Abstract
During the 20th century, seawater temperatures have significantly increased, leading to profound alterations in biogeochemical cycles and ecosystem processes. Elevated temperatures have also caused massive bleaching (symbiont/pigment loss) of autotrophic symbioses, such as in coral-dinoflagellate association. As symbionts provide most nutrients to the host, their expulsion during bleaching induces host starvation. However, with the exception of carbon, the nutritional impact of bleaching on corals is still unknown, due to the poorly understood requirements in inorganic nutrients during stress. We therefore assessed the uptake rates of nitrogen and phosphate by five coral species maintained under normal and thermal stress conditions. Our results showed that nitrogen acquisition rates were significantly reduced during thermal stress, while phosphorus uptake rates were significantly increased in most species, suggesting a key role of this nutrient. Additional experiments showed that during thermal stress, phosphorus was required to maintain symbiont density and photosynthetic rates, as well as to enhance the translocation and retention of carbon within the host tissue. These findings shed new light on the interactions existing between corals and inorganic nutrients during thermal stress and highlight the importance of phosphorus for symbiont health.
Similar content being viewed by others
Introduction
Mutualistic marine symbioses, which are widely distributed both geographically and taxonomically1 involve partnerships between ectothermic organisms2. These associations, which are thus particularly sensitive to thermal stress, are already strongly affected by climate change3. Among them, the coral-dinoflagellate symbioses, which dominate tropical benthic ecosystems, experience severe bleaching events in warming waters4.
Bleaching results from the breakdown of the symbiosis and the subsequent expulsion of the corals’ endosymbiotic dinoflagellates and/or the reduction in photosynthetic pigments5. However, dinoflagellates play a central role in the coral host nutrition6. They photosynthesize energy-rich compounds (called photosynthates), which are massively transferred to the host for its own energetic requirements and growth6. They also recycle the host metabolic wastes7 and take up dissolved inorganic nitrogen (DIN) and phosphorus (DIP) from seawater7,8,9. Dissolved nutrients are then transformed into essential organic molecules either respired by or stored in the symbiotic association10. In turn, the coral host, like all animals, is able to prey on a wide range of detrital and living particles11 and shares this nutrient source with its algae12. As most corals rely on their endosymbionts for their nutrition and survival, the increasing frequency in bleaching events may lead to a global collapse of coral reefs all around the world oceans by the 2050s5. There is therefore a crucial need to discern which factors, in addition to thermal stress, reduce or exacerbate the bleaching susceptibilities of scleractinian corals.
Nutrient availability is one of the key biological factors, which has to be taken into account to further explain the corals’ response to thermal stress11,13,14: it is indeed quite clear now that consumption of particulate organic nutrients such as plankton (i.e. heterotrophic feeding)11 allows corals to overcome the decrease in autotrophic nutrient acquisition by symbionts15 during or after thermal stress.
Therefore, species that are able to increase their heterotrophic capacities will have a higher resistance and resilience to bleaching15,16,17. Availability in dissolved inorganic nitrogen and phosphorus (DIN and DIP respectively) in reef environments can also strongly affect coral bleaching susceptibility18,14. However, whether these nutrients reduce or exacerbate bleaching is still subject to debate, because of the following contradicting results; on the one hand, a moderate DIN and DIP enrichment was shown to reduce bleaching and enhance coral photosynthetic performance and calcification under non-stressful18,19 and stressful conditions13,20,21,22. On the other hand, high DIN concentrations can also stimulate symbiont cell division23,24, which in turn may induce a cascading of events (such as higher retention of photosynthates in symbiont cells and CO2 limitation) resulting in coral bleaching14,25. Wiedenmann et al.26 finally pooled information together and proposed a conceptual model, in which the N:P ratio of inorganic nutrients in seawater was the most important factor in explaining coral bleaching. In particular, these authors showed that nitrate enrichment, coupled with phosphorus starvation resulted in an increase of sulpho- to phospholipid ratios in Symbiodinium and caused a destabilization of their thylakoid membranes, increasing the corals’ bleaching susceptibility.
Significant changes in seawater N:P ratios will occur in the future, due to ocean warming and/or eutrophication of coastal waters27,28. Rising sea surface temperatures can indeed enhance near surface stratification, which will inhibit vertical mixing and reduce access to nutrients by corals29. Conversely, nitrogen fixation activity of coral-associated microorganisms will increase28, as well as nutrient concentrations in eutrophicated coastal areas30. Whilst corals will not be able to change their nutritive environment, they can modulate their interaction with it.
Yet, little is known about the corals’ ability to regulate DIN and DIP uptake rates under thermal stress. In particular, no study has explored the corals’ capacity to decrease their nitrate and increase their phosphorus uptake rates under thermal stress, to keep a balanced N:P ratio inside the holobiont tissue26.
The first aim of this study was therefore to test whether thermal stress affects nitrogen and phosphorus uptake in different coral-symbiont (holobiont) associations, representative of common coral species in tropical reefs worldwide. For this purpose, we used a set of aquaria either maintained at 25 °C or 30 °C and we measured the thermally induced variations in DIN and DIP uptake rates by four scleractinian coral species (Turbinaria reniformis, Pocillopora damicornis, Pavona cactus and Galaxea fascicularis) in symbiosis with clade C1 of Symbiodinium and one soft coral Heteroxenia fuscescens, in symbiosis with clade D1. We tested the hypotheses that (i) nitrogen and phosphorus uptake rates will vary among the different holobionts under normal growth conditions; (ii) the rhythmic pulsations of H. fuscescens, which enhance photosynthesis31, will also stimulate nutrient uptake rates32, (iii) thermal stress will decrease nitrogen and increase phosphate uptake rates, to avoid phosphorus limitation, which may exacerbate bleaching26. The second aim of this study was to assess the importance of phosphorus availability for the coral resistance to thermal stress. For this purpose, we exposed one of the above species (Pocillopora damicornis) to a 2 μM DIP enrichment and compared its physiology and stress resistance to non-enriched corals. We tested the hypothesis that phosphorus enrichment will improve the resistance of corals to thermal stress. Broadly, our results provide detailed insights into the changes in the acquisition of inorganic nutrients by scleractinian and soft coral species and further confirm the relevance of studying shifts in N:P ratios to assess the response of reef corals to future climate change.
Results
First experiment
Physiological parameters
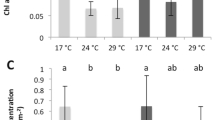
Significant differences were observed between species and temperature for symbiont density and chlorophyll content per mg protein (ANOVA, p = 0.008 and p = 0.0106, Fig. 1a,b). At 25 °C, H. fuscescens presented the highest concentration of chlorophyll (Tukey HSD, p < 0.04, Fig. 1a), while G. fascicularis had the highest symbiont density (Tukey HSD, p < 0.05). At 30 °C, all coral species experienced a decrease in either symbiont density or chlorophyll content. Chlorophyll concentrations indeed decreased for all species compared to 25 °C (ANOVA, p < 0.0001) except for P. cactus (Tukey HSD, p = 0.11). Symbiont density was significantly reduced compared to control conditions in P. cactus, P. damicornis and T. reniformis (Tukey HSD, p < 0.03). A significant effect of species and temperature was observed for the rates of respiration (R), net photosynthesis (Pn) and gross photosynthesis (Pg) normalized to protein content (ANOVA, p1 < 0.0001; p2 = 0.005, p3 < 0.0001, Fig. 1c,d). Both at 25 °C and 30 °C, P. cactus showed the lowest rate of Pn and R (and thus Pg) (Tukey HSD, p < 0.03 for Pn and p < 0.001 for R), while T. reniformis showed higher respiration rates compared to the other species (Tukey HSD, p < 0.008) except H. fuscescens (Tukey HSD, p > 0.05). At 30 °C, P. cactus and P. damicornis showed a significant reduction in Pn compared to 25 °C (Tukey HSD, p = 0.002 and p = 0.006 respectively).
(a) Total chlorophyll content (μg chl (a + c2) (mg prot)−1) and (b) Zooxanthellae density (nb Zoox (mg prot)−1) of the five different coral species at 25 °C (white color) and 30 °C (black color), (c) Rates of net photosynthesis (μmol O2 h−1 (mg prot)−1) and (d) Respiration (μmol O2 h−1 (mg prot)−1) for the different coral species at 25 °C and 30 °C. Data are expressed by means ± standard deviations. Significant effects of the temperature factor were highlighted by an asterisk (*).
Uptake of inorganic nutrients at 25 °C and 30 °C
Species and temperature significantly affected nutrient uptake rates normalized to protein content (ANOVA, p = 0.008, Figs 2 and 3), or to symbiont density (ANOVA, p < 0.039, see Supplementary Table S2).
(a) Uptake rates of ammonium (μmol NH4 h−1 (mg prot)−1) (b) nitrate (μmol NO3 h−1 (mg prot)−1) and (c) phosphorus (μmol PO4 h−1 (mg prot)−1) for the different species at 25 °C (white color) and 30 °C (black color). Data are expressed by means ± standard deviations. Significant effects of the temperature factor were highlighted by an asterisk (*).
Radar plots highlighting the behavior of each coral species according to the different temperature steps (25 °C, green color and 30 °C, red color) and the studied parameters: Rates of NH4, NO3, PO4 uptake, chlorophyll and zooxanthellae concentrations as well as net photosynthesis and respiration rates (μmol O2 h−1 (mg prot)−1).
At 25 °C, P. cactus presented one of the highest NO3 uptake rate per protein content (Tukey HSD, p < 0.03, Figs 2b and 3) and one of the highest PO4 uptake rate per symbiont cell (Tukey HSD, p < 0.014, Supplementary Table S2). Ammonium uptake rate per symbiont cell was higher for P. damicornis compared to P. cactus, G. fascicularis and H. fuscescens. P. damicornis presented higher N-uptake:P-uptake ratios, around 9, compared to T. reniformis and H. fuscescens (Tukey HSD, p < 0.004, Supplementary Table S2).
Thermal stress had a significant effect on nutrient uptake rates, normalized to protein. At 30 °C, NH4 uptake rates significantly decreased in P. damicornis (Tukey HSD, p < 0.03, Fig. 2a), while NO3 uptake rates decreased in P. damicornis, P. cactus and G. fascicularis (Tukey HSD, p < 0.02). Effects of thermal stress on nitrogen uptake rates normalized to symbiont cell were significant in the presence of nitrate, with a net observed decrease in the acquisition rate of NO3 in P. cactus (Supplementary Table S8, Tukey HSD, p < 0.05) and a further but not significant decrease for T. reniformis and G. fascicularis (Tukey HSD, p > 0.05). PO4 uptake rates significantly increased in T. reniformis, G. fascicularis, H. fuscescens and P. damicornis when normalized to protein content (Tukey HSD, p < 0.04, Figs 2c and 3) and in the first three species when normalized to symbiont cell (Tukey HSD, p < 0.01, see Supplementary Table S2). PO4 uptake rates however significantly decreased for P. cactus (Tukey HSD, p < 0.05). Regarding the effect of tentacle pulsation of the soft coral H. fuscescens on nutrient uptake, pulsation rates did not vary significantly between 25 °C and 30 °C (10 puls/min at 25 °C to 8.5 puls/min at 30 °C; T-test, p > 0.05). However, nitrogen uptake rates significantly decreased as tentacles stopped pulsating (ANOVA, pNH4 = 0.009; pNO3 = 0.033, see Supplementary Table S4). A similar but non-significant trend was observed for the PO4 uptake rate (ANOVA, pPO4 > 0.05). P. damicornis and G. fascicularis showed a drastic decrease in their N-uptake:P-uptake ratios (Tukey HSD, p < 0.0074, Supplementary Fig. S3).
2nd Experiment: Effect of thermal stress and phosphate enrichment on the rates of carbon assimilation and translocation
At 25 °C, no significant difference was observed between control and P-enriched corals regarding the symbiont density, the rETR max and NPQmax and the gross photosynthesis expressed in equivalent carbon (PgC) (Tukey HSD, p > 0.05). After one week at 30 °C, control corals showed a significant decrease in symbiont density per surface area (−20%, Tukey HSD, p = 0.008, Supplementary Fig. S6), in the rates of PgC (Tukey HSD, p = 0.049, Supplementary Fig. S5) and NPQmax (Tukey HSD, p = 0.048, from 0.36 to 0.25). Conversely, corals maintained under PO43− enrichment did not show any significant sign of bleaching nor change in their rates of gross photosynthesis compared to those at 25 °C (Tukey HSD, p > 0.05). They maintained high values of rETRmax (74 against 55 in control corals) and NPQmax (0.33 against 0.25 in control nubbins) with increasing temperature, higher than control corals (Tukey HSD, prETR = 0.023; pNPQ = 0.021, Fig. 4c). In addition, at 25 °C, 13C-bicarbonate labelling showed significant interactions between compartments (either zooxanthellae or animal tissue) and treatment for the rate of carbon assimilation (ANOVA, p < 0.0001). While carbon was equally incorporated in symbiont and animal tissue in control corals (Tukey HSD, p > 0.05, Fig. 4a), tissues from corals maintained under phosphorus enrichment showed a higher carbon incorporation rate and percentage (50% against 14%) (Tukey HSD, p < 0.001). The same trend was observed at 30 °C (34% against 12%; Tukey HSD, p < 0.001). Carbon translocation rates were also higher in P-enriched corals at 30 °C compared with control corals (81% against 67%; Tukey HSD p = 0.013, Fig. 4b). Symbiont respiration rates were two times higher at 25 °C and 30 °C for control corals compared to enriched nubbins, with values of 0.5 μg C cm−2 h−1 against 1–1.3 μg C cm−2 h−1 for control (Tukey HSD, p25 °C = 0.017; p30 °C = 0.0014), while the percentage of carbon respired was higher in enriched corals than control nubbins at 30 °C (44% against 25%; Tukey HSD, p < 0.05).
(a) Carbon assimilated (μg C h−1 cm−2) either in the symbiont or animal tissue fractions, (b) Percentage of carbon transferred from the symbiont to the animal tissue and (c) Maximum relative electron transport rate (rETRmax) according to the different temperature steps and the nutrient treatments (Control corals: light grey, Enriched corals: dark grey).
Finally, no significant differences were observed for the calcification rates either at 25 °C and 30 °C, with equivalent values from 14 to 16 μg C cm−2 h−1 at 25 °C and from 11 to 13 μg C cm−2 h−1 at 30 °C for control and enriched nubbins (ANOVA, p > 0.05).
Discussion
Our analysis of the DIN and DIP uptake rates of four scleractinian and one soft tropical coral species shows that coral holobionts regulate the uptake of nutrients and points to higher DIP uptake rates under thermal stress. In addition, corals maintained under DIP enrichment avoided bleaching and maintained high rates of photosynthesis, conversely to control, non-enriched corals, which experienced both a decrease in symbiont density and in the acquisition of inorganic carbon. All together, these observations strongly suggest that phosphorus is an essential nutrient for the symbiosis during stress events. Conversely, nitrogen uptake rates significantly decreased under thermal stress, either due to coral bleaching and/or to avoid excess nitrogen in the coral tissue. Our results highlight the importance of nitrogen and phosphorus availability in the coral response to thermal stress.
Under control conditions, DIP and DIN uptake rates normalized to symbiont cell or holobiont biomass, as well as the ratios of DIN:DIP uptake, presented a threefold variation among the coral species tested, potentially reflecting different nutritional requirements and ecological trade-offs in the allocation of nitrogen and phosphorus amongst macromolecules associated with diverse functions33. Furthermore, we show that H. fuscescens exhibited higher DIN uptake rates during its pulsating activity, as previously demonstrated for the jellyfish Cassiopeia sp34. Therefore, rhythmic pulsations not only improve H. fuscescens photosynthesis by preventing seawater re-filtration31, but also enhanced its acquisition of essential nutrients.
All scleractinian coral species bleached in response to thermal stress with a reduction in rates of net photosynthesis for P. cactus and P. damicornis.
Despite this loss of symbionts and/or chlorophyll, rates of phosphate acquisition per protein content or symbiont cell were significantly enhanced in four out of the five species investigated and for both clades C1 and D1. Since the same trend was observed with S. pistillata in symbiosis with another Symbiodinium clade (clade A)35, increased phosphate uptake seems to be a general response of corals to thermal stress, independently of the clade genotype or on the polyp activity. Although it might only be due to a temperature dependency of the phosphorus transporters at the cell membranes36, our results tend to confirm the fact that phosphorus is actively needed in corals to offset the negative effect of thermal stress26 and that corals underpin an active control mechanism for phosphorus uptake and allocation, as observed in plants37. Indeed, our second experiment, in which DIP enrichment of the seawater prevented bleaching and increased the photosynthetic efficiency and the translocation of photosynthates during thermal stress, highlights the importance of phosphorus to maintain the physiological functions in corals under thermal stress. Wiedenmann et al.26 also demonstrated that limited phosphorus availability induces a shift from phospholipids to sulpholipids in symbiont membranes and increases the susceptibility of corals to temperature-induced bleaching38. In addition to being a structural membrane component, phosphorus takes part in the synthesis of nucleic acids and other energy generation processes39. It is also involved in cellular signalling via thylakoid protein phosphorylation40. It has to be noticed that although corals are able to increase their rates of phosphorus acquisition under warming conditions, a minimal concentration of inorganic phosphorus, which remains to be determined, is required to avoid bleaching.
In contrast to phosphorus, three coral species (P. cactus, P. damicornis and G. fascicularis) out of the five investigated showed a significant decrease in nitrogen uptake rates normalized to protein content. Nitrogen uptake rates normalized to symbiont cell, however, remained unchanged between 25 °C and 30 °C, except for nitrate in P. cactus.
A similar finding was observed with the scleractinian coral S. pistillata associated with clade A1 symbionts35, which significantly decreased both nitrate and ammonium uptake rates down to zero, at 33 °C. These results suggest that the reduction in nitrogen uptake is a general response of corals to thermal stress, independent of the clade genotype. Whether this reduction is a consequence of bleaching (and reduced symbiont activity), or is due to a physiological control or feedback aimed at decreasing the amount of nitrogen entering the symbiotic association, requires further investigation. Both hypotheses have pros and cons. The first hypothesis is in agreement with studies showing that nitrogen uptake is mostly driven by the symbionts8,9,41,42. In H. fuscescens, which did not experience any bleaching or decrease in photosynthetic activity, due to its pulsating activity and/or its association with the thermally resistant clade D143,44,45, no decrease in nitrogen uptake rate was indeed observed. A decrease in nitrogen uptake rates in the scleractinian coral species may lead to nitrogen limitation, which in turn may impair protein repair23, weaken the photosynthetic capacities and decrease carbon fixation, as already noted in mulberry leafs46. This may explain why a supplementation of seawater with ammonium or nitrate was shown to increase the resilience of coral species to stress-induced bleaching21,22,47. On the other hand, the second hypothesis is in agreement with studies showing an increased bleaching susceptibility of heat-stressed corals in presence of high nitrogen and low phosphorus concentrations26,48,49,50. High nitrogen availability for symbionts during thermal stress tends to favour symbiont growth and reduce the amount of photosynthates transferred to the host14, together with inducing a higher phosphorus limitation26. This could explain the reduction in nitrogen uptake rates by the symbiotic association during thermal stress, in order to reduce the amount of nitrogen available to symbionts.
Overall, the decrease in the N:P uptake ratios during thermal stress, which is consistent for most coral species, may imply that the corals internally experience an increase in their N:P ratio, maybe due to reallocation of nitrogen that was stored in tissue reserves10. The relevance of the N:P ratio for terrestrial and marine symbiotic associations has recently been discussed through a meta-analysis50 which showed that a decoupling in mutualism performance occurs whenever phototrophs benefit from nutrient enrichment at the expense of their heterotrophic partners. It has also been demonstrated in recent studies26,51, in which changes in seawater nutrient ratios induced changes in carbon acquisition, translocation and allocation within the symbiosis. The benefits that the symbiotic association retrieves from a low N:P ratio condition are highlighted in our second experiment, in which the supplementation of seawater with 2 μM phosphate and the subsequent decrease in the N:P ratio, prevented bleaching, increased the photosynthetic capacities, the carbon translocation, as well as the carbon retained into animal biomass during thermal stress.
Understanding how corals respond to alterations in seawater N:P ratio, or how they adjust their internal N:P ratio in response to environmental changes, is an important task for coral reef research today. Our results clearly show that corals are phosphorus-limited during thermal stress and that they are able to increase their acquisition of phosphorus independently of the Symbiodinium clade involved in the symbiotic association. Further investigations, which will trace the fate of nitrogen and phosphorus within the symbiosis at elevated temperatures, are however needed to bring detailed insights into the processes involved in the nutrient acquisition and allocation by corals under thermal stress.
Materials and Methods
First experiment: experimental design and biological material
The first experiment was conducted with four tropical scleractinian coral species, belonging to different families, Turbinaria reniformis (Dendrophylliidae), Pocillopora damicornis (Pocilloporidae), Pavona cactus (Agariciidae), Galaxea fascicularis (Oculinidae) and the soft coral species Heteroxenia fuscescens (Xeniidae), all originating from the Red Sea. These corals species were selected for their different morphology types and because they are good representatives of common coral families in tropical reefs worldwide52,53. Coral colonies were maintained in aquaria for several months at 25 °C ± 0.5. The total daily light received by our corals in these conditions (13 E m−2 d−1 which corresponds to 150 μmol photons m−2 s−1) was equivalent to the one received on reefs at 3–5 m depth (a typical ideal day with no cloud generates a total of 14 E m−2d−1 day at 3 m depth, while a cloudy day generated 6.2 E m−2d−1 54. A total of 48 nubbins per species was prepared by cutting 4 parental colonies (12 nubbins per colony) with a bone cutter for hard corals or scissors for soft corals. Nubbins were then evenly divided in twelve 20 L tanks (with 4 nubbins per species per tank) in order to maximize the genetic diversity. Aquaria received a continuous supply of oligotrophic seawater (<0.5 μM dissolved inorganic nitrogen and <0.1 μM inorganic phosphorus) at a flow rate of 20 L h−1 (i.e. one tank renewal every hour).
Control conditions were maintained using submersible resistance heaters (Visi-ThermH Deluxe, Aquarium Systems, France) and metal halide lamps (Philips, HPIT 400 W, Distrilamp, Bossee, France). Submersible pumps ensured proper water mixing inside each tank. Nubbins were fed twice a week for 2 weeks with Artemia salina nauplii to improve their recovery. Three weeks prior to the experiments, feeding was stopped to minimize the impact of organic nutrients on the uptake rates of inorganic nutrients (described below).
Before the measurements, four nubbins from each species (one per colony) were frozen at −80 °C for the Symbiodinium clade determination. DNA was extracted using the DNeasy Plant mini Kit (Qiagen) for H. fuscescens and the PowerSoil DNA Isolation Kit (MO-BIO) for the other species. For the determination of symbiont phylogeny, we followed the protocol of Santos et al.55 and sequenced a 600 to 700 bps fragment of chloroplastic 23S ribosomal gene. All coral species harboured Symbiodinium clade C1, with the exception of H. fuscescens, which hosted clade D1 (see S1).
Uptake of inorganic nutrients at 25 °C and 30 °C
Six aquaria were kept under control conditions. In the remaining six tanks, seawater temperatures were raised from 25 °C ± 0.5 °C (time T0 −25 °C) to 30 °C ± 0.5 °C over a 10 day-period (0.5 °C per day) and maintained constant for 10 days (time T1 −30 °C). This thermal stress was chosen according to the highest temperature monitored in the northern Red Sea56. Samples from the 5 coral species were collected from both the control and experimental tanks at T0 and T1 to assess their photosynthetic performances as well as their uptake rates of DIN and DIP (NH4, NO3, PO4) as described below. Nubbins were then frozen at −20 °C prior to the determination of the chlorophyll and protein content as well as the symbiont density. Throughout the experiment, inorganic nutrient concentrations were measured in the different aquaria twice a week using an Autoanalyzer (Alliance Instrument, AMS, France), following the protocol of Le Corre & Tréguer57. Concentrations remained below 0.5 μM for total DIN and below 0.1 μM for DIP.
Net photosynthesis (Pn) and respiration (R) rates were assessed at 0 and 150 μmol photons m−2 s−1 on four coral nubbins per species (from 4 different colonies) and temperature using the respirometry technique58.
Gross photosynthesis (Pg) was assessed by adding the absolute value of R to Pn. Data were normalized to total protein content and surface area for hard coral species. For protein and chlorophyll concentrations (a and c2) as well as symbiont density, tissue was removed from the skeleton in filtered seawater (FSW) using an air-brush, homogenized using a potter grinder and divided into three aliquots for each measurement, performed according to Rodolfo-Metalpa et al.59 for symbionts, Jeffrey & Humphrey60 for chl a and c2 and Smith et al.61 using a BCAssay kit (Interchim, Montluçon, France) for protein content determination. Surface area of nubbins were assessed using the wax dipping technic62.
Inorganic nutrient uptake was assessed, at the same time for the control and experimental tanks, at T0 and T1, by the depletion technique63. In short, for each nutrient and incubation time, four nubbins per species (one nubbin per colony) were placed in individual 200-mL beakers maintained in a water bath at the respective experimental temperatures (25 °C or 30 °C) and light intensity (150 μmol photons m−2 s−1). Three control beakers, without coral nubbin, were used to monitor for potential changes in water chemistry due to adsorption onto beaker surface, consumption by microbial activity, or to air contamination. After a 30 min acclimation period for corals, seawater was enriched with a stock solution of either 10 mM ammonium chloride (NH4Cl), Sodium nitrate (NaNO3), or sodium dihydrogen phosphate (NaH2PO4), in order to reach a final concentration of 3 μM for all nutrients. Stirring bars were added to ensure a proper homogenization of the nutrient mixture in the beakers. Water samples (10 mL) were collected from each beaker and filtered through a 45 micron syringe filter immediately after the enrichment and then subsequently after 15 min, 30 min, 45 min, 60 min and 90 min. Ammonium concentrations were determined immediately after sampling, according to the spectrofluorimetric method of Holmes et al.64. Samples for nitrate and phosphate were frozen, prior to measurements using an autoanalyzer (Alliance Instrument, AMS, France).
Uptake rates of each inorganic nutrient were calculated as the difference in concentration between the 0 and 90 minutes time points, after data correction for the diminution of the beakers’ volume and verification of the linearity in the depletion during the experiment. Uptake rates were normalized to the symbiont density (which changed during the thermal stress) or to the total protein content, which did not change during the whole experiment. N-uptake:P-uptake ratios were calculated at T0 and T1 by dividing the total amount of nitrogen (NH4 and NO3) by the amount of phosphorus taken up.
Effect of soft coral pulsation on the uptake rates of inorganic nutrients
Polyp pulsation by H. fuscescens under non stressful conditions was shown to improve photosynthesis, by enhancing oxygen and inorganic carbon exchange between seawater and polyp tissue31 and was suggested to favor uptake of inorganic nutrients32. To investigate whether thermal stress had an effect on the rhythmic pulsation of this species, we monitored the number of tentacle pulsation per minute of four nubbins of H. fuscescens maintained under non-stressful (25 °C) and thermal stress (30 °C) conditions. In addition, we also assessed the effect of polyp pulsation on the uptake rates of inorganic nutrients (NH4, NO3 and PO4). For this purpose, additional 32 coral nubbins (8 nubbins per colony) were cut from 4 parental colonies, different from the ones used for the uptake rates assessment. A clove oil solution (Phytosun aroms; Omega Pharma, France) was chosen to anaesthetize corals and inhibit their movements. For this experiment, each coral nubbin was only maintained for 1 h30 under a diluted clove oil solution (0.2 ppt). A concentration lower than 0.5 ppt was shown to be completely harmless to corals65,66. In addition, preliminary tests were performed to test the harmlessness of the clove oil at different dilutions, by comparing rates of photosynthesis and respiration as well as chlorophyll and symbionts concentrations with and without clove oil addition.
We didn’t observe any effect of clove oil on the above parameters for concentrations below 0.5 ppt (results not shown). Following optimization, eight nubbins per nutrient were disposed in distinct 200-mL beakers, half of them were placed in FSW with clove oil and the other half in FSW only (control condition). When coral tentacles were totally immobilized by the clove oil, NH4, NO3 or PO4 solutions were added to reach a final concentration of 3 μM, N or P and nutrient depletion was followed as described above. Nutrient uptake rates were normalized to the total protein concentration of each nubbin.
Second experiment: Carbon budget under PO4 3− supply
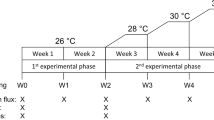
Based on the results obtained in the first experiment, we tested the effects of phosphate enrichment and thermal stress on the carbon budget of Pocillopora damicornis, which was chosen as a model species. We used 13C-bicarbonate, to follow the inorganic carbon incorporation rate and transfer between the host and symbionts, as described in Tremblay et al.67. 160 nubbins were generated from four parental colonies and equally distributed among 8 aquaria maintained under control conditions as previously described. Corals were exposed to: 1) natural seawater, called control conditions, with low nutrient levels (0.5 μM N and 0.1 μM P; 4 aquaria) and 2) phosphorus enriched conditions, called “P” (0.5 μM N and 2 μM P; 4 aquaria). Tanks were continuously supplied with a solution of NaH2PO4 pumped from a stock solution via a peristaltic pump. The stock solution was renewed every three days and added to the tanks at a constant flow rate of 0.3 L h−1.
Nutrient concentrations in the tanks were monitored twice a week using an Autoanalyzer (Alliance Instrument, AMS, France). Nubbins were maintained under these conditions at 25 °C (T25) for four weeks. Seawater temperatures were then raised from 25 °C ± 0.5 °C to 30 °C ± 0.5 °C over a 10 day-period in half of the tanks and was kept constant for 7 days (T30).
Rates of net photosynthesis (Pn), R and Pg, as well as zooxanthellae density, were measured as previously described at 0 and 150 μmol photons m−2 s−1 on six nubbins per treatment at T25 and T30. Symbiont respiration rates were assessed on six freshly isolated symbiont samples according to Tremblay et al.67. The resulting oxygen fluxes were converted to carbon equivalent based on molar weights according to Anthony & Fabricius68. The maximum relative electron transport rate (rETRmax) and the non-photochemical quenching (NPQmax) were assessed on the same six coral nubbins, at 25 °C and 30 °C at different light levels using a Pulse Amplitude Modulation (PAM) fluorometer (Walz, Germany) according to Ralph and Gademann69. Calcification rates were measured on six nubbins per treatment using the buoyant weight technique70. All results were normalized to the surface area of each respective nubbin.
The carbon budget was assessed at the end of T25 and T30 using the H13CO3 labelling technique according to Tremblay et al.67. For each treatment, eight nubbins (2 per colony) were disposed in individual beakers filled with 200 mL FSW, enriched with a final concentration of 0.6 mM of NaH13CO3 (98 atom %13C, no. 372382, Sigma-Aldrich, St Louis, MO, USA). Nubbins were incubated for 5 h and transferred to beakers containing non-enriched seawater (FSW) for two chase periods (0 and 24 h). At the end of each chase period, 4 nubbins were directly frozen at −20 °C to assess 13C enrichment in animal tissue and symbionts. Additionally, four control nubbins were incubated in non 13C- enriched seawater. The resulting %13C enrichment was quantified using a Delta Plus Mass spectrometer coupled to a C/N analyser (Thermofisher Scientific, Bremen, Germany).
Statistical analyses
Statistical analyses were performed using Statistica 10. Data were checked for normality using the Kolmogorov-Smirnov test with Lilliefors correction and for Homoscedasticity using Levene’s test. Data were log-transformed if necessary. Two ways ANOVAs were used to test (i) the effect of temperature and coral species (independent factors) on nutrient uptake rates and other physiological parameters measured in the first experiment; (ii) the effect of temperature and phosphorus enrichment (independent factors) on all parameters measured in the second experiment. Calcification rates and the maximum rETR and NPQ were analysed using a general linear model for parametric repeated measures analysis of variance with “Temperature” as dependent variable and “Treatment” as categorical predictor. In case the assumption of sphericity (independency of the repeated measures) was not fulfilled, the hypotheses were tested using the multivariate approach (Wilks test) for repeated measurements. Differences in the rates of carbon assimilation and the percentage of carbon remaining within the symbiosis, were assessed using three-ways ANOVA (with temperature, phosphorus enrichment and host-symbiont compartments as independent factors). A t-test was finally processed to test the effect of clove oil on nutrient uptake rates by H. fuscescens. In case significant differences were observed between treatments, analyses were followed by a posteriori test (Tukey’s test). Differences were considered significant when p < 0.05.
Additional Information
How to cite this article: Ezzat, L. et al. Limited phosphorus availability is the Achilles heel of tropical reef corals in a warming ocean. Sci. Rep. 6, 31768; doi: 10.1038/srep31768 (2016).
References
Saffo, M. B. In Encyclopedia of Life Sciences, doi: 10.1002/9780470015902.a0003281.pub2. (John Wiley & Sons Ltd, 2001).
Six, D. L. Climate change and mutualism. Nat. Rev. Microbiol. 7, 686–686 (2009).
Doney, S. C. et al. Climate change impacts on marine ecosystems. Mar. Sci. 4, 11–37, doi: 10.1146/annurev-marine-041911-111611 (2012).
Roth, M. S., Goericke, R. & Deheyn, D. D. Cold induces acute stress but heat is ultimately more deleterious for the reef-building coral Acropora yongei. Sci. Rep. 2, 240, doi: 10.1038/srep00240 (2012).
Hoegh-Guldberg, O. et al. Coral reefs under rapid climate change and ocean acidification. Science 318, 1737–1742 (2007).
Muscatine, L., Falkowski, P., Porter, J. & Dubinsky, Z. Fate of photosynthetic fixed carbon in light-and shade-adapted colonies of the symbiotic coral Stylophora pistillata. Proc. R. Soc. B. 222, 181–202 (1984).
Rosset, S., D’Angelo, C. & Wiedenmann, J. Ultrastructural Biomarkers in Symbiotic Algae Reflect the Availability of Dissolved Inorganic Nutrients and Particulate Food to the Reef Coral Holobiont. Front. Mar. Sci. 2, 103, doi: 10.3389/fmars.2015.00103 (2015).
Grover, R., Maguer, J.-F., Allemand, D. & Ferrier-Pages, C. Nitrate uptake in the scleractinian coral Stylophora pistillata. Limnol. Oceanogr. 48, 2266–2274 (2003).
Pernice, M. et al. A single-cell view of ammonium assimilation in coral–dinoflagellate symbiosis. ISME J. 6, 1314–1324 (2012).
Miller, D. & Yellowlees, D. Inorganic nitrogen uptake by symbiotic marine cnidarians: a critical review. Proc. R. Soc. B. 237, 109–125 (1989).
Houlbreque, F. & Ferrier‐Pagès, C. Heterotrophy in tropical scleractinian corals. Biol. Rev. 84, 1–17 (2009).
Tremblay, P., Grover, R., Maguer, J.-F., Hoogenboom, M. & Ferrier-Pagès, C. Carbon translocation from symbiont to host depends on irradiance and food availability in the tropical coral Stylophora pistillata. Coral Reefs 33, 1–13 (2014).
Langdon, C. & Atkinson, M. Effect of elevated pCO2 on photosynthesis and calcification of corals and interactions with seasonal change in temperature/irradiance and nutrient enrichment. J. Geophys. Res. Oceans 110, doi: 10.1029/2004JC002576 (2005).
Wooldridge, S. Breakdown of the coral-algae symbiosis: towards formalising a linkage between warm-water bleaching thresholds and the growth rate of the intracellular zooxanthellae. Biogeosciences 10, 1647–1658 (2013).
Grottoli, A. G., Rodrigues, L. J. & Palardy, J. E. Heterotrophic plasticity and resilience in bleached corals. Nature 440, 1186–1189 (2006).
Palardy, J. E., Grottoli, A. G. & Matthews, K. A. Effects of upwelling, depth, morphology and polyp size on feeding in three species of Panamanian corals. Mar. Ecol. Prog. Ser. 300, 79–89 (2005).
Ferrier-Pagès, C., Rottier, C., Beraud, E. & Levy, O. Experimental assessment of the feeding effort of three scleractinian coral species during a thermal stress: Effect on the rates of photosynthesis. J. Exp. Mar. Biol. Ecol. 390, 118–124 (2010).
Tanaka, Y., Miyajima, T., Koike, I., Hayashibara, T. & Ogawa, H. Imbalanced coral growth between organic tissue and carbonate skeleton caused by nutrient enrichment. Limnolo. Oceanogr. 52, 1139–1146 (2007).
Godinot, C., Ferrier-Pagès, C. & Grover, R. Control of phosphate uptake by zooxanthellae and host cells in the scleractinian coral Stylophora pistillata. Limnolo. Oceanogr. 54, 1627–1633 (2009).
Holcomb, M., McCorkle, D. C. & Cohen, A. L. Long-term effects of nutrient and CO 2 enrichment on the temperate coral Astrangia poculata (Ellis and Solander, 1786). J. Exp. Mar. Biol. Ecol. 386, 27–33 (2010).
Chauvin, A., Denis, V. & Cuet, P. Is the response of coral calcification to seawater acidification related to nutrient loading? Coral Reefs 30, 911–923 (2011).
Béraud, E., Gevaert, F., Rottier, C. & Ferrier-Pagès, C. The response of the scleractinian coral Turbinaria reniformis to thermal stress depends on the nitrogen status of the coral holobiont. J. Exp. Biol. 216, 2665–2674 (2013).
Falkowski, P. G., Dubinsky, Z., Muscatine, L. & McCloskey, L. Population control in symbiotic corals. Bioscience 43, 606–611 (1993).
Cunning, R. & Baker, A. C. Excess algal symbionts increase the susceptibility of reef corals to bleaching. Nat. Clim. Change 3, 259–262 (2013).
Rädecker, N., Pogoreutz, C., Voolstra, C. R., Wiedenmann, J. & Wild, C. Nitrogen cycling in corals: the key to understanding holobiont functioning? Trends Microbiol. 23, 490–497 (2015).
Wiedenmann, J. et al. Nutrient enrichment can increase the susceptibility of reef corals to bleaching. Nat. Clim. Change 3, 160–164 (2013).
Van de Waal, D. B., Verschoor, A. M., Verspagen, J. M., van Donk, E. & Huisman, J. Climate-driven changes in the ecological stoichiometry of aquatic ecosystems. Front. Ecol. Environ. 8, 145–152 (2009).
Santos, H. F. et al. Climate change affects key nitrogen-fixing bacterial populations on coral reefs. ISME J. 8, 2272–2279 (2014).
Tyrrell, T. The relative influences of nitrogen and phosphorus on oceanic primary production. Nature 400, 525–531 (1999).
Johnson, A. & Harrison, M. The increasing problem of nutrient runoff on the coast. Am. Sci. 103, 98, doi: 10.1511/2015.113.98 (2015).
Kremien, M., Shavit, U., Mass, T. & Genin, A. Benefit of pulsation in soft corals. Proc. Natl. Acad. Sci. USA 110, 8978–8983 (2013).
Wild, C. & Naumann, M. S. Effect of active water movement on energy and nutrient acquisition in coral reef-associated benthic organisms. Proc. Natl. Acad. Sci. USA 110, 8767–8768 (2013).
Moore, C. et al. Processes and patterns of oceanic nutrient limitation. Nat. Geosci. 6, 701–710 (2013).
Jantzen, C., Wild, C., Rasheed, M., El-Zibdah, M. & Richter, C. Enhanced pore water nutrient fluxes by the upside-down jellyfish Cassiopeia sp. in a Red Sea coral reef. Mar. Ecol. Prog. Ser. 411, 117–125 (2010).
Godinot, C., Houlbrèque, F., Grover, R. & Ferrier-Pagès, C. Coral uptake of inorganic phosphorus and nitrogen negatively affected by simultaneous changes in temperature and pH. PLoS One 6, e25024 (2011).
D’Elia, C. F. The uptake and release of dissolved phosphorus by reef coralsl'2. Limnol. Oceanogr. 22, 301–315 (1977).
Amtmann, A. & Blatt, M. R. Regulation of macronutrient transport. New Phytol. 181, 35–52 (2009).
Tchernov, D. et al. Membrane lipids of symbiotic algae are diagnostic of sensitivity to thermal bleaching in corals. Proc. Natl. Acad. Sci. USA 101, 13531–13535 (2004).
Beardall, J., Sobrino, C. & Stojkovic, S. Interactions between the impacts of ultraviolet radiation, elevated CO2 and nutrient limitation on marine primary producers. Photochem. Photobiol. Sci. 8, 1257–1265 (2009).
Vener, A. V. Environmentally modulated phosphorylation and dynamics of proteins in photosynthetic membranes. Biochim. Biophys. Acta. BBA. 1767, 449–457 (2007).
Grover, R., Maguer, J.-F., Reynaud-Vaganay, S. & Ferrier-Pages, C. Uptake of ammonium by the scleractinian coral Stylophora pistillata: effect of feeding, light and ammonium concentrations. Limnol. Oceanogr. 47, 782–790 (2002).
D’Elia, C., Domotor, S. & Webb, K. Nutrient uptake kinetics of freshly isolated zooxanthellae. Mar. Biol. 75, 157–167 (1983).
Jones, A. M., Berkelmans, R., van Oppen, M. J., Mieog, J. C. & Sinclair, W. A community change in the algal endosymbionts of a scleractinian coral following a natural bleaching event: field evidence of acclimatization. Proc. R. Soc. B. 275, 1359–1365 (2008).
LaJeunesse, T. C. et al. Host–symbiont recombination versus natural selection in the response of coral–dinoflagellate symbioses to environmental disturbance. Proc. R. Soc. B. 277, 2925–2934, doi: 10.1098/rspb.2010.0385 (2010).
Baker, A. C. Ecosystems: reef corals bleach to survive change. Nature 411, 765–766 (2001).
Chaitanya, K., Sundar, D. & Reddy, A. R. Mulberry leaf metabolism under high temperature stress. Biol. Plantarum 44, 379–384 (2001).
Tanaka, Y. et al. Nutrient availability affects the response of juvenile corals and the endosymbionts to ocean acidification. Limnol. Oceanogr. 59, 1468–1476 (2014).
Nordemar, I., Nyström, M. & Dizon, R. Effects of elevated seawater temperature and nitrate enrichment on the branching coral Porites cylindrica in the absence of particulate food. Mar. Biol. 142, 669–677 (2003).
Schlöder, C. & D’Croz, L. Responses of massive and branching coral species to the combined effects of water temperature and nitrate enrichment. J. Exp. Mar. Biol. Ecol. 313, 255–268 (2004).
Shantz, A. A., Lemoine, N. P. & Burkepile, D. E. Nutrient loading alters the performance of key nutrient exchange mutualisms. Ecol. lett. 19, 20–28 (2016).
Ezzat, L., Maguer, J.-F., Grover, R. & Ferrier-Pagès, C. New insights into carbon acquisition and exchanges within the coral–dinoflagellate symbiosis under NH4+ and NO3− supply. Proc. R. Soc. B. 282, doi: 10.1098/rspb.2015.0610 (2015).
Veron, J. E. N. Corals of the World. (Odyssey Publishing, 2000).
Lieske, E., Fiedler, K. E. & Myers, R. F. Coral Reef Guide: Red Sea to Gulf of Aden, South Oman;[the Definitive Guide to Over 1200 Species of Underwater Life]. (Collins, 2004).
Davies, P. S. Effect of daylight variations on the energy budgets of shallow-water corals. Mar. Biol. 108, 137–144 (1991).
Santos, S. R. et al. Molecular phylogeny of symbiotic dinoflagellates inferred from partial chloroplast large subunit (23S)-rDNA sequences. Mol. Phylogenet. Evol. 23, 97–111 (2002).
Sawall, Y., Al-Sofyani, A., Banguera-Hinestroza, E. & Voolstra, C. R. Spatio-temporal analyses of Symbiodinium physiology of the coral Pocillopora verrucosa along large-scale nutrient and temperature gradients in the Red Sea. PloS one 9, e103179 (2014).
Le Corre, P. & Tréguer, P. Dosage de l’ammonium dans l’eau de mer: comparaison entre deux méthodes d’analyse automatique. J. Conseil 38, 147–153 (1978).
Hoogenboom, M., Rodolfo-Metalpa, R. & Ferrier-Pagès, C. Co-variation between autotrophy and heterotrophy in the Mediterranean coral Cladocora caespitosa. J. Expl. Biol. 213, 2399–2409 (2010).
Rodolfo-Metalpa, R. et al. Response of zooxanthellae in symbiosis with the Mediterranean corals Cladocora caespitosa and Oculina patagonica to elevated temperatures. Mar. Biol. 150, 45–55 (2006).
Jeffrey, S. & Humphrey, G. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem. Physiol. Pfl. 167, 191–194 (1975).
Smith, P. et al. Measurement of protein using bicinchoninic acid. Anal. Biochem. 150, 76–85 (1985).
Veal, C., Carmi, M., Fine, M. & Hoegh-Guldberg, O. Increasing the accuracy of surface area estimation using single wax dipping of coral fragments. Coral Reefs 29, 893–897 (2010).
Godinot, C., Grover, R., Allemand, D. & Ferrier-Pagès, C. High phosphate uptake requirements of the scleractinian coral Stylophora pistillata. J. Exp. Biol. 214, 2749–2754, doi: 10.1242/jeb.054239 (2011).
Holmes, R. M., Aminot, A., Kérouel, R., Hooker, B. A. & Peterson, B. J. A simple and precise method for measuring ammonium in marine and freshwater ecosystems. Can. J. Fish. Aquat. Sci. 56, 1801–1808 (1999).
Boyer, S., White, J., Stier, A. & Osenberg, C. Effects of the fish anesthetic, clove oil (eugenol), on coral health and growth. J. Exp. Mar. Biol. Ecol. 369, 53–57 (2009).
Frisch, A. J., Ulstrup, K. E. & Hobbs, J.-P. A. The effects of clove oil on coral: an experimental evaluation using Pocillopora damicornis (Linnaeus). J. Exp. Mar. Biol. Ecol. 345, 101–109 (2007).
Tremblay, P., Grover, R., Maguer, J. F., Legendre, L. & Ferrier-Pagès, C. Autotrophic carbon budget in coral tissue: a new 13C-based model of photosynthate translocation. J. Exp. Biol. 215, 1384–1393 (2012).
Anthony, K. R. & Fabricius, K. E. Shifting roles of heterotrophy and autotrophy in coral energetics under varying turbidity. J. Exp. Mar. Biol. Ecol. 252, 221–253, doi: 10.1016/S0022-0981(00)00237-9 (2000).
Ralph, P. J. & Gademann, R. Rapid light curves: a powerful tool to assess photosynthetic activity. Aquat. Bot. 82, 222–237 (2005).
Davies, P. S. Short-term growth measurements of corals using an accurate buoyant weighing technique. Mar. Biol. 101, 389–395 (1989).
Acknowledgements
We would like to thank C. Rottier, A. Labbe and D. Desgre for laboratory assistance and technical help during the experimental setup as well as Robin Cristofari, Jeroen van de Water, Jérôme Durivault, Didier Zoccola, Nathalie Le Roy and Rémy Melkonian for their advices throughout the experiment. Many thanks to Pr. Denis Allemand, Director of the Centre Scientifique de Monaco for scientific support. Finally, we are grateful to four anonymous reviewers as well as the Editorial board for their constructive and helpful comments on the manuscript. This project was supported by the CSM and the Institut Européen de la mer. Financial support to L.E. was provided by the Centre Scientifique de Monaco.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: C.F.P., L.E. and R.G. Performed the experiments: L.E. and J.F.M. Analysed the data: L.E. and C.F.P. Contributed reagents/materials/analysis tools: C.F.P., R.G. and J.F.M. Wrote the paper: L.E., C.F.P. and R.G.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ezzat, L., Maguer, JF., Grover, R. et al. Limited phosphorus availability is the Achilles heel of tropical reef corals in a warming ocean. Sci Rep 6, 31768 (2016). https://doi.org/10.1038/srep31768
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep31768
This article is cited by
-
Meta-organism gene expression reveals that the impact of nitrate enrichment on coral larvae is mediated by their associated Symbiodiniaceae and prokaryotic assemblages
Microbiome (2023)
-
Spatiotemporal variations and impact factors of nutrients in the Sanya Bay, northern South China Sea
Environmental Science and Pollution Research (2023)
-
Alkaline phosphatase activity in an equatorial coral reef ecosystem unrelated to seasonal nutrient dynamics
Coral Reefs (2023)
-
The widely distributed soft coral Xenia umbellata exhibits high resistance against phosphate enrichment and temperature increase
Scientific Reports (2022)
-
Species-specific elementomes for scleractinian coral hosts and their associated Symbiodiniaceae
Coral Reefs (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.