Abstract
The calcium-activated potassium channel KCa3.1 controls different cellular processes such as proliferation and volume homeostasis. We investigated the role of KCa3.1 in experimental and human liver fibrosis. KCa3.1 gene expression was investigated in healthy and injured human and rodent liver. Effect of genetic depletion and pharmacological inhibition of KCa3.1 was evaluated in mice during carbon tetrachloride induced hepatic fibrogenesis. Transcription, protein expression and localisation of KCa3.1 was analysed by reverse transcription polymerase chain reaction, Western blot and immunohistochemistry. Hemodynamic effects of KCa3.1 inhibition were investigated in bile duct-ligated and carbon tetrachloride intoxicated rats. In vitro experiments were performed in rat hepatic stellate cells and hepatocytes. KCa3.1 expression was increased in rodent and human liver fibrosis and was predominantly observed in the hepatocytes. Inhibition of KCa3.1 aggravated liver fibrosis during carbon tetrachloride challenge but did not change hemodynamic parameters in portal hypertensive rats. In vitro, KCa3.1 inhibition leads to increased hepatocyte apoptosis and DNA damage, whereas proliferation of hepatic stellate cells was stimulated by KCa3.1 inhibition. Our data identifies KCa3.1 channels as important modulators in hepatocellular homeostasis. In contrast to previous studies in vitro and other tissues this channel appears to be anti-fibrotic and protective during liver injury.
Similar content being viewed by others
Introduction
Hepatic fibrosis is the common response to any form of chronic liver injury and upon progression it undermines liver function and initiates portal hypertension. Activated and myofibroblast-like hepatic stellate cells (HSC), instigated and perpetuated by inflammation secondary to hepatocellular damage, are considered the main cellular source for the extracellular matrix during liver fibrogenesis1. Nevertheless, quiescent HSC were reported to contribute to progenitor cells and liver regeneration2. Hepatocytes show a high regenerative potential upon injury and frequently react with adaption of the cell volume, which is suggested to be controlled by the calcium-activated potassium channel KCa3.13. This channel is activated by Ca2+ binding to constitutively bound calmodulin and the K+-efflux and concomitant water efflux leads to cell volume reduction. In hepatoma cell-lines KCa3.1 activation seems to be crucial for volume restoration after cell swelling caused by hypotonic stress and thereby for hepatocyte volume control3.
Besides its role in cell volume regulation, the KCa3.1 channel modulates other cellular processes such as cell proliferation and endothelium-dependent vasorelaxation4,5,6,7,8 and was shown to be pathomechanistically implicated in renal and pulmonary fibrosis9,10,11,12. Indeed, KCa3.1 deficiency in mice blunted renal fibrosis13 and the KCa3.1 inhibitor TRAM-34 was shown to suppress fibrogenic properties of myofibroblasts in vitro14. Yet, the pathomechanistic roles of KCa3.1 in liver fibrosis especially in experimental studies and in human situation remains unclear, especially in regards to the potential effect of the channel on the high hepatic regenerative potential compared to other parenchymal organs.
Phenotypically, KCa3.1 deficient mice are characterized by a sizable splenomegaly, which is linked to KCa3.1’s well-established effect on erythrocyte volume regulation15. Furthermore, the mice have a mild arterial hypertension16, are hyperactive and have higher bodyweights than wild type littermates17.
In this study, we hypothesized pathomechanistic roles of KCa3.1 in liver injury and fibrosis and tested whether genetic deficiency or pharmacological blockade alters fibrotic remodelling and expression of profibrotic mediators in experimental rodent models.
Materials and Methods
Human liver specimen
Liver biopsies were performed using the Menghini method (17G Menghini needle, Hepafix, Braun, Germany). Biopsies were collected from 54 patients at the Department of Medical Gastroenterology, Odense University Hospital. We included patients, who for clinical reasons were scheduled to undergo liver biopsy and used the excess tissue for further analyses. Oral and written informed consent was given prior to biopsy and all experiments were performed in accordance with the approved guidelines and regulations. The study was approved by The Regional Scientific Ethical Committees for Southern Denmark (case number S-20110115) and was in agreement with the 1975 Declaration of Helsinki.
Samples were immediately stored in 4% formalin and sent to routine histological staining and evaluation. METAVIR score was evaluated blinded by a hepatopathologist and performed on all biopsies. Patients were classified into groups based on fibrosis score; non-fibrotic (n = 12), F1–F2 fibrosis (n = 20) and F3–F4 fibrosis (n = 22) (Table 1).
Animal studies
All animals within this study received human care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals. Animals were housed in a room with controlled temperature and humidity in a 12-hour light cycle. KCa3−/− mice, also known as Kcnn4 deficient mice, were obtained from own breeding colonies5. The genetic background for the KCa3.1 deficient mice is C57BLJ/6 × 129 Ola. KCa3.1 knockout mice were bred by homozygote breeding and KCa3.1 deficiency was furthermore verified by quantitative polymerase chain reaction (qPCR). We used KCa3.1+/+ or KCa3.1+/− animals as wt controls. Experimental protocols were approved by the Danish Animal Experience Inspectorate (2012-15.2934-00264).
For rat studies we used Sprague-Dawley rats. The responsible committee for animal studies in North Rhine-Westphalia approved this part of the study (LANUV 8.87-50.10.31.08.28).
Mouse models of fibrosis
We used KCa3.1−/− mice and corresponding wt littermates at age 12–16 weeks. The mice were challenged with carbon tetrachloride (CCl4) (1 ml/kg i.p.) twice weekly. Animals were grouped according to a CCl4 exposure time of 8 weeks to induce fibrosis18 and 12 weeks to induce advanced fibrosis or cirrhosis19. For each exposure group we included 8–10 wt and 8–10 KCa3.1 knockout mice. Age and sex matched untreated wt and KCa3.1−/− mice (n = 10 wt, 10 KCa3.1−/−) served as controls.
Rat models of fibrosis
For CCl4 intoxication in rats we exposed rats (n = 4) with an initial body weight of 100–120 g to CCl4 inhalation 1 l/min for 14–16 week as described previously20,21. For thioacetamide (TAA) induced injury we administered TTA in 200–250 g rats (n = 4) in their drinking water for 18 weeks as previously described22.
Bile duct ligation (BDL) and transection was performed under anaesthesia with ketamine/xylazine as previously described20,21. Hemodynamic experiments were conducted four weeks after BDL surgery in 3 rats.
Treatment with the KCa3.1-blocker Senicapoc
For pharmacological studies, wt mice were used. Animals were exposed to CCl4 for 12 weeks and allocated to receive either normal chow (placebo) (n = 10) or the oral KCa3.1 antagonist Senicapoc (n = 10). Senicapoc (ICA-17043) was synthesized as previously described23 and animals received a dosage of 30 mg/kg/day mixed in the chow. Senicapoc was administered in the fodder and initiated 48 hours prior to the first CCl4 injection. Food intake was measured weekly until termination. Prior to termination, animals were anaesthetised with fentanyl/fluanisone and midazolam, blood was collected through penetration of the retro-orbital sinus and plasma was separated by centrifuge and stored at −80 degrees Celsius until analysed. The concentration of Senicapoc was measured by mass spectrometry (TSQ Quantum Access MAX, ThermoFisher Scientific, Waltham, MA, USA).
Hemodynamic studies
In vivo measurements of portal pressure (PP), mean arterial pressure (MAP) and heart rate (HR) were performed in BDL (n = 3) and CCl4 exposed (n = 3) rats during anaesthesia with ketaminen/xylazine (78 and 12.5 mg/kg i.m. respectively) as previously described20,21,24,25. Briefly, following midline laparotomy, a PE-50 catheter was inserted into the ileocecal vein and progressed to the portal vein. This was followed by insertion of a similar catheter in the femoral artery for MAP measurement. Both catheters were connected to a pressure transducer (Hugo Sachs Electronic, March-Hugstetten, Germany). Hemodynamic stabilization was awaited before proceeding and subsequently 100 nM of the KCa3.1 antagonist TRAM-34 was injected as bolus. Continuous monitoring of HR, MAP and PP was performed for the following 20 minutes.
Quantitative RT-PCR
For murine samples RNA was isolated using TRIZOL reagent (Invitrogen, United Kingdom) and DNAse digestion using RNase free DNase 1 (Thermo Scientific, CA, USA) according to manufactures instructions. cDNA was synthesized with iScript cDNA Synthesis Kit (Bio-Rad, CA, USA). qRT-PCR was performed using SYBR Green Supermix (Bio-Rad, CA, USA) on the Stratagene MX3000 qPCR instrument (Agilent Technologies, CA, USA). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as endogenous control.
For gene expression analyses in rat and cell studies, RNA isolation, reverse transcription and detection by RT-PCR were performed by TaqMan method as described previously20,21,26. 18S rRNA served as endogenous control.
Results are expressed as 2−ΔΔCt and express the x-fold increase of gene expression compared to the control group and in murine studies results were compared to untreated wt.
Western blotting
Western blot was performed on samples from untreated and fibrotic wt and KCa3.1−/− mice (12 weeks CCl4 exposure) for Proliferating Cell Nuclear Antigen (PCNA; Santa Cruz-Biotechnology, Santa Cruz, USA). Proto-oncogene tyrosine-protein kinase Src (c-Src) activity was analysed by phosphorylation either at Thr418 or at Thr530 (both Santa Cruz-Biotechnology, Santa Cruz, USA). Activation of c-Jun N-terminal kinases 1 and 2 (pJNK1&2; Life technologies, Darmstadt, Germany) and total JNK (Santa Cruz-Biotechnology, Santa Cruz, USA) was assessed by their phosphorylation at Thr183, respectively Tyr185. GAPDH served as endogenous control.
Snap frozen liver samples were processed as previously described using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE)20,21,26. Ponceau S staining was used to confirm equal protein loading. Membranes were blocked and incubated with respective antibodies. Thereafter, membranes were incubated with the corresponding secondary peroxidase-coupled antibodies (Santa Cruz-Biotechnology, Santa Cruz, USA).
After enhanced chemiluminescence (ECL; Amersham, Bucks, UK), digital detection was evaluated using Chemi-Smart (PeqLab Biotechnologies, Erlangen, Germany). Data are expressed as means ± standard error of the mean (SEM) with values of controls normalized to 100%.
Hepatic hydroxyproline content
Analogue segments (200 mg) of snap-frozen murine livers were hydrolysed in HCl (6 N). Hepatic hydroxyproline content was determined photometrically in liver hydrolysates as previously described26,27.
Isolation of primary rat HSC and hepatocytes
Primary rat HSCs were isolated as described previously21,26. In brief, primary HSC were isolated in a two-step pronase-collagenase perfusion from the livers of healthy rats (n = 5) and fractionated by density gradient centrifugation. Viability and purity were controlled and were systematically over 95%. Cells were seeded on uncoated plastic culture dishes. Experiments were performed 1 day after isolation to investigate effects in quiescent HSCs and 7 days after isolation or after the first passage (10 days) when HSCs were fully activated. Primary hepatocytes were isolated from male rats (n = 5) as described previously28.
Furthermore, these cells were incubated with the KCa3.1 inhibitor TRAM-34 and activator SKA-31 for 24 hours respectively and mRNA expression for collagen1, α-SMA and TGFβ were measured. To evaluate effects on cell cycle progression, mRNA expression of cyclin E1 (indicating S-phase initiation) and cyclin A2 (S-phase progression) was measured in 3 different isolations of hepatocytes and HSC incubated with TRAM-34 and SKA-31.
To evaluate effect of KCa3.1 inhibition and activation on cell volume, rat hepatocytes and HSC were incubated with TRAM-34 and SKA-31 for 24 hours. Hepatocyte and HSC cell area representative sections of the cell culture dish were photographed under 20x magnifications using a Nikon Eclipse TS100 inverted microscope with a Nikon DS-Vi1 head camera (Nikon, Düsseldorf, Germany). The measurement of cell area was performed using ImageJ (NIH, USA) and 5–10 cells were analysed per representative section.
Apoptosis assay
FACS analyses were performed to analyse apoptosis in rat hepatocytes by using the Annexin V Apoptosis Detectin Kit (KD Biosciences, Heidelberg, Germany) as previously described29.
DNA isolation and electrophoresis
DNA from rat hepatocyte samples incubated with agonist, antagonist or left untreated, was isolated using Apoptotic DNA ladder extraction Kit (BioVision, Mountain View, USA) according to the manufacturer’s guidelines. Samples were run in agarose gel, stained with ethidium bromide and visualized by transillumination with UV light, as described previously30.
Histology and immunohistochemical analyses
All analyses were performed on 5 μm paraffin-embedded sections. Positive and negative controls were used to exclude non-specific staining. Immunohistochemistry (IHC) and immunofluorescent (IF) staining of KCa3.1 channels were performed in human samples. In brief, following paraffin removal and dehydration, sections were incubated with a primary antibody (anti-KCNN4 #HPA05384, Sigma-Aldrich, St. Louis, MO, USA). For KCa3.1 IHC we used the PowerVision Poly-Horseradish peroxidase anti-Rabbit IgG (Dako, Glostrup, Denmark) as secondary antibody. For IF protocols we used an Alexa Fluor 488-labeled secondary antibody (Life Technologies, Darmstadt, Germany) for visualization. Finally, sections were counterstained with haematoxylin for IHC or diamidino-2-phenylindole dihydrochloride (DAPI) for IF. Human stained sections were reviewed with an Olympus BX51 microscope and pictures were captured with an Olympus DP26 camera in the cellSens program (Olympus, Hamburg, Germany).
For the detection of collagen fibres in mice, liver samples were stained with 0.1% Sirius red F3B in saturated picric acid (Chroma, Münster, Germany) using standard methods as described previously27. Further immunofluorescent staining was performed with collagen1 on murine samples. In human tissue paraffin was removed and sections were dehydrated. Subsequently, sections were incubated with a primary antibody (anti-Collagen, ab34710, Abcam, Cambridge, United Kingdom) and an Alexa Fluor 568-labeled secondary antibody (Life Technologies, Darmstadt, Germany) was used for visualization. Additionally, immunohistochemical analyses were performed for αSMA, which is a surrogate marker for hepatic stellate cell activation. The sections were incubated with a mouse-anti-SMA antibody (Actin clone 1A4; Dako, Hamburg, Germany). Thereafter, a biotinylated goat-anti-mouse (Dako, Hamburg, Germany) secondary antibody was used. Finally, sections were counterstained with haematoxylin.
The histological stainings of the murine samples were digitalised using Panoramic MIDI (3DHistech, Budapest, Hungary). The amount of positive staining was evaluated by computational analysis (Histoquant; 3DHistech, Budapest, Hungary). Large bile ducts and vessels were excluded. The analysis was performed following the principles of computational analysis as described previously31,32.
Statistical analysis
Unless otherwise stated, data are given as means ± standard error of mean (SEM) and student’s t-test was used for group comparison when appropriate. Otherwise nonparametric tests were used. Wilcoxon test was used for paired comparison in the same group. Statistical analysis was performed using Prism 5 (GraphPad, La Jolla, USA) software for Mac Os X. P values < 0.05 were considered statistical significant.
Results
KCa3.1 channels are upregulated in hepatocytes of fibrotic and cirrhotic human liver
Using immunohistochemical and immunofluorescence stainings, we evaluated the expression of KCa3.1 in non-fibrotic, fibrotic and cirrhotic human liver biopsies (Fig. 1). The corresponding general characteristics of the patients included are summarised in Table 1.
Distribution of KCa3.1 channel in non-fibrotic, fibrotic and cirrhotic human liver.
KCa3.1 channel expression evaluated by IHC top panel (I) (x10 magnification) and immunofluorescent stain (II) in bottom panel (x20 magnification). KCa3.1 channels are predominantly expressed in hepatocytes (a) of fibrotic and cirrhotic livers. Furthermore, KCa3.1 was detected in the vessel endothelium (b). To a minor degree KCa3.1 positive staining is seen in the fibrotic septae of cirrhotic livers (c), probably in activated HSCs.
In non-fibrotic livers, regardless of the presence of inflammation, the KCa3.1 channels were only detectable in erythrocytes and vessel endothelium. KCa3.1 was predominantly expressed in hepatocytes of fibrotic livers and were seen to be overexpressed in hepatocytes of cirrhotic livers. In virtually all the hepatocytes KCa3.1 was distributed in the cirrhotic noduli and to a lesser degree in fibrotic septae, (Fig. 1).
KCa3.1 channels are upregulated in fibrotic livers in rodents
To confirm the findings in human tissue samples from diseased livers, KCa3.1 mRNA expression was analysed in mice following 8 and 12 weeks of CCl4 induced hepatic injury. Overall, KCa3.1 gene expression increased during liver fibrogenesis and increased with progression of hepatic injury, thereby confirming our data in patients (Fig. 2A). This effect was independent from the fibrosis model, since KCa3.1 expression was also significantly increased in TAA, CCl4 intoxicated and BDL rat livers (Fig. 2B).
KCa3.1 expression in untreated and fibrotic rodents, cellular differences in distribution and effects.
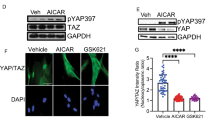
Similar to the differences between normal and fibrotic human livers, KCa3.1 channels are upregulated in fibrotic livers of mice (A) and rats (B) compared to untreated or sham animals. KCa3.1 is higher expressed in isolated hepatocytes compared to HSC (C). Incubation with the KCa3.1 inhibitor TRAM-34 or with the KCa3.1 agonist did not affect significantly the cell cycle progression of hepatocytes (D). TRAM-34 directly stimulates hepatocyte apoptosis, as evaluated by increased DNA degradation and annexin V (E). KCa3.1 activation with SKA-31 showed no effects on DNA degradation and apoptosis (E). KCa3.1 inhibition with TRAM-34 showed a clear trend towards an increase in the hepatocyte cell volume, whereas activation with SKA-31 induced cellular shrinkage (F).
In addition, in vitro, the expression of the KCa3.1 channel was significantly higher in hepatocytes compared to activated HSC (Fig. 2C), thereby validating our findings from immune staining of the human liver.
KCa3.1 inhibition induces hepatocyte apoptosis and alters hepatocyte volume
To dissect the effects of KCa3.1 in inhibition or activation of the cell cycle in hepatocytes, we measured the gene expression of the cell cycle mediators cyclin E1 (driving the release from quiescence) and cyclin A2 (controlling S-phase progression)33. Neither KCa3.1 inhibition with TRAM-34 nor activation with SKA-31 changed cyclin E1 and A2 expression significantly and hence showed no direct effect on cell cycle progression (Fig. 2D). However, the KCa3.1 inhibitor TRAM-34, but not the activator SKA-31, induced apoptosis of hepatocytes, as shown by DNA ladder formation and FACS analyses of annexin V (Fig. 2E).
Pharmacologic manipulation of the KCa3.1 channel directly affected the cell volume of hepatocytes. Inhibition with TRAM-34 showed a clear trend towards increase in hepatocyte volume, whereas activation with SKA-31 significantly reduced the cell volume (Fig. 2F).
KCa3.1 channel deficiency aggravates CCl4 induced liver fibrosis in mice
The KCa3.1−/− mice showed higher levels of hepatic hydroxyproline (Fig. 3A) and a higher deposition of collagen evaluated by Sirius red staining (Fig. 3B+C) and collagen-1 staining (Fig. 3C). Similarly, the hepatic mRNA levels of TGFβ1 and TNFα were increased in KCa3.1−/− mice after CCl4 intoxication compared to their respective wt littermates (Fig. 3D).
Effect of genetic KCa3.1 depletion in CCl4 induced liver fibrosis in mice.
KCa3.1−/− mice show increased fibrosis by the evaluation of hepatic hydroxyproline (A) and collagen, as evaluated by Sirius red and fluorescent stain for collagen1 (B,C) compared to wt littermates. These differences were especially pronounced in severe fibrosis, seen after 12 weeks of CCl4 exposure. Similarly, knockouts have higher expression of TGFβ mRNA and increased inflammatory activity, as evaluated by the higher transcription of the TNFα gene (D). Representative Western blot analysis of the hepatic protein-expression of PCNA, JNK, pJNK, p-c-Src Thr418 and 530 were analysed in fibrotic mice following 12 weeks CCl4 exposure. The hepatic expression of PCNA was higher in KCa3.1−/−. They similarly demonstrated increased pJNK and p-c-SrcThr418 expression, but lower Thr530 (E+F), suggesting a direct effect on c-Src and volume control (E,F).
Hepatic protein expression of Proliferating Cell Nuclear Antigen (PCNA) was highly increased in KCa3.1 deficient mice compared to wt mice after 12 weeks of CCl4 exposure (Fig. 3E+F). Activation, but not expression of hepatic JNK, was significantly increased in fibrotic KCa3.1−/− mice compared to wt littermates (Fig. 3E+F). Moreover, the hepatic protein expression of the c-Src activating phosphorylation (p-c-SrcThr418) was higher, whereas the protein expression of the c-Src deactivating phosphorylation (p-c-SrcThr530) was lower in knockout animals after 12 weeks of CCl4 compared to the respective wt mice (Fig. 3E+F). These analyses suggest that proliferation (PCNA) and volume control (c-Src activation) are dysregulated in knockout animals after hepatic injury.
KCa3.1 inhibition or deficiency stimulates HSC activity in vitro and in vivo
To better understand the reason for increased fibrosis in KCa3.1 deficient mice, activated hepatic stellate cells (HSC) were incubated with the KCa3.1 inhibitor TRAM-34 in vitro. TRAM-34 significantly suppressed collagen mRNA expression in activated HSC, however no significant effects could be observed on α-SMA and TGFβ mRNA expression (Fig. 4A).
Effect of KCa3.1 deficiency and inhibition on activated HSC.
In vivo, HSCs were incubated with TRAM-34 and SKA-31 and effect on profibrotic markers evaluated. Incubation with the KCa3.1 inhibitor TRAM-34 suppressed collagen-1 transcription. A similarly trend towards reduced expression was seen for TGF-β. α-SMA was reduced at levels similar to KCa3.1 activation with SKA-31, but was not significantly altered from control (A). Incubation with TRAM-34 was followed by an increase in cyclins E1 and A2 in HSCs (B), suggesting enhanced cell cycle progression. This was supported by increased protein expression of PCNA in isolated HSC (C). Similarly to findings in hepatocytes, TRAM-34 increased HSC volume, but contrary SKA-31 was not able to reduce volume (D). CCl4 intoxication induces hepatic injury and leads to increased activation of HSCs in Kca3.1−/− mice, as evaluated by α-SMA immunohistochemistry (E+F).
Furthermore, TRAM-34 induced the cyclin E1 and A2 mRNA expression in activated HSC (Fig. 4B), which marks cell cycle progression. KCa3.1 channel stimulation, using SKA-31, however did not affect cell cycle progression (Fig. 4B). PCNA protein expression was increased in isolated HSC after KCa3.1 inhibition with TRAM-34, while activation with SKA-31 did not impact proliferation (Fig. 4C).
Similarly to findings in hepatocytes, TRAM-34 increased cell volume in HSC, but contrary to the hepatocytes, activation with SKA-31 showed no significant reduction in volume (Fig. 4D).
The in vitro results were confirmed by the in vivo observation of increased α-SMA positive staining, as a sign of increased HSC activation in fibrotic KCa3.1 deficient mice (Fig. 4E+F).
KCa3.1 inhibition with Senicapoc worsens hepatic fibrosis
Since the genetic deficiency of the KCa3.1 channel might have different effects than the pharmacological inhibition in vivo, the KCa3.1 blocker Senicapoc was tested in CCl4 induced liver injury in wt mice by oral administration. Senicapoc treated mice ate less than the respective placebo (18,6 g/week vs. 22,9 g/week), but sufficient absorption of the drug was observed in all animals with average p-Senicapoc levels of 587.1 nM.
Senicapoc treated mice showed increased deposition of collagen, as evaluated by Sirius red (Fig. 5A+B) and collagen1 mRNA levels (Fig. 5C). This was associated with higher HSC activation, as assessed by α-SMA staining (Fig. 5D+E) and α-SMA gene transcription (Fig. 5F).
Effects of the KCa3.1 channel inhibitor Senicapoc in CCl4 induced hepatic injury in wt mice.
Pharmacological KCa3.1 inhibition in CCl4 induced liver injury in mice led to increased liver fibrosis compared to wt littermates receiving placebo (A–C) and was similarly to the effects seen in KCa3.1−/− mice. Fibrosis was more severe as shown by Sirius red staining (A+B) and collagen-1 gene expression (C). Senicapoc treatment led to increased HSC activation as shown by immunohistochemiacal staining (D+E), as well as by gene expression levels (F) of surrogate marker α-SMA.
KCa3.1 inhibition does not alter portal pressure in vivo
Portal pressure measurements were performed to evaluate the direct in vivo effect of TRAM-34 on portal hemodynamics in BDL and CCl4 cirrhotic rats. However, no significant changes in portal pressure, mean arterial pressure or heart rate were observed (Fig. 6).
In vivo effects on hemodynamics of the KCa3.1 blocker TRAM-34 in bile duct ligated rats.
In vivo, invasive measurement of portal pressure was performed in BDL and CCl4 cirrhotic rats before and after i.v. injection of TRAM-34 (100 nM). Following a 20 minutes observation period, no significant change in portal pressure (mmHg) could be observed (A). Measurements of mean arterial pressure (mmHg) and heart rate (beats/min) did not show any alterations compared to prior to TRAM-34 bolus (B+C).
Discussion
This study highlights the importance of the KCa3.1 channel in response to liver injury in humans and rodents. We demonstrate for the first time that the KCa3.1 channel is upregulated in hepatocytes with increasing liver fibrosis, while its absence or inhibition aggravates liver fibrosis.
The KCa3.1 channel is widely distributed in the organism, including the red and white blood cell linage34,35,36, epithelial cells of airway, colon and salivary glands, fibrocytes and vascular endothelial cells4,5,6,7,8, where as it’s hepatic expression is sparsely explored. A recent histopathological study based on stainings of only a few fibrotic liver samples reported that these channels are upregulated in hepatic fibrosis and the channels might be located in activated HSC14. We confirmed that the KCa3.1 channel is expressed in HSCs. However, KCa3.1 was greatly upregulated in the damaged hepatocytes compared activated HSC of fibrotic livers. Similarly, we showed a significantly higher expression of KCa3.1 mRNA in healthy hepatocytes compared to HSC in vitro, however there seems to a discrepancy between mRNA and transcription to protein, as expression of KCa3.1 evaluated by IHC, was absent in healthy human liver. The suggested anti-fibrotic effect of KCa3.1 inhibition in other fibroproliferative disorders has been linked to a suppressed effect on myofibroblast proliferation. In vitro, we were similarly able to show suppression of collagen1 mRNA in isolated HSC. However, although the fibrogenic pathways in all fibroproliferative diseases are highly similar, the liver differs from other organs by its unique highly regenerative potential – a capacity linked directly to the physiological roles of the hepatocytes. Upon injury, presence of apoptotic hepatocytes stimulates the inflammatory response and hence fibrogenesis37. As KCa3.1 channels in the liver show a high hepatocyte expression compared to the expression seen in HSC, the mediated effect on hepatocytes may blunt any potential anti-fibrotic effect on HSC. The reason why KCa3.1 is found in activated HSC might be the profound changes that occur in these cells showing an increased proliferation, protein expression and migration. KCa3.1 might be one of the requirements of activated HSC to maintain their activated phenotype, since inhibiting it led to proliferation, but not to higher pro-fibrotic activity. The hepatocyte KCa3.1 expression and effect on hepatocyte volume recovery seems important and explains why the roles of KCa3.1 in the liver differ from effects seen in other parenchymal organs. Therefore, the deficiency or inhibition of KCa3.1 in vivo in models of liver injury is associated with hepatocellular damage and inflammation, which drives fibrogenesis, while the mild antifibrotic effects of KCa3.1-inhibition in activated HSC is not relevant in the in vivo setting.
The expression of KCa3.1 increased with the severity of liver fibrosis in humans and in three different rodent models of liver fibrosis, suggesting that this is a common pathway irrespective of the species and type of liver injury. Although limited by the small number of animals in the rat experiments, these studies however support the findings from murine and human experiments. The attempt to demonstrate effects of KCa3.1 in a cholestatic mouse model (BDL) was not successful. A pilot study demonstrated a high mortality (>80%) in operated KCa3.1 knockout mice (data not shown), which was linked to a high number of post-operative complications, especially caused by suture rupture induced by the mice – the latter which may be linked to the hyperactive phenotype of KCa3.1 knockouts17.
Based on our findings, the exact role of KCa3.1 in hepatic injury and fibrogenesis is unknown. In several cell types (e.g. cancer cell lines, myofibroblasts) channel activation leads to calcium influx and thereby stimulates cell cycle progression and cellular proliferation6,38,39. In other cell types (e.g. colon epithelial cells, erythrocytes) the channel regulates cell volume. Channel activation induces outflow of chloride and water, which leads to shrinkage40,41,42,43. In in vitro experiments with hepatoma cells, KCa3.1 regulated the cell volume3. This present study clearly supports these findings and shows that KCa3.1 is crucial for the homeostasis of hepatocytes upon injury. Supporting this hypothesis, we have shown that KCa3.1 activation was able to induce shrinkage of hepatocytes, as demonstrated by the reduction in cell volume. Approaches blocking this channel lead to cell cycle dysregulation and apoptosis in vitro, whereas the underlying mechanism might be a lack in the capacity to shrink after hepatocyte injury44. In vivo, activation of JNK and c-Src kinase was increased in KCa3.1 deficient fibrotic mice compared to the wild types assessed by their phosphorylation. It has been shown previously in vitro and in the isolated perfused rat liver, as reviewed in44,45,46,47, that hepatocyte swelling is directly linked to c-Src kinase, extracellular regulated kinase (Erk)-1/-2 and epidermal growth factor receptor (EGFR) activation in a α5β1-integrin-dependent manner resulting in hepatocyte proliferation. It is also a well-established concept that hepatocyte cell swelling is linked to proliferation, whereas hepatocyte shrinkage ends up in JNK- and CD95 death receptor-mediated apoptosis44,45,46,47. Interestingly, it has recently been reported, that KCa3.1 inhibition in human hepatoma HepG2 cell line cells, which most likely is accompanied by cell swelling, results in apoptosis48. Thus it might be an interesting speculation whether an overwhelming hepatocyte swelling in the context of liver regeneration after hepatic injury by either CCl4 or BDL in knockouts results in hepatocyte apoptosis and liver fibrosis. This is supported by our findings that in the KCa3.1 knockout mice a more pronounced c-Src and an increased hepatocyte injury became detectable. These findings account for a phenomenon associated with swelling since c-Src is usually de-activated in liver fibrosis as shown recently by our group49. The abundant KCa3.1 expression seen in human cirrhotic liver tissue may also be explained by a repair mechanism of the hepatocytes, which attempt to restore cell volume in the case of hepatocytes swelling caused by chronic liver injury. Our view is encouraged by recent studies in a model of alveolar epithelial repair in which a complementary role of the swelling activated β1-integrin and KCa3.1 was described50. This again points to the importance of KCa3.1 in hepatocyte volume homeostasis and differentiated Src family kinase activation51 as it might protect hepatocytes from an overwhelming cell swelling in the context of hepatocyte proliferation.
Since the hepatocellular damage is more pronounced in fibrotic mice when KCa3.1 is deficient or blocked, the concomitant increased activation of HSC and fibrosis accumulation is expected. The mechanism by which the HSC get more activated after inhibition of KCa3.1 might be dual. On the one side the hepatocellular damage leads to more hepatic inflammation as shown by TNFα expression; on the other side the blocking the KCa3.1 channel as shown in vitro promotes proliferation. Furthermore, it has previously been reported that in quiescent HSC, in contrast to the hepatocytes, addition of death receptor ligands such as CD95L or TNFα are linked to proliferative signalling pathways. This might contribute to the increased activation of HSC after KCa3.1 inhibition as reported in this study and is probably at least partly JNK-mediated52.
HSCs might not be as sensitive as hepatocytes in response to cell volume47. Interestingly we found that KCa3.1 inhibition induced swelling in HSC, whereas activation was not able to induce shrinkage in these cells. Therefore, KCa3.1-inactivation might even lead to proliferation of these cells. In vivo, these harmful effects of KCa3.1 deficiency or inhibition on liver fibrosis could not be ameliorated by a slight decrease in collagen transcription in vitro.
A previous study showed that acute administration of a pharmacological KCa3.1-blocker reduced hepatic resistance in in situ liver perfusion experiments14. This effect could not be confirmed in our set of in vivo experiments. Therefore this is a strict in vitro effect without relevance for in vivo conditions when evaluating the effects on acute administration of the KCa3.1 blocker. Furthermore, the use of TRAM-34 in experimental settings may be challenged by the lack of selectivity of the blocker towards KCa3.153.
KCa3.1 blockers seem to be beneficial in renal and pulmonary fibrosis or other investigated disorders6,7,9,11,12,13. This study demonstrates, that in liver fibrosis, KCa3.1 expression and proper function is protective for hepatocellular damage and fibrosis. Therefore, the activation of KCa3.1 might be advantageous in the context of liver injury.
Furthermore, the quantification of KCa3.1 expression could be a read-out for hepatocellular homeostasis during liver injury and under anti-fibrotic strategies.
In conclusion, our findings demonstrate that hepatic KCa3.1 activation is an important anti-fibrotic pathway and protects hepatocytes during liver injury and liver fibrosis.
Additional Information
How to cite this article: Møller, L. S. et al. The calcium-activated potassium channel KCa3.1 is an important modulator of hepatic injury. Sci. Rep. 6, 28770; doi: 10.1038/srep28770 (2016).
References
Mederacke, I. et al. Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nature communications 4, 2823, doi: 10.1038/ncomms3823 (2013).
Kordes, C., Sawitza, I., Gotze, S., Herebian, D. & Haussinger, D. Hepatic stellate cells contribute to progenitor cells and liver regeneration. The Journal of clinical investigation 124, 5503–5515, doi: 10.1172/jci74119 (2014).
Barfod, E. T., Moore, A. L., Roe, M. W. & Lidofsky, S. D. Ca2+-activated IK1 channels associate with lipid rafts upon cell swelling and mediate volume recovery. The Journal of biological chemistry 282, 8984–8993, doi: 10.1074/jbc.M607730200 (2007).
Greger, R. et al. The role of K+ channels in colonic Cl- secretion. Comparative biochemistry and physiology. Part A, Physiology 118, 271–275 (1997).
Si, H. et al. Impaired endothelium-derived hyperpolarizing factor-mediated dilations and increased blood pressure in mice deficient of the intermediate-conductance Ca2+-activated K+ channel. Circulation research 99, 537–544, doi: 10.1161/01.RES.0000238377.08219.0c (2006).
Shepherd, M. C. et al. KCa3.1 Ca2+ activated K+ channels regulate human airway smooth muscle proliferation. American journal of respiratory cell and molecular biology 37, 525–531, doi: 10.1165/rcmb.2006-0358OC (2007).
Cruse, G. et al. Functional KCa3.1 K+ channels are required for human fibrocyte migration. J. Allergy Clin. Immunol. 128, 1303–1309.e1302, doi: 10.1016/j.jaci.2011.07.047 (2011).
Damkjaer, M. et al. Pharmacological activation of KCa3.1/KCa2.3 channels produces endothelial hyperpolarization and lowers blood pressure in conscious dogs. British journal of pharmacology 165, 223–234, doi: 10.1111/j.1476-5381.2011.01546.x (2012).
Huang, C. et al. KCa3.1 mediates activation of fibroblasts in diabetic renal interstitial fibrosis. Nephrol. Dial. Transplant. 29, 313–324, doi: 10.1093/ndt/gft431 (2014).
Fu, R. G. et al. Inhibition of the K+ channel K(Ca)3.1 reduces TGF-beta1-induced premature senescence, myofibroblast phenotype transition and proliferation of mesangial cells. PLoS One 9, e87410, doi: 10.1371/journal.pone.0087410 (2014).
Roach, K. M. et al. The K+ channel KCa3.1 as a novel target for idiopathic pulmonary fibrosis. PLoS One 8, e85244, doi: 10.1371/journal.pone.0085244 (2013).
Huang, C., Day, M. L., Poronnik, P., Pollock, C. A. & Chen, X. M. Inhibition of KCa3.1 suppresses TGF-beta1 induced MCP-1 expression in human proximal tubular cells through Smad3, p38 and ERK1/2 signaling pathways. Int. J. Biochem. Cell Biol. 47, 1–10, doi: 10.1016/j.biocel.2013.11.017 (2014).
Grgic, I. et al. Renal fibrosis is attenuated by targeted disruption of KCa3.1 potassium channels. Proc. Natl. Acad. Sci. USA 106, 14518–14523, doi: 10.1073/pnas.0903458106 (2009).
Freise, C. et al. K -channel inhibition reduces portal perfusion pressure in fibrotic rats and fibrosis associated characteristics of hepatic stellate cells. Liver Int, doi: 10.1111/liv.12681 (2014).
Grgic, I. et al. Disruption of the Gardos channel (KCa3.1) in mice causes subtle erythrocyte macrocytosis and progressive splenomegaly. Pflugers Archiv : European journal of physiology 458, 291–302, doi: 10.1007/s00424-008-0619-x (2009).
Brahler, S. et al. Genetic deficit of SK3 and IK1 channels disrupts the endothelium-derived hyperpolarizing factor vasodilator pathway and causes hypertension. Circulation 119, 2323–2332, doi: 10.1161/circulationaha.108.846634 (2009).
Lambertsen, K. L. et al. Genetic KCa3.1-deficiency produces locomotor hyperactivity and alterations in cerebral monoamine levels. PloS one 7, e47744, doi: 10.1371/journal.pone.0047744 (2012).
Liedtke, C. et al. Experimental liver fibrosis research: update on animal models, legal issues and translational aspects. Fibrogenesis & tissue repair 6, 19, doi: 10.1186/1755-1536-6-19 (2013).
Constandinou, C., Henderson, N. & Iredale, J. P. Modeling liver fibrosis in rodents. Methods in molecular medicine 117, 237–250, doi: 10.1385/1-59259-940-0:237 (2005).
Trebicka, J. et al. Atorvastatin lowers portal pressure in cirrhotic rats by inhibition of RhoA/Rho-kinase and activation of endothelial nitric oxide synthase. Hepatology 46, 242–253 (2007).
Trebicka, J. et al. Role of beta3-adrenoceptors for intrahepatic resistance and portal hypertension in liver cirrhosis. Hepatology 50, 1924–1935, doi: 10.1002/hep.23222 (2009).
Laleman, W. et al. A stable model of cirrhotic portal hypertension in the rat: thioacetamide revisited. European journal of clinical investigation 36, 242–249, doi: 10.1111/j.1365-2362.2006.01620.x (2006).
Stocker, J. W. et al. ICA-17043, a novel Gardos channel blocker, prevents sickled red blood cell dehydration in vitro and in vivo in SAD mice. Blood 101, 2412–2418, doi: 10.1182/blood-2002-05-1433 (2003).
Trebicka, J. et al. Hemodynamic effects of urotensin II and its specific receptor antagonist palosuran in cirrhotic rats. Hepatology 47, 1264–1276, doi: 10.1002/hep.22170 (2008).
Grace, J. A. et al. Activation of the MAS receptor by angiotensin-(1–7) in the renin-angiotensin system mediates mesenteric vasodilatation in cirrhosis. Gastroenterology 145, 874–884.e875, doi: 10.1053/j.gastro.2013.06.036 (2013).
Granzow, M. et al. Angiotensin-II type 1 receptor-mediated Janus kinase 2 activation induces liver fibrosis. Hepatology 60, 334–348, doi: 10.1002/hep.27117 (2014).
Trebicka, J. et al. Atorvastatin attenuates hepatic fibrosis in rats after bile duct ligation via decreased turnover of hepatic stellate cells. Journal of hepatology 53, 702–712, doi: 10.1016/j.jhep.2010.04.025 (2010).
Wojtalla, A. et al. The endocannabinoid N-arachidonoyl dopamine (NADA) selectively induces oxidative stress-mediated cell death in hepatic stellate cells but not in hepatocytes. American journal of physiology. Gastrointestinal and liver physiology 302, G873–887, doi: 10.1152/ajpgi.00241.2011 (2012).
Klein, S. et al. Atorvastatin inhibits proliferation and apoptosis, but induces senescence in hepatic myofibroblasts and thereby attenuates hepatic fibrosis in rats. Laboratory investigation; a journal of technical methods and pathology 92, 1440–1450, doi: 10.1038/labinvest.2012.106 (2012).
Hennenberg, M. et al. Hepatic and HSC-specific sorafenib effects in rats with established secondary biliary cirrhosis. Lab. Invest. 91, 241–251, doi: 10.1038/labinvest.2010.148 (2011).
Trebicka, J. et al. Role of cannabinoid receptors in alcoholic hepatic injury: steatosis and fibrogenesis are increased in CB2 receptor-deficient mice and decreased in CB1 receptor knockouts. Liver international: official journal of the International Association for the Study of the Liver 31, 860–870, doi: 10.1111/j.1478-3231.2011.02496.x (2011).
Huss, S. et al. Development and evaluation of an open source Delphi-based software for morphometric quantification of liver fibrosis. Fibrogenesis & tissue repair 3, 10, doi: 10.1186/1755-1536-3-10 (2010).
Nevzorova, Y. A. et al. Cyclin E1 controls proliferation of hepatic stellate cells and is essential for liver fibrogenesis in mice. Hepatology (Baltimore, Md.) 56, 1140–1149, doi: 10.1002/hep.25736 (2012).
Hoffman, J. F. et al. The hSK4 (KCNN4) isoform is the Ca2+-activated K+ channel (Gardos channel) in human red blood cells. Proceedings of the National Academy of Sciences of the United States of America 100, 7366–7371, doi: 10.1073/pnas.1232342100 (2003).
Logsdon, N. J., Kang, J., Togo, J. A., Christian, E. P. & Aiyar, J. A novel gene, hKCa4, encodes the calcium-activated potassium channel in human T lymphocytes. The Journal of biological chemistry 272, 32723–32726 (1997).
Wulff, H., Knaus, H. G., Pennington, M. & Chandy, K. G. K+ channel expression during B cell differentiation: implications for immunomodulation and autoimmunity. Journal of immunology (Baltimore, Md. : 1950) 173, 776–786 (2004).
Canbay, A., Friedman, S. & Gores, G. J. Apoptosis: the nexus of liver injury and fibrosis. Hepatology (Baltimore, Md.) 39, 273–278, doi: 10.1002/hep.20051 (2004).
Millership, J. E. et al. Calcium-activated K+ channels increase cell proliferation independent of K+ conductance. American journal of physiology. Cell physiology 300, C792–802, doi: 10.1152/ajpcell.00274.2010 (2011).
Parihar, A. S., Coghlan, M. J., Gopalakrishnan, M. & Shieh, C. C. Effects of intermediate-conductance Ca2+-activated K+ channel modulators on human prostate cancer cell proliferation. European journal of pharmacology 471, 157–164 (2003).
Brugnara, C., de Franceschi, L. & Alper, S. L. Inhibition of Ca(2+)-dependent K+ transport and cell dehydration in sickle erythrocytes by clotrimazole and other imidazole derivatives. The Journal of clinical investigation 92, 520–526, doi: 10.1172/jci116597 (1993).
Elliott, J. I. & Higgins, C. F. IKCa1 activity is required for cell shrinkage, phosphatidylserine translocation and death in T lymphocyte apoptosis. EMBO reports 4, 189–194, doi: 10.1038/sj.embor.embor722 (2003).
Albaqumi, M. et al. KCa3.1 potassium channels are critical for cAMP-dependent chloride secretion and cyst growth in autosomal-dominant polycystic kidney disease. Kidney international 74, 740–749, doi: 10.1038/ki.2008.246 (2008).
Matos, J. E. et al. Role of cholinergic-activated KCa1.1 (BK), KCa3.1 (SK4) and KV7.1 (KCNQ1) channels in mouse colonic Cl- secretion. Acta physiologica (Oxford, England) 189, 251–258, doi: 10.1111/j.1748-1716.2006.01646.x (2007).
Haussinger, D. & Reinehr, R. Osmotic regulation of bile acid transport, apoptosis and proliferation in rat liver. Cell Physiol Biochem 28, 1089–1098, doi: 10.1159/000335845 (2011).
Haussinger, D., Reinehr, R. & Schliess, F. The hepatocyte integrin system and cell volume sensing. Acta physiologica (Oxford, England) 187, 249–255, doi: 10.1111/j.1748-1716.2006.01542.x (2006).
Reinehr, R. & Haussinger, D. CD95 death receptor and epidermal growth factor receptor (EGFR) in liver cell apoptosis and regeneration. Arch Biochem Biophys 518, 2–7, doi: 10.1016/j.abb.2011.12.004 (2012).
Reinehr, R. & Haussinger, D. Epidermal growth factor receptor signaling in liver cell proliferation and apoptosis. Biol Chem 390, 1033–1037, doi: 10.1515/bc.2009.106 (2009).
Liu, Y. et al. The Blockage of KCa3.1 Channel Inhibited Proliferation, Migration and Promoted Apoptosis of Human Hepatocellular Carcinoma Cells. Journal of Cancer 6, 643–651, doi: 10.7150/jca.11913 (2015).
Gortzen, J. et al. Interplay of Matrix Stiffness and c-SRC in Hepatic Fibrosis. Frontiers in physiology 6, 359, doi: 10.3389/fphys.2015.00359 (2015).
Girault, A. et al. Complementary roles of KCa3.1 channels and beta1-integrin during alveolar epithelial repair. Respiratory research 16, 100, doi: 10.1186/s12931-015-0263-x (2015).
Reinehr, R., Sommerfeld, A. & Haussinger, D. The Src family kinases: distinct functions of c-Src, Yes and Fyn in the liver. Biomolecular concepts 4, 129–142, doi: 10.1515/bmc-2012-0047 (2013).
Reinehr, R., Sommerfeld, A. & Haussinger, D. CD95 ligand is a proliferative and antiapoptotic signal in quiescent hepatic stellate cells. Gastroenterology 134, 1494–1506, doi: 10.1053/j.gastro.2008.02.021 (2008).
Schilling, T. & Eder, C. TRAM-34 inhibits nonselective cation channels. Pflugers Archiv : European journal of physiology 454, 559–563, doi: 10.1007/s00424-007-0232-4 (2007).
Acknowledgements
The authors thank G. Hack, S. Bellinghausen and I. Nissen for excellent technical assistance. The study was supported by grants from the A.P Møller Foundation for the Advancements in Medical Science, The Hede Nielsen Family Foundation and Prosector Axel Emil Søeborg Ohlsen and Spouse Memorial Fund (to LSM). The Odense University Hospital Research Council (to ADF and SDM), The Lundbeck Foundation (RK) and Deutsche Forschungsgemeinschaft (SFB TRR57) (to CL, JT).
Author information
Authors and Affiliations
Contributions
L.S.-M.: study concept and design, acquisition, analysis and interpretation of data, drafting of the manuscript, obtaining funding, administrative support. A.D.F. and O.B.S.d.M.: study concept and design, acquisition, analysis and interpretation of data, obtaining funding, study supervision. R.S., M.B., C.L., S.K., W.R., L.K.H. and M.R.: acquisition, analysis and interpretation of data. W.L.: acquisition, providing important animal models. V.S., J.S. and J.O.: providing support for animal studies. R.R.: analysis and interpretation of data, drafting of the manuscript. R.K.: study concept and design, acquisition, obtaining funding, administrative support. J.T.: study concept and design, acquisition, analysis and interpretation of data, drafting of the manuscript, obtaining funding, administrative support, study supervision.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sevelsted Møller, L., Fialla, A., Schierwagen, R. et al. The calcium-activated potassium channel KCa3.1 is an important modulator of hepatic injury. Sci Rep 6, 28770 (2016). https://doi.org/10.1038/srep28770
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep28770
This article is cited by
-
Oxidative stress promotes myocardial fibrosis by upregulating KCa3.1 channel expression in AGT-REN double transgenic hypertensive mice
Pflügers Archiv - European Journal of Physiology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.