Abstract
Adiponectin regulates glucose and lipid metabolism, acting against metabolic syndrome and atherosclerosis. Accumulating evidence suggest that adiponectin acts on the brain including hypothalamic arcuate nucleus (ARC), where proopiomelanocortin (POMC) neurons play key roles in feeding regulation. Several studies have examined intracerebroventricular (ICV) injection of adiponectin and reported opposite effects, increase or decrease of food intake. These reports used different nutritional states. The present study aimed to clarify whether adiponectin exerts distinct effects on food intake and ARC POMC neurons depending on the glucose concentration. Adiponectin was ICV injected with or without glucose for feeding experiments and administered to ARC slices with high or low glucose for patch clamp experiments. We found that adiponectin at high glucose inhibited POMC neurons and increased food intake while at low glucose it exerted opposite effects. The results demonstrate that glucose level determines excitatory or inhibitory effects of adiponectin on arcuate POMC neuron activity and feeding.
Similar content being viewed by others
Introduction
Adipokines are the adipose tissue-derived hormones. Among them, leptin plays an important role in regulating energy metabolism and body weight. Adiponectin, another major adipokine, regulates glucose and lipid metabolism and acts against metabolic syndrome and atherosclerosis1,2,3. Accumulating evidence also suggest that adiponectin acts on the central nervous system (CNS). Peripheral adiponectin could penetrate the cerebrospinal fluid (CSF) from circulation4. The adiponectin receptors, AdipoR1/2, are located in the brain including hypothalamus4,5. Adiponectin regulates neuronal activity in the paraventricular nucleus (PVN) of the hypothalamus and the nucleus of solitary tract of the brain stem6,7,8. Intracerebroventricular (ICV) injection of adiponectin has been shown to regulate food intake4,9, energy metabolism4,10, glucose metabolism11,12, bone metabolism4,9 and circulation systems13 and to induce phosphorylation of AMP-activated protein kinase (AMPK) pathway4, insulin receptor substrate (IRS)1/2–Akt–forkhead box protein O1 (FOXO1) pathway and janus activating kinase 2 (JAK2)-signal transducer and activator of transcription 3 (STAT3) pathway9 via AdipoR1 in the mediobasal hypothalamus.
The proopiomelanocortin (POMC)-expressing neurons in the arcuate nucleus (ARC) are activated by hormonal and nutritional satiety signals such as leptin and glucose, which reflect systemic energy states14,15,16. POMC neurons, once activated, release α-melanocyte stimulating hormone, which interacts with melanocortin receptor 3/4, leading to suppression of food intake and promotion of energy expenditure17,18. AdipoR1/2, the adiponectin receptors, are located in the ARC POMC neurons4,5. These findings raise an important question whether adiponectin could regulate ARC POMC neurons.
The effect of ICV injection of adiponectin on food intake has been investigated. In one report, ICV injection of adiponectin increased food intake for 6 hr following 3 hr refeeding condition4. In another report, ICV adiponectin injection at late light phase decreased food intake during dark phase9. Thus, apparently opposite effects of ICV adiponectin on food intake have been observed. These reports used different timing of adiponectin injection, either before or during eating, which is associated with different metabolic states. Feeding-associated changes in nutritional and hormonal states are known to influence brain functions.
In the present study, we hypothesized that adiponectin could exert distinct effects on the central feeding center including ARC POMC neurons depending on nutritional and hormonal states. Among them, we focused on the glucose concentration, which regulates activities of both glucose-responsive and glucose-inhibited neurons in the brain including ARC19,20,21. Adiponectin was injected ICV with or without glucose to mimic fed or fasted states, respectively and examined the effect on food intake and POMC neuron activity in mice. We found that ICV injection of adiponectin without or with glucose exerted reciprocal effects on the POMC neuron activity and feeding.
Results
ICV adiponectin injection glucose-dependently reciprocally regulates food intake
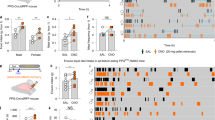
Previous studies used different experimental conditions, including animal species and timing of adiponectin injection4,9. In the present study, mouse hexametric or middle molecular weight (MMW, 180 kDa) adiponectin (150 ng) or saline was ICV injected at the end of light phase (19:30) after fasting for 3 hr (Fig. 1A), following the procedure reported by Coope et al.9. This condition of 3 hr fasting at late light phase is considered to produce a moderate appetite, though it is milder than the intense starvation produced by overnight or 24 hr fasting. On the other hand, to produce a condition with considerable satiety, the same time course and food-deprived procedure were used but glucose (100 μg) was ICV injected together with adiponectin (Fig. 1A).
Central adiponectin suppressed or promoted food intake in glucose dependent manner.
(A) Protocol for food intake measurement after ICV injection of adiponectin and/or glucose. (B) Cumulative food intake after ICV injection of saline or adiponectin with or without glucose at the end of light phase following 3 hr fasting (n = 6~12).*p < 0.05 (unpaired t-test).
Glucose injection decreased food intake (Fig. 1B). ICV injection of adiponectin together with glucose significantly increased cumulative food intake at 6 hr after injection (glucose: 1.61 ± 0.06 g, glucose + adiponectin: 2.25 ± 0.10 g, p < 0.05, Fig. 1B). This result indicated that adiponectin increased food intake in the condition with elevated glucose in CNS.
After 3 hr fasting, a condition considered to lower the glucose concentration in the brain, single injection of adiponectin, compared to saline injection, significantly (p < 0.05) decreased food intake at 2, 3 and 6 hr after injection (Fig. 1B). This result demonstrated that adiponectin decreased food intake in the condition with low glucose in CNS, in consistent with previous study in rats9. This also indicated that species difference is not observed in the adiponectin effect on food intake.
Adiponectin at high glucose hyperpolarizes hypothalamic ARC POMC neurons via AMPK
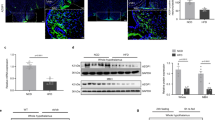
To investigate whether adiponectin regulates the activity of POMC neurons in ARC, membrane potentials and action potentials of ARC POMC neurons in the acute hypothalamic slice isolated from POMC-hrGFP transgenic mice were recorded by patch clamp experiments under whole-cell current clamp mode. In the high glucose condition, artificial CSF (aCSF) contained 10 mM glucose. Glucose at this concentration inhibits glucose-inhibited neurons implicated in stimulation of food intake20,22 and activates glucose-responsive neurons implicated in inhibition of food intake23. Bath application of hexametric form adiponectin (0.56 nM) for 4 min hyperpolarized the membrane potential from −48.0 ± 1.9 mV to −55.4 ± 3.2 mV (Fig. 2A,C) in nine of 12 POMC neurons (75%) and significantly decreased firing rate of action potential (Fig. 2A,D). The hyperpolarization continued to be observed at least for 20 min after washout of adiponectin. In the presence of 1 μM tetrodotoxin (TTX) that prevents presynaptic action potentials, five of seven POMC neurons (70%) were hyperpolarized from −53.7 ± 4.1 mV to −60.6 ± 2.7 mV (Fig. 2B,E) by adiponectin. Thus, TTX altered neither the incidence nor the amplitude of adiponectin-induced hyperpolarization in POMC neurons, indicating that the hyperpolarization was not mediated by altered neuronal transmission onto POMC neurons. These results indicate that, in the presence of high (10 mM) glucose, adiponectin directly interacts with POMC neurons to suppress their electrical activity.
Adiponectin hyperpolarized POMC neurons via AMPK under high glucose condition.
(A,B) Representative trace of current clamp recording with 10 mM glucose in aCSF in the absence (A) or presence (B) of TTX. (C,D) Membrane potential (C, n = 9) and action potential firing rate normalized by baseline (D, n = 9) before, during and after application of 0.56 nM adiponectin. (E) Membrane potential before, during and after application of adiponectin in the presence of 1 μM TTX. (F) Representative trace of current clamp recording with 10 mM glucose and 30 μM Compound C in aCSF. (G,H) Membrane potential (G, n = 6) and action potential firing rate normalized by baseline (H, n = 5) before, during and after administration of adiponectin in the presence of Compound C.
We examined possible involvement of AMPK in the downstream signal pathway of adiponectin in POMC neurons. Since adiponectin reportedly regulates AMPK via AdipoR1 in peripheral organs24,25,26,27 and also in mediobasal hypothalamus including ARC4, whole-cell current clamp was performed with AMPK inhibitor, Compound C (30 μM). Adiponectin application induced hyperpolarization in none of eight POMC neurons (0%) (baseline: −54.5 ± 2.9 mV, adiponectin: −51.9 ± 3.1 mV, Fig. 2F,G). These results indicate that adiponectin under 10 mM glucose condition inhibits POMC neurons via a mechanism involving AMPK. These results suggest that adiponectin at high glucose may inactivate POMC neurons and increase food intake. These results are consistent with previous report that ICV adiponectin injected under conditions with mild satiety increases food intake.
Adiponectin at low glucose depolarizes hypothalamic ARC POMC neurons via phosphoinositide 3-kinase (PI3K)
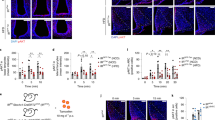
Current clamp recording was performed with lower glucose concentration of 5 mM in aCSF. Surprisingly, adiponectin application depolarized eight of 12 POMC neurons (67%) from −56.1 ± 2.9 mV to −49.2 ± 3.7 mV (Fig. 3A,C), while none of neurons were hyperpolarized. Action potential firing rate tended to increase in response to adiponectin (p = 0.08) (Fig. 3A expanded scale and 3D). In the presence of TTX, five of seven POMC neurons (71%) were depolarized from −55.3 ± 3.8 mV to −49.6 ± 5.1 mV (Fig. 3B,E). The incidence and amplitude of adiponectin-induced depolarization in POMC neurons were similar without and with TTX (Fig. 3C vs. 3E), indicating that the depolarization was not mediated by neuronal transmission onto POMC neurons. In the presence of 2.5 mM glucose in aCSF, adiponectin depolarized seven of 12 POMC neurons (58%) from −50.4 ± 2.5 mV to −40.9 ± 2.6 mV (Supplemental Fig. 1). Thus, similar results were obtained with 5 and 2.5 mM glucose. These data indicate that adiponectin directly interacts with POMC neurons to increase their electrical activity at low glucose, being consistent with the report by Coope et al.9 When POMC neurons were pretreated with PI3K inhibitor LY200492 (50 μM) for 1 hr, adiponectin failed to depolarize them and, instead, rather induced mild hyperpolarization (Fig. 3F) and reduced firing rates in some POMC neurons (Fig. 3F,H). This result is in consistent with previous report that adiponectin stimulates IRS1/2–Akt–FOXO1 pathway in mediobasal hypothalamus including ARC9. These results suggest that adiponectin depolarizes POMC neurons via a mechanism involving PI3K pathway.
Adiponectin depolarized POMC neurons via PI3K pathway under lower glucose condition.
(A,B), Representative trace of current clamp recording with 5 mM glucose in aCSF in the absence (A) or presence (B) of 1 μM TTX. (C,D), Membrane potential (C, n = 8) and action potential firing rate normalized by baseline (D, n = 5) before, during and after administration of 0.56 nM adiponectin. (E) Membrane potential before, during and after administration of adiponectin in the presence of TTX (n = 5). (F) Representative trace of current clamp recording with 10 mM glucose in aCSF with the pretreatment of 50 μM LY200492, PI3K inhibitor. (G,H) Membrane potential (G, n = 8) and action potential firing rate normalized by baseline (H, n = 4)) before, during and after administration of adiponectin with the pretreatment of LY200492.
Discussion
We have demonstrated that ICV injection of adiponectin increases food intake when the glucose level in the brain is high, whereas it suppresses food intake when the glucose level in the brain is low. In parallel, adiponectin decreases the POMC neuron activity at high glucose (10 mM), whereas it increases the POMC neuron activity at low glucose (2.5~5 mM). Thus, adiponectin alters the ARC POMC neuron activity and food intake toward opposite directions depending on the energy state or glucose concentration in the brain. These results reveal level determines excitatory or inhibitory effects of adiponectin on the ARC POMC neuron activity and feeding. Furthermore, the current data suggest that adiponectin inhibits POMC neurons at high glucose via signaling involving AMPK (Fig. 2), while it activates POMC neurons at low glucose via signaling involving PI3K (Fig. 3). These signaling pathways may serve to couple adiponectin receptor to POMC neuron activity. The energy state- or glucose-dependent dual effects of adiponectin on POMC neurons with distinct signaling cascades may solve the apparent discrepancy in previous reports conducted under different nutritional states4,9.
Our data suggest a possible link of the adiponectin reception to AMPK and PI3K signaling in ARC neurons. This is in agreement with earlier studies. AdipoR1-derived signaling pathway includes AMPK in ARC neurons as well as in peripheral organs such as the liver and skeletal muscle24,25,26,27. Activation of AMPK hyperpolarizes POMC neurons28. Under conditions with low CSF glucose in the late light phase or after moderate fasting, ICV adiponectin injection phosphorylates IRS2-PI3K-Akt pathway in the mediobasal hypothalamus in rats9 and Lepob/ob mice11. Activation of PI3K-Akt pathway by leptin29 and insulin30,31 depolarizes POMC neurons via TRPC32,33. The leptin activation is blunted in POMC neurons deleted PDK1, a signaling molecule downstream of PI3K-Akt34,35.
In the presence of AMPK inhibitor, adiponectin at high glucose failed to hyperpolarize but tended to depolarize POMC neurons, showing the reversal of the adiponectin effect (Fig. 2G,H). Conversely, after pretreatment with PI3K inhibitor, adiponectin at low glucose failed to depolarize but moderately hyperpolarized POMC neurons, showing a reversed effect (Fig. 3G,H). These results suggest that adiponectin simultaneously induces two signaling pathways that function toward opposite directions in POMC neurons. It is suggested that at high glucose, adiponectin activation of PI3K pathway is less influential than that of AMPK pathway but becomes obvious when AMPK pathway is inhibited. Likewise, at low glucose, adiponectin activation of AMPK pathway is less influential than that of PI3K pathway but becomes obvious when AMPK pathway is inhibited. Additionally, previous studies suggest that AMPK and PI3K pathways may act competitively or reciprocally. Constitutive active form of AMPK suppressed the leptin pathway involving PI3K36. AMPK binds and phosphorylates 794Ser of IRS1, which may inhibit PI3K pathway37. Furthermore, AMPK phosphorylate TSC2 and Raptor which are suppressor of mTORC138,39. On the other hand, Akt or mTOR-S6 kinase, downstream target of PI3K, phosphorylate 485Ser and 491Ser of α1AMPK, possibly suppressing AMPK activity40,41,42. These reports support that glucose-induced suppression of AMPK could activate PI3K pathway. Based on our present finding together with these previous reports, we propose that high vs. low glucose shifts the dominant signaling pathway between AMPK and PI3K and that the yet poorly stimulated signaling pathway under each glucose concentration could be preferentially and fully activated by adiponectin, which results in glucose-dependent reciprocal effects of adiponectin on POMC neurons and food intake (Fig. 4). Under high glucose condition, PI3K in POMC neuron is already activated by high glucose, insulin and/or leptin and adiponectin cannot influence it further but mainly activates AMPK pathway to hyperpolarize POMC neurons. Under low glucose condition, AMPK in POMC neuron is already phosphorylated by low glucose and associated increase of AMP/ATP ratio and adiponectin cannot stimulate it further but chiefly activates PI3K pathway to depolarize POMC neurons. Based on these findings, we propose that adiponectin moderately counteracts with both high glucose and low glucose, serving as an attenuator of profound effect of glucose change on POMC neuron activity and feeding.
Hypothetical model of central adiponectin effect on POMC neurons and food intake.
Under higher glucose condition, high level of ATP suppresses AMPK pathway, which leads to promotion of PI3K pathway. Adiponectin stimulate AMPK, but not PI3K pathway that is already activated. Activation of AMPK hyperpolarizes POMC neurons and thereby promotes food intake. Under lower glucose condition, high level of AMP activate AMPK pathway and inactivate PI3K pathway. Adiponectin stimulate PI3K pathway, but not AMPK pathway which is already activated. Activation of PI3K pathway depolarizes POMC neurons and thereby suppresses food intake. Red arrow/line indicates adiponectin-stimulated pathway.
In current clamp recordings, recovery from adiponectin effect was not obtained till 15~20 min after its washout. The reason may be the sticky nature of adiponectin due to its collagen domain43. Therefore, we could not test whether adiponectin glucose-dependently depolarize and/or hyperpolarize the same or distinct subpopulations of POMC neurons. However, the experiments with AMPK inhibitor or PI3K inhibitor showed that adiponectin can simultaneously induce dual effects on POMC neurons toward opposite directions (Figs 2G,H and 3G,H). This result suggest that adiponectin can stimulate both AMPK pathway and PI3K pathway on the same POMC neuron subpopulation and that the final effect is determined by the glucose-dependent balance between hyperpolarization partly medicated by AMPK and depolarization partly medicated by PI3K.
In the present study, ICV adiponectin co-injected with glucose did not significantly increase food intake in early phase (0~3 hr), while adiponectin rapidly inhibited the electrical activity of POMC neurons by current clamp recording. The mechanism underlying this apparent dissociation remains to be elucidated. However, we speculate that in in vivo experiment, since adiponectin and glucose were injected simultaneously, it may require time for adiponectin’s orexigenic effect to overcome the glucose-induced anorexigenic effect. On the other hand, in vitro electrical recording was performed in POMC neurons that had been exposed to 10 mM glucose for more than 1.5 hr, in which no acute effect of glucose was present and hence the adiponectin administration could rapidly activate AMPK and hyperpolarize POMC neurons.
In the present study, we found glucose- or energy state-dependent opposing effects of adiponectin on POMC neuron activity and feeding. The physiological significance of this reciprocal action of adiponectin remains to be clarified. However, we can speculate its possible role from the point of adaptation and evolution. In the basal energy state, adiponectin suppresses food intake. However, once food is available, adiponectin stimulates food intake. Under wild circumstance with restricted food, it has been essential for living species to store more energy. This may be the reason why adiponectin stimulates food intake in positive energy states. Thus, adiponectin may have contributed to the adaptation and survival of living species, as well as regulation of metabolism. In fact, adiponectin and their receptor homolog have been highly conserved from yeast to human44,45.
Methods
Food intake measurement
Male C57black6/J mice aged 8–10 weeks were maintained in a 12/12 hrs light/dark cycle. For ICV injection in mice, a gauge guide cannula (type; OM205-113, diameter; 0.2 mm; Unique medical, Tokyo, Japan) was placed stereotaxically into 3V, at 1.5 mm caudal to the bregma in the midline and 5 mm below the surface of the skull, under anesthesia with tribromoethanol (200 mg/kg). Mouse was allowed to recover from the operation for 1 week while they were habituated to handling. On the day of experiments, food was removed form cages at 16:00. At 19:30, mouse received an injection of 150 μg mouse purified MMW-adiponectin46 without or with 100 μg glucose dissolved in vehicle (sterile saline; 0.9% NaCl). The food was returned to cages and food intake at 0.5, 1, 2, 3, 6, 16 hr were measured. At the end of the experiments, sections of the hypothalamus were histologically examined to verify the position of the cannulas. The animal protocols for this study were approved by Jichi Medical University Institute of Animal Care and Use Committee. and all experiments were carried out in accordance with the approved protocols.
Acute slice preparation
POMC-hrGFP transgenic mice aged 7–10 weeks were maintained in a 12/12 h light/dark cycle. Their brains were then removed rapidly from mouse anesthetized with tribromoethanol (200 mg/kg) and placed in an ice-cold, carboxygenated (95% O2 and 5% CO2) high mannitol solution that contained (in mM) 229 mannitol, 3 KCl, 6 MgCl2, 0.5 CaCl2, 1 NaH2PO4, 26 NaHCO3 and 10 glucose, pH7.4 (with an osmolarity of 300–305 mOsm) with 0.5 μM tetrodotoxin. A block of tissue containing the hypothalamus was isolated and coronal slices (300 μm) were cut on a Vibratome. After a 1–2 hrs recovery period, slices were moved to a recording chamber mounted on a BX51WI upright microscope (Olympus) equipped with video-enhanced infrared-differential interference contrast (DIC) and fluorescence. Slices were perfused with a continuous flow of carboxygenated aCSF that contained (in mM) 127 NaCl, 2.5 KCl, 2 MgCl2, 2 CaCl2, 1.23 NaH2PO4, 26 NaHCO3 and 2.5–10 glucose, pH 7.4 Neurons were visualized with an Olympus Optical 40x water-immersion lens.
Patch-clamp recording
Whole-cell current-clamp recordings were performed as previously reported47 Briefly, pipettes were used with 3–9 MΩ resistance after being filled with pipette solution. Pipettes were made of borosilicate glass (Narishige) using a PP-83 vertical puller (Narishige) or a Sutter micropipette puller (P-1000). The pipettes were used with 3–9 MΩ resistance after being filled with pipette solution. The composition of the pipette solution was as follows (in mM): 135 K-gluconate, MgCl2 2, HEPES 10, EGTA 1.1, Mg-ATP 2.5, Na2-GTP 0.3 and Na2-phosphocreatine 10, pH 7.3 with KOH (with an osmolarity of 290–295 mOsm). An Axopatch 200B amplifier and Clampex 9.2 or 10 software (Axon Instruments) were used for data acquisition. Pclamp 9.2 or 10 (Axon Instruments) software was used for analysis. Liquid junction potential correction was performed off-line. Access resistance was continuously monitored during the experiments. Only those cells in which access resistance was stable (changes ~30%) were included in the analysis. The data was analyzed by Clamp fit 9.2 or 10 (Axon instruments) software and GraphPad Prism6 software. The response by adiponectin was defined as a change over 2 times standard deviation of membrane potential for 2 min before applying adiponectin.
Statistical Analysis
One-way ANOVA followed by Dunnet multiple range tests were used to compare multiple test groups and unpaired Student’s t tests were used for two groups.
Additional Information
How to cite this article: Suyama, S. et al. Glucose level determines excitatory or inhibitory effects of adiponectin on arcuate POMC neuron activity and feeding. Sci. Rep. 6, 30796; doi: 10.1038/srep30796 (2016).
References
Kadowaki, T. & Yamauchi, T. Adiponectin and adiponectin receptors. Endocr Rev 26, 439–451 (2005).
Yamauchi, T. & Kadowaki, T. Physiological and pathophysiological roles of adiponectin and adiponectin receptors in the integrated regulation of metabolic and cardiovascular diseases. Int J Obes (Lond) 32 Suppl 7, S13–S18 (2008).
Okamoto, Y. et al. An adipocyte-derived plasma protein, adiponectin, adheres to injured vascular walls. Horm. Metab. Res. 32, 47–50 (2000).
Kubota, N. et al. Adiponectin stimulates AMP-activated protein kinase in the hypothalamus and increases food intake. Cell Metab 6, 55–68 (2007).
Guillod-Maximin, E. et al. Adiponectin receptors are expressed in hypothalamus and colocalized with proopiomelanocortin and neuropeptide Y in rodent arcuate neurons. J Endocrinol 200, 93–105 (2009).
Hoyda, T. D., Fry, M., Ahima, R. S. & Ferguson, A. V. Adiponectin selectively inhibits oxytocin neurons of the paraventricular nucleus of the hypothalamus. The Journal of Physiology 585, 805–816 (2007).
Hoyda, T., Samson, W. & Ferguson, A. Adiponectin depolarizes parvocellular paraventricular nucleus neurons controlling neuroendocrine and autonomic function. Endocrinology 150, 832 (2009).
Hoyda, T. D. & Ferguson, A. V. Adiponectin modulates excitability of rat paraventricular nucleus neurons by differential modulation of potassium currents. Endocrinology 151, 3154–3162 (2010).
Coope, A. et al. AdipoR1 mediates the anorexigenic and insulin/leptin-like actions of adiponectin in the hypothalamus. FEBS Lett 582, 1471–1476 (2008).
Qi, Y. et al. Adiponectin acts in the brain to decrease body weight. Nature Medicine 10, 524–529 (2004).
Koch, C. E. et al. Central adiponectin acutely improves glucose tolerance in male mice. Endocrinology 155, 1806–1816 (2014).
Kajimura, D. et al. Adiponectin regulates bone mass via opposite central and peripheral mechanisms through FoxO1. Cell Metab 17, 901–915 (2013).
Hoyda, T. D., Smith, P. M. & Ferguson, A. V. Adiponectin acts in the nucleus of the solitary tract to decrease blood pressure by modulating the excitability of neuropeptide Y neurons. Brain Research 1256, 76–84 (2009).
Schwartz, M. W., Woods, S. C., Porte, D., Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Gao, Q. & Horvath, T. Neurobiology of feeding and energy expenditure. (2007).
Lee, D. K., Jeong, J. H., Chun, S. K., Chua, S., Jr. & Jo, Y.-H. Interplay between glucose and leptin signalling determines the strength of GABAergic synapses at POMC neurons. Nat Commun 6, 6618 (2015).
Gao, Q. & Horvath, T. L. Neuronal control of energy homeostasis. FEBS Lett 582, 132–141 (2008).
Plum, L. et al. The obesity susceptibility gene Cpe links FoxO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. Nature Medicine 15, 1195–1201 (2009).
Fioramonti, X. et al. Characterization of glucosensing neuron subpopulations in the arcuate nucleus: integration in neuropeptide Y and pro-opio melanocortin networks? Diabetes 56, 1219–1227 (2007).
Kurita, H. et al. Arcuate Na+,K+-ATPase senses systemic energy states and regulates feeding behavior through glucose-inhibited neurons. Am J Physiol Endocrinol Metab 309, E320–E333 (2015).
Routh, V. H., Hao, L., Santiago, A. M., Sheng, Z. & & Zhou, C. Hypothalamic glucose sensing: making ends meet. Front Syst Neurosci 8, 236 (2014).
Muroya, S., Yada, T., Shioda, S. & Takigawa, M. Glucose-sensitive neurons in the rat arcuate nucleus contain neuropeptide Y. Neuroscience letters 264, 113–116 (1999).
Muroya, S. et al. Orexins (hypocretins) directly interact with neuropeptide Y, POMC and glucose-responsive neurons to regulate Ca 2 signaling in a reciprocal manner to leptin: orexigenic neuronal pathways in the mediobasal hypothalamus. Eur J Neurosci 19, 1524–1534 (2004).
Yamauchi, T., Iwabu, M., Okada-Iwabu, M. & Kadowaki, T. Adiponectin receptors: a review of their structure, function and how they work. Best Pract. Res. Clin. Endocrinol. Metab. 28, 15–23 (2014).
Iwabu, M. et al. Adiponectin and AdipoR1 regulate PGC-1alpha and mitochondria by Ca(2+) and AMPK/SIRT1. Nature 464, 1313–1319 (2010).
Yamauchi, T. et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nature Medicine 13, 332–339 (2007).
Kahn, B. B., Alquier, T., Carling, D. & Hardie, D. G. AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab 1, 15–25 (2005).
Claret, M. et al. AMPK is essential for energy homeostasis regulation and glucose sensing by POMC and AgRP neurons. J Clin Invest 117, 2325–2336 (2007).
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature. Nature 411, 480–484 (2001).
Hill, J. et al. Acute effects of leptin require PI3K signaling in hypothalamic proopiomelanocortin neurons in mice. J Clin Invest 118, 1796 (2008).
Al-Qassab, H. et al. Dominant role of the p110beta isoform of PI3K over p110alpha in energy homeostasis regulation by POMC and AgRP neurons. Cell Metab 10, 343–354 (2009).
Qiu, J., Fang, Y., Rønnekleiv, O. K. & Kelly, M. J. Leptin excites proopiomelanocortin neurons via activation of TRPC channels. J Neurosci 30, 1560–1565 (2010).
Qiu, J. et al. Insulin excites anorexigenic proopiomelanocortin neurons via activation of canonical transient receptor potential channels. Cell Metab 19, 682–693 (2014).
Iskandar, K. et al. PDK-1/FoxO1 pathway in POMC neurons regulates Pomc expression and food intake. Am J Physiol Endocrinol Metab 298, E787–E798 (2010).
Belgardt, B. F. et al. PDK1 deficiency in POMC-expressing cells reveals FOXO1-dependent and -independent pathways in control of energy homeostasis and stress response. Cell Metab 7, 291–301 (2008).
AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. 428, 569–574 (2004).
Tzatsos, A. & Tsichlis, P. N. Energy depletion inhibits phosphatidylinositol 3-kinase/Akt signaling and induces apoptosis via AMP-activated protein kinase-dependent phosphorylation of IRS-1 at Ser-794. J Biol Chem 282, 18069–18082 (2007).
Inoki, K., Zhu, T. & Guan, K.-L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115, 577–590 (2003).
Gwinn, D. M. et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 30, 214–226 (2008).
Horman, S. et al. Insulin antagonizes ischemia-induced Thr172 phosphorylation of AMP-activated protein kinase alpha-subunits in heart via hierarchical phosphorylation of Ser485/491. J Biol Chem 281, 5335–5340 (2006).
Hardie, D. G. & Ashford, M. L. J. AMPK: Regulating Energy Balance at the Cellular and Whole Body Levels. Physiology 29, 99–107 (2014).
Dagon, Y. et al. p70S6 kinase phosphorylates AMPK on serine 491 to mediate leptin’s effect on food intake. Cell Metab 16, 104–112 (2012).
Maeda, K. et al. cDNA cloning and expression of a novel adipose specific collagen-like factor, apM1 (AdiPose Most abundant Gene transcript 1). Biochem Biophys Res Commun 221, 286–289 (1996).
Narasimhan, M. L. et al. Osmotin is a homolog of mammalian adiponectin and controls apoptosis in yeast through a homolog of mammalian adiponectin receptor. Mol. Cell 17, 171–180 (2005).
Karpichev, I. V., Cornivelli, L. & Small, G. M. Multiple regulatory roles of a novel Saccharomyces cerevisiae protein, encoded by YOL002c, in lipid and phosphate metabolism. J Biol Chem 277, 19609–19617 (2002).
Hada, Y. et al. Selective purification and characterization of adiponectin multimer species from human plasma. Biochem Biophys Res Commun 356, 487–493 (2007).
Suyama, S. et al. Fasted/fed states regulate postsynaptic hub protein DYNLL2 and glutamatergic transmission in oxytocin neurons in the hypothalamic paraventricular nucleus. Neuropeptides. 10.1016/j.npep.2015.08.008 (2015).
Acknowledgements
We thank Dr. Joel Elmquist for providing POMC-hrGFP transgenic mice and Dr. Hiroyuki Ebinuma for providing purified mouse MMW-adiponectin. This study was supported by Grant-in-Aid for Scientific Research (B) (23390044) and for Challenging Exploratory Research (24659101, 26670453) from Japan Society for the Promotion of Science (JSPS), Strategic Research Program for Brain Sciences (10036069) by the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT), MEXT-Supported Programs for Strategic Research Foundation at Private Universities (S1101022) 2011–2015 (Cooperative Basic and Clinical Research on Circadian Medicine) and (S1311029) 2013–2017, a grant from Salt Science Research Foundation, No. 1434 and a grant from Lilly-Incretin Basic Research Grant from Japan Diabetes Foundation to TY. This study was subsidized by JKA through its promotion funds from KEIRIN RACE to TY.
Author information
Authors and Affiliations
Contributions
S.S., F.M. and Y.M. performed experiments. S.S. and T.Y. designed study and wrote manuscript. N.K. and T.K. provided adiponectin and contributed to discussion. T.Y. managed the project.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Suyama, S., Maekawa, F., Maejima, Y. et al. Glucose level determines excitatory or inhibitory effects of adiponectin on arcuate POMC neuron activity and feeding. Sci Rep 6, 30796 (2016). https://doi.org/10.1038/srep30796
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep30796
This article is cited by
-
Influence of diet and body weight in treatment-resistant acquired partial lipodystrophy after hematopoietic stem cell transplantation and its potential for metabolic improvement
Diabetology International (2023)
-
Hypothalamic primary cilium: A hub for metabolic homeostasis
Experimental & Molecular Medicine (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.