Abstract
Fatty alcohols are value-added chemicals and important components of a variety of industries, which have a >3 billion-dollar global market annually. Long chain fatty alcohols (>C12) are mainly used in surfactants, lubricants, detergents, pharmaceuticals and cosmetics while medium chain fatty alcohols (C6–C12) could be used as diesel-like biofuels. Microbial production of fatty alcohols from renewable feedstock stands as a promising strategy to enable sustainable supply of fatty alcohols. In this study, we report, for the first time, that medium chain fatty alcohols could be produced in yeast via targeted expression of a fatty acyl-CoA reductase (TaFAR) in the peroxisome of Saccharomyces cerevisiae. By tagging TaFAR enzyme with peroxisomal targeting signal peptides, the TaFAR could be compartmentalized into the matrix of the peroxisome to hijack the medium chain fatty acyl-CoA generated from the beta-oxidation pathway and convert them to versatile medium chain fatty alcohols (C10 & C12). The overexpression of genes encoding PEX7 and acetyl-CoA carboxylase further improved fatty alcohol production by 1.4-fold. After medium optimization in fed-batch fermentation using glucose as the sole carbon source, fatty alcohols were produced at 1.3 g/L, including 6.9% 1-decanol, 27.5% 1-dodecanol, 2.9% 1-tetradecanol and 62.7% 1-hexadecanol. This work revealed that peroxisome could be engineered as a compartmentalized organelle for producing fatty acid-derived chemicals in S. cerevisiae.
Similar content being viewed by others
Introduction
As an important raw chemical material, fatty alcohols have been widely used to produce detergents, emulsifiers, lubricants, cosmetics and fuels1. In 2006, over 1.3 million tons of fatty alcohols were used worldwide each year2. As a whole, the industry represents a >3-billion-dollar market. The majority of the fatty alcohol market targets on medium chain (C6-C12) or short chain fatty alcohols (<C6). Currently, fatty alcohols are mainly produced in two ways. One is direct extraction from natural plant oils3 and the other is chemical synthesis from petrochemical sources. These methods have limitations due to the competition with the food supply4 and generation of environmental concerns5.
Recently, with the development of metabolic engineering, microbial production of fatty alcohols from renewable feedstock has been achieved successfully in both Escherichia coli2,6 and Saccharomyces cerevisiae7,8. In E. coli, fatty alcohols have been produced by introducing heterologous enzymes such as carboxylic acid reductase (CAR)9, a bifunctional fatty acyl-CoA reductase (FAR)10 or combined expressing an fatty aldehyde-forming fatty acyl-CoA reductase (ACR) with the endogenous aldehyde reductases (AR)2,11. Recent research showed that a high level of 1-dodecanol and 1-tetradecanol could be produced in E. coli by overexpression an acyl-ACP thioesterase (BTE), an acyl-CoA ligase (FadD) and an acyl-CoA/aldehyde reductase (MAACR)2. Compared to E. coli, yeast, especially S. cerevisiae, is also a well characterized and robust industrial host but can be cultivated at lower pH and various harsh fermentation conditions12. In addition, S. cerevisiae uses type I fatty acids synthetase (FAS) rather than type II FAS as that used by E. coli. Compared to that in E. coli, the synthetic route of fatty acyl-CoA is shorter in yeast, which could allow more efficient conversion of carbohydrate substrates to fatty acids and fatty acid-derived biofuels6. Therefore, there has been an increasing interest on developing yeast, such as S. cerevisiae, as a cell factory for production of fatty acid-derived chemicals. In S. cerevisiae, a mouse FAR has been expressed to produce 1-hexadecanol. Through over-expression of acetyl-CoA carboxylase (ACC1) and fatty acid synthases (FAS1 and FAS2), the engineered S. cerevisiae strain produced 98.0 mg/L 1-hexadecanol from 20 g/L hexose in batch fermentation in minimal medium7. In our previous study, we found that by manipulating the structural genes in yeast lipid metabolism, tuning the regulation of phospholipid synthesis and increasing the supply of key precursors, 1-hexadecanol was produced at 1.1 g/L as the sole fatty alcohol product using glucose as the sole carbon source in a fed-batch fermentation8.
Despite the success on engineering S. cerevisiae to produce long chain fatty alcohols, no success, to our best knowledge, has been achieved to engineer S. cerevisiae to produce medium chain fatty alcohols. This is mainly because of the lack of the precursor (i.e., medium chain fatty acyl-CoA) for producing medium chain fatty alcohols in S. cerevisiae, since the majority of fatty acyl-CoAs in native S. cerevisiae have long chains13. Recently, several pioneering studies have engineered S. cerevisiae to produce medium and short chain fatty acids by replacing the native type I FAS with heterologous type II FAS14 or disrupting the beta-oxidation15,16. However, the titers of medium and short chain fatty acids in these engineered yeast strains were still too low (<200 mg/L) to meet the demands of fatty acid-derived chemicals such as medium chain fatty alcohols.
In this study, we developed a novel strategy to engineer S. cerevisiae to produce medium chain fatty alcohols by compartmentalizing the fatty alcohol synthesis pathway in peroxisome. In native S. cerevisiae, the pool of shortened fatty acyl-CoAs only exist in peroxisome as the intermediates of the beta-oxidation cycle, which catalyzes the chain shortening of fatty acyl-CoA esters between carbons 2 and 3, yielding products as chain-shortened acyl-CoA and acetyl-CoA or propionyl-CoA, depending on substrates17. We hence assumed that by expressing TaFAR in peroxisome, TaFAR could hijack the chain-shortened acyl-CoAs to produce medium or even short chain fatty alcohols (Fig. 1). Indeed, when we introduced a series of peroxisomal targeting signal peptide to import TaFAR into peroxisome, versatile medium chain fatty alcohols, including 1-decanol, 1-dodecanol and 1-tetradecanol, could be detected in the engineered S. cerevisiae strains. We then manipulated the structural proteins in peroxisome (e.g., PEX7) to enhance the efficiency of enzyme location into peroxisome and overexpressed ACC1 to overcome the rate limiting steps in fatty acid synthesis. Over 1.3 g/L medium chain and long chain fatty alcohols were produced in total in a fed-batch fermentation using an optimized medium, which represents the highest titer of fatty alcohol reported in S. cerevisiae so far. To our best knowledge, it is first time that yeast was engineered to produce medium chain fatty alcohols. Our work demonstrated that the compartmentalization of metabolic pathways stands as a promising strategy to produce fatty acid-derived chemicals.
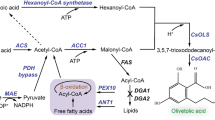
Scheme of engineering yeast peroxisome for medium chain fatty alcohols production.
Fatty alcohols can be produced by expressing a fatty acyl-CoA reductase from Tyto alba (TaFAR). TaFAR was then introduced into matrix of peroxisome by tagging with PTS1 or PTS2 signals to convert shortened fatty acyl-CoAs to produce medium chain fatty alcohols. The peroxisomal membrane protein PEX5 and PEX7 was overexpressed to enhance the efficiency of cytosol TaFAR introduction. PEX3 and PEX19 were engineered to help the formation of peroxisomes. In addition, acetyl-CoA carboxylase (ACC1) and ATP-dependent citrate lyase (ACL) from Yarrowi lipolytica were overexpressed to enhance the supply of precursors in fatty acid synthesis.
Results
Using PTS signaling pathway for targeted expression of FAR in peroxisome
In S. cerevisiae, the cytosol type I FAS, which is a highly integrated multi-enzyme complex, mainly produces long chain fatty acyl-CoA (e.g., C16:0) in the cell. Therefore, when expressing a FAR gene, such as TaFAR from barn owl Tyto alba, in yeast, the produced fatty alcohol was mainly in the form of 1-hexadecanol8. To produce medium chain fatty alcohols, the shorter chain fatty acyl-CoAs are required as the precursors. The shortened fatty acyl-CoAs could be generated in peroxisome as important intermediates in the beta-oxidation cycle that degrades fatty acids and exclusively takes place in peroxisomes of fungi and plants18. In this case, we assumed that when TaFAR was introduced into peroxisomes, it could use the shortened acyl-CoAs as the precursors to produce medium chain fatty alcohols.
The targeted expression of proteins in peroxisomes requires signal sequences known as peroxisomal targeting signal (PTS). Two types of PTS have been discovered: type I (PTS1) and type II (PTS2)19. The PTS1, used by the majority of peroxisomal matrix proteins, is located at the extreme C-terminus and was initially discovered in firefly luciferase as the tripeptide SKL20. The PTS1 generally fits the consensus sequence (S/A/C)-(K/R/H)-(L/M)21. The PTS2 is a conserved sequence which is located near the N-terminus of a protein and is comprised in some species within a pre-sequence that is cleaved off after import into the peroxisomal matrix. Sequence comparisons of the signal sequences showed the conserved nonapeptide of PTS2 as (R/K)-(L/V/I)-X5-(H/Q)-(L/A/F)22.
To prove the concept that TaFAR can be expressed in peroxisomes of S. cerevisiae and produce medium chain fatty alcohols by tagging with a PTS peptide, we selected two PTS peptides: SKL (i.e., a PTS1 peptide) and KL-X5-QL (i.e., a PTS2 peptide), fused them to the C-terminal or N-terminal of TaFAR respectively and measured the fatty alcohols profiles by GC-MS (Fig. 2). As expected, it was found that both the TaFAR with PTS1 and PTS2 peptides produced long chain and medium chain fatty alcohols (7.7% 1-decanol, 28.3% 1-dodecanol and 64.0% 1-hexadeconal), while the wild type TaFAR only produced 1-hexadeconal (Fig. 2).
The profile of fatty alcohols produced by the engineered S. cerevisiae strains.
(A) The GC spectrum of fatty alcohols produced by the strains with wild type TaFAR (S. cerevisiae INVSc1 with plasmid pFAR) and the TaFAR with PTS1 (S. cerevisiae INVSc1 with plasmid pFARPTS01) or PTS2 (S. cerevisiae INVSc1 with plasmid pFAR PTS09) peptides. (B) The MS spectrum of the fatty alcohols produced by S. cerevisiae in comparison with the standards.
In order to select the PTS peptide with the highest efficiency of targeted expression of TaFAR, we next screened 7 PTS1 peptides (-SKL, -SKF -SFL, SKV, -LKL, -FKL, -SHL) and 4 PTS2 peptides (RV-X5-QL, KL-X5-QL, KI-X5-QL, KV-X5-HL) and evaluated the fatty alcohols production by GC-MS (Fig. 3). We found that all of the PTS peptides could lead to the production of medium chain fatty alcohols. Among the PTS1 peptides, TaFAR with SKL had the highest total fatty alcohols production, while among the PTS2 signals KL-QL led to the highest titers of fatty alcohols. We also fused both SKL and KL-X5-QL with TaFAR in C- and N- terminals. Such “double PTS peptide” led to a further 15% improvement of the total fatty alcohols production (Fig. 3). In addition, we used the fluorescence microscopy and confirmed that the “double PTS peptide”, in which TaFAR was tagged with SKL peptide on C-terminal and KL-X5-QL on N-terminal, was indeed expressed in the peroxisome (Fig. 4).
Fluorescence microscopy images of engineered strain PEXFLUO (S. cerevisiae BY4741 with plasmids pPEX14GFP, pFARCHERRY) showed the co-localization of the peroxisomal membrane protein PEX14 (fused with GFP) and TaFAR-mCherry tagged with SKL peptide on C-terminal and KL-QL on N-terminal.
PEX14 was used as a marker of the peroxisomal matrix. (A) The bright field image of S. cerevisiae cells. (B) The green fluorescence of PEX14 protein fused with GFP. (C) The red fluorescence of TaFAR-mCherry protein tagged with SKL and KL-QL. (D) The merged image of green fluorescence and red fluorescence. (E) The merged image of both fluorescence and bright field. The overlapped green fluorescence and red fluorescence indicated the same location of PEX14 and TaFAR-mCherry, which confirmed the targeted expression of TaFAR in peroxisomes.
Engineer the cargo and structural protein in peroxisome to enhance the production of medium chain fatty alcohols
Encouraged by the success on engineering S. cerevisiae to produce medium chain fatty alcohols, we next aimed to systemically engineer the cargo and structural proteins of peroxisome to improve the fatty alcohol production. In general, peroxisomal matrix proteins synthesized in the cytosol are bound by peroxisomal receptors – PEX523 in the case of PTS1 and PEX724 in the case of PTS2. These receptor–cargo complexes then move to the peroxisome membrane where they dock with protein sub-complexes that are in or on the membrane. In addition to PEX5 and PEX 7, PEX3 and PEX1925 are also essential for peroxisome biogenesis. PEX19 binds newly synthesized peroxisomal membrane proteins post-translationally and directs them to peroxisomes by engaging PEX3, a protein anchored in the peroxisome membrane26. Working together, PEX3 and PEX19 mediate the import of membrane proteins as well as the de novo formation of peroxisomes. Therefore, overexpression of PEX5, PEX7, PEX3 and PEX19 was hypothesized to enhance the targeted expression of TaFAR and thus increase the titer of fatty alcohols.
To evaluate the effects of PEX5, PEX7, PEX3 and PEX19 on production of medium chain fatty alcohols, we chose SKL and KL-QL as our model PTS1 and PTS2 peptides since they showed the most positive effects on producing medium chain fatty alcohols and tested different combinations of PTS peptides and overexpression of PEX proteins in batch fermentation. We found that the overexpression of PEX5 with PTS1 peptide could not improve fatty alcohol production, while on the other hand, the overexpression of PEX7 with PTS2 peptide increased fatty alcohol titer by 37.1%, reaching 782.7 ± 77.6 mg/L. The highest fatty alcohol titer was achieved in batch fermentation at 817.9 ± 39.1 mg/L when TaFAR was fused with PTS2 peptide and PEX7, PEX3 and PEX 19 were overexpressed simultaneously (Fig. 5). Considering the similar titer of the two constructs (i.e., PTS2 with overexpression of PEX7 and PTS2 with overexpression of PEX7, PEX3, PEX19), we chose the strain that overexpressed TaFAR fused with PTS2 and its receptor PEX7 as our target strain for further engineering to avoid the possible high metabolic burdens caused by overexpression of multiple proteins. We also evaluated the effects of knocking out a peripheral peroxisomal membrane protein PEX2727, which is a negative regulator in controlling peroxisome size and number, but did not observe the improvement of fatty alcohol titer. It was also worth noting that while the that PTS1&2 led to higher production of fatty alcohols than PTS1 (Fig. 3) when we screened different PTS peptides in INVSc1 strain, the PTS1&2 tagged TaFAR led to similar amount of fatty alcohols as that of PTS1 tagged TaFAR (Fig. 5) when we expressed the same plasmids in BY4741 strain. The different genetic background of S. cerevisiae INVSc1 and BY4741 strains could be the reason for such conflict.
Host engineering to enhance the production of fatty alcohols
In our previous studies8 on engineering S. cerevisiae to produce 1-hexadecanol in cytosol, we found that the highest titer of fatty alcohol was achieved when using the strategy of overexpressing ACC1 (a well-known bottleneck in fatty acid synthesis), knocking out RPD3 (a negative regulator for phospholipid synthesis) and introducing a heterologous ATP-dependent citrate lyase (ACL) to over-produce cytosolic acetyl-CoA. Inspired by the previous success on host engineering to enhance the production of long chain fatty alcohols, we used the same strategies to try to increase the production of medium chain fatty alcohols. We chose the strain expressing PEX7 and PTS2-TaFAR as our starting point and comprehensively evaluate the effects of overexpressing ACC1, knocking out RPD3 and the heterologous expression of ACL (Fig. 6). We found that overexpressing ACC1 could slightly increased the production of fatty alcohols, leading to the highest titer at 832.7 ± 73 mg/L. However, knocking out RPD3 and heterologous expression of ACL did not have beneficial effects on improving the fatty alcohol production. Interestingly, we could detect small amount of 1-tetradecanol (<3% of the total fatty alcohol) when engineering ACC1, RPD3 and ACL. As shown in the GC spectrum in Figure S1, A tiny peak of 1-tetradecanol could be detected in the strain with ACC1 over-expressed. However, no match of 1-tetradecanol could be found in the strain without overexpression of ACC1 when searching against the NIST MS library. Overall, the host engineering clearly indicated the shift of the bottlenecks for fatty alcohol production when compartmentalizing pathways in peroxisome.
Fed-batch fermentation for fatty alcohol production
With the strain overexpressing TaFAR tagged with a PTS peptide, PEX7 and ACC1 as our best strain (MFAOH37) to produce fatty alcohols, we next characterized the fatty alcohol production in fed-batch fermentation with different media. We identified two parameters (C/N ratio and K+ ions) that could potentially affect the fatty alcohol production based on previous reports28,29. In general, it has been found that the production of fatty acid and the derived chemicals could be enhanced30 at high C/N ratio (i.e., nitrogen limited). When K+ ions were added in the form of KOH and KCl to the culture medium, it was reported the change on the elemental balance could significantly enhance product accumulation in S. cerevisiae28. Therefore, we designed and tested four media: 1) normal SC medium with C/N ratio at 3:1; 2) nitrogen limited medium with C/N ratio at 50:1; 3) normal SC medium (C/N = 3:1) with K+ (a mixture of 40 mM KCl and 10 mM KOH); and 4) nitrogen limited medium (C/N = 50:1) with K+ (a mixture of 40 mM KCl and 10 mM KOH).
In fed-batch fermentation, we used a resting cell fermentation, in which the cells had marginal net growth rate but rather served as biocatalysts to convert glucose to fatty alcohols. We found that the titer of fatty alcohols using the normal SC medium was 0.98 ± 0.07 g/L; the titer of fatty alcohols using the normal SC medium with K+ supplement was 1.11 ± 0.07 g/L and the titer of fatty alcohols using the nitrogen limited SC medium with K+ supplement was 1.02 ± 0.08 g/L (Figure S2). The nitrogen limited medium without K+ supplied in the fed batch fermentation led to the highest the titer of total fatty alcohols at 1.30 ± 0.10 g/L with 6.9% 1-decanol, 27.5% 1-dodecanol, 2.9% 1-tetradecanol and 62.7% 1-hexadecanol, which represents the highest fatty alcohol titer reported in S. cerevisiae (Fig. 7). However, a drop of fatty alcohol concentration was observed together with the OD decrease at 36 h (Fig. 7), which was likely due to the starvation caused by nutrient limitation. The fermentation results confirmed the positive effects of nitrogen limitation and K+ supplement on fatty alcohol production in S. cerevisiae. Nevertheless, no synergistic effects were found between nitrogen limitation and K+ supplement.
Fed-batch fermentation of strain MFAOH37 (S. cerevisiae INVSc1 with plasmids pFARPEX and pACC1) using the nitrogen limited (C:N = 50:1) SC medium.
(A) The production of fatty alcohols. (B) Sugar consumption and byproduct production. Black square: OD600; Blue circle: the total glucose consumed; Red triangle: The ethanol produced.
Discussion
Pathway compartmentalization has been recently demonstrated as a novel approach for enhancing the production of a series of chemicals, including isobutanol31, isopentanol31, 2-methyl-1-butanol31, itaconic acid32, terpenoids33, acetoin34 and fatty acids35. Compared to the conventional engineering of cytosolic pathways, the compartmentalizing of pathways has several advantages such as favoring faster reaction rates due to the concentrated precursors in the smaller volume of compartments and avoiding repressive regulatory responses and competing pathways31. Till now, most of the studies on pathway compartmentalization focused on mitochondria. In this study, we demonstrated that peroxisome could be another promising candidate to compartmentalize metabolic pathways for synthesis of novel chemicals, especially fatty acid-derived chemicals such as medium chain fatty alcohols, considering the abundance and versatility of fatty acyl-CoA generated from the beta-oxidation in peroxisome. Another unique advantage of using peroxisome for pathway compartmentalization is the easiness in manipulating the peroxisome as a whole. As discovered in this study, the modification of PTS peptides as well as cargo and structural peroxisomal proteins led to the most significant improvement of medium chain fatty alcohols production (from 0 mg/L to ~800 mg/L). On the other hand, the conventional host engineering strategy only marginally improved the peroxisomal production of fatty alcohol. This indicated a paradigm shift when adopting the strategy of pathway compartmentalization with a new set of bottlenecks arising. Compared to the study on functionality of cytosolic pathways, our understanding on peroxisomal pathways is still limited. However, with the in-depth analysis using systems biology approaches (e.g., -omics analysis), we can envision a much better mechanistic understanding on peroxisomal pathways and use such knowledge to further improve the biochemical production in peroxisome.
During the engineering of peroxisomal pathways, we found that 1-hexadecanol was still produced as the main products (>60%) despite of targeted expression of TaFAR. This was not unexpected since the main fatty acid produced by S. cerevisiae was palmitic acid, which could generate palmitoyl-CoA as the most abundant fatty acyl-CoA in peroxisome. Therefore, when TaFAR was expressed in peroxisome, it would mainly use palmitoyl-CoA to produce 1-hexadecanol. Interestingly, we could not detect 1-tetradecanol when engineering the PTS peptides or peroxisomal proteins, but observed tiny amount of 1-tetradecanol during host engineering when enhancing fatty acid synthesis. This could be attributed to the fact that the overproduction of fatty acids led to more tetradecanoic acid synthesized, which was further diverted to peroxisome and used as the precursor for 1-tetradecanol production. Also, the substrate specificity of TaFAR could be another reason for the low concentration of 1-tetradecanol. It was worth noting that some other FAR enzymes such as AdFAR1 was reported to prefer medium-chain fatty acyl-CoAs36. In fact, as shown in Figure S3, we have characterized the profile of fatty alcohol produced by AdFAR1. In general, we constructed a new pAdFARPEX plasmid in the same way as pFARPEX plasmid, in which the AdFAR was expressed under constitutive TEF1 promoter and tagged with the PTS2 peptide (KL-X5-QL) and the PEX7 was expressed under constitutive TPI1 promoter. The reason that we chose this combination of this PTS2 peptide and PEX7 is that they led to the highest fatty alcohol titer when using TaFAR. By transforming the pAdFARPEX plasmid into BY4741 strain, it was shown that the AdFAR led to the similar titer of fatty alcohols as that of TaFAR (812 ± 92 mg/L in AdFAR and 783 ± 78 mg/L in TaFAR), but different profiles of fatty alcohols (Figure S3). In addition to 1-decanol, 1-dodecanol and 1-hexadecanol, 1-tetradecanol was also produced (8.1% of total fatty alcohols produced) when expressing AdFAR1 and culturing the recombinant yeast in SC medium for 24h in batch fermentation. This indicates by expressing different FARs, the profile of fatty alcohols produced by yeast could vary.
In summary, by compartmentalizing fatty alcohol synthesis pathway in peroxisome, we successfully engineered S. cerevisiae to produce medium chain fatty alcohols for the first time. The current production of fatty alcohols reached 1.3 g/L in titer, 36 mg/L/h in productivity and 43 mg/g in yield. Recently, it was found that when coupled with the enhanced precursor supply, the over-production of NADPH could lead to over five-fold increase of 3-hydroxypropionic acid production37. Therefore, it is possible that by coupling with NADPH overproduction and the pathway compartmentalization, the production of fatty alcohols could be further improved. We are currently trying to engineering the cytosolic and peroxisomal NADPH supply, together with our established strategies of targeted expression of TaFAR, overexpressing PEX7 and ACC1 to further increase the production of fatty alcohols.
Materials and Methods
Yeast strains, media and transformation
The yeast strains used in this study were derived from both BY4741 and INVSc1. The cells were stored in a 15% v/v glycerol solution at −80 °C. E. coli DH5α strain was used for maintaining and amplifying plasmids and recombinant strains were cultured at 37 °C in Luria-Bertani (LB) broth. Ampicillin at a concentration of 100 μg/mL was added to the LB medium when required. The BY4741 and INVSc1 strains were cultured in YPAD medium. Yeast cells were transformed with plasmids using the LiAc/PEG method as described previously38. For selection of the yeast transformants, a synthetic complete (SC) medium was used, which contains 0.17% yeast nitrogen base, 0.5% ammonium sulfate and the appropriate amino acid dropout mix (MP Biomedicals, Solon, OH). A single colony was picked and cultured in 5 mL SC media containing 20 g/L glucose. The cells were cultured at 30 °C in disposable culture tubes shaken at 250 rpm for 2 days.
Plasmid construction
An established, yeast homologous recombination-based method, DNA assembler, was used to construct the recombinant plasmids39. Basically, DNA fragments sharing homologous regions to adjacent DNA fragments were co-transformed into S. cerevisiae along with linearized backbone to assemble several elements in a single step40. Oligonucleotides used in this study were listed in Table S1 and the recombinant plasmids constructed in this study were listed in Table 1.
Fluorescence microscopy for visualizing enzyme localization
To confirm TaFAR was being transported into the peroxisome fluorescence microscopy was used. Two fluorescent proteins were used in the imaging: Green Fluorescent Protein (GFP) and mCherry. To highlight the membrane of the peroxisome, the GFP was fused to PEX14, a protein known to be embedded in the peroxisomal membrane. To highlight the interior of the peroxisome, TaFAR-mCherry was tagged with SKL peptide on C-terminal and KL-X5-QL on N-terminal. Yeast cells were transformed using the methods previously described with the plasmids containing PEX14-GFP and TaFAR-mCherry and grown in 3 mL of SC media containing all appropriate amino acids, with 20 g/L glucose for 2 days. Then, 1 mL of the yeast cells were centrifuged, washed and re-suspended in 1 mL distilled water. 5 μL of the yeast cells were mixed with 10 μL of 1% melted agarose and then overlaid with a glass cover slip to fix the cells in place. To visualize the PEX14-GFP protein, the excite wavelength was 488 nm and emission wavelength was 525 nm. To visualize the TaFAR-mCherry protein, the excitation wavelength was 587 nm and emission wavelength was 610 nm. The overlapped green fluorescence and red fluorescence indicated the same location of PEX14 and TaFAR and confirmed the targeted expression of TaFAR in peroxisomes.
Measurement of fatty alcohol production
The fatty alcohol production of the engineering yeast cells was screened in triplicate following a published protocol8. In general, to screen for the production of fatty alcohols, the engineered yeast strains were pre-cultured in disposable culture tubes with 3 mL of synthetic complete (SC) media with all appropriate amino acids and 2% glucose for three days until saturation. Then, 0.5 mL of the cells were transferred to glass tubes containing 4.5 mL of fresh SC media overlaid with 10% (v/v) dodecane to extract the fatty alcohols. The concentrations of fatty alcohols were quantified at 48 h. The glass tubes of yeast culture were allowed to sit for several minutes until the organic layers were clearly visible. Then, 3 μL of dodecane was extracted from the organic layer and diluted 100 times using ethyl acetate containing 20 mg/L tridecane as the internal standard. Of this mixture, 60 μL was added to 40 μL BSTFA with 1% TMCS solution and incubated at 70 °C for 30 minutes. This solution was centrifuged and analyzed by GC-MS (Shimadzu GC-MS-QP2010) with a DB-Wax column with 0.25 μm film thickness, 0.25 mm diameter and 30 m length (Agilent Inc., Palo Alto, CA). The GC program was as follows: an initial temperature of 50 °C was maintained for 1.5 min, followed by ramping to 180 °C at a rate of 25 °C/min. The temperature was then ramped to 250 °C at a rate of 10 °C/min, where the temperature was held for 3 min. The final titer of fatty alcohol (mg/L) was calculated as 10 × GC data. All the titers we reported were the results that were adjusted to final titer (i.e., mg/L fatty alcohol produced in the fermenter). Since we were not sure what type of fatty alcohols (e.g., odd chain fatty alcohols) could be produced when expressing FARs in peroxisome, we decided to choose tridecane instead of tridecanol or pentadecanol as our internal standard. We found a very linear correlation between the fatty alcohol concentration (i.e., 1-decanol, 1-dodecanol, 1-tetradecanol and 1-hexadecanol) and the ratio of GC peak area of tridecane to fatty alcohol standards, with the R2 reaching 0.90 or higher.
Medium optimization and fed-batch fermentation
For fed-batch fermentation, the engineered yeast strains were first grown in 40 mL SC media including all appropriate amino acids, with 20 g/L glucose and allowed to grow for 48 h. Then, 25 mL of the cell cultures were centrifuged, washed twice with sterilized water, re-suspended with 5 mL of SC media in glass tubes and overlaid with 10% dodecane to extract the fatty alcohol. The correlation between the OD measurement in our lab and the DCW was found as 1 OD ≈ 4 g/L DCW. Therefore, in the fed-batch fermentation, there were 8~10 g/L DCW. This number (i.e., 8~10 g/L DCW) was still lower than the theoretical biomass yield from glucose (i.e., ~0.5 g/g). This is mainly because of the metabolic burden when expressing multiple plasmids, which was found to slow down the cell growth in this study. Therefore, when we harvested the cells at 48 h from the batch culture, the OD of the cells had not yet reached the saturation. Fermentations were performed in triplicate and tested every 12 h. Fatty alcohol levels were measured using the GC-MS procedure described above, remaining glucose levels and ethanol production were measured by HPLC and OD was measured using the plate reader. Additional glucose (0.5 mL with concentration of 200 g/L) and dodecane (0.05 mL) were fed every 24 h. To optimize the medium in the fed-batch fermentation, four different types of media were used to evaluate fatty alcohol production: 1) Normal SC medium (control condition with C:N ratio at 3:1), with the glucose concentration at 20 g/L and yeast nitrogen base (without amino acids) concentration at 6.7 g/L; 2) Nitrogen limited SC medium with C:N ratio at 50:1, with the glucose concentration at 20 g/L and yeast nitrogen base (without amino acids) concentration at 0.4 g/L; 3) Normal SC medium with supplement of 40 mM KCl and 10 mM KOH; and 4) Nitrogen limited SC medium with C:N ratio at 50:1, supplement of 40 mM KCl and 10 mM KOH. Triplicates of each type of medium were performed. The same fed-batch procedure and protocol of fatty alcohol measurement were used as described above.
Additional Information
How to cite this article: Sheng, J. et al. Pathway Compartmentalization in Peroxisome of Saccharomyces cerevisiae to Produce Versatile Medium Chain Fatty Alcohols. Sci. Rep. 6, 26884; doi: 10.1038/srep26884 (2016).
References
Mudge, S. M., Belanger, S. E. & Nielsen, A. M. (The Royal Society of Chemistry, Cambridge, UK, 2008).
Youngquist, J. T. et al. Production of medium chain length fatty alcohols from glucose in Escherichia coli. Metab Eng 20, 177–186, doi: 10.1016/j.ymben.2013.10.006 (2013).
Rupilius, W. & Ahmad, S. Palm oil and palm kernel oil as raw materials for basic oleochemicals and biodiesel. Eur J Lipid Sci Technol 109, 433–439 (2007).
Fitzherbert, E. et al. How will oil palm expansion affect biodiversity? Trends Ecol Evol 23, 538–545 (2008).
Hill, J., Nelson, E., Tilman, D., Polasky, S. & Tiffany, D. Environmental, economic and energetic costs and benefits of biodiesel and ethanol biofuels. Proceedings of the National Academy of Sciences of the United States of America 103, 11206–11210, doi: 10.1073/pnas.0604600103 (2006).
Zhang, F., Rodriguez, S. & Keasling, J. D. Metabolic engineering of microbial pathways for advanced biofuels production. Current opinion in biotechnology 22, 775–783, doi: 10.1016/j.copbio.2011.04.024 (2011).
Runguphan, W. & Keasling, J. D. Metabolic engineering of Saccharomyces cerevisiae for production of fatty acid-derived biofuels and chemicals. Metab Eng 21, 103–113, doi: 10.1016/j.ymben.2013.07.003 (2014).
Feng, X., Lian, J. & Zhao, H. Metabolic engineering of Saccharomyces cerevisiae to improve 1-hexadecanol production. Metab Eng 27, 10–19, doi: 10.1016/j.ymben.2014.10.001 (2015).
Akhtar, M., Turner, N. & Jones, P. Carboxylic acid reductase is a versatile enzyme for the conversion of fatty acids into fuels and chemical commodities. Proc Natl Acad Sci 110, 87–92 (2013).
Zheng, Y. et al. Optimization of fatty alcohol biosynthesis pathway for selectively enhanced production of C12/14 and C16/18 fatty alcohols in engineered Escherichia coli. Microbial cell factories 11, 65 (2012).
Reiser, S. & Somerville, C. Isolation of mutants of Acinetobacter calcoaceticus deficient in wax ester synthesis and complementation of one mutation with a gene encoding a fatty acyl coenzyme A reductase. J Bacteriol 179, (1997).
Hong, K. K. & Nielsen, J. Metabolic engineering of Saccharomyces cerevisiae: a key cell factory platform for future biorefineries. Cellular and molecular life sciences : CMLS 69, 2671–2690, doi: 10.1007/s00018-012-0945-1 (2012).
Tang, X., Lee, J. & Chen, W. N. Engineering the fatty acid metabolic pathway in Saccharomyces cerevisiae for advanced biofuel production. Metabolic Engineering Communications 2, 58–66, doi: 10.1016/j.meteno.2015.06.005 (2015).
Fernandez-Moya, R., Leber, C., Cardenas, J. & Da Silva, N. A. Functional replacement of the Saccharomyces cerevisiae fatty acid synthase with a bacterial type II system allows flexible product profiles. Biotechnol Bioeng 112, 2618–2623, doi: 10.1002/bit.25679 (2015).
Leber, C. & Da Silva, N. A. Engineering of Saccharomyces cerevisiae for the synthesis of short chain fatty acids. Biotechnol Bioeng 111, 347–358, doi: 10.1002/bit.25021 (2014).
Leber, C., Choi, J. W., Polson, B. & Da Silva, N. A. Disrupted short chain specific beta-oxidation and improved synthase expression increase synthesis of short chain fatty acids in Saccharomyces cerevisiae. Biotechnol Bioeng, doi: 10.1002/bit.25839 (2015).
Schulz, H. Beta oxidation of fatty acids. Biochimica et Biophysica Acta (BBA) - Lipids and Lipid Metabolism 1081, 109–120, doi: 10.1016/0005-2760(91)90015-A (1991).
van der Klei, I. J. & Veenhuis, M. Yeast peroxisomes: function and biogenesis of a versatile cell organelle. Trends Microbiol 5, 502–509, doi: 10.1016/s0966-842x(97)01156-6 (1997).
Rucktaschel, R., Girzalsky, W. & Erdmann, R. Protein import machineries of peroxisomes. Biochim Biophys Acta 1808, 892–900, doi: 10.1016/j.bbamem.2010.07.020 (2011).
Gould, S. J., Keller, G. A. & Subramani, S. Identification of peroxisomal targeting signals located at the carboxy terminus of four peroxisomal proteins. The Journal of cell biology 107, 897–905 (1988).
Elgersma, Y. et al. Analysis of the carboxyl-terminal peroxisomal targeting signal 1 in a homologous context in Saccharomyces cerevisiae. J Biol Chem 271, 26375–26382 (1996).
Schafer, A., Kerssen, D., Veenhuis, M., Kunau, W. H. & Schliebs, W. Functional similarity between the peroxisomal PTS2 receptor binding protein Pex18p and the N-terminal half of the PTS1 receptor Pex5p. Molecular and cellular biology 24, 8895–8906, doi: 10.1128/mcb.24.20.8895-8906.2004 (2004).
Miyata, N. & Fujiki, Y. Shuttling mechanism of peroxisome targeting signal type 1 receptor Pex5: ATP-independent import and ATP-dependent export. Molecular and cellular biology 25, 10822–10832, doi: 10.1128/mcb.25.24.10822-10832.2005 (2005).
Kunze, M. et al. Structural requirements for interaction of peroxisomal targeting signal 2 and its receptor PEX7. J Biol Chem 286, 45048–45062, doi: 10.1074/jbc.M111.301853 (2011).
Schmidt, F. et al. Insights into peroxisome function from the structure of PEX3 in complex with a soluble fragment of PEX19. J Biol Chem 285, 25410–25417, doi: 10.1074/jbc.M110.138503 (2010).
Hettema, E. H., Girzalsky, W., van den Berg, M., Erdmann, R. & Distel, B. Saccharomyces cerevisiae Pex3p and Pex19p are required for proper localization and stability of peroxisomal membrane proteins. The EMBO Journal 19, 223–233, doi: 10.1093/emboj/19.2.223 (2000).
Yan, M., Rayapuram, N. & Subramani, S. The control of peroxisome number and size during division and proliferation. Current opinion in cell biology 17, 376–383, doi: 10.1016/j.ceb.2005.06.003 (2005).
Lam, F. H., Ghaderi, A., Fink, G. R. & Stephanopoulos, G. Engineering alcohol tolerance in yeast. Science 346, 71–75, doi: 10.1126/science.1257859 (2014).
Thompson, R. A. & Trinh, C. T. Enhancing fatty acid ethyl ester production in Saccharomyces cerevisiae through metabolic engineering and medium optimization. Biotechnology and bioengineering 111, 2200–2208, doi: 10.1002/bit.25292 (2014).
Kamisaka, Y., Tomita, N., Kimura, K., Kainou, K. & Uemura, H. DGA1 (diacylglycerol acyltransferase gene) overexpression and leucine biosynthesis significantly increase lipid accumulation in the Δsnf2 disruptant of Saccharomyces cerevisiae. The Biochemical journal 408, 61–68, doi: 10.1042/BJ20070449 (2007).
Avalos, J. L., Fink, G. R. & Stephanopoulos, G. Compartmentalization of metabolic pathways in yeast mitochondria improves the production of branched-chain alcohols. Nat Biotech 31, 335–341 (2013).
Blumhoff, M. L., Steiger, M. G., Mattanovich, D. & Sauer, M. Targeting enzymes to the right compartment: Metabolic engineering for itaconic acid production by Aspergillus niger. Metabolic engineering 19, 26–32, doi: 10.1016/j.ymben.2013.05.003 (2013).
Farhi, M. et al. Harnessing yeast subcellular compartments for the production of plant terpenoids. Metabolic engineering 13, 474–481, doi: 10.1016/j.ymben.2011.05.001 (2011).
Li, S., Liu, L. & Chen, J. Compartmentalizing metabolic pathway in Candida glabrata for acetoin production. Metabolic engineering 28, 1–7, doi: 10.1016/j.ymben.2014.11.008 (2015).
Chen, L., Zhang, J. & Chen, W. N. Engineering the Saccharomyces cerevisiae beta-oxidation pathway to increase medium chain fatty acid production as potential biofuel. PLoS One 9, e84853, doi: 10.1371/journal.pone.0084853 (2014).
Hellenbrand, J., Biester, E.-M., Gruber, J., Hamberg, M. & Frentzen, M. Fatty acyl-CoA reductases of birds. BMC biochemistry 12:64 (2011).
Chen, Y., Bao, J., Kim, I., Siewers, V. & Nielsen, J. Coupled incremental precursor and co-factor supply improves 3-hydroxypropionic acid production in Saccharomyces cerevisiae. Metab Eng 22C, 104–109 (2014).
Gietz, R. & Schiestl, R. High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nature protocols 2, 31–34 (2007).
Shao, Z., Zhao, H. & Zhao, H. DNA assembler, an in vivo genetic method for rapid construction of biochemical pathways. Nucleic Acids Res 27, e16 (2009).
Shao, Z., Luo, Y. & Zhao, H. Rapid characterization and engineering of natural product biosynthetic pathways via DNA assembler. Molecular bioSystems 7, 1056–1059, doi: 10.1039/c0mb00338g (2011).
Lian, J., Si, T., Nair, N. U. & Zhao, H. Design and construction of acetyl-CoA overproducing Saccharomyces cerevisiae strains. Metab Eng 24, 139–149, doi: 10.1016/j.ymben.2014.05.010 (2014).
Acknowledgements
We thank the writing center in Virginia Tech for improving the language of the paper. This study was supported by start-up fund (#175323) from Virginia Tech (X.F.).
Author information
Authors and Affiliations
Contributions
X.F. conceived the project. J.S. and X.F. designed the research and coordinated the project. J.S. and J.S. conducted metabolic engineering experiments and analyzed the data. J.S., J.S. and X.F. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sheng, J., Stevens, J. & Feng, X. Pathway Compartmentalization in Peroxisome of Saccharomyces cerevisiae to Produce Versatile Medium Chain Fatty Alcohols. Sci Rep 6, 26884 (2016). https://doi.org/10.1038/srep26884
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep26884
This article is cited by
-
Microbial synthesis of long-chain α-alkenes from methanol by engineering Pichia pastoris
Bioresources and Bioprocessing (2022)
-
Spatial–temporal regulation of fatty alcohol biosynthesis in yeast
Biotechnology for Biofuels and Bioproducts (2022)
-
High production of fatty alcohols in Yarrowia lipolytica by coordination with glycolysis
Science China Chemistry (2019)
-
An engineered fatty acid synthase combined with a carboxylic acid reductase enables de novo production of 1-octanol in Saccharomyces cerevisiae
Biotechnology for Biofuels (2018)
-
Biosynthesis of nervonic acid and perspectives for its production by microalgae and other microorganisms
Applied Microbiology and Biotechnology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.