Abstract
Increasing evidence indicates that the neutrophil to lymphocyte ratio (NLR) is a useful biomarker of long-term outcomes in patients with cholangiocarcinoma. However, the prognostic role of NLR in patients with cholangiocarcinoma remains unclear. Thus, the current meta-analysis was undertaken to clarify the correlation between NLR and overall survival (OS) in cholangiocarcinoma and a comprehensive literature research was conducted to understand the association of NLR and prognosis of cholangiocarcinoma. The hazard ratio (HR) with 95% confidence interval (CI) was used to assess OS. The synthesized HR of 1.449 (95% CI: 1.296–1.619, P < 0.001) indicated that a high NLR had an unfavourable effect on OS. Overall, this meta-analysis suggested that elevated preoperative NLR is associated with poorer rates of survival in cholangiocarcinoma patients.
Similar content being viewed by others
Introduction
The incidence and mortality rates of the malignant tumour cholangiocarcinoma (CCA) are increasing worldwide1,2, with approximately 5000 CCA-related deaths occurring per year3. Although patients undergo curative-intent surgery or adjuvant therapies including systemic chemotherapy and radiotherapy for palliation of CCA, their clinical outcomes remain poor4. Several trials have shown that clinicopathological factors including tumour size5,6, intrahepatic satellite lesions6,7, lymph node metastasis8, vascular invasion6 and resection margin involvement are associated with poor survival9. However, the available data are largely derived from retrospective observational studies mostly from small, single-institution series, so they are not applicable to clinical practice and cannot be validated externally. Recently published evidence suggests that systemic inflammation is related to poor survival in patients with various types of malignancies10,11,12. The neutrophil to lymphocyte ratio (NLR) is one of the inflammatory parameters that has been reported to be of prognostic value for some solid tumours, including CCA13,14. However, because of variation in study design and limited sample sizes, studies have yielded conflicting results on the use of NLR to predict OS in patients with CCA13,15. Thus, a meta-analysis to estimate the prognostic value of NLR in these patient groups is of significance.
Results
Selection and characteristics of included studies
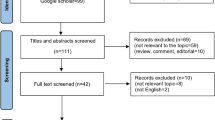
The flowchart of the study selection process is shown in Fig. 1. A total of 121 records were identified from an initial comprehensive literature research. Then, 99 of these 121 articles were included after removing duplicates. Eighty studies were excluded after their titles and abstracts were screened. After full text assessment, 2 more studies were excluded because they did not provide adequate data for calculating the HR and 95% CI and 8 studies were excluded because they were either conference abstracts or not relevant. Thus, 9 studies that met our selection criteria with a total of 2093 patients with CCA13,16,17,18,19,20,21,22,23 were finally included in this meta-analysis. Among the 9 articles included, 1 article by Hamed et al. could be divided into two “sub-groups,” given that it provided survival data related to NLR and OS categorized as ampullary carcinoma and cholangiocarcinoma depending on the anatomic location of the tumour; thus, we designated them as Hamed1 and Hamed2. Similarly, 1 article by McNamara et al. could be segmented into three “sub-groups” including three cohorts and reported the HR and 95% CI; we designated them McNamara1, McNamara2 and McNamara3. The main characteristics of the 9 articles, which included 12 studies, are summarized in Table 1. Seven studies came from Western countries, including the United Kingdom, Canada and Romania. The remaining 5 studies were from Japan, Korea and China. NLR was recorded from the pre-treatment data in all studies. HRs and 95% CIs had been obtained via multivariate analysis in 7 studies and were recorded from the original literature. The HRs for the 5 remaining studies were deduced from survival curves or other data. The scores of study quality estimated using the Newcastle Ottawa Scale (NOS) for quality assessment ranged from 5 to 7.
Meta-analysis results
No obvious between-study heterogeneity was detected (I2 = 0.0%, P = 0.47) and thus, a fixed-effects model was applied to estimate pooled HR. The combined HR of 1.449 (95% CI: 1.296–1.619, P < 0.001) suggested that patients with elevated NLR tended to have poor OS. The forest plot for this analysis is shown in Fig. 2.
Subgroup analysis was performed by therapeutic intervention (surgical and mixed) and the pooled estimates displayed that elevated pre-treatment NLRs predicted poor prognosis for patients both in Western countries (HR = 1.360, 95% CI: 1.112–1.609) and Eastern countries (HR = 1.421, 95% CI: 1.211–1.631). Further, high NLR predicted a poor prognosis in patients treated with both surgical (HR = 1.353, 95% CI: 1.099–1.607) and mixed (surgical and non-surgical) interventions (HR = 1.424, 95% CI: 1.217–1.691). On performing subgroup analyses stratified by cut-off value, we found that increased NLR was a negative predictor for patients with cut-off values ≥4 (HR = 1.724, 95% CI: 1.215–2.233) and cut-off value <4 (HR = 1.360, 95% CI: 1.191–1.529). Subgroup analysis by the NOS score of the studies showed that a high NLR indicated poorer OS in CCA patients for studies with both NOS score ≥7 (HR = 1.396, 95% CI: 1.235–1.556) and NOS score < 7 (HR = 1.311, 95% CI: 1.078–1.544). Finally, stratification by sample size showed that the combined HR was 1.394 (95% CI: 1.212–1.576) for studies with more than 200 cases and 1.402 (95% CI: 1.062–1.743) for those with less than 200 cases (Table 2).
Publication bias
Begg’s funnel plot and Egger’s test linear regression test suggested the visual assessment of overt publication bias had statistical significance for the included studies (P > |t| = 0.008; Fig. 3). Therefore, we further performed a “trim and fill” analysis and found that filling 4 unpublished studies did not significantly change the recalculated combined HRs of OS (HR = 1.402, 95% CI: 1.260–1.560; P < 0.001; Fig. 4).
Discussion
The meta-analysis conducted in the present study on 12 studies with a total of 2093 patients with CCA demonstrated that a high NLR is associated with significantly poor OS. Similar to our study, 2 recent meta-analyses confirmed the prognostic value of NLR for pancreatic cancer and non-small cell lung cancer24,25. To our knowledge, ours is the first meta-analysis assessing the prognostic role of NLR in CCA.
Inflammation plays an important role in tumour growth, including matrix degradation and cancer progression and causes immunosuppression and enhances angiogenesis26. This microenvironment potentiates and enhances the neoplastic risk and ultimately promotes metastatic spread12. Neutrophils and immunocytes related to inflammation mediate communication between the microenvironment and tumour cells. Different categories of cells play distinct roles in the systemic inflammation response. Studies have shown that neutrophils promote the survival and proliferation of cancer cells by secreting many inflammation mediators such as tumour necrosis factor, interleukin 1, interleukin 6 and vascular endothelial growth factor27,28,29. However, lymphopenia is vital in the immune defence against tumour cells30. The infiltration of CD4+ T cells triggers the immune activation of CD8+ T cells31 and activated CD8+ T cells cause apoptosis of cancer cells by releasing cytotoxic factors32. These findings collectively indicate that it is reasonable to assume that neutrophilia and lymphocytopenia are a potential indicator of prognosis for estimating the systemic inflammatory response and outcome of individual patients. As an indicator of the balance between tumour destruction and tumour protection, NLR is a significant prognostic factor.
Although we comprehensively evaluated the association between the NLR and CCA, this meta-analysis has some limitations. First, a publication bias obviously exists since small-scale studies are prone to remain unpublished and could account for language limitations in the inclusion criterion or selective publication. Second, a selection bias is impossible to avoid because most of the studies included in this meta-analysis were retrospective. Third, the included articles use different NLR cutoff levels and determine these levels using various methods. Therefore, the threshold value of NLR should be standardised in future trials and in clinical practice. Fourth, the data were not adequate for us to examine the relationship between NLR and the clinicopathological parameters of the tumour. Finally, the HRs and Cis had to be deduced from survival curves in 6 studies because these studies did not report these parameters directly.
In conclusion, our study demonstrated the importance of NLR as a predictor of OS in patients with CCA. The NLR is easily determine from the widely available findings of routine blood tests, so it can be extensively used as a novel predictive factor for cholangiocarcinoma. Further large-scale research and standardised investigations are warranted to confirm our findings.
Methods
Literature research
We performed a search of articles published in PubMed and EMBASE up to March 2, 2016. The relevant studies were identified using the following search terms:
-
1
“neutrophil to lymphocyte ratio,” “neutrophil lymphocyte ratio,” “neutrophil-to-lymphocyte ratio”;
-
2
Bile duct OR cholangio* OR biliary tract OR Klatskin OR “ampulla of Vater”;
-
3
Cancer OR adenocarcinoma OR carcinoma;
-
4
#1 AND #2 AND #3.
Study selection criteria
The entire selection process was performed independently by two authors (T.D.W. and F.Y.) and a third author (S.Q.) was consulted to resolve any discrepancies. We included studies that met the following selection criteria: (1) investigation of the prognostic value of NLR in CCA; (2) data available for calculating survival estimates, such as HR with 95% CIs, or P values and other data that could be used to calculate these values33; (3) and availability of full text. Abstracts, meetings or case reports were excluded.
Data extraction and quality assessment
Data extraction and quality assessment were conducted independently by two authors (T.D.W. and G.M.J.). Any disagreement was resolved by discussion and consensus. The investigators extracted the following data from the 12 studies: names of the first authors, publication year, sample sizes, participant characteristics and endpoints with their corresponding HRs and 95% CIs. The NOS was used to evaluate study quality.
Data synthesis and analysis
HRs and 95% CIs from each study were used to calculate pooled HRs. Cochran’s Q test and Higgins’ I-squared statistics were used to test the heterogeneity of the combined HRs. If heterogeneity was observed, the random effects model (Der Simonian and Laird method) was applied for analysis; otherwise, the HRs were pooled using a fixed-effects model. We tried to contact the authors of published studies and failed to do so for subgroup analysis and subgroup analysis and meta-regression analyses were performed to detect and explain the heterogeneity among the results of various studies. Sensitivity analyses were performed to confirm the robustness of the study. Egger’s linear regression test and Begg’s funnel plot test were used to evaluate publication bias34. The trim and fill method was applied to estimate asymmetry in the funnel plot35. Statistical significance was set at 0.05. All statistical analyses were performed using STATA version 12.0 (StataCorp, College Station, TX, USA).
Additional Information
How to cite this article: Tan, D.-W. et al. Prognostic Significance of Neutrophil to Lymphocyte Ratio in Oncologic Outcomes of Cholangiocarcinoma: a Meta-analysis. Sci. Rep. 6, 33789; doi: 10.1038/srep33789 (2016).
References
Shaib, Y. & El-Serag, H. B. The epidemiology of cholangiocarcinoma. Semin Liver Dis 24, 115–125, doi: 10.1055/s-2004-828889 (2004).
Patel, T. Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States. Hepatology 33, 1353–1357, doi: 10.1053/jhep.2001.25087 (2001).
Lazaridis, K. N. & Gores, G. J. Cholangiocarcinoma. Gastroenterology 128, 1655–1667 (2005).
Khan, S. A. et al. Guidelines for the diagnosis and treatment of cholangiocarcinoma: an update. Gut 61, 1657–1669, doi: 10.1136/gutjnl-2011-301748 (2012).
Roayaie, S. et al. Aggressive surgical treatment of intrahepatic cholangiocarcinoma: predictors of outcomes. J Am Coll Surg 187, 365–372 (1998).
Weber, S. M. et al. Intrahepatic cholangiocarcinoma: resectability, recurrence pattern and outcomes. J Am Coll Surg 193, 384–391 (2001).
Ohtsuka, M. et al. Extended hepatic resection and outcomes in intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Surg 10, 259–264, doi: 10.1007/s00534-002-0724-8 (2003).
Uenishi, T. et al. Clinicopathological factors predicting outcome after resection of mass-forming intrahepatic cholangiocarcinoma. Br J Surg 88, 969–974, doi: 10.1046/j.0007-1323.2001.01784.x (2001).
Okuno, M. et al. Evaluation of inflammation-based prognostic scores in patients undergoing hepatobiliary resection for perihilar cholangiocarcinoma. J Gastroenterol 51, 153–161, doi: 10.1007/s00535-015-1103-y (2016).
Keizman, D. et al. Active smoking may negatively affect response rate, progression-free survival and overall survival of patients with metastatic renal cell carcinoma treated with sunitinib. Oncologist 19, 51–60, doi: 10.1634/theoncologist.2012-0335 (2014).
Guthrie, G. J., Roxburgh, C. S., Farhan-Alanie, O. M., Horgan, P. G. & McMillan, D. C. Comparison of the prognostic value of longitudinal measurements of systemic inflammation in patients undergoing curative resection of colorectal cancer. Br J Cancer 109, 24–28, doi: 10.1038/bjc.2013.330 (2013).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867, doi: 10.1038/nature01322 (2002).
Lee, B. S. et al. Neutrophil-lymphocyte ratio predicts survival in patients with advanced cholangiocarcinoma on chemotherapy. Cancer Immunol Immunother 65, 141–150, doi: 10.1007/s00262-015-1780-7 (2016).
Templeton, A. J. et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst 106, dju124, doi: 10.1093/jnci/dju124 (2014).
Hakeem, A. R. et al. Does the extent of lymphadenectomy, number of lymph nodes, positive lymph node ratio and neutrophil-lymphocyte ratio impact surgical outcome of perihilar cholangiocarcinoma? Eur J Gastroenterol Hepatol 26, 1047–1054, doi: 10.1097/meg.0000000000000162 (2014).
Okuno, M. et al. Evaluation of inflammation-based prognostic scores in patients undergoing hepatobiliary resection for perihilar cholangiocarcinoma. Journal of Gastroenterology 51, 153–161 (2016).
Haruki, K. et al. Neutrophil to lymphocyte ratio predicts therapeutic outcome after pancreaticoduodenectomy for carcinoma of the ampulla of vater. HPB 17, 94 (2015).
Chen, Q. et al. The elevated preoperative neutrophil-to-lymphocyte ratio predicts poor prognosis in intrahepatic cholangiocarcinoma patients undergoing hepatectomy. Tumor Biology 36, 5283–5289 (2015).
McNamara, M. G. et al. Neutrophil/lymphocyte ratio as a prognostic factor in biliary tract cancer. European Journal of Cancer 50, 1581–1589 (2014).
Iwaku, A. et al. The Glasgow Prognostic Score accurately predicts survival in patients with biliary tract cancer not indicated for surgical resection. Medical Oncology 31 (2014).
Hamed, M. O., Roberts, K. J., Smith, A. M. & Stiff, G. M. Elevated pre-operative neutrophil to lymphocyte ratio predicts disease free survival following pancreatic resection for periampullary carcinomas. Pancreatology 13, 534–538 (2013).
Dumitrascu, T., Chirita, D., Ionescu, M. & Popescu, I. Resection for Hilar Cholangiocarcinoma: Analysis of Prognostic Factors and the Impact of Systemic Inflammation on Long-term Outcome. Journal of Gastrointestinal Surgery 17, 913–924 (2013).
Gomez, D., Morris-Stiff, G., Toogood, G. J., Lodge, J. P. A. & Prasad, K. R. Impact of systemic inflammation on outcome following resection for intrahepatic cholangiocarcinoma. Journal of Surgical Oncology 97, 513–518 (2008).
Gu, X. B., Tian, T., Tian, X. J. & Zhang, X. J. Prognostic significance of neutrophil-to-lymphocyte ratio in non-small cell lung cancer: a meta-analysis. Sci Rep 5, 12493, doi: 10.1038/srep12493 (2015).
Cheng, H. et al. Prognostic role of the neutrophil-to-lymphocyte ratio in pancreatic cancer: a meta-analysis. Sci Rep 5, 11026, doi: 10.1038/srep11026 (2015).
Sparmann, A. & Bar-Sagi, D. Ras-induced interleukin-8 expression plays a critical role in tumor growth and angiogenesis. Cancer Cell 6, 447–458, doi: 10.1016/j.ccr.2004.09.028 (2004).
Schreiber, R. D., Old, L. J. & Smyth, M. J. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331, 1565–1570, doi: 10.1126/science.1203486 (2011).
Jablonska, J., Leschner, S., Westphal, K., Lienenklaus, S. & Weiss, S. Neutrophils responsive to endogenous IFN-beta regulate tumor angiogenesis and growth in a mouse tumor model. J Clin Invest 120, 1151–1164, doi: 10.1172/jci37223 (2010).
Hofman, P. M. Pathobiology of the neutrophil-intestinal epithelial cell interaction: role in carcinogenesis. World J Gastroenterol 16, 5790–5800 (2010).
Dunn, G. P., Old, L. J. & Schreiber, R. D. The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21, 137–148, doi: 10.1016/j.immuni.2004.07.017 (2004).
Rosenberg, S. A. Progress in human tumour immunology and immunotherapy. Nature 411, 380–384, doi: 10.1038/35077246 (2001).
Zikos, T. A., Donnenberg, A. D., Landreneau, R. J., Luketich, J. D. & Donnenberg, V. S. Lung T-cell subset composition at the time of surgical resection is a prognostic indicator in non-small cell lung cancer. Cancer Immunol Immunother 60, 819–827, doi: 10.1007/s00262-011-0996-4 (2011).
Parmar, M. K., Torri, V. & Stewart, L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 17, 2815–2834 (1998).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Duval, S. & Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56, 455–463 (2000).
Author information
Authors and Affiliations
Contributions
Conceived and designed the analyses: H.-L.W. and D.-W.T. Reference collection and data management: D.-W.T., Y.F. and M.-J.G. Performed the analyses: D.-W.T., Y.F., S.-Q.W. and P.K. Wrote and reviewed the manuscript: D.-W.T., Y.F., H.-L.W., Q.S., M.-J.G., P.K. and S.-Q.W. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Tan, DW., Fu, Y., Su, Q. et al. Prognostic Significance of Neutrophil to Lymphocyte Ratio in Oncologic Outcomes of Cholangiocarcinoma: A Meta-analysis. Sci Rep 6, 33789 (2016). https://doi.org/10.1038/srep33789
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep33789
This article is cited by
-
Neutrophils and polymorphonuclear myeloid-derived suppressor cells: an emerging battleground in cancer therapy
Oncogenesis (2022)
-
The prognostic value of neutrophil-to-lymphocyte ratio in cholangiocarcinoma: a systematic review and meta-analysis
Scientific Reports (2022)
-
High Neutrophil–Lymphocyte Ratio and Delta Neutrophil–Lymphocyte Ratio Are Associated with Increased Mortality in Patients with Hepatocellular Cancer
Digestive Diseases and Sciences (2022)
-
The Limitations of Standard Clinicopathologic Features to Accurately Risk-Stratify Prognosis after Resection of Intrahepatic Cholangiocarcinoma
Journal of Gastrointestinal Surgery (2018)
-
Predictive value of preoperative peripheral blood neutrophil/lymphocyte ratio for lymph node metastasis in patients of resectable pancreatic neuroendocrine tumors: a nomogram-based study
World Journal of Surgical Oncology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.