Abstract
Green CaCl2-based chemical sorbent has been widely used in sorption refrigeration, air purification and air desiccation. Methods to improve the sorption rate have been extensively investigated, but the corresponding theoretical formulations have not been reported. In this paper, a sorption system of solid-liquid coexistence is established based on the hypothesis of steady-state sorption. The combination of theoretical analysis and experimental results indicates that the system can be described by steady-state sorption process. The steady-state sorption equation, μ = (η − γT)  , was obtained in consideration of humidity, temperature and the surface area. Based on engineering applications and this equation, two methods including an increase of specific surface area and adjustment of the critical relative humidity (γ) for chemical sorbents, have been proposed to increase the sorption rate. The results indicate that the CaCl2/CNTs composite with a large specific surface area can be obtained by coating CaCl2 powder on the surface of carbon nanotubes (CNTs). The composite reached sorption equilibrium within only 4 h and the sorption capacity was improved by 75% compared with pure CaCl2 powder. Furthermore, the addition of NaCl powder to saturated CaCl2 solution could significantly lower the solution’s γ. The sorption rate was improved by 30% under the same environment.
, was obtained in consideration of humidity, temperature and the surface area. Based on engineering applications and this equation, two methods including an increase of specific surface area and adjustment of the critical relative humidity (γ) for chemical sorbents, have been proposed to increase the sorption rate. The results indicate that the CaCl2/CNTs composite with a large specific surface area can be obtained by coating CaCl2 powder on the surface of carbon nanotubes (CNTs). The composite reached sorption equilibrium within only 4 h and the sorption capacity was improved by 75% compared with pure CaCl2 powder. Furthermore, the addition of NaCl powder to saturated CaCl2 solution could significantly lower the solution’s γ. The sorption rate was improved by 30% under the same environment.
Similar content being viewed by others
Introduction
Sustainable water supply is important for social and economic development. However, fresh water that can be directly used has been exhausting rapidly due to gradual natural environmental degradation, especially the serious pollution of lakes and rivers1,2,3,4. The atmosphere stores approximately 14, 000 km3 fresh water, 10 times more than the surface fresh water on the Earth5. Obtaining sustainable fresh water from air has attracted much attention of many researchers. At present, methods of mechanical compression/condensation6,7 and adsorption8,9,10,11 are frequently-used to obtain fresh water from air. The adsorption of water vapour from atmosphere has been widely researched due to its high efficiency, low energy consumption, friendliness to the environment, low-cost and renewable characteristics. The common sorbents are physical12,13,14,15 and chemical sorbents16,17. Chemical adsorbents present large adsorption capacities and thermal energy.
Among the various chemical sorbents, green CaCl2 has been widely applied to air desiccation, thermal energy storage and gas purification, because of its large water adsorption capacity, excellent chemical and thermal stability, excellent cycle performance, low cost and absence of environmental pollution. For the other chemical sorbents, LiCl with crystal water produces HCl when drying temperature is over 98 °C; when the environment temperature rises up to 132 °C, Ca(NO3)2 decomposes quickly, producing oxygen and carcinogenic nitrite; CuSO4 and MgSO4 display a low water uptake. It means that the reusability of the CaCl2-based chemical sorbents can effectively reduce the overall cost of the sorbent for practical application in comparison with other chemical sorbents.
In the adsorption process, pure CaCl2 can generate CaCl2·0.33H2O, CaCl2·H2O, CaCl2·2H2O and many other types of stable hydrates. Its equilibrium water adsorption capacity exceeds 100 g/100 g, more than three times larger than that of the silica gel physical sorbent. A large amount of heat is released in the water adsorption process by CaCl2. The condensation heat of the water vapour is 2.443 kJ/g at room temperature; the solution heat of the CaCl2 hydrates such as CaCl2·2H2O, CaCl2·4H2O and CaCl2·6H2O are 87.2, 134 and 198 J/g, respectively18. The CaCl2/silica gel composite sorbent reported by Zhu et al.19 exhibited a thermal storage capacity of up to 1 kJ/g and a storage efficiency of 0.78. Analogous to other chemical sorbents such as LiCl and LiBr, CaCl2 easily agglomerates; a liquid phase is formed during its adsorption process. The defects lead to a low adsorption rate of the CaCl2 powder. To improve the adsorption rate, CaCl2 has typically been added to porous media such as silica gel and activated alumina so that composite sorbents with large specific surface areas can be obtained20. Aristov et al.21 reported the synthesis of a CaCl2/silica gel composite sorbent. Its water adsorption capacity exceeded 75 g/100 g and its desorption temperature was as low as 70 °C. By combining CaCl2 with the nanostructured mesoporous MCM-41, Wang et al.22 prepared a composite sorbent with 30–58 wt% CaCl2. The maximum water uptake of the sorbent was as high as 175 g/100 g. The water loss of the composite material could reach 90% at 80 °C. Because CaCl2 blocked the mass transport channels in MCM-41 for water vapour, the adsorption time for CaCl2 to reach sorption equilibrium exceeded 30 h. By impregnating CaCl2 into macroporous silica gels with an average pore size of 4.61 nm, Zhu et al.19 obtained a CaCl2/silica gel composite sorbent. The sorption time was less than 7 h. To increase the sorption rate, Spiridon et al.23 coated CaCl2 with carbon materials having huge specific surface area (680 m2/g) and porosity (0.27 cm3/g). The initial adsorption rate of the composite sorbent was 0.0275 kg/m3/s and the time was approximately 2 h to reach adsorption equilibrium.
The surface adsorption processes of solid materials have been extensively studied, producing the classic monolayer adsorption model (the Langmuir model for the adsorption isotherm)24,25,26 and the multilayer adsorption models (the Polanyi adsorption potential theory and the Brunauer–Emmett–Teller (BET) theory)27,28. Six types of adsorption isotherms have been proposed and widely used in various research areas. Classical adsorption models are based on the adsorption processes that occur on the surface of solid materials. However, chemical sorbents such as CaCl2 and LiCl can rapidly adsorb water vapour, which leads to the formation of an aqueous film on the powder surface. As a result, their sorption process originates from a saturated solution. In addition, the vapour sorption process involves the diffusion of water vapour in air, the dissolution of CaCl2 powder and the diffusion of water molecules, Cl− and Ca2+ in the liquid phase. Because of these factors, classical isotherm adsorption models are not applicable to vapour sorption by chemical sorbents.
In this paper, a sorption model of solid-liquid coexistence is established based on the definition of steady-state sorption. Both theoretical analysis and experimental results indicate that the apparent sorption rate (μ) is not dependent on sorption time, suggesting that it can be modelled using a steady-state sorption model. This result implies that the ion concentration at the surface layer remains constant. It is easily understanded that the evaporation of liquid water can be viewed as a desorption process in which the ion concentration in solution is zero at all times. The evaporation equation of distilled water is similar to that of the saturated CaCl2 solution for the sorption process. Based on the reported evaporation equation for pure water, the steady-state sorption equation,  , is herein proposed and validated. Based on the steady-state sorption equation, two methods have been proposed to increase the sorption rate, including increasing the specific surface area of chemical sorbent and adjusting the critical relative humidity (γ) of solution. Two types of composite sorbents have been prepared by using these methods: (1) it has been combined with carbon nanotubes to increase the specific surface area of the CaCl2 powder; (2) to decease the γ of CaCl2, NaCl with no sorption capability was added to the saturated CaCl2 solution.
, is herein proposed and validated. Based on the steady-state sorption equation, two methods have been proposed to increase the sorption rate, including increasing the specific surface area of chemical sorbent and adjusting the critical relative humidity (γ) of solution. Two types of composite sorbents have been prepared by using these methods: (1) it has been combined with carbon nanotubes to increase the specific surface area of the CaCl2 powder; (2) to decease the γ of CaCl2, NaCl with no sorption capability was added to the saturated CaCl2 solution.
Results and Discussions
Steady-state sorption equation
The sorption process is classified as solution sorption for CaCl2 chemical sorbent. It shares a similar sorption/desorption equation with the evaporation of liquid water. Dalton proposed the evaporation equation of liquid water in 1802 as following:

where μ is the sorption rate, g/(m2s);  , Pw represent the saturated vapor pressure and partial pressure of water vapour, respectively, Pa; and f (u) is the function of wind speed. Currently, the most widely used evaporation equation29,30 takes the following form:
, Pw represent the saturated vapor pressure and partial pressure of water vapour, respectively, Pa; and f (u) is the function of wind speed. Currently, the most widely used evaporation equation29,30 takes the following form:

where R is the gas constant, J/(molK); and T stands for environmental temperature, K. Based on thses equation, the steady-state sorption equation for saturated solution can be deduced:

where f (T) and f (S) represent the functions of temperature and surface area of the solution, respectively and Pa are the vapour pressure of the saturated solution, respectively. Because the partial pressure of water vapour (Pw) equals the product of the relative humidity (η) and the saturated vapour pressure of water vapour (PT*), the sorption equation can be transformed to

where γ, critical relative humidity, characterizes the critical condition of sorption/desorption of the solution. It is a function of the type and concentration of the solutes only.
To ensure a constant surface area of the aqueous film along with easy and accurate measurements throughout the experiment, we have employed a liquid seal for CaCl2 powder to construct a system of solid-liquid coexistence. The sorption of the resultant system of solid-liquid coexistence can be classified as a steady-state sorption process; the theoretical proof can be found in the Supporting Information. This conclusion is also corroborated by the isotherm sorption experiments. As shown in Fig. 1a, the sorption curves of the saturated solution were measured at 15, 20 and 25 °C under the η of 50%. At 20 °C, the sorption amount was 8.62, 17.5, 25.41 and 34.88 mg at a sorption time of 10, 20, 30 and 40 min, respectively. The corresponding sorption amount per unit time was 0.862, 0.875, 0.847 and 0.847 mg/min. The results indicate that the sorption rate remains constant if the testing environment remains the same. Both theoretical analysis and experimental results demonstrate that sorption in the system of solid-liquid coexistence can be classified as a steady-state sorption process. Therefore, the system of solid-liquid coexistence was modelled with the steady-state sorption equation.
The effect of temperature and humidity on the sorption rate
To investigate the effect of temperature and humidity on the sorption rate, the sorption curves of the saturated solution were measured at different humidities and temperatures, as shown in Fig. 1b. At 20 °C, the sorption rate of the saturated solution was 10.8, 20.7, 29.5 and 40.4 mg/(m2s) under η conditions of 50%, 60%, 70% and 80%, respectively, which indicates that the sorption rate is linearly correlated with the η. It can be observed from the sorption equation (Equation (4)) at constant temperature, the γ is a constant and the sorption rate is a linear function of the η. This result agrees well with the experimental data.
In particular, as shown in Fig. 1c, the γ of the CaCl2 chemical sorbent and  were linearly correlated with temperature and the fitting formula are:
were linearly correlated with temperature and the fitting formula are:


The results indicate that the sorption rate of the saturated CaCl2 solution is a quadratic function of temperature. The fitted equation of the steady-state sorption is as follows:

Aqueous surface area and the rate of isothermal sorption
To investigate the relationship between the aqueous surface area and the rate of isothermal sorption, the masking method was used to adjust the aqueous surface area. As shown in Fig. 2, the sorption rate curves of three saturated CaCl2 solutions with different surface areas were measured. Here, S1 is the cross-sectional area of the beaker (15 cm2) and S2 and S3 are the difference between the cross-sectional areas of the beaker and the mask (10.6 and 7.2 cm2, respectively). It can be observed from the inset graph that under the same η value, the sorption rate is linearly correlated with the specific surface area. According to this relationship, the area of the mask is calculated to be 10.3 and 7.4 cm2, respectively, representing a decrease of only 3.0% and 2.8% compared to the measured values. This finding further confirms that the sorption rate is directly proportional to the aqueous surface area.
Based on the above results, the steady-state sorption equation is as follows:

In particular, the sorption rate of the saturated CaCl2 solution is a quadratic function of temperature. The fitted equation of the steady-state sorption is as follows:

The application conditions of the equation are as follows: temperature (°C) T ϵ (10, 27) and η (%) ϵ (50, 80).
Application of the steady-state sorption equation
It can be observed from the steady-state sorption equation (9, 10) that when both the temperature and humidity are kept constant, the sorption rate of sorbent solution can be significantly improved by increasing the surface area and decreasing the γ of the solution. Chemical sorbents (CaCl2, LiCl and LiBr) can rapidly adsorb water vapour, which leads to the formation of an aqueous sorption film on the surface of their powder. As a result, increasing the surface area of the powder will increase that of the aqueous solution. In addition, according to hydration theory31,32, hydrated salts such as NaCl and KaNO3 can form hydrated ion clusters in solution and can thus significantly decrease the saturated vapour pressure (Pa) and γ ( ). The application of the steady-state sorption equation was investigated based on these two aspects.
). The application of the steady-state sorption equation was investigated based on these two aspects.
Increasing the specific surface area of powder
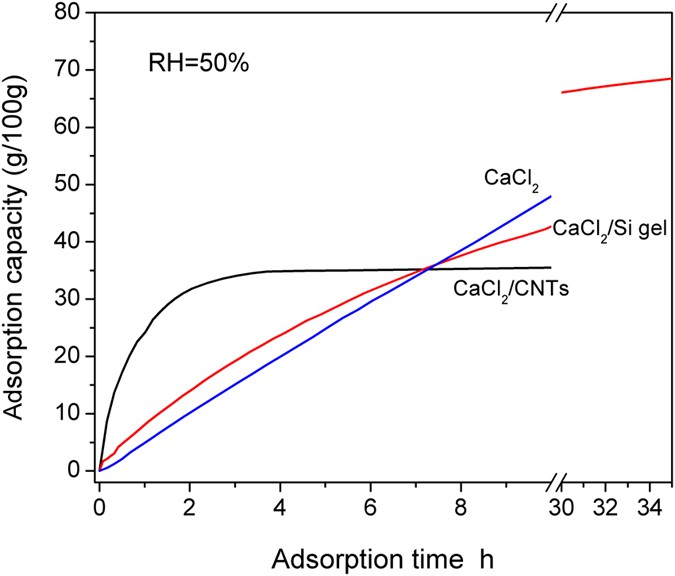
With the progression of sorption, the surface area of the chemical sorbents became progressively smaller, resulting in reduced sorption performance. To obtain CaCl2-based sorbents with high specific surface area and stable structure, two modification methods are reported in this paper: (1) CaCl2 was impregnated into silica gel to prepare an SWS-type composite sorbent (CaCl2/Si-gel); (2) CaCl2 was coated on the surface of carbon nanotubes (CNTs) to prepare a coating-modified composite sorbent (CaCl2/CNTs). Figure 3 shows the sorption curves of the CaCl2, CaCl2/Si-gel and CaCl2/CNT composite materials. The sorption amount was 10.0, 20.0 and 29.5 g/100 g, respectively, at an sorption time of 2, 4 and 6 h. For the CaCl2/Si-gel composite sorbent and the same sorption time, the corresponding sorption amount was 13.8, 23.8 and 31.4 g/100 g, which was larger than the case of pure CaCl2 by 38%, 19% and 6.4%, respectively. The results indicate that the CaCl2/Si-gel composite material enhanced sorption performance at the initial stage of sorption. However, the CaCl2 in the pores of the silica gel gradually formed a solution, blocking the mass transport channel of water vapour and resulting in a rapid decrease in the sorption rate. Because of this decrease, the time exceeded 35 h to reach sorption equilibrium. In contrast, the CaCl2/CNT sorbent can reach sorption equilibrium within 4 h, corresponding to an equilibrium sorption amount of 35.0 g/100 g. The sorption amount was 24.2, 31.8 and 34.0 g/100 g at an sorption time of 1, 2 and 3 h, respectively, accounting for 69.1%, 90.8% and 97.1% of the sorption amount at equilibrium. Therefore, the two modification methods (improving the specific surface area of CaCl2 powder) can significantly increase the sorption rate at the early stage of sorption. Moreover, CaCl2/CNTs exhibited more outstanding performance.
To analyse the microstructure and stability of the coating-modified CaCl2/CNT sorbent, the CNTs and composite were characterised by SEM, as shown in Fig. 4. The CNTs exhibited a network structure and were uniform with a diameter of 10–20 nm. For the CaCl2/CNT composite sorbent, part of the CaCl2 was adsorbed on the surface of the CNTs and part was deposited in the mesh resulting from the CNT network. Therefore, the surface area of the composite sorbent (48 m2/g) was markedly lower than that of pure CNTs (142 m2/g). When the CaCl2/CNT composite sorbent was recycled 20 times with an η of 50%, its microstructure did not experience any detectable change. The results indicate that the CaCl2/CNT composite sorbent had a very stable microstructure.
Adjusting the critical relative humidity solution
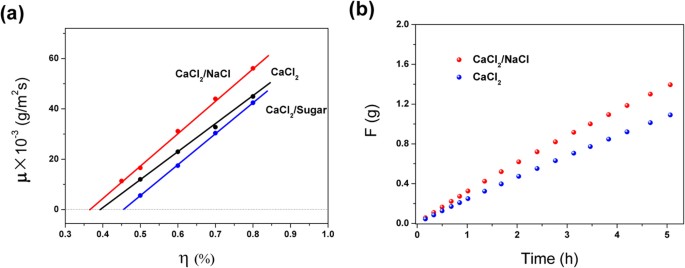
According to the steady-state sorption equation, the sorption performance can be significantly improved by decreasing the γ of the solution. The γ can be adjusted by the type and concentration of solutes. Based on this approach, the strong electrolyte NaCl (NaCl:CaCl2 solution = 1:10) and the non-electrolyte sucrose (sucrose:CaCl2 solution = 1:10) were used to adjust the sorption performance of the saturated CaCl2 solution. The testing results are shown in Fig. 5. It can be observed from Fig. 5a that when the η was 50%, the respective sorption rates of CaCl2/NaCl, CaCl2/sugar and saturated solution of CaCl2 were 16.6, 12.0 and 5.6 g/(m2s), respectively. When the η was increased to 80%, their sorption rates increased to 56.1, 44.9 and 42.4 g/(m2s), respectively. Thus, the NaCl strong electrolyte improved the sorption performance, whereas the sucrose non-electrolyte decreased the sorption performance. According to hydration theory, there exist large amounts of hydrated ion clusters, Na+(H2O)1–6, in NaCl solution33,34. The Na+(H2O)1–6 clusters can decrease the molecular kinetic energy of water molecules, thus leading to the decrease of the ability of water molecules to escape from the aqueous surface. Therefore, the strong electrolyte, NaCl, can decrease the γ of the CaCl2 solution and improve its sorption rate. To further confirm these results, 0.29 and 0.28 g of CaCl2 were placed in the CaCl2 and CaCl2/NaCl solutions, respectively. Their sorption curves were measured under the conditions of a temperature of 20 °C and η of 80%, as shown in Fig. 5b. The sorption mass by the saturated CaCl2 solution was 0.249, 0.679 and 1.09 g, respectively, at an sorption time of 1, 3 and 5 h. For the modified CaCl2 solution, at the same sorption time, the corresponding sorption amount was 0.325, 0.81 and 1.39 g, respectively, which was larger than that of the saturated CaCl2 solution by 30.6%, 29.9% and 27.9%. Therefore, the strong electrolyte, NaCl, can significantly improve the sorption rate of the saturated CaCl2 solution.
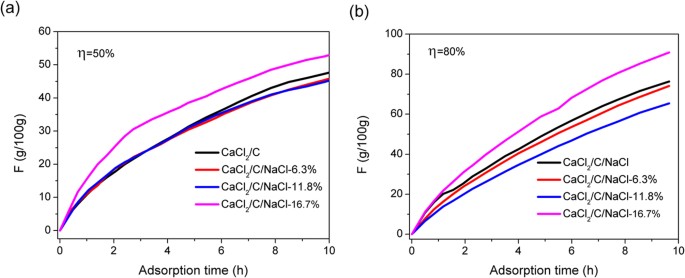
These experiments demonstrate that the sorption performance of saturated solution can be significantly improved by decreasing the γ of CaCl2 solution. To confirm that this method is effective for powder material, the CaCl2, activated carbon and NaCl with certain ratios were homogeneously mixed to prepare a composite sorbent. The sorption curve of the resultant composite sorbent is shown in Fig. 6. The sorption performance was obviously improved for the composite with an addition of 16.7 wt% NaCl. At an η of 50%, the sorption amount by CaCl2/C (CaCl2:activated carbon = 1:1) samples was 23.1, 36.4 and 45.8 g/100 g at an sorption time of 3, 6 and 9 h, respectively. The sorption amount of the CaCl2/C/NaCl-16.7% sample was 51.0 g/100 g at 9 h, an 11.5% increase compared with that of the CaCl2/C sample. When the η was increased to 80%, the sorption amount of the CaCl2/C, CaCl2/C/NaCl-6.3%, CaCl2/C/NaCl-11.8% and CaCl2/C/NaCl-16.7% samples at 9 h was 73.5, 71.0, 62.8 and 87.8 g/100 g, respectively. The results indicate that an appropriate amount of NaCl powder can significantly improve the sorption rate of composite materials.
Extension of application objects
The aforementioned experimental results indicate that the sorption performance can be significantly improved by increasing the specific surface area or decreasing the γ. To study whether the steady-state sorption equation can be applied to powder sorbents, the sorption curve of copper sulphate powder was measured, as shown in Fig. 7. The dashed line is the tangent of the sorption curve at the origin; the value of its slope,  , is the initial sorption rate of copper sulphate crystal. It can be observed from the figure that when η is equal to 50%, the testing results overlapped with the corresponding dashed line in the first 1.5 h. The results indicate that the measured sorption rate (1.7 g/100 g/h) remained constant in the first 1.5 h of the initial sorption process. Similarly, when η = 80%, the sorption rate of the copper sulphate crystals remained constant in the first 1.5 h of the initial sorption. These results suggest that the initial sorption process of copper sulphate powder can also be modelled as a steady-state sorption process. In addition, the measured sorption amount was markedly lower than the values corresponding to the dashed line after 1.5 h. This phenomenon occurs because the thickness increases for the water-containing crystals covering the surface of anhydrous copper sulphate, resulting in an increase of resistance to the mass transport of water molecules. Therefore, the corrected results for the steady-state sorption equation can be applied to the early-stage sorption process for the powder chemical sorbents. For the intermediate and final sorption stages, the effect of bulk mass transport on the sorption rate has to be taken into account.
, is the initial sorption rate of copper sulphate crystal. It can be observed from the figure that when η is equal to 50%, the testing results overlapped with the corresponding dashed line in the first 1.5 h. The results indicate that the measured sorption rate (1.7 g/100 g/h) remained constant in the first 1.5 h of the initial sorption process. Similarly, when η = 80%, the sorption rate of the copper sulphate crystals remained constant in the first 1.5 h of the initial sorption. These results suggest that the initial sorption process of copper sulphate powder can also be modelled as a steady-state sorption process. In addition, the measured sorption amount was markedly lower than the values corresponding to the dashed line after 1.5 h. This phenomenon occurs because the thickness increases for the water-containing crystals covering the surface of anhydrous copper sulphate, resulting in an increase of resistance to the mass transport of water molecules. Therefore, the corrected results for the steady-state sorption equation can be applied to the early-stage sorption process for the powder chemical sorbents. For the intermediate and final sorption stages, the effect of bulk mass transport on the sorption rate has to be taken into account.
Conclusions
If the sorption rate also remains constant, the sorption process can be considered as steady-state sorption. A simple system of solid-liquid coexistence was established based on the steady-state sorption hypothesis. Through theoretical analysis and experimental results, it has been demonstrated that the sorption process of this model behaves as a steady-state sorption process. By measuring the relationship between sorption rate and testing temperature, humidity and aqueous surface area, the steady-state sorption equation was obtained. Lastly, based on the steady-state sorption equation, two methods were proposed to increase the sorption rate, including increasing the specific surface area and adjusting the γ of materials.
The CaCl2/Si-gel and CaCl2/CNT composite materials dispaly higher specific surface area in comparion with that of the pure CaCl2 powder. For the CaCl2/Si-gel material, the CaCl2 solution formed in the sorption process blocks the pores of silica gel support, thus inducing resistance to the mass transport of water vapour. The sorption amount at 6 h was improved by only 6.4%. For the CaCl2/CNT composite, CaCl2 powder could coat the surface of the CNTs. The composite material reached sorption equilibrium within only 4 h and the sorption capacity was improved by 75% compared with pure CaCl2 powder within the same time. As a result, compared with pure CaCl2 powder, their sorption rates were dramatically improved.
The simple method, adding NaCl with no sorption capability into the saturated CaCl2 solution, could effectively decrease the γ of solution. The sorption rate of the mixture was improved by 30% under the same testing conditions. The method was verified to be applicable to powder materials.
Methods
Preparation of composites
CaCl2 powder can rapidly adsorb water vapour in air, which leads to the formation of an aqueous film on the surface of the powder. With the progression of sorption, CaCl2 powder is submerged by the solution formed on its surfaces, resulting in the change of the surface area of the aqueous film. To ensure a constant surface area of the aqueous film along with easy and accurate measurements throughout the experiment, we have employed a liquid seal for CaCl2 powder to construct a system of solid-liquid coexistence. Briefly, an appropriate amount of CaCl2 powder was submerged in a saturated solution of CaCl2; the air contained in the powder was removed by stirring the solution. A system of solid-liquid coexistence was obtained after sitting for 12 h in order to test its sorption performances. Similarly, the systems of solid-liquid coexistence of CaCl2/NaCl (NaCl:CaCl2 solution = 1:10) and CaCl2/sugar (sucrose:CaCl2 solution = 1:10) were also prepared.
Two methods have been used to prepare the CaCl2 composite materials with large specific surface area. Porous silica gels were immersed in CaCl2 solution for 24 h and the SWS-type CaCl2/Si-gel composite sorbent was obtained after being dried. In addition, CaCl2 and CNTs were mixed with mass ratio of 1:2.5 for the preparation of CaCl2/CNT composite sorbent.
Lastly, CaCl2, activated carbon and NaCl were mixed with certain ratios for the preparation of CaCl2/C/NaCl composite sorbent. The composites with 6.3%, 11.8% and 16.7% (mass percentage) NaCl were labelled as CaCl2/C/NaCl-6.3%, CaCl2/C/NaCl-11.8% and CaCl2/C/NaCl-16.7%, respectively.
Material characterization
The specific surface area of the composite material was measured through nitrogen sorption at 77 K using a Micro-meritics ASAP 2040 Analyzer in the range of the relative pressures P/P0 = 0.06–0.99 with a step of 0.015. The morphologies of the samples were observed using a field-emission scanning electron microscope (FE-SEM, Hitachi, S3400N). The sorption equilibrium between water vapour and the composite was studied in thermostat-humidistat chamber. In the thermostat-humidistat chamber, as-synthesized sorbent was put in 50 ml beakers with the cross-section of 15 cm2. The testing environment was set at the relative humidity of 40 to 80% at room temperature from 15 to 25 °C. The amount of water adsorbed by composite materials was measured by an electronic scale during the process until the mass reaches steady state.
Additional Information
How to cite this article: Zhang, H. et al. Steady-state equation of water vapor sorption for CaCl2-based chemical sorbents and its application. Sci. Rep. 6, 34115; doi: 10.1038/srep34115 (2016).
References
Holbach, A., Bi, Y., Yuan, Y., Wang, L. & Zheng, B. Environmental water body characteristics in a major tributary backwater of the unique and strongly seasonal Three Gorges Reservoir, China. Environ. Sci.: Processes & Impacts 17, 1641–1653 (2015).
Jiang, S. & Yuan, Z. Phosphorus Flow Patterns in the Chaohu Watershed from 1978 to 2012. Environ. Sci. & Technol. 49, 13973–13982 (2015).
Li, W., Gao, L., Shi, Y., Liu, J. & Cai, Y. Occurrence, distribution and risks of antibiotics in urban surface water in Beijing, China. Environ. Sci.: Processes & Impacts 17, 1611–1619 (2015).
Mattsson, K., Hansson, L. A. & Cedervall, T. Nano-plastics in the aquatic environment. Environ. Sci.: Processes & Impacts 17, 1712–1721 (2015).
Fairbanks, R. G. A 17, 000-year glacio-eustatic sea level record: influence of glacial melting rates on the Younger Dryas event and deep-ocean circulation. Nature 342, 637–642 (1989).
Khalil, A. Dehumidification of atmospheric air as a potential source of fresh water in the UAE. Desalination 93, 587–596 (1993).
Sundqvist, H. A parameterization scheme for non-convective condensation including prediction of cloud water content. Q. J. Roy. Meteor. Soc. 104, 677–690 (1978).
Zhang, H., Yuan, Y., Yang, F., Zhang, N. & Cao, X. Inorganic composite sorbent CaCl2/MWNT for water vapor sorption. RSC Advances 5, 38630–38639 (2015).
Furukawa, H., Gándara, F., Zhang, Y. B., Jiang, J. & Queen, W. L. Water sorption in porous metal-organic frameworks and related materials. J. Am. Chem. Soc. 136, 4369–4381 (2014).
Hasell, T., Schmidtmann, M., Stone, C. A., Smith, M. W. & Cooper, A. I. Reversible water uptake by a stable imine-based porous organic cage. Chem. Commun. 48, 4689–4691 (2012).
Wang, D., Zhang, J., Tian, X., Liu, D. & Sumathy, K. Progress in silica gel–water sorption refrigeration technology. Renew. Sust. Energy Rev. 30, 85–104 (2014).
Cortés, F., Chejne, F., Carrasco-Marín, F., Moreno-Castilla, C. & Pérez-Cadenas, A. Water sorption on zeolite 13X: comparison of the two methods based on mass spectrometry and thermogravimetry. Sorption 16, 141–146 (2010).
Li, X. Y., Uehara, M., Enomoto, N. & Hojo, J. Synthesis of titania-doped mesoporous silica and its gas adsorbability. J. Ceram. Soc. Jpn. 109, 818–822 (2001).
Benkoula, S. Water sorption on TiO2 surfaces probed by soft X-ray spectroscopies: bulk materials vs. isolated nanoparticles. Sci. Rep. 5, 15088 (2015).
Bi, H. Ultrahigh humidity sensitivity of graphene oxide. Sci. Rep. 3, 2714 (2013).
Gordeeva, L. G., Grekova, A. D., Krieger, T. A. & Aristov, Y. I. Sorption properties of composite materials (LiCl + LiBr)/silica. Micropor. Mesopor. Mat. 126, 262–267 (2009).
Kiplagat, J., Wang, R., Oliveira, R. & Li, T. Lithium chloride–expanded graphite composite sorbent for solar powered ice maker. Sol. Energy 84, 1587–1594 (2010).
Yuan, Y., Zhang, H., Yang, F., Zhang, N. & Cao, X. Inorganic composite sorbents for water vapor sorption: A research progress. Renew. Sust. Energy Rev. 54, 761–776 (2016).
Zhu, D., Wu, H. & Wang, S. Experimental study on composite silica gel supported CaCl2 sorbent for low grade heat storage. Int. J. Therm. Sci. 45, 804–813 (2006).
Wang, J. Y., Wang, R. Z. & Wang, L. W. Water vapor sorption performance of ACF-CaCl2 and silica gel-CaCl2 composite sorbents. Appl. Therm. Eng. 100, 893–901 (2016).
Aristov, Y. I., Restuccia, G., Cacciola, G. & Parmon, V. A family of new working materials for solid sorption air conditioning systems. Appl. Therm. Eng. 22, 191–204 (2002).
Ji, J., Wang, R. & Li, L. New composite sorbent for solar-driven fresh water production from the atmosphere. Desalination 212, 176–182 (2007).
Spiridon, M., Hauta, O. R., Secula, M. S. & Petrescu, S. Preparation and Characterization of Some Porous Composite Materials for Water Vapor Sorption. Revista de Chimie 63, 711–714 (2012).
Hoory, S. & Prausnitz, J. Monolayer sorption of gas mixtures on homogeneous and heterogeneous solids. Chem. Eng. Sci. 22, 1025–1033 (1967).
MacDonald, J. R. & Barlow, Jr, C. Work function change on monolayer sorption. J. Chem. Phys. 39, 412–422 (1963).
Olsen, S. R. & Watanabe, F. S. A method to determine a phosphorus sorption maximum of soils as measured by the Langmuir isotherm. Soil Sci. Soc. Am. J. 21, 144–149 (1957).
Dubinin, M. The potential theory of sorption of gases and vapors for sorbents with energetically nonuniform surfaces. Chem. Rev. 60, 235–241 (1960).
Walker, W. C. & Zettlemoyer, A. C. A dual-surface BET sorption theory. J. Phys. Chem. 52, 47–58 (1948).
Cai, L. Influence of forced air volume on water evaporation during sewage sludge bio-drying. Water Res. 47, 4767–4773 (2013).
Pashley, E. L., Zhang, Y., Lockwood, P. E., Rueggeberg, F. A. & Pashley, D. H. Effects of HEMA on water evaporation from water-HEMA mixtures. Dent. Mater. 14, 6–10 (1998).
Guggenheim, E. A. L. The specific thermodynamic properties of aqueous solutions of strong electrolytes. The London, Edinburgh and Dublin Philosophical Magazine and Journal of Science 19, 588–643 (1935).
Kumar, A. & Patwardhan, V. S. Prediction of vapour pressure of aqueous solutions of single and mixed electrolytes. Can. J. Chem. Eng. 64, 831–838 (1986).
Uddin, N. & Choi, C. H. Comparative Atomic Charges on Na+(H2O)n (n = 1–6) Clusters. B. Kor. Chem. Soc. 36, 827–831 (2015).
Merrill, G. N., Webb, S. P. & Bivin, D. B. Formation of Alkali Metal/Alkaline Earth Cation Water Clusters M(H2O)1−6, M = Li+, Na+, K+, Mg2+ and Ca2+: An Effective Fragment Potential (EFP) Case Study. J. Phys. Chem. A 107, 386–396 (2003).
Acknowledgements
We thank the Program of Natural Science Foundation of China (No: 51378426) and the Youth Science and Technology Innovation Team of Sichuan Province of Building Environment and Energy Efficiency (No: 2015TD0015).
Author information
Authors and Affiliations
Contributions
H.Z. conceived the idea, designed the research, performed the experiments, analyzed the data and wrote the main manuscript. Y.Y. proposed and designed the experiments. Q.S. carried out sorption profiles and performed the SEM characterization. X.C. analysed the data and modified the manuscript. L.S. supported the discussion and interpretation of results. All the authors participated in discussions of the research.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, H., Yuan, Y., Sun, Q. et al. Steady-state equation of water vapor sorption for CaCl2-based chemical sorbents and its application. Sci Rep 6, 34115 (2016). https://doi.org/10.1038/srep34115
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep34115
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.





 and γ curves.
and γ curves.