Abstract
The integrated stress response (ISR) is a cytoprotective pathway initiated upon phosphorylation of the eukaryotic translation initiation factor 2 (eIF2α) residue designated serine-51, which is critical for translational control in response to various stress conditions. Four eIF2α kinases, namely heme-regulated inhibitor (HRI), protein kinase R (PKR), PKR-like endoplasmic reticulum kinase, (PERK) and general control non-depressible 2 (GCN2), have been identified thus far, and they are known to be activated by heme depletion, viral infection, endoplasmic reticulum stress, and amino acid starvation, respectively. Because eIF2α is phosphorylated under various stress conditions, the existence of an additional eIF2α kinase has been suggested. To validate the existence of the unidentified eIF2α kinase, we constructed an eIF2α kinase quadruple knockout cells (4KO cells) in which the four known eIF2α kinase genes were deleted using the CRISPR/Cas9-mediated genome editing. Phosphorylation of eIF2α was completely abolished in the 4KO cells by various stress stimulations. Our data suggests that the four known eIF2α kinases are sufficient for ISR and that there are no additional eIF2α kinases in vertebrates.
Similar content being viewed by others
Introduction
The α subunit of eukaryotic translation initiation factor 2 (eIF2α) is known to be phosphorylated by diverse stress stimuli1. There are several phosphorylation sites in eIF2α, but serine 51 is critical for translational control2. eIF2α phosphorylation causes transient attenuation of the translation of most mRNAs and transcriptional induction by selective translation of transcription factors. These signalling programs allow cells to adapt to various stress conditions, referred to as the integrated stress response (ISR)3,4. Recent studies have shown that ISR plays physiological roles in the regulation of intermediary metabolism5,6, memory7,8, tumorigenesis9, and immunity10. Therefore, it is important to understand the mechanism in which the physiological or pathological stimuli are coupled to eIF2α phosphorylation.
Four distinct eIF2α kinases have been identified in vertebrates. Each kinase is activated by stimulatory factors that bind to its regulatory domains, which promote the active state dimer configuration of its catalytic kinase domains. For example, heme-regulated inhibitor (HRI/EIF2AK1) is activated during heme deficiency by the release of heme from its kinase insert domains11,12. Protein kinase R (PKR/EIF2AK2) is activated during a viral infection by the binding of double-stranded RNA (dsRNA) to its two conserved dsRNA binding domains13,14. PKR-like endoplasmic reticulum (ER) kinase (PERK/EIF2AK3) is activated during ER stress by the release of binding immunoglobulin protein from its ER luminal domains15,16. General control non-depressible 2 (GCN2/EIF2AK4) is activated under amino acid deprivation by the binding of uncharged tRNA to the regulatory domains17,18. Although much is known about the structural and functional features of these four eIF2α kinases, there are key questions that remain to be elucidated, such as what eIF2α kinases are responsible for certain stress stimuli and are there additional eIF2α kinases that have not been identified. Therefore, the primary aim of the present study was to validate the existence of additional eIF2α kinase in vertebrates.
We observed 14 types of diverse stress stimuli that induced eIF2α phosphorylation. Surprisingly, this eIF2α phosphorylation was completely abolished in quadruple knockout cells (4KO cells) for all four known eIF2α kinases, suggesting that the four known eIF2α kinases are sufficient for ISR and that there are no additional eIF2α kinases in vertebrates.
Results
Possibility of additional eIF2α kinases among eukaryotes
The completion of genomic sequencing has permitted classifications and comparisons with model organism kinomes, which has identified putative protein kinase genes. Kinases were classified into 10 groups primarily by sequence comparison of their catalytic domains, aided by knowledge of the biological functions19,20. The four known eIF2α kinases, namely as HRI, PKR, PERK, and GCN2, are categorized as eukaryotic protein kinases (ePKs), but they do not fit into any of the 10 ePK groups. Therefore, these eIF2α kinases are categorized into the other group and no criterion for classifying the eIF2α kinases has been established. The classification has grown as new subfamilies have been added for kinases that are conserved among multiple organisms. To gain insights into additional eIF2α kinases, phylogenetic comparison of eIF2α kinomes among plants, yeast, parasites, worms, flies, fish, mice, and humans was conducted (Fig. 1). Kinases were first clustered as a family by sequence similarity in the kinase domain and additional information from subcellular localizations and known functions.
Phylogenetic analysis of the eukaryotic translation initiation factor 2 (eIF2α) kinomes.
The amino acid sequences of kinases that are homologous to eIF2α kinases were aligned using CLUSTAL W 2.1 and the unrooted phylogenetic tree was created via the neighbour-joining method using MEGA 5.05. The scale bar represents 0.2 substitutions per site. The species abbreviations used in the Figure were as follows: Ha Hammondia hammondi (Hh), Toxoplasma gondii (Tg), Arabidopsis thaliana (At), Schizosaccharomyces pombe (Sp), Caenorhabditis elegans (Ce), Drosophila melanogaster, Danio rerio (Dr) and Mus musculus (Mm).
GCN2 is conserved throughout eukaryotes from plants such as Arabidopsis thaliana to animals. HRI is conserved in fungi, such as Schizosaccharomyces pombe, and many vertebrates from Danio rerio to Homo sapiens, although some exceptions have been observed, such as Drosophila melanogaster and Caenorhabditis elegans. However, it is not found in plants or protists, such as Hammondia hammondi and Toxoplasma gondii. PKR is only conserved in vertebrates, whereas it is not found in plants, fungi, protists, and invertebrates. PERK is conserved in protists, invertebrates, and vertebrates but not in plants and fungi. Interestingly, newly identified protist eIF2α kinases21,22,23 formed a fifth eIF2α kinase cluster independent of the other four kinases. The kinase classification remains imperfect and incomplete. Indeed, several new subfamilies have been added for kinases that are conserved among multiple phyla. Therefore, we hypothesized that an additional kinase may exist in vertebrates.
Various stress conditions cause ISR
The eIF2α kinases are known to be activated primarily by specific stressors. Hence, to validate the existence of additional eIF2α kinases, it is important to know what types of stressors can activate ISR. Mouse embryonic fibroblasts (MEFs) treated with 12 distinct stressors for 1 h were concomitantly analysed for the phosphorylation of eIF2α (Fig. 2A). We found that all 12 distinct stressors phosphorylated eIF2α (Fig. 2A). The degree of eIF2α phosphorylation was quantified by the ratio of phosphorylated/non-phosphorylated eIF2α. NaCl, ultraviolet light (UV), and thapsigargin (Tg) caused strong eIF2α phosphorylation; cold shock (Cold), H2O2, heat shock (Heat), low glucose (LG), arsenate (Ars), and histidinol (His) caused moderate phosphorylation; and polyinosinic-polycytidylic acid (pIC), anoxia (Ano), and serum starvation (SS) caused mild phosphorylation (Fig. 2A).
Eukaryotic translation initiation factor 2 (eIF2α) is phosphorylated by various stresses in mouse embryonic fibroblasts (MEFs).
(A) Representative immunoblots of phosphorylated eIF2α, total eIF2α and ribophorin (Rb) 1 h after treatment with the indicated stress stimulus in the wild-type MEFs. The ratio of autophosphorylated versus total eIF2α is indicated. (B) Representative immunoblots of phosphorylated PKR, PERK, GCN2 and Rb 1 h after treatment with the indicated stress stimulus in MEFs. The stress condition abbreviations used in the Figure were as follows: untreated (UT), 400 μM arsenate (Ars), 10 ng/μl polyinosinic-polycytidylic acid (pIC), 200 nM thapsigargin (Tg), 2 mM histidinol (His), 1 mM H2O2 (H2O2), 500 mM NaCl (NaCl), 254 nm 200 J/m2 ultraviolet irradiation (UV), 42 °C heat shock (heat), cold shock (cold), low glucose (LG), serum starvation (SS) and anoxia (Ano).
The expression of three eIF2α kinases (PKR, PERK, and GCN2) (Fig. 2B) was assessed by immunoblot analysis. Because none of the two antibodies against HRI that we purchased could recognize mouse HRI under our conditions, we could not evaluate the phosphorylation of HRI. The activation of PKR by an arsenic compound in mammalian cells was described in an earlier paper24, but this activation was refuted by other groups using PKR knockout cells, which reported the activation of HRI by an arsenic compound25,26. In contrast, a report was published stating that an arsenic compound activated HRI and GCN2 in yeast27. When we treated MEFs with 400 μM Ars, the phosphorylation of eIF2α and GCN2 was observed, but the phosphorylation of PKR or PERK was not detected by a phosphorylation-dependent mobility shift assay on standard or phos-tag SDS-PAGE gel (Fig. 2A,B). Thus, it was suggested that Ars activated HRI and GCN2 in accordance with previous reports. A synthetic analog of dsRNA, polyinosinic-polycytidylic acid (polyIC), mimics RNA viral infection, which is known to activate PKR28. Tg is an inhibitor of the sarcoplasmic/endoplasmic reticulum Ca2+ ATPase, which is known to induce ER stress and PERK activation15. The histidine analog histidinol (His) inhibits histidyl-tRNA synthetase activity, which is known to increase uncharged tRNA levels and activate GCN229. In agreement with previous reports, MEFs treated with 10 ng/μl polyIC, 200 nM Tg, or 2 mM histidinol displayed increased phosphorylation of eIF2α and activation of PKR, PERK, and GCN2, respectively (Fig. 2A,B).
We examined the activation of eIF2α kinases by H2O2, UV, heat shock, cold shock, low glucose, serum starvation, and anoxia, for which it was not obvious which eIF2α kinase was activated. We found that 1 mM H2O2 induced phosphorylation of eIF2α and GCN2, but phosphorylation of PKR or PERK was not observed (Fig. 2A,B). As H2O2 has been reported to activate HRI in S. pombe27, H2O2 can possibly activate two eIF2α kinases, GCN2 and HRI. Under hyperosmotic stress induced by NaCl, phosphorylation of eIF2α via HRI has been reported but not the involvement of any other kinases25. We observed strong phosphorylation of eIF2α by 500 mM NaCl, in line with previous reports, and activation of PKR, PERK and GCN2 (Fig. 2A,B and Supplemental Fig. 1). UV irradiation has been reported to activate PERK30 and GCN231. We found that exposure to UV (254 nM) induced strong phosphorylation of eIF2α and activated GCN2 as previously reported but not PERK nor PKR (Fig. 2A,B). It has been reported that heat shock induces eIF2α via HRI but not any other kinases25. We observed weak phosphorylation of eIF2α in response to heat shock at 42 °C, which did not induce the activation of PKR, but weak activation of PERK and GCN2 was noted (Fig. 2A,B). Upon cold shock on ice, we observed phosphorylation of eIF2α, activation of PERK, as previously reported32, and weak activation of GCN2 and PKR. Low glucose (1 g/l glucose) induced phosphorylation of eIF2α, but it did not induce PERK activation, which was previously reported33,34 (Fig. 2A,B). Low glucose did not activate PKR, but it was found to activate GCN2 (Fig. 2A,B). Serum starvation induced weak phosphorylation of eIF2α, GCN2 activation, as previously reported35,36, and weak activation of PERK, but it did not induce PKR activation (Fig. 2A,B). Anoxia induced weak phosphorylation of eIF2α and activation of PERK and GCN2, as previously reported37,38, but it did not induce PKR activation (Fig. 2A,B). From the results, it was verified that among various stress conditions that induce eIF2α phosphorylation, there are stresses that specifically activate one of the known eIF2α kinases and those that activate two or more kinases. However, our experiments were conducted at a specific concentration of stress agent, a specific time point, and the analysis was in a specific cell type: MEF cells. Therefore, there are many variables that can account for the differences between the results presented in this paper and in other published reports.
Establishment of a 4KO cell line lacking the four known kinases
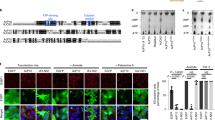
The recently developed genome engineering strategy using CRISPR/Cas9 systems39,40 enables two or more genes to be destroyed simultaneously, which has been a difficult, time-consuming, and laborious task. We established an MEF line that lacks all four eIF2α kinases using CRISPR/Cas9 technology. Upon DNA sequencing, we confirmed that the four gRNAs for the eIF2α kinases efficiently induced indel mutations in the respective genes in the 4KO cells (Fig. 3A and Supplemental Fig. 2). Via immunoblotting, we ascertained that PKR, PERK, and GCN2 were not expressed in the 4KO cells at the protein level (Fig. 3B). Since there was no antibody available that could detect endogenous HRI, we confirmed by reverse transcription-quantitative polymerase chain reaction (RT-qPCR) that the mRNA expression of the gene was remarkably low (Fig. 3C).
Generation of quadruple eukaryotic translation initiation factor 2 (eIF2α) kinase knockout mouse embryonic fibroblasts (MEFs) (4KO) by CRISPR/Cas9-mediated genome editing.
(A) Schematic of each targeting locus of the four Eif2ak kinases with the target sequence underscored, PAM in bold type and exons in block boxes. Representative sequencing results showing the indel mutations generated in 4KO cells. (B) Representative immunoblots of protein kinase R (PKR), PKR-like ER kinase (PERK), general control non-depressible 2 (GCN2) and ribophorin (Rb) in the wild-type and 4KO MEFs. (C) RT-qPCR analysis (mean ± SD, n = 4, *p < 0.01) of the expression of heme-regulated inhibitor (Hri) mRNA in the wild-type and 4KO MEFs.
There are no additional eIF2α kinases other than the known four kinases
As the phylogenetic tree suggests, phosphorylation of eIF2α should be observed in the established 4KO cells upon exposure to some type of stress condition if there is any additional eIF2α kinase other than the known four kinases. We applied twelve different stress stimuli that are known to induce phosphorylation of eIF2α to the established 4KO cells and analysed eIF2α phosphorylation. Against our hypothesis, phosphorylation of eIF2α completely disappeared for all twelve different stress stimuli (Fig. 4A). To verify that the disappearance of eIF2α phosphorylation was not attributable to genome engineering due to an off-target effect of the CRISPR/Cas9 systems, a ligand-activatable PERK kinase called Fv2E-PERK was overexpressed in the 4KO cells. Fv2E-PERK consists of a cytosolic PERK kinase domain and an artificial dimerization domain (Fv2E) (Fig. 4B). The application of the specific dimerizer AP20187 (AP) can activate its PERK kinase domain. AP-dependent phosphorylation of eIF2α was observed, denying the possibility that genome engineering was the cause of the disappearance of eIF2α phosphorylation in the 4KO cells (Fig. 4C). From these results, it can be deduced that there are no additional eIF2α kinases other than HRI, PKR, PERK, and GCN2 and that these four eIF2α kinases are entirely responsible for ISRs against various stress conditions.
Eukaryotic translation initiation factor 2 (eIF2α) phosphorylation was completely abolished in quadruple eIF2α kinase knockout mouse embryonic fibroblasts (4KO MEFs) under the various stress conditions.
(A) Representative immunoblots of phosphorylated eIF2α, total eIF2α and ribophorin (Rb) 1 h after treatment with the indicated stress stimulus in the wild-type and 4KO MEFs. The ratio of autophosphorylated versus total eIF2α is indicated. The stress conditions abbreviations used in the Figure are as follows: untreated (UT), 400 μM arsenate (Ars), 10 ng/μl polyIC (pIC), 200 nM thapsigargin (Tg), 2 mM histidinol (His), 1 mM H2O2 (H2O2), 500 mM NaCl (NaCl), 254 nm 200 J/m2 ultraviolet irradiation (UV), 42 °C heat shock (heat), cold shock (cold), low glucose (LG), serum starvation (SS) and anoxia (Ano). (B) Schematic representation of the structure and activation of the Fv2E-PERK fusion protein. AP20187(AP)-induced Fv2E-PERK homodimerization causes the activation of Fv2E-PERK, subsequently phosphorylating eIF2α. (C) Representative immunoblots of phosphorylated eIF2α, total eIF2α and Fv2E-PERK in the wild-type and 4KO MEFs overexpressing Fv2E-PERK after exposure to Mock (Ctrl) or 1 nM AP for 1 h.
The 4KO cells represent an effective tool for identifying the kinase responsible for ISR activation
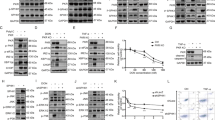
Cells are likely to acquire stress tolerance against various stress stimuli via the four eIF2α kinases responding in an ingenious overlapping manner. There is a possibility that a stress stimulus activates two or more eIF2α kinases. However, the magnitude of the activation of the primary kinase overshadows that of the secondary kinase, making the latter kinase difficult to detect. To overcome this problem, we established eIF2α kinase rescue 4KO cell lines in which a single eIF2α kinase was rescued. All eIF2α kinases that respond to a stress stimulus can be identified by applying the stimulus to the cell lines and observing the phosphorylation of eIF2α. In the 4KO cell lines, an eIF2α kinase carrying a FLAG tag was re-introduced using a retrovirus. By immunoblotting using FLAG antibody, we confirmed that the expression level of the eIF2α kinase in the four eIF2α kinase rescue 4KO cell lines was mutually similar (Fig. 5A). Via immunoblotting or RT-qPCR, we confirmed that the eIF2α kinases carrying the FLAG tag were overexpressed in the 4KO cells compared with the wild-type endogenous eIF2α kinases (Fig. 5B,C).
Quadruple eukaryotic translation initiation factor 2 (eIF2α) kinase knockout mouse embryonic fibroblasts (4KO MEFs) overexpressing the indicated Flag-tagged eIF2α kinases are useful tools for validating the activation of each eIF2α kinase by various stressors.
(A) Representative immunoblots showing ectopically expression of the indicated Flag-tagged eIF2α kinases and endogenous ribophorin (Rb) in the six types of MEFs (wild-type, 4KO and 4KO overexpressing the indicated Flag-tagged eIF2α kinases). (B) Representative immunoblots of protein kinase R (PKR), PKR-like ER kinase (PERK), general control non-depressible 2 (GCN2) and Rb in the six types of MEFs (wild-type, 4KO and 4KO overexpressing the indicated Flag-tagged eIF2α kinases). (C) RT-qPCR analysis (mean ± SD, n = 4, *p < 0.01) of the mRNA expression of heme-regulated inhibitor (Hri) in the six types of MEFs (wild-type, 4KO and 4KO overexpressing the indicated Flag-tagged eIF2α kinases). (D) Representative immunoblots of phosphorylated eIF2α, total eIF2α and Rb 1 h after treatment with the indicated stress stimulus in the six types of MEFs (wild-type, 4KO and 4KO overexpressing the indicated Flag-tagged eIF2α kinases).
The involvement of ISR in oxidative, osmotic, and mitochondrial oxidative stresses, which has not been clarified, was investigated using the eIF2α kinase rescue 4KO cell lines (Fig. 5D). The phosphorylation of eIF2α in response to 400 μM Ars was recovered only when HRI and GCN2 were expressed, which was consistent with a previous finding in yeast27. Phosphorylation of eIF2α in response to 1 mM H2O2 was recovered only when HRI and GCN2 were expressed. Interestingly, we found that it is possible to recover eIF2α phosphorylation in response to 500 mM NaCl, which causes ionic and hyperosmotic stress, or 500 mM sucrose, which causes non-ionic and hyperosmotic stress, by rescuing any of the four eIF2α kinases. Furthermore, eIF2α phosphorylation induced by 25 μM FCCP, which blocks mitochondrial oxidative phosphorylation, could be recovered only by the expression of HRI (Fig. 5D). The 4KO cell lines established in this study were demonstrated to represent effective tools for identifying which eIF2α kinase is the primary or secondary kinase for activating ISR.
Discussion
ISR is a highly conserved adaptation to stress. However, the type of eIF2α kinase differs among the kingdoms. In plants, GCN2 is the sole eIF2α kinase41. Invertebrates and fungi have two eIF2α kinases and protists and vertebrates carry four eIF2α kinases. Protists lack eIF2α kinases that correspond to HRI and PKR but have two eIF2α kinases in addition to the orthologs of GCN2 and PERK. This evidence suggests that the number of eIF2α kinases increased in the course of evolution among plants, fungi, and animals to permit proper responses to diverse stresses. Protists are assumed to have undergone a peculiar course of evolution to survive as a parasite in its metazoan host and acquired two types of eIF2α kinases that differ from those of other eukaryotes. The results of our analysis using 4KO cells derived from a vertebrate suggest that there is no eIF2α kinase universally present in the cells of vertebrates other than the four known kinases. This is in agreement with the disappearance of eIF2α phosphorylation, which was slightly observed in the 4KO cells under non-stressed conditions. However, as our experiments were conducted in a specific cell type (MEF cells), at a specific concentration of stress agents, and at a specific time point (1 hour), there is still the possibility that a cell-specific eIF2α kinase is present in vertebrates. As ISR is involved in the regulation of diverse biological functions, it is worth testing further stress conditions in a future study.
eIF2α kinases have a mutually similar catalytic domain. They have a common characteristic in which their phospho-transfer function is regulated by dimerization. The four kinases have mutually different regulatory domains and this likely explains why they can respond to different stress stimuli. On the contrary, it is interesting that oxidative and osmotic stresses activate two or more eIF2α kinases. This suggests the existence of a molecular mechanism that activates ligands other than the primary target ligand of activation. For example, heme is a natural negative regulator of HRI, and it inhibits the conformation change necessary for autophosphorylation by binding with the HRI dimer. Both nitric oxide (NO) and carbon monoxide (CO) bind to the N-terminal heme-binding domain of HRI. NO acts as an activator of HRI and CO serves as a suppressor of NO-induced HRI activation42. Similarly to these gases, a stimulus could possibly act directly on a regulatory domain and indirectly on a regulatory domain via a binding protein. For example, PACT (a protein activator) has been reported to bind to the dsRNA binding domain of PKR and upon phosphorylation, activate PKR in a process not associated with dsRNA24. For drug development by ISR regulation, it is essential to unveil the mechanisms by which various stress stimuli activate eIF2α kinases. ISR is likely to be involved in the regulation of diverse biological functions and the onset of diseases. Compounds that regulate eIF2α kinase activity, such as via HTS, have been discovered43, but it is important to understand eIF2α kinase selectivity of the compounds. Our developed 4KO cells and eIF2α kinase rescue 4KO cells, which express individual eIF2α kinases, are effective tools for accelerating studies on the molecular mechanism of eIF2α kinase activity.
Methods
Cell culture, transfection, and transduction
SV40 large T-antigen immortalized MEFs were cultured in DMEM-high glucose supplemented with 10% FBS, 2 mM l-glutamine, 55 μM 2-mercaptoethanol, and nonessential amino acids (Invitrogen). PlatGP cells were cultured in DMEM-high glucose supplemented with 10% FBS. Transfection was conducted using the Neon Transfection System (Invitrogen) at 1400 V and 30 ms or polyethylenimine (PEI) (Polysciences). Mouse Hri, Pkr, Perk, and Gcn2 were cloned in-frame with a COOH-terminal 3X-FLAG into the retroviral expression vector pMXs-IG or pMXs-IP (kindly provided by Dr. T. Kitamura). Transfection for retrovirus production was performed using PEI. Retroviral transduction was performed using a previously published protocol with modifications44.
Stress conditions
To validate the ISR activation, MEFs were treated for 1 hour with 400 μM arsenate (Ars), 10 ng/μl polyinosinic-polycytidylic acid (pIC), 200 nM thapsigargin (Tg), 2 mM histidinol (His), 1 mM H2O2 (H2O2), 500 mM NaCl (NaCl), 254 nm 200 J/m2 ultraviolet irradiation (UV), 42 °C heat shock (heat), 4 °C cold shock (cold), low glucose (LG), serum starvation (SS), and anoxia (Ano). Transfection with polyIC was performed using PEI. UV (254 nm) irradiation (200 J/m2) was performed in a CL-1000 UV cross-linker (UVP). Anoxia was induced by placing cells in am MIC-101 modular incubator chamber (Billups-Rothenberg Inc.) flushed with 100% nitrogen gas at 37 °C.
Genome editing using the CRISPR-Cas9 system
The specific gRNA sequences were selected using the CRISPR Design Tool (http://crispr.mit.edu/) or E-CRISP (http://e-crisp-test.dkfz.de/E-CRISP/) and cloned into the gRNA/Cas9 dual expression vector pSpCas9(BB)-2A-GFP or pSpCas9(BB)-2A-Puro45. After transfection, single cells were selected by puromycin resistance or sorted using the GFP signal by fluorescence-activated cell sorting using an S3e cell sorter (Bio-Rad) or JSAN desktop cell sorter (Bay Bioscience) and grown to confluence. The resulting clones were sequenced for verification using an ABI 3130 sequencer (Applied Biosystems).
Real-time RT-qPCR analysis
Total RNA was subjected to RT using RiverTra Ace qPCR RT Master Mix with a gDNA Remover kit (TOYOBO Life Science) according to the manufacturer’s protocol. Real-time PCR was performed using the StepOnePlus Real-Time PCR System (Applied Biosystems) with Power SYBR Green PCR Master Mix (Applied Biosystems). The primer sequences were as follows: mHRI.SP1: 5′-AACCCGCTCCACTCCAAACA-3′; mHRI.AP1: 5′-CTCTGTTGTGGTGGAGTCTCA-3′; mActinβ.SP1: 5′-CTAAGGCCAACCGTGAAAAG-3′ and mActinβ.AP1: 5′-ACCAGAGGCATACAGGGACA-3′.
Immunoblot analysis
Cells were lysed in TNT buffer (50 mM Tris, pH 7.5, 150 mM NaCl, 10% glycerol, 1% Triton X-100) with protease inhibitor cocktail (Nacalai Tesque), phosphatase inhibitor cocktail (Biotool), and 10 μM MG132 for standard SDS-PAGE or Phos-tag SDS-PAGE. Immunoblot analysis was performed as previously described using Blocking One (Nacalai Tesque) or Blocking One-P (Nacalai Tesque) and WesternSure ECL Substrate (Li-Cor Biosciences)46. Protein was visualized by Ez-Capture II (ATTO Corp) and the band intensities were quantified using Image Studio software (Li-Cor Biosciences). The sources of antibodies were as follows: Phospho-Ser51-eIF2α (D9G8) (Cell Signaling Technology); eIF2α (D7D3) (Cell Signaling Technology); anti-PERK (C33E10) (Cell Signaling Technology); anti-GCN2 (Cell Signaling Technology); anti-PKR (M-515) (Santa Cruz Biotechnology); anti-FLAG M2 (Sigma Aldrich); anti-HRI (sc-30143) (Santa Cruz Biotechnology) and anti-HRI (ab84980) (Abcam).
Phylogenetic analysis
To generate phylogenetic trees for the eIF2AK family, the amino acid sequences of the eIF2AK family were retrieved from GenBank (http://www.ncbi.nlm.nih.gov/genbank/) as listed in Table 1. The sequences were aligned using CLUSTAL W 2.147. The phylogenetic trees were constructed via the neighbour-joining method using MEGA 5.0548. Internal branch support was estimated with 1000 bootstrap replicates.
Statistical analysis
Statistical analysis was performed by Student’s t-test. Data are expressed as the mean ± SD. P < 0.05 was defined as the threshold of significance, unless otherwise stated.
Additional Information
How to cite this article: Taniuchi, S. et al. Integrated stress response of vertebrates is regulated by four eIF2α kinases. Sci. Rep. 6, 32886; doi: 10.1038/srep32886 (2016).
References
Wek, R. C., Jiang, H. Y. & Anthony, T. G. Coping with stress: eIF2 kinases and translational control. Biochem Soc Trans 34, 7–11, doi: 10.1042/BST20060007 (2006).
Pathak, V. K., Schindler, D. & Hershey, J. W. Generation of a mutant form of protein synthesis initiation factor eIF-2 lacking the site of phosphorylation by eIF-2 kinases. Mol Cell Biol 8, 993–995 (1988).
Harding, H. P. et al. An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol Cell 11, 619–633 (2003).
Ron, D. Translational control in the endoplasmic reticulum stress response. Journal of Clinical Investigation 110, 1383–1388, doi: 10.1172/jc0200216784 (2002).
Oyadomari, S., Harding, H. P., Zhang, Y., Oyadomari, M. & Ron, D. Dephosphorylation of translation initiation factor 2alpha enhances glucose tolerance and attenuates hepatosteatosis in mice. Cell Metab 7, 520–532, doi: 10.1016/j.cmet.2008.04.011 (2008).
Baird, T. D. & Wek, R. C. Eukaryotic initiation factor 2 phosphorylation and translational control in metabolism. Adv Nutr 3, 307–321, doi: 10.3945/an.112.002113 (2012).
Costa-Mattioli, M. et al. Translational control of hippocampal synaptic plasticity and memory by the eIF2alpha kinase GCN2. Nature 436, 1166–1173, doi: 10.1038/nature03897 (2005).
Sidrauski, C. et al. Pharmacological brake-release of mRNA translation enhances cognitive memory. Elife 2, e00498, doi: 10.7554/eLife.00498 (2013).
Dey, S. et al. ATF4-dependent induction of heme oxygenase 1 prevents anoikis and promotes metastasis. J Clin Invest 125, 2592–2608, doi: 10.1172/JCI78031 (2015).
Munn, D. H. et al. GCN2 kinase in T cells mediates proliferative arrest and anergy induction in response to indoleamine 2,3-dioxygenase. Immunity 22, 633–642, doi: 10.1016/j.immuni.2005.03.013 (2005).
Chen, J. J. & London, I. M. Regulation of protein synthesis by heme-regulated eIF-2 alpha kinase. Trends Biochem Sci 20, 105–108 (1995).
Chen, J. J. et al. Cloning of the cDNA of the heme-regulated eukaryotic initiation factor 2 alpha (eIF-2 alpha) kinase of rabbit reticulocytes: homology to yeast GCN2 protein kinase and human double-stranded-RNA-dependent eIF-2 alpha kinase. Proc Natl Acad Sci USA 88, 7729–7733 (1991).
Levin, D. H., Petryshyn, R. & London, I. M. Characterization of double-stranded-RNA-activated kinase that phosphorylates alpha subunit of eukaryotic initiation factor 2 (eIF-2 alpha) in reticulocyte lysates. Proc Natl Acad Sci USA 77, 832–836 (1980).
Meurs, E. et al. Molecular cloning and characterization of the human double-stranded RNA-activated protein kinase induced by interferon. Cell 62, 379–390 (1990).
Harding, H. P., Zhang, Y. & Ron, D. Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 397, 271–274, doi: 10.1038/16729 (1999).
Bertolotti, A., Zhang, Y., Hendershot, L. M., Harding, H. P. & Ron, D. Dynamic interaction of BiP and ER stress transducers in the unfolded-protein response. Nat Cell Biol 2, 326–332, doi: 10.1038/35014014 (2000).
Dever, T. E. et al. Phosphorylation of initiation factor 2 alpha by protein kinase GCN2 mediates gene-specific translational control of GCN4 in yeast. Cell 68, 585–596 (1992).
Zhang, P. et al. The GCN2 eIF2alpha kinase is required for adaptation to amino acid deprivation in mice. Mol Cell Biol 22, 6681–6688 (2002).
Manning, G., Whyte, D. B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934, doi: 10.1126/science.1075762 (2002).
Manning, G., Plowman, G. D., Hunter, T. & Sudarsanam, S. Evolution of protein kinase signaling from yeast to man. Trends Biochem Sci 27, 514–520 (2002).
Narasimhan, J. et al. Translation regulation by eukaryotic initiation factor-2 kinases in the development of latent cysts in Toxoplasma gondii. Journal of Biological Chemistry 283, 16591–16601, doi: 10.1074/jbc.M800681200 (2008).
Konrad, C., Wek, R. C. & Sullivan, W. J. A GCN2-Like Eukaryotic Initiation Factor 2 Kinase Increases the Viability of Extracellular Toxoplasma gondii Parasites. Eukaryotic Cell 10, 1403–1412, doi: 10.1128/ec.05117-11 (2011).
Konrad, C., Wek, R. C. & Sullivan, W. J. GCN2-like eIF2 alpha kinase manages the amino acid starvation response in Toxoplasma gondii. International Journal for Parasitology 44, 139–146, doi: 10.1016/j.ijpara.2013.08.005 (2014).
Patel, C. V., Handy, I., Goldsmith, T. & Patel, R. C. PACT, a stress-modulated cellular activator of interferon-induced double-stranded RNA-activated protein kinase, PKR. Journal of Biological Chemistry 275, 37993–37998, doi: 10.1074/jbc.M004762200 (2000).
Lu, L. R., Han, A. P. & Chen, J. J. Translation initiation control by heme-regulated eukaryotic initiation factor 2 alpha kinase in erythroid cells under cytoplasmic stresses. Molecular and Cellular Biology 21, 7971–7980, doi: 10.1128/mcb.21.23.7971-7980.2001 (2001).
McEwen, E. et al. Heme-regulated inhibitor kinase-mediated phosphorylation of eukaryotic translation initiation factor 2 inhibits translation, induces stress granule formation, and mediates survival upon arsenite exposure. Journal of Biological Chemistry 280, 16925–16933, doi: 10.1074/jbc.M412882200 (2005).
Zhan, K., Narasimhan, J. & Wek, R. C. Differential activation of eIF2 kinases in response to cellular stresses in Schizosaccharomyces pombe. Genetics 168, 1867–1875, doi: 10.1534/genetics.104.031443 (2004).
Cosentino, G. P. et al. Double-stranded-RNA-dependent protein kinase and TAR RNA-binding protein form homo- and heterodimers in vivo. Proc Natl Acad Sci USA 92, 9445–9449 (1995).
Kimball, S. R., Antonetti, D. A., Brawley, R. M. & Jefferson, L. S. Mechanism of inhibition of peptide chain initiation by amino acid deprivation in perfused rat liver. Regulation involving inhibition of eukaryotic initiation factor 2 alpha phosphatase activity. J Biol Chem 266, 1969–1976 (1991).
Wu, S. Y. et al. Ultraviolet light inhibits translation through activation of the unfolded protein response kinase PERK in the lumen of the endoplasmic reticulum. Journal of Biological Chemistry 277, 18077–18083, doi: 10.1074/jbc.M110164200 (2002).
Deng, J. et al. Activation of GCN2 in UV-irradiated cells inhibits translation. Current Biology 12, 1279–1286, doi: 10.1016/s0960-9822(02)01037-0 (2002).
Hofmann, S., Cherkasova, V., Bankhead, P., Bukau, B. & Stoecklin, G. Translation suppression promotes stress granule formation and cell survival in response to cold shock. Molecular Biology of the Cell 23, 3786–3800, doi: 10.1091/mbc.E12-04-0296 (2012).
Gomez, E., Powell, M. L., Bevington, A. & Herbert, T. P. A decrease in cellular energy status stimulates PERK-dependent eIF2 alpha phosphorylation and regulates protein synthesis in pancreatic beta-cells. Biochemical Journal 410, 485–493, doi: 10.1042/bj20071367 (2008).
Moore, C. E., Omikorede, O., Gomez, E., Willars, G. B. & Herbert, T. P. PERK Activation at Low Glucose Concentration Is Mediated by SERCA Pump Inhibition and Confers Preemptive Cytoprotection to Pancreatic beta-Cells. Molecular Endocrinology 25, 315–326, doi: 10.1210/me.2010-0309 (2011).
Berlanga, J. J., Santoyo, J. & de Haro, C. Characterization of a mammalian homolog of the GCN2 eukaryotic initiation factor 2 alpha kinase. European Journal of Biochemistry 265, 754–762, doi: 10.1046/j.1432-1327.1999.00780.x (1999).
Srivastava, S. P., Kumar, K. U. & Kaufman, R. J. Phosphorylation of eukaryotic translation initiation factor 2 mediates apoptosis in response to activation of the double-stranded RNA-dependent protein kinase. Journal of Biological Chemistry 273, 2416–2423, doi: 10.1074/jbc.273.4.2416 (1998).
Koumenis, C. et al. Regulation of protein synthesis by hypoxia via activation of the endoplasmic reticulum kinase PERK and phosphorylation of the translation initiation factor eIF2 alpha. Molecular and Cellular Biology 22, 7405–7416, doi: 10.1128/mcb.22.21.7405-7416.2002 (2002).
Liu, Y. et al. Regulation of G(1) arrest and apoptosis in hypoxia by PERK and GCN2-mediated eIF2alpha phosphorylation. Neoplasia 12, 61–68 (2010).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823, doi: 10.1126/science.1231143 (2013).
Wang, H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918, doi: 10.1016/j.cell.2013.04.025 (2013).
Lageix, S. et al. Arabidopsis eIF2 alpha kinase GCN2 is essential for growth in stress conditions and is activated by wounding. Bmc Plant Biology 8, 9, doi: 10.1186/1471-2229-8-134 (2008).
Uma, S., Yun, B. G. & Matts, R. L. The heme-regulated eukaryotic initiation factor 2alpha kinase. A potential regulatory target for control of protein synthesis by diffusible gases. J Biol Chem 276, 14875–14883, doi: 10.1074/jbc.M011476200 (2001).
Joshi, M., Kulkarni, A. & Pal, J. K. Small molecule modulators of eukaryotic initiation factor 2alpha kinases, the key regulators of protein synthesis. Biochimie 95, 1980–1990, doi: 10.1016/j.biochi.2013.07.030 (2013).
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp Hematol 31, 1007–1014 (2003).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat Protoc 8, 2281–2308, doi: 10.1038/nprot.2013.143 (2013).
Oyadomari, S. et al. Cotranslocational degradation protects the stressed endoplasmic reticulum from protein overload. Cell 126, 727–739, doi: 10.1016/j.cell.2006.06.051 (2006).
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22, 4673–4680 (1994).
Kumar, S., Tamura, K., Jakobsen, I. B. & Nei, M. MEGA2: molecular evolutionary genetics analysis software. Bioinformatics 17, 1244–1245 (2001).
Acknowledgements
We thank C. Kimura (Tokushima University) for the manuscript preparation and D. Ron (University of Cambridge) and E. Kitamura (University of Tokyo) for providing Fv2E-PERK and pMXs vectors, respectively. This study was supported by the Support Center for Advanced Medical Sciences, Institute of Biomedical Sciences, Tokushima University Graduate School. This work was also supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Culture (MEXT) of Japan to S.O.
Author information
Authors and Affiliations
Contributions
S.O. conceived of the study and wrote the manuscript. S.T. performed most experiments assisted by M.M., K.T. and M.O.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Taniuchi, S., Miyake, M., Tsugawa, K. et al. Integrated stress response of vertebrates is regulated by four eIF2α kinases. Sci Rep 6, 32886 (2016). https://doi.org/10.1038/srep32886
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep32886
This article is cited by
-
Brown adipose tissue CoQ deficiency activates the integrated stress response and FGF21-dependent mitohormesis
The EMBO Journal (2024)
-
Tachykinin-related peptides modulate immune-gene expression in the mealworm beetle Tenebrio molitor L.
Scientific Reports (2022)
-
Homeostasis of the ER redox state subsequent to proteasome inhibition
Scientific Reports (2021)
-
Hepatocyte-specific deletion of XBP1 sensitizes mice to liver injury through hyperactivation of IRE1α
Cell Death & Differentiation (2021)
-
A KDM6 inhibitor potently induces ATF4 and its target gene expression through HRI activation and by UTX inhibition
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.