree species without or with AMF inoculation from July to September. Values represented means ± SE. (a–d) Indicated wild cherry, cerasus humilis, shiny leaf Yellowh orn and apricot of tree species, respectively.
Effects of AMF inoculation on carbon-linked parameters
In control of wild cherry, cerasus humilis, shiny leaf yellowh orn and apricot, the production of OC, EE-GRSP and T-GRSP almost did not change with time (Table 2). In contrast, the production of soil OC, EE-GRSP and T-GRSP in AMF treated of four species increased significantly over time (Table 2). After three months of treatment, the increment of OC, EE-GRSP and T-GRSP in AMF treated groups were all significant (27–37%, 34–45% and 19–22%, respectively (Table 2). After five months of AMF treatments, compared to the control, the levels of OC, EE-GRSP and T-GRSP in AMF treated groups were remarkable enhanced by 52–61%, 55–70% and 36–44%, respectively (Table 2). Collectively, AMF had a positively impacted on soil carbon-linked parameters over time in all of the four tree species (Table 2).
Discussion
The present study firstly examined photosynthetic ability, Rs and carbon storage in respond to AMF inoculation in the coalfields. Under AMF treatment, carbon storage was derived from belowground parts of the tree species. Meanwhile, the temporal variability strongly responded to Rs and annual cumulative carbon with exponential models.
Pn and Rs responses to AMF inoculation
AMF inoculation incurred a series changes in Pn, Rs and carbon storage via close symbiosis with host plants, leading to enhanced carbon production and improved nutrition status against the detrimental effects14,23,24,25,26,27. Meanwhile, AMF accelerated the organic matter (OM) decomposition rates28 and provided some raw materials, which elevated CO2 concentration in micro circumstance29,30 and facilitated soil carbon turnover in the agro-or grassland via CO2 stimulation effects31. In the moist forest, abundant and diverse AMF (e.g. tropical forests) species created a transient carbon sink via N transferring to reduce carbon lost. This implied that AMF promotes carbon production and storage via increasing Pn, which ameliorated the negative effect caused by unfavorable environment32. Recently, strong evidences showed that the colonized plants have higher Pn and ribulose-1,5-bisphosphate carboxylase/oxygenase activity than non-colonized plants, which compensated greater photosynthate33,34. Carbon was produced directly from Pn and AMF perhaps supported the higher metabolism of plants and greater productivity of carbon in the harsh environment35,36,37,38,39. Furthermore, soil temperature decreasing inhibited substrate movement, lowing microbal activity, which decreased plant Pn and belowground carbon allocation. Temperature also reduced Pn and the belowground coupling, which potentially affected the substrate availability and carbon cycling40. The results demonstrated that AMF can increase Pn of the plant, thus increasing carbon allocation.
Carbon partitioning responses to AMF inocluation
Due to its ability to enhance carbon allocation, AMF has been used in agricultural field and grasslands, but information about carbon storage is omitted in mining soil. Carbon accumulation tended to be higher with AMF inoculation in this study in the growing season. AMF sequestered greater carbon via enhanced Pn and photosynthate production, which was transferred from aboveground to roots or microbes. AMF counteracted carbon loss of respiration, due to increase productivity and nutrient acquisition, especially carbon sequestration35,41. In the non-growing seasons, AMF perferentially allocated photosynthate from tree branchs to belowground parts, increasing the EE-GRSP and T-GRSP of carbon stocks14,38. Estimately, AMF utilized a large proporation (about 20%) of photosynthates19,42,43,44,45, leading to more carbon retention via symbiosis46,47,48,49,50. Consequently, AMF inoculation resulted in remarkable greater carbon sequestration.
Nonlinear response to Rs
AMF affected carbon storage and partition, which has been discussed in the previous study51. However, no data have shown whether interactions between species and temperature affect AMF facilitated carbon sequestration52. As literatures indicated, carbon productivity of the plants was greater with AMF, due to higher availability of nutrients from the soil environments53. The beneficial effect of AMF in carbon sequsteration was considered a result of improved availability of nutrients and altered carbon allocation caused by AMF54,55. The exponential models successfully illustrated the changes of Rs in response to soil temperature variation and captured monthly dynamics of all tree species used in the exepriment.
Hysteresis between Rs and soil temperature exists in various ecosystems and vegetations56,57. In general, the decoupling of Rs in response to soil temperature is attributed to confound effect, such as precipitation or physiological drought caused hysteresis, which leads to decreased AMF activity and subsequently lower CO2 production57. Over all, the Rs-temperature relationship was affected by Pn, litterfall and soil microbe activities58,59,60,61. However, the detailed mechanisms remain unclear. In this study, due to the existence of hystersis, Rs in control groups was higher than AMF inoculation groups under a given temperature, which was in agreement with the previous hypothesis62. Most likely, AMF plants accumulated more fresh litter in a given temperature than non-colonized plants. Q10 was often used to represent the temperature measurement depth in greenhouse or field exprimental models14,63. In this work, apparent Q10 increased in AMF treated groups, indicating that AMF increases the sensitivity of Rs to soil temperature. Apparent Q10 value was lower in summer than in cooler season, which might be because of higher substrate availability and severe soil moisture limitation in summer. In consistent with the previous publication14, the experiment data showed that Rs strongly depends on soil temperature in the study.
Previous studies have used exponential model but ingored the function of AMF64. The results suggested that AMF plays a key role in Rs and carbon stocks. AMF and soil temperature in combination modulate apparent Q10 in coalfields.
Conclusions
This study provided new insights into the impacts of AMF on Rs and carbon storage in coalfields. AMF enhances carbon sequestration and the sensitivity of Rs to temperature. Higher AMF infection rate significantly enhances Rs and Pn, the positive AMF function is through promoting plant growth, especially increasing leaf area, chlorophyll content and the Q10 value. These results clearly demonstrated that AMF infection increased organic matter and glomalin which may be associated with the enhancement of carbon storage in soil. The model of Rs to soil temperature should be reassessed to take into account of the interaction between the soil volumetric moisture and mycorrhizal fungus. Thus, further study will focus on long-term AMF effect on carbon stock in a tree species specific manner.
Materials and Methods
Study site and experimental set-up
This experiment was conducted at the ecological reclaimed region, which located in Daliuta town (39°18′N, 110°4′E), Shenmu County, Yulin City, Shaanxi Province, Northwest China at 1,200 m height above sea level. This site was a typical junction area of Shanxi, Shaanxi and Inner Mongolia of three Provinces and the south margin of Mu us desert in the loess plateau transition zone. About 70% precipitation falls from June to September, the 10-year (2005–2014) average total precipitation and the potential evaporation were about 150 mm and 2000 mm according to Shenmu Meteorological Station near the experiment site. The experimental location had a typical characteristic of continental climate, with annual average temperature about 8°C. The cumulative temperature above 0 °C and 10 °C were 3,550 °C and 3,210 °C, respectively; the frost-free period is 150 days and total solar radiation is 6,000 MJ m−2 year−1. The local soil was classified as Aeolian sandy (FAO/UNESCO, 1988) which contained 75% sand, 22% silt and 3% clay. The topsoil physicochemical properties were: soil OM 4.5 g kg−1, total N 0.21 g kg−1, Olsen P 5.3 mg kg−1, exchangeable K 37.8 mg kg−1 and pH value (1:2.5 soil: distilled water) 7.9.
All experiments were carried out in three replicates. The sub-plots of four tree species, wild cherry (Prunus discadenia Koebne L.), cerasus humilis (Prunus dictyneura Diels L.), shiny leaf Yellow horn (Xanthoceras sorbifolium Bunge L.) and apricot (Armeniaca sibirica L.) were used in the experiments. Each species was divided into two groups: AMF group and control group. AMF group received the inoculation of AMF at 100 g inoculums/seedling, while the control group did not contain any AMF inoculums.
The four seedlings were obtained from the forest bureau of Shenmu County, Yulin City, Shaanxi Province and Northwest China. Each seedling was transplanted into the plot at temperatures ranging from about 10–15 °C in March 25, 2014 to April 5, 2014. The AMF colonization rate of wild cherry, cerasus humilis, shiny leaf Yellow horn and apricot were 4.8%, 6.3%, 5.4% and 7.2% before transplanting and 78.1%, 80.2%, 76.4% and 81.7% at the end of monitoring stage. Two months after transplantation, over 90% of the seedlings were colonized with mycorrhizal fungus.
Each plot had an area of 20 × 12 m2, which consisted of 6 rows, with 10 seedlings per row at 2 m row spacing. 100 g AMF was inoculated in root of AMF plants at seedling transplantation. Funneliformis mosseae BGCXJ01 inoculums (Supplied by Beijing Academy of Agriculture and Forestry Sciences) were propagated on Trifolium repens (clover) for 12 weeks. 100 g inoculums included spores (10–20 spores/g), colonized root fragments (40 root fragments per gram inoculums, 85% root colonization) and external mycelium65.
Rs determination
Rs was measured from July to September in individual plot with three replicates. Each measurement event was made in the middle of individual month (about 15th) using an Automated Soil Gas Flux System (Li-8100A, Li-cor Inc., Lincoln, Nebraska, USA) coupled with the small PVC chamber (20 cm × 10 cm). The instrument was permanently installed between two rows with identical distance. In each plot, measurements of Rs were taken inside three replicate PVC chambers with 20 cm in diameter and 10 cm in height, inserted to a depth of 2–3 cm in soil, capturing respirations. The CO2 efflux from this collar mainly derived from soil organic matter decomposition and root and microbial respiration. Meanwhile, soil temperature and volumetric moisture at the top of 5 cm was measured with the corresponding probes. Specifically, Rs was always measured between 8:00 and 12:00 in the morning in the sunny day without wind66.
The cumulative Rs values were calculated by the method of Bremer et al.67. Additionally, diel respiration (every 2 hours over 24-hour cycles) was periodically measured in all plots and individual collars. All of the measurements were made in four seasons from 2014 to 2015 due to time constraints. The diel measurements were used to calculate the annual growing season (July to September) cumulative Rs. Briefly, respiration measured during the daytime was assumed to be the daily maximum soil CO2 efflux. Diel measurements were used to calculate the daily minimum efflux as a percentage of maximum efflux. The daily maximum and minimum efflux were used to calculate the average daily efflux. The cumulative flux as the product of average daily flux and the number of days was estimated between each measurement.
Pn determination
Plants assimilated CO2 via Pn. To quantify photosynthetic performance of plant species, the measurement was with two AMF inoculation levels using a portable open system infrared gas analyzer for net CO2 assimilation rate of leaves in individual treatment (Li-6400, Li-cor Inc., Lincoln, Nebraska, USA). The experiment was performed from July to September in 2014, including three candidate trees within individual plot. Each measurement event was carried out at 08:00 to 18:00 in sunny day every 2 h, with three fully expanded healthy sun-exposed leaves of each species. Leaves were carefully positioned in the leaf chamber when they were exposed to the 1000 μmol m−2 s−1 saturating quantum flux level. Parameters including the instantaneous Pn, transpiration rate (Tr), stomatal conductance (Gs), intercellular CO2 concentration (Ci), photosynthetically active radiation (PAR), atmosphere CO2 concentration (Ca), atmosphere temperature (Ta), leaf temperature (Tleaf) and relative air humidity (RH) were tested.
Diurnal Pn amount was the area which surrounded by the curves of net photosynthetic rate > 0 and the time transverse in the diurnal curves variations of species Pn. According to the principle, the diurnal net hotosynthetic amount was calculated as follows:

where P was the diurnal total net assimilation amount (mmol.m−2.d−1), pi and pi+1 (μmol.m−2.s−1) was the instantaneous photosynthetic rate of the beginning and next measuring point, respectively; ti and ti+1 was the time duration of the beginning and next measuring point; n was the numbers of determination times; one hour equaled to 3600 seconds; 1 mmol equaled to 1000 μmol.
Generally, the 20% diurnal assimilation photosynthates production was consumed by dark respiration and converted the assimilation into the CO2 diurnal sequestration as follows:

where 44 was CO2 mole mass;  was CO2 sequestration mass in the per unit leaf area (g.m−2.d−1).
was CO2 sequestration mass in the per unit leaf area (g.m−2.d−1).
According to the Pn reaction equation CO2 + 4H2O → CH2O + 3H2O + O2, which the formula can be calculate the daily CO2 absorption in the per unit land area of per plant as follow:

 was the daily CO2 sequestration in the per unit land area of per plant species (g.m−2.d−1).
was the daily CO2 sequestration in the per unit land area of per plant species (g.m−2.d−1).
The average CO2 uptake per day of individual plants as follow:


where  was the daily average CO2 sequestration of each plant (g.d−1); Paverage was the daily assimilation in the per unit land area (mmol.m−2.d−1); Y was the total leaf area of each plant (m−2).
was the daily average CO2 sequestration of each plant (g.d−1); Paverage was the daily assimilation in the per unit land area (mmol.m−2.d−1); Y was the total leaf area of each plant (m−2).
Soil OC, EE-GRSP and T-GRSP fractions
Soil samples were collected from 0–20 cm of the profile using an auger (35 mm diameter) in each corresponding monitoring stage in 2014. Three cores were collected from individual plot and combined to give one composite sample per plot of each tree species. The composite soil samples were arid-dried and sieved through a 2 mm mesh and placed in plastic bags for chemical analysis.
A simple method for routine determination of soil OC by a modified Mebius procedure was described. It involved a digestion of the soil sample with an acidified dichromate (K2Cr2O7-H2SO4) solution for 30 minutes in a Pyrex digestion tube in (a) 40 tube block digester preheated to 170 °C and (b) estimation of the un-reacted dichromate by titration of the cooled digest with an acidified solution of ferrous ammonium sulfate using the N-phenylanthranilic acid as an indicator.
0.25 g composite sample was extracted with 2 ml of extractant. EE-GRSP was extracted with 20 mM citrate solution; pH 7.0 at 121 °C for 30 min. T-GRSP was extracted with 50 mM citrate solution, pH 8.0 at 121 °C. 90 min was required for one soil sample and six additional sequential extractions. For the sequential extractions, the supernatant was removed by centrifugation at 10,000 × g for 5 min, 2 ml of 50 mm citrate, pH 8.0 was added to the residue and samples were autoclaved for 60 min. Extraction of a sample continued until the supernatant showed no red-brown color typical of glomalin. Extracts from each replicate were pooled and then analyzed.
Citrate extractants were added to soil samples and disrupted by a brief (3 min) autoclave cycle. When necessary, the extractant was adjusted with HCl solution until the pH stabilized at 7.0 for 20 mM citrate or 8.0 for 50 mM citrate solution. Samples were then subjected to 121 °C for 90 min to extract T-GRSP or 30 min to extract EE-GRSP.
After extraction cycles completed, samples were centrifuged to remove soil particles (10,000 × g for 5 min) and protein in the supernatant was determined by the Bradford dye-binding assay with bovine serum albumin as standard. Concentration of glomalin was extrapolated to mg/g of soil particles by correcting the dry weight of coarse fragments >0.25 mm included in the weight of aggregates and the volume of extractant.
Data analysis
To assess the four plant species with and without AMF inoculation treatments to long-term exposure and potentially elucidate the role of AMF in previous response, the relationship of Rs and Pn, Rs and soil temperature and carbon storage and AMF inoculation of the key issues have been a focus of this study.
The experiment was at two AMF inoculation levels with four plant species. To determine main effects of AMF inoculation on soil temperature and Rs variables, mixed model restricted maximum likelihood estimation with repeated measures was used (PROC MIXED; version 8.0; SAS Institute Inc., Cary, NC, USA, 2003). Turkey’s HSD multiple comparison was used to identify the differences between two AMF inoculation levels.
An exponential model was used to calculate soil temperature sensitivity on Rs68,69.

where Rs is soil CO2 efflux (μmol m−2 s−1), T is soil temperature (°C) at 0–5 cm depth, a is the basal Rs, b is temperature sensitivity of CO2 efflux. The respiratory quotient (Q10) was calculated as Q10 = e10b.
Regression coefficients, correlations, figures and curves were obtained by the Sigma-Plot software package (version 11.0, San Jose, California, USA). Analysis was performed to reveal the differences of two AMF inoculation levels with the relevant parameters; differences in means were revealed by LSD (P < 0.05) with SAS software package. To determine the effect of AMF on photosynthetic parameters, the data set was divided into two parts; the results were insensitive to variation in species groupings as the sample size was not unduly restricted. Differences in slopes were determined by a dummy variable representing the interaction between independent variable (AMF inoculation level) and plant species in the regression analyses.
Cumulative carbon calculations
To assess the effect of soil volumetric moisture on carbon accumulation, the cumulative carbon (Cumulative-C) was fitted using a combined exponential and quadratic function as follows:

where θv is the volumetric moisture content (the minimum soil volumetric water content of our data set was 3.1% and the maximum was 37.6%, respectively) and c is the coefficient for soil moisture.
Additional Information
How to cite this article: Wang, Z.-G. et al. Arbuscular mycorrhizal fungi enhance soil carbon sequestration in the coalfields, northwest China. Sci. Rep. 6, 34336; doi: 10.1038/srep34336 (2016).
References
Hu, Z. Q., Wei, Z. Y. & Qin P. Concept and methods for soil reconstruction in mined land reclamation. Soils. 37(1), 8–12 (In Chinese) (2005).
Zhang, F. W. et al. The effect of coal-mining subsidence on water environment in the shenfu-dongsheng mining area. Acta Geoscientica Sinca. 28(6), 521–527 (In Chinese)] (2007).
McSweetney, K. & Jansen, I. J. Soil structure and associated rooting behavior in minesoils. Soil Sci Soc of Am J. 48, 607–612, doi: 10.2136/sssaj03615995004800030028x (1984).
Vitousek, P. M. et al. Human domination of Earth’s ecosystems. Science 277, 494–499, doi: 10.1126/science. 277.5325.494 (1997).
Levy, M. A. & Cumming J. R. Development of soils and communities of plants and arbuscular mycorrhizal fungi on West Virginia surface mines. Environ Manage. 54(5), 1153–1162, doi: 10.1007/s00267-014-0365-0 (2014).
Sinha, S. et al. Rhizosphere soil microbial index of tree species in a coal mining ecosystem. Soil Biol Biochem. 41, 1824–1832, doi: 10.1016/j.soilbio. 2008.11.022 (2009).
Ussiri, D. A. N., Lal, R. & Jacinthe, P. A. Soil properties and carbon sequestration of afforested pastures in reclaimed minesoils of Ohio. Soil Sci Soc of Am J. 70, 1797–1806, doi: 10.2136/2005.0352 (2006).
Pedrol, N. et al. Soil fertility and spontaneous revegetation in lignite spoil banks under different amendments. Soil Till Res. 110, 134–142, doi: 10.1016/2010. 07.005 (2010).
Friedlingstein, P. et al. Climate-carbon cycle feedback analysis: results from the C4 MIP model intercomparison. J Climate. 19, 3337–3353, doi: 10.1175/3800.1 (2006).
Bahn, M. et al. Soil respiration in European grasslands in relation to climate and assimilate supply. Ecosystems. 11, 1352–1367, doi: 10.1007/s10021-008-9198-0 (2008).
Luo, Y. Q. & Zhou, X. H. Soil respiration and the environment. Academic Press, San Diego, California, USA (2010).
Qi, Y., Xu, M. & Wu, J. G. Temperature sensitivity of soil respiration and its effects on ecosystem carbon budget: nonlinearity begets surprises. Ecol Model. 153, 131–142, doi: 10.1016/S0304-3800(01)00506-3 (2002).
Lee, X. H. et al. Rapid and transient response of soil respiration to rain. Global Change Biol. 10, 1017–1026, doi: 10.1111/j.1529-8817.2003.00787.x (2004).
Vicca, S. et al. Arbuscular mycorrhizal fungi may mitigate the influence of a joint rise of temperature and atmosphere CO2 on soil respiration in grasslands. International Journal of Ecology. doi: 10.1155/2009/209768 (2009).
Sanders, I. R. et al. Increased allocation to external hyphae of arbuscular mycorrhzial fungi under CO2 enrichment. Oecologia 117(4), 496–503, doi: 10.1007/s004420050685 (1998).
Chapin, F. S., Schulze, E. D. & Mooney, H. A. The ecology and economics of storage in plants. Annu Rev Ecol Syst. 21, 423–447, doi: http://www.jstor.org/stable/2097032 (1990).
Treseder, K. K. A meta-analysis of mycorrhizal responses to nitrogen, phosphorus and atmospheric CO2 in field studies. New Phytol. 164(2), 347–355, doi: 10.1111/j1469-8137.2004.01159.x (2004).
Diaz, S. Effects of elevated CO2 at the community level mediated by root symbionts. Plant Soil 187(2), 309–320, doi: 10.1007/BF00017096 (1996).
Wilson, G. W. et al. Soil aggregation and carbon sequestration are tightly correlated with the abundance of arbuscular mycorrhizal fungi: results from long-term field experiments. Ecol Lett. 12(5), 452–461, doi: 10.1111/j.1461-0248.2009.01303.x (2009).
Rillig, M. C. et al. Rise in carbon dioxide changes soil structure. Nature 400(6745), 628, doi: 10.1038/23168 (1999).
Heinemeyer, A. & Fitter, A. H. Impact of temperature on the arbuscular mycorrhizal (AM) symbiosis: growth responses of the host plant and its AM fungal partner. J Exp Bot. 55(396), 525–534, doi: 10.1093/jxb/erh049 (2004).
Hawkes, C. V. et al. Soil temperature affects carbon allocation within arbuscular mycorrhizal networks and carbon transport from plant to fungus. Global Change Biol. 14(5), 1181–1190, doi: 10.1111/j.1365-2486.2007.01535.x (2008).
Kaschuk, G. et al. Are the rates of photosynthesis stimulated by the catbon sink strengh of rhizobial and arbuscular mycorrhizal symbiosis? Soil Biol Biochem. 41, 1233–1244, doi: 10.1016/j.soilbio.2009.03.005 (2009).
Tian, Y. H. et al. Synergistic effect of colonization with arbuscular mycorrhizal fungi improves growth and drought tolerance of Plukenetia volubilis seedlings. Acta Physiol Plant. 35, 687–696, doi: 10.1007/s11738-012-1109-5 (2013).
Cavagnaro, T. R. et al. Growth, nutrition and soil respiration of a mycorrhiza-defective tomato mutant and its mycorrhizal wild-type progenitor. Funct Plant Biol. 35, 228–235, 10.1071/FP07281 (2008).
Moyano, F. E., Kutsch, W. L. & Schulze, E. D. Response of mycorrhizal, rhizosphere and soil basal respiration to temperature and photosynthesis in a barley field. Soil Biol Biochem. 39, 843–853, doi: 10.1016/j. soilbio. 2006. 10.001 (2007).
Verbruggen, E. et al. Arbuscular mycorrhizal fungi-short-term liability but long-term benefits for soil carbon storage? New Phytol. 197, 366–368, doi: 10. 1111/nph.12079 (2013).
Zhang, J. et al. Glomalin-related soil protein responses to elevated CO2 and nitrogen addition in a subtropical forest: Potential consequences for soil carbon accumulation. Soil Biol Biochem. 83, 142–149, doi: 10.1016/j.soilbio 2015.01.023 (2015).
Wang, X. R. et al. Effect of co-inoculation with arbuscular mycorrhzial fungi and rhizobia on soybean growth as related to root architecture and availability of N and P. Mycorrhiza 21, 173–181, doi: 10.1007/s00572-010-0319-1 (2010).
Cheng, L. et al. Arbuscular mycorrhizal fungi increase organic carbon decomposition under elevated CO2 . Science 337, 1084–1087, doi: 10.1126/science. 1224304 (2012).
van Groenigen, K. J., Osenberg, C. W. & Hungate, B. A. Increased soil emissions of potent greenhouse gases under increased atmospheric CO2 . Nature 475(7355), 214–216, doi: 10.1038/nature10176 (2011).
Reich, P. B. et al. Nitrogen limitation constrains sustainability of ecosystem response to CO2 . Nature 440(7086), 922–925, doi: 10.1038/nature 04486 (2006).
Nemec, S. & Vu J. V. C. Effect of soil phosphorus and Glomus intraradices on growth, nonstructural carbohydrates and photosynthetic activity of Citrus aurantium. Plant Soil 128, 257–263, doi: 10.1007/BF00011117 (1990).
Smith, S. E. & Read D. J. Mycorrhizal Symbiosis. 2nd ed. London: Academic Press (1997).
Douds, D. D. Jr., Pfeffeer, P. E. & Schachar-Hill, Y. Carbon partitioning, cost and metabolism of arbuscular mycorrhizas.-In Kapulnik, Y. & Douds, D. D., Jr. (ed.): Arbuscular Mycotthizas: Physiology and Function. Kluwer Academic Publ., Dordrecht; pp. 107–130 (2000).
Davies, F. T., Potter, J. R. & Linuerman, R. G. Drought resistance of mycorrhizal pepper plants independent of leaf P concentration-response in gas exchange and water relations. Physiol Plantarum 87(1), 45–53, doi: 10.1111/j.1399-3054.1993.tb08789.x (1993).
Koide, R. T. Nutrient supply, nutrient demand and plant response to mycorrhizal infection. New Phytol. 117, 365–386, doi: 10.1111/j.1469-8137.1991.tb00001.x (1991).
Wu, Q. S. et al. Arbuscular mycorrhiza mediates glomalin-related soil protein production and soil enzyme activities in the rhizosphere of trifoliate orange grown under different P levels. Mycorrhiza 25, 121–130, doi: 10.1007/s00572- 014-0594-3 (2015).
Augé, R. M. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 11, 361–365, doi: 10.1007/s005720100097 (2001).
Ruehr, N. K. et al. Drought effects on allocation of recent carbon: from beech leaves to soil CO2 efflux. New Photol. 184, 950–961, doi: 10.1111/j.1469-8137.2009.03044.x (2009).
Sönmez, F. & Gülser, F. Effects of arbuscular mycorrhizal fungus and salicylic acid on nutrient uptake by maize (Zea mays L.) seedlings in cadmium contaminated media. J. Int. Environmental Application & Science 9(5), 608–613, doi: 280620985 (2014).
Kiers, E. T. et al. Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333(6044), 880–882, doi: 10.1126/science.1208473 (2011).
Drigo, B. et al. Shifting carbon flow from roots into associated microbial communities in response to elevated atmospheric CO2 . Proc Natl Acad Sci USA 107(24), 10938–10942, doi: 10.1073/pnas. 0912421107 (2010).
Tisdall, J. M., Smith, S. E. & Rengasamy, P. Aggregation of soil by fungal hyphae. Aust J Soil Res. 35(1), 54–60, doi: 10723. 35400006225255.0050 (1997).
Sanders, I. R. et al. Increased allocation to external hyphae of arbuscular mycorrhizal fungi under CO2 enrichment. Oecologia 117(4), 496–503, doi: 10.1007/s004420050685 (1998).
Treseder, K. K. & Allen, M. F. Mycorrhizal fungi have a potential role in soil carbon storage under elevated CO2 and nitrogen deposition. New Phytol. 147(1), 189–200, doi: 10.1046/j.1469-8137.2000.00690.x (2000).
Alberton, O., Kuyper, T. W. & Gorissen, A. Taking mycocentrism seriously: mycorrhizal fungal and plant responses to elevated CO2 . New Phytol. 167(3), 859–868, doi: 10.1046/j.1469-8137.2005.01458.x (2005).
Rillig, M. C. et al. Long-term CO2 elevation affects soil structure of natural ecosystems. Nature 400, 628, doi: 10.1038/23169 (1999).
Hu, S. et al. Nitrogen limitation of microbial decomposition in a grassland under elevated CO2 . Nature 409(6817), 188–191, doi: 10.1038/35051576 (2001).
Orwin, K. H. et al. Organic nutrient uptake by mycorrhizal fungi enhances ecosystem carbon storage: a model-based assessment. Ecol Lett. 14(5), 493–502, doi: 10.1111/j. 1461-0248.2011.01611.x (2011).
Sanders, I. R. et al. Increased allocation to external hyphae of arbuscular mycorrhizal fungi under CO2 enrichment. Oecologia 117(4), 496–503, doi: 10.1007/s004420050685 (1998).
Treseder, K. K. & Allen, M. F. Mycorrhizal fungi have a potential role in soil carbon storage under elevated CO2 and nitrogen deposition. New Phytol. 147(1), 189–200, doi: 10.1046/j.1469-8137.2000.00690.x (2000).
Alberton, O., Kuyper, T. W. & Gorissen, A. Taking mycocentrism seriously: mycorrhizal fungal and plant responses to elevated CO2 . New Phytol. 167(3), 859–868, doi: 10.1046/j.1469-8137.2005.01458.x (2005).
Rillig, M. C. et al. Long-term CO2 elevation affects soil structure of natural ecosystems. Nature 400, 628, doi: 10.1038/23169 (1999).
Hu, S. et al. Nitrogen limitation of microbial decomposition in a grassland under elevated CO2 . Nature 409(6817), 188–191, doi: 10.1038/35051576 (2001).
Orwin, K. H. et al. Organic nutrient uptake by mycorrhizal fungi enhances ecosystem carbon storage: a model-based assessment. Ecol Lett. 14(5), 493–502, doi: 10.1111/j. 1461-0248.2011.01611.x (2011).
Shan, J., Morris, L. A. & Hendrick, R. L. The effects of management on soil and plant carbon sequestration in slash pine plantations. J Appl Ecol. 38(5), 932–941, doi: 0.1046/j.1365-2664.2001.00648.x (2001).
Rillig, M. C. & Mummey, D. L. Mycorrhizas and soil structure. New Phytol. 171(1), 41–53, doi: 10.1111/j. 1469-8137.2006. 01750.x (2006).
van der Heijden, M. G. et al. Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396(6706), 69–72, doi: 10.1038/23932 (1998).
Langley, J. A. & Hungate, B. A. Mycorrhizal controls on belowground litter quality. Ecology 84(9), 2302–2312, doi: 10.1890/02-0282 (2003).
Iqbal, J. et al. Fungal endophyte infection increases carbon sequestration potential of southeastern USA tall fescue stands. Soil Biol Biochem. 44(1), 81–92, doi: 10.1007/s00442-014-5 (2012).
Tang, J., Baldocchi, D. D. & Xu L. Tree photosynthesis modulates soil respiration on a diurnal time scale. Global Change Biol. 11, 1298–1304, doi: 10.1111/j.1365-2486.2005.00978.x (2005).
Baath, E. & Wallander, H. Soil and rhizosphere microorganisms have the same Q10 for respiration in a model system. Global Change Biol. 9(12), 1788–1791, doi: 10.1046/j.1365-2486.2003.00692.x (2003).
Reichstein, M. & Janssens, I. A. Semi-empirical modeling of the response of soil respiration to environmental factors in laboratory and field conditions. In W. Kutsch, M. Bahn, A. Heinemeyer (Eds), Soil Carbon Dynamics-an Integrated Methodology. Cambridge University Press, Cambridge, UK (pp. 207–220), http://hdl.handle.net/11858/00-001M-0000-000E-D8B7-6 (2009).
Plench, C. & Morel, C. External phosphorus requirement of mycorhizal and non-mycorrhizal barley and soybean plants. Biol Fert Soils. 21(4), 303–308, doi: 10.1007/BF00334907 (1996).
Heinemeyer, A. et al. Respiration of the mycelium in the arbuscular mycorrhizal symbiosis shows strong dependence on recent phytosynthesis and acclimation to temperature. New Phytol. 171(1), 159–170, doi: 10.1111/j.1469-8137.2006.01730.x (2006).
Bremer D. J. et al. Responses of soil respiration to clipping and grazing in a tallgrass prairie. J Environ Qual. 27(6), 1539–1548, doi: 10.2134/jeq1998.00472425002700060034x (1998).
Zhou, X., Wan, S. Q. & Luo, Y. Q. Source components and interannual variability of soil CO2 efflux under experimental warming and clipping in a grassland ecosystem. Global Change Biol. 13, 761–775, doi: 10.1111/j.1365-2486.2007.01333.x (2007).
Wan, S. Q. & Luo, Y. Q. Substrate regulation of soil respiration in a tallgrass prairie: results of a clipping and shading experiment. Global Biogeochem Cy. 17(2), 1–23, doi: 10.1029/2002GB001971 (2003).
Acknowledgements
The study was supported by the National Science Foundation of China (51574253) and the Fund for 863 Programs (2013AA102904) of the Ministry of the Science and Technology, PR China and the Bilateral Corporation on the Prospectus of the Open Research Project (SKLCRSM16KFA01) between Kazakhstan and PR China.
Author information
Authors and Affiliations
Contributions
Z.-G.W. initialized the study; Conceived and designed the experiments: Z.-G.W and Y.-L.B. performed the experiments: Z.-G.W., B.J., Y.Z., W.-W.L., H.L., Z.-G.W. and Y.-L.B. performed the data analysis; Z.-G.W. and Y.-L.B. drafted the manuscript; contributed reagents/materials/analysis tools: Z.-G.W., Y.-L.B., B.J., Y.Z., S.-P.P., W.-W.L. and H.L.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wang, ZG., Bi, YL., Jiang, B. et al. Arbuscular mycorrhizal fungi enhance soil carbon sequestration in the coalfields, northwest China. Sci Rep 6, 34336 (2016). https://doi.org/10.1038/srep34336
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep34336
This article is cited by
-
Hedgerows increase the diversity and modify the composition of arbuscular mycorrhizal fungi in Mediterranean agricultural landscapes
Mycorrhiza (2022)
-
Arbuscular mycorrhizal symbiosis facilitates apricot seedling (Prunus sibirica L.) growth and photosynthesis in northwest China
International Journal of Coal Science & Technology (2021)
-
Exploring economic assessment of the arbuscular mycorrhizal symbiosis
Symbiosis (2021)
-
Forage biomass and soil aggregate carbon under fodder banks with contrasting management regimes
Agroforestry Systems (2020)
-
The potential of arbuscular mycorrhizal fungi in C cycling: a review
Archives of Microbiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.