Abstract
Recently, pyruvate kinase M2 (PKM2) has been implicated in the progression of certain cancers and might play pivotal roles in the formation of malignancy. However, the role of PKM2 in gallbladder cancer had not been well investigated. This study analyzed associations between PKM2 expression status with various clinical and pathologic parameters in a large cohort of gallbladder cancer (GBC) patients from a long term follow up results. The expression level of pyruvate kinase isotypes in GBC tissues and their adjacent normal gallbladder tissues were estimated by qRT-PCR and Western blot. PKM2 mRNA level were significantly high in gallbladder cancer tissues than in adjacent noncancerous tissues (P < 0.001). High expression of the PKM2 was detected in 55.71% paraffin-embedded GBC tissue. The high PKM2 expression was independently associated with poorer overall survival in patients with GBC (median survival 11.9 vs 30.1 months; hazard ratio 2.79; 95% CI = 1.18 to 6.55; P = 0.02). These findings indicated elevated expression of PKM2 is a prognostic factor for poor GBC clinical outcomes, implied involving of PKM2 in GBC progression.
Similar content being viewed by others
Introduction
Gallbladder cancer (GBC) is the most frequently occurring malignancy of the biliary system, it is highly aggressive and associated with an extremely poor prognosis1. Radical surgical resection is the only potentially curative therapy for GBC2,3. However, metastasis prevents GBC patients from obtaining long-term curative effects. The 5-year survival rate of GBC patients who underwent radical surgical resection with negative nodal involvement greatly differed from that of patients with lymph nodes metastasis or with local invasion of adjacent organs duration4.
Moreover, the prognosis of patients with GBC were varied diversely. Clinically, patients with similar clinico-pathological tumor stage, especially in the early stage, who accepted the same treatment, exhibit great difference in prognosis, indicating their innate different biological characteristics. Locating genetic alterations of GBC may help to estimate the prognosis and to identify potential targets5. Several relevant target genes, such as Sox4, NLK, and ERBB3, have recently been reported to have prognostic value in GBC6,7,8. However, the molecular mechanisms of GBC are far from clear; further efforts to discover the frequency of altered gene expression might help in understanding the intrinsic characteristics of GBC and the consequent difference in clinical manifestations.
Recently, pyruvate kinase (PK) M2 (PKM2) was identified as a clinically relevant prognostic marker for gastric cancer, tongue cancer, hepatic cancer and esophageal squamous cell carcinoma9,10,11,12. PKM2 overexpression is believed to be closely related to the cancer cell-specific glucose metabolism characteristics of aerobic glycolysis, or the Warburg effect13,14. A recent study found that PKM2 has nuclear transcriptional regulator activity, indicating a complicated cellular functional activity15. However, PKM2 had distinct functions concerning different carcinoma diseases reported. Furthermore, the function of PKM2 in GBC has not been well studied, which prompted us to investigate the clinical pathological significance of PKM2 and its function in GBC.
Results
PKM2 protein was highly expressed in majority of GBC tissues and in GBC cell lines
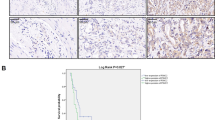
Paraffin-embedded tissue specimens of 70 GBC tissues and 25 non-cancerous gallbladder tissues were obtained and built into a TMA. Positive stain of the PKM2 protein was found in part of normal gallbladder tissues and majority of the GBC tissues. PKM2 protein was observed in both the cytoplasm and nucleus of the gallbladder cancer cells (Fig. 1A–E).
(A) Well-differentiated adenocarcinoma exhibited low PKM2 staining and was given a score of 2. (B) Moderately differentiated adenocarcinoma exhibited moderate PKM2 expression and was given a score of 3. (C) Poorly differentiated adenocarcinoma exhibited strong PKM2 expression and was given a score of 4. (D) Gallbladder tissue was used as the negative control as it exhibited negative staining for PKM2 and was given a score of 0. (E) Gallbladder tissue which were positive staining for PKM2 and was given a score of 1 (F) Western blot with an antiPKM2 antibody revealed positive expression of PKM2 in GBC-SD, NOZ and SGC-996 cells. The red arrow in (A–C,E) point to the positive stained cell which were dyed brown. Original magnification: ×200.
PKM2 was significantly highly expressed in the tissue samples of advanced stage GBC
The immunohistochemistry score for PKM2 was divided into low expression and high expression according to the intensity and proportion of PKM2 staining among tumor cells as described in the Materials and Methods. The clinico-pathological significance of PKM2 expression was examined based on the immunohistochemical staining pattern of this protein. The relationship between the PKM2 protein expression and clinico-pathological characteristics is summarized in Table 1. Of the 70 GBC patients, 39 (55.7%) had high PKM2 expression. The protein expression of PKM2 was significantly associated with pathological stage (Fig. 2A–D), whereas other characteristics, including histological grade and neurovascular infiltration, were not. We divided the GBC patients into two groups: an advanced stage group (TNM stage III–IV) and an early stage group (TNM stage I–II); the PKM2 expression, as measured by Image-Pro Plus software 6.0, was relatively high in the advanced group (P = 0.008, Fig. 2E). However, 0.16% (4/25) cases of normal gallbladder tissue were found PKM2 high expression. It was significantly low portion of high PKM2 expression were found in normal gallbladder tissues than in GBC tissues (P = 0.0007, Fig. 2F). This finding suggested that high PKM2 expression in GBC was related to carcinogenesis and malignant outcomes.
(A–D) The percentage of patients with low PKM2 expression decreased gradually as the disease progressed from pT stage 1 to 4 and pN stage 0 to 3 and when non-metastatic disease progressed to metastatic disease; percentages are also summarized according to TNM stages I to IV. (E) Comparison of the dot distribution of the optical density of PKM2 protein staining in gallbladder tumor tissues according to the TNM stage (TNM I–II are regarded as early stage, and III–IV are regarded as advanced stage; ***P = 0.0008, Mann-Whitney U test). (F) Comparison of the dot distribution of the optical density of PKM2 protein staining in gallbladder tumor tissues with normal gallbladder tissues ***P = 0.0007, Mann-Whitney U test).
PKM2 expression level was correlated with OS and DFS significantly in GBC patients
All patients had follow-up records for 1–72 months, with a median of 17.4 (23 ± 18.8) months. The OS and DFS were analyzed with PKM2 expression level and the clinico-pathologic factors using the Kaplan-Meier method and Cox’s proportional hazards regression model, the significance was determined by the log-rank test (Table 2). For Kaplan-Meier method, estimation showed that lymph node metastasis condition, advanced TNM stage and high PKM2 immuno-reactivity level were significantly associated with worse OS and a short time relapse. The group with high PKM2 expression presented with a significantly poorer prognosis for OS (P = 0.0001, log-rank test) (Fig. 3A) and DFS (P = 0.0003, log-rank test) (Fig. 3B) than the low expression group. We numerically scored the combined risk factor according to PKM2 expression (0 represents low expression and 1 represents high expression) and TNM stage (1 represents early stage and 2 represents advanced stage); the combined risk factor score was calculated using the PKM2 expression level and TNM stage classifications. The GBC patients were divided into three groups: a low risk group (score 0), a middle risk group (score 1) and a high risk group score (score 2). Kaplan-Meier analysis showed that the high risk group had an extremely poor OS compared with the middle risk group, and the OS of the middle risk group was worse than the low risk group (P = 0.0004, log-rank test) (Fig. 3C). The median OS of high risk patients was 11.6 months significantly short than 41.1 months of low risk patients (P = 0.0002, log-rank test). Although the DFS analysis showed a similarity between the high and middle risk groups, both were worse than the low risk group patients (P = 0.002, log-rank test) (Fig. 3D).
(A,B) Kaplan-Meier survival estimates revealed that high PKM2 immunoreactivity in gallbladder adenocarcinoma was significantly associated with worse overall survival and worse disease-free survival according to the dichotomy of the extent of PKM2 staining intensity. (C,D) Kaplan-Meier survival estimates showed that the trichotomy of the combined risk scores was significantly associated with corresponding overall survival prognosis. A high-risk score represents worse overall survival (OS) and disease-free survival (DFS), whereas a low-risk score represents relatively improved OS and DFS rates. However, a median-risk score predicts median OS but worse DFS.
PKM2 protein level is a potential prognostic factor in GBC patients
For the Cox proportional hazard regression model, univariate analyses demonstrated that PKM2 protein expression, pN category, distant metastasis and the stage of TNM classification were significantly associated with cause-specific survival (Table 2). When the data were stratified for multivariate analysis using both forward and backward stepwise Cox regression procedures, the PKM2 immunoreactivity in tumor cells remained significant at P = 0.001, estimated hazard ratio (HR) 2.786 (95% CI 1.184–6.553) for OS and P = 0.019, HR 4.797 (95% CI 1.922–11.972) for DFS in all patients, suggesting that PKM2 immunoreactivity is an independent predictor of OS and DFS. Multivariate Cox analysis of the risk factors was conducted using the clinical and pathological data; the PKM2 expression level, the combined risk factor, and the predictive values of OS and disease-free relapse interval in GBC patients were compared using the forest plot show in Fig. 4A,B. Generally, pN stage, PKM2 level and the combined risk factor had prognostic value in predicting OS and DFS; in particular, PKM2 showed outstanding prognostic value predicting OS and DFS in early stage patients; the combined risk factor had superior predictive value in predicting DFS compared with the other factors. The predictive value of the combined risk factor was very effective for stratifying the GBC patients into three groups, favorable prognosis (average OS = 41.1 months), middle prognosis (19.9 months), and poor prognosis (11.6 months), according to the OS and the dichotomy-classified favorable DFS (37.6 months) and poor DFS (13.5 months), see Fig. 4C–F.
(A) A multivariate Cox analysis of the clinical and pathological factors showed predictive value with respect to OS in gallbladder cancer patients. Generally, PKM2 and pN stage showed prognostic value for predicting OS. Specifically, PKM2 demonstrated prominent prognostic value in patients with early stage gallbladder cancer (i.e., TNM stages I and II), and the combination of the TNM stages and the PKM2 staining demonstrated better predictive value than either alone. (B) A multivariate Cox analysis of the clinical and pathological factors demonstrated predictive value with respect to DFS in gallbladder cancer patients. Generally, PKM2 and pN stage showed prognostic value for predicting DFS. Specifically, PKM2 exhibited better prognostic value in patients with early stage gallbladder cancer (i.e., TNM stages I and II). The combination of TNM stage and PKM2 staining showed better predictive value than either alone. (C–F) A rank-sum test compared PKM2 expression levels and the combined risk factors to predict OS and DFS.
To further exam the predictive value of the PKM2 expression in different TNM stage GBC patients, we stratified the GBC patients into an early stage group and an advanced stage group. We analyzed association between PKM2 expression levels and OS using the COX regression model in each group and in the whole population (Table 3); the HR was 12.43 (95% CI 1.26–122.96) in early stage patients, whereas the HR was 2.64 (95% CI 1.08–6.45) in advanced stage patients and 2.79 (95% CI 1.18–6.55) in the whole population (see Fig. 2E). The estimated HR for DFS was 13.30 (95% CI 1.43–124.07) in the early stage group, 2.41 (95% CI 0.95–6.10) in the advanced stage patients, and 4.80 (95% CI 1.92–11.97) in the whole population, Fig. 4B. These data suggest that the PKM2 expression level had better predictive efficacy particularly in early stage GBC patients.
PKM2 level was increased while PKM1 was decreased in GBC tissues
The expression of pyruvate kinase isotypes was examined in 11 pair of gallbladder cancer tissue and their adjacent normal gallbladder tissue by qPCR. We found that M1, M2 and L type PK isotype mRNAs were expressed in gallbladder cancer tissue as well as in noncancerous gallbladder tissues. The major PK isotype was PKM1 in noncancerous gallbladder tissues (P = 0.0022, Friedman test) and PKM2 in cancer tissues (P < 0.0001, Friedman test). PKM2 mRNA was significantly highly expressed in gallbladder cancer tissues than in adjacent noncancerous tissues (P = 0.001, Wilcoxon test), while PKM1 mRNA level was less in gallbladder cancer tissues than in noncancerous gallbladder tissues (P = 0.0029, Wilcoxon test), Fig. 5A. No significant difference was observed between the expression level of PKR and PKL mRNA (P = 0.70 and P = 0.28 respectively, Wilcoxon test). We further examined the protein expression of isotypes of pyruvate kinase by Western blot and immunohistochemistry. It was identified that PKM2 protein level were enhanced while PKM1 decreased in 5 GBC patients’ fresh tissue samples, Fig. 5B. The anti-PKM1, anti-PKM2 and anti-PKLR antibody were used to examine the pyruvate isotypes protein expression in paraffin-embedded GBC tissues, PKM1, PKM2 and PKLR were positively stained in the GBC tissues while it was strong stained while using anti-PKM2 antibody, Fig. 5C. We further compare the PKM1 expression using TMA IHC, the expression of PKM1 were significantly lower in GBC tissues than in normal gallbladder tissues, (P = 0.0011, Mann–Whitney test), Fig. 5D. However, there were no significant correlation of PKM1 expression with TNM stage, P > 0.05.
(A) Compare the expression of PKM1 and PKM2 mRNA in gallbladder cancer tissue with adjacent normal gallbladder tissues. (B) Compare the protein level of pyruvate kinase isotypes in 5 paired gallbladder cancer tissues and normal counterpart by Western blot using the indicated protein antibody. (C) Compare the protein level of pyruvate kinase isotypes in paraffin-embedded gallbladder cancer tissues by immunohistochemistry using the indicated protein antibody. The red arrow in the figure point to the positive stained cell which were dyed brown. Original magnification: ×200. (D) Compare the intensity of PKM1 in the tissue microarray samples by immunohistochemistry. Normal, normal gallbladder tissues; GBC, gallbladder cancer.
Methods
Study population
The patients with suspected GBC who were aged 18–75 years old and without severe comorbidities or blood abnormalities, and had an estimated Eastern Cooperative Oncology Group (ECOG) performance status of 0–1, were recruited prospectively during January 2004 to December 2010 to observe the surgical outcome of radical resection versus extended radical resection. A total of 70 consecutive patients with primary GBC resected at Xinhua Hospital were included in this retrospective study. GBC disease stage was defined using the American Joint Committee on Cancer Tumor, Node, Metastasis (TNM) classification system16. Demographic and clinical patient information relevant to prognosis include: age, sex, tumor differentiation, invasion depth, lymph node metastasis, distant metastasis, neurovascular infiltration and TNM stage was extracted from medical records. Follow-up was conducted using direct interviews in outpatient clinics or telephone interviews with patients or their family when patients were lost to follow-up, disease-free survival (DFS) and OS period were calculated. Patient follow-up data were collected up to July 2014. Among the patients, there were 18 males and 52 females with a mean age of 65 years (range 39–75 years). Histologically, 6 cases were well-differentiated, 51 cases were moderately differentiated, and 13 cases were poorly differentiated. Well preserved 25 paraffin-embedded non-cancerous gallbladder tissues were obtained from patients accepted cholecystectomy for cholecystitis with gallstones were include as normal gallbladder tissue control. The patients were pathological confirmed and recruited with written agreement informed consent form for our study. The study is approved by the ethics committee of the Xinhua Hospital, Shanghai Jiao Tong University School of Medicine. Additionally, institutional review board approval was obtained for the molecular analysis of the tumor blocks and the correlation with clinical variables performed in our study. The study was carried out in accordance with the approved guideline and the Helsinki Declaration.
Tissue microarray construction
Tissue blocks were collected from the surgically resected specimens of the 70 GBC patients, and from 25 non-cancerous gallbladder tissues obtained by cholecystectomy from patients with cholecystitis with gallstones. All tissues were formalin-fixed and paraffin-embedded. After hematoxylin and eosin-stained slides had been screened for optimal tumor content, tissue microarrays (TMAs) were constructed from the diagnosis-confirmed formalin-fixed paraffin-embedded tissues. TMA blocks were prepared from GBC and noncancerous gallbladder mucosae using the method described previously17. Briefly, 2 mm diameter tissue cylinders were punched out of the targeted area of each donor tissue block and transferred to a recipient block using a TMA instrument (Beecher Instruments, Silver Spring, MD, USA). Multiple 4-μm sections were cut consecutively from each block using a Leica microtome (Leica Microsystems Inc., Bannockburn, IL, USA) and were transferred to adhesive-coated slides (Leica Microsystems Inc.) for immunohistochemical staining. One section from each block was hematoxylin and eosin stained for histological verification of the adequacy of the arrayed tumor tissues. Qualified samples were defined as those in which the tumor tissue occupied >10% of the core area.
Immunohistochemistry
Immunohistochemistry was performed to investigate PK isoforms’ expression in the TMA, as described previously18. Briefly, the tissue sections were dehydrated with ethanol, washed three times with phosphate-buffered saline (PBS) (pH 7.4) and boiled for 8 min in a pressure cooker for antigen retrieval. Endogenous peroxidase activity was blocked by incubation in 3% hydrogen peroxide for 10 min at 26 °C. The sections were further blocked with 3% normal goat serum for 10 min. After the serum had been discarded, the sections were incubated overnight with Primary rabbit anti-human PKM1, PKM2 and PKLR antibody (Proteintech, Chicago, IL, USA) in a humid chamber at 4 °C. The following day, the sections were incubated with secondary antibody-coated polymer peroxidase complexes (Abcam, Cambridge, UK) for 30 min at room temperature. After three 3 min washes with PBS, the sections were developed using diaminobenzidine (Abcam), and slides were counterstained with hematoxylin for long-term storage. Negative controls were treated identically, but the primary antibody was omitted.
Evaluation of PKM1 and PKM2 expression
PKM2 proteins were stained yellowish-brown in the tumor cell nucleus, cytoplasm. However, PKM1 were stained mainly in cytoplasm. The intensity of the staining was scored by optical density using the semi-quantitative software Image-Pro Plus software 6.0 (Media Cybernetics Co., USA) as described previously19,20,21. In these cases, an optical density of less than 0.01 was regarded as low PKM2 expression and a score of 0.01 or above was regarded as high PKM2 expression. The results were double-evaluated independently by two gastroenterology pathologists with regards to the strength, position and portion of staining. The PKM2 expression, including both the cytoplasmic and nuclear expression, was evaluated. For the manual evaluation, cases showing negative to weak staining were regarded as low PKM2 expression, whereas cases showing moderate to strong staining were regarded as high PKM2 expression.
Fresh tissue specimens
Fresh gallbladder cancer tissues and the adjacent normal gallbladder tissues specimens were retrieved from the tissue bank of General surgery department, Xinhua hospital. The tissue specimens were resected into 1 cm in diameter per piece and washed with saline (10% heparin) then transferred to liquid nitrogen within 5 min of surgical resection, and stored at −80 °C. Twelve consecutive pair specimens were used for RNA isolation in qPCR and 5 pair were used for protein isolation in Western blot. All participants provided fresh kept tissue specimens had signed written informed consent for the collection of samples and subsequent analysis.
Quantitative real-time PCR (qPCR)
Total RNA was extracted from the cultured cells using the Trizol reagent (Takara, Shiga, Japan). cDNA was synthesized from 2 μg of total RNA using random primers and MMLV Reverse Transcriptase (Invitrogen, Carlsbad, CA). The following primers were used for detection of relevant gene expression: PKM1 forward: 5′-CGAGCCTCAAGTCACTCCAC-3′, PKM1 reverse: 5′-GTGAGCAGACCTGCCAGACT-3′; PKM2 forward: 5′-ATTATTTGAGGAACTCCGCCGCCT-3′, PKM2 reverse: 5′-ATTCCGGGTCACAGCAATGATGG-3′; PKL, forward: 5′-AGAGCCTCCCGTGTGTTAAA-3′, PKL, reverse: 5′-GTGTCACCACTGTCTCCTGTTC-3′; PKR, forward: 5 GAGGTTCCACCGAGATTTGG-3′, PKR, reverse: 5 GCAGGCGCAAACATTAGATG-3′; β-tubulin (sense): 5′- CCGTTGCGTTTGCACCTC-3′, and β-tubulin (antisense): 5′-TCAAGGTATGTATGGGGCAAGA-3′. The relative expression level of the target gene was calculated by 2−ΔCT (ΔCT = CTtarget gene − CTβ-tubulin) and normalized to the relative expression detected in the corresponding control cells.
Western blotting
After the treatments, the cellular protein lysates were harvest and blotted onto polyvinylidene difluoride (PVDF) membranes as described previously. The membranes were blocked and then probed with the primary antibodies against PKM1, PKM2 and PKLR (Proteintech) or GAPDH (Cell Signaling Technology, Danvers, MA, USA). The membranes were then incubated with horseradish peroxidase-conjugated secondary antibodies. The formed immunocomplexes were visualized using the enhanced chemiluminescent (ECL) detection reagent from Pierce (Rockford, IL, USA).
Statistical analysis
The clinico-pathological variables pertaining to each patient group according to PKM2 expression level were analyzed for significance using χ2 or Fisher’s exact test. For the analysis of survival, Kaplan-Meier survival curves were constructed for groups based on univariate predictors, and differences between the groups were tested with the log-rank test. Univariate and multivariate survival analyses were performed using the likelihood ratio test of the stratified Cox proportional hazards model. Differences between subgroups were tested with the non-parametric Mann–Whitney U-test. Parameters correlation was tested by nonparametric Spearman correlation analysis and fit lined by linear regression. The difference in mRNA expression of PK isotypes were assessed using Wilcoxon matched-pairs signed rank test for single gene between tissues samples and Dunn’s multiple comparison test for multiple genes expression in the same type of tissue. Friedman test was used to assess the significance of the concerned gene expression different to other genes in the same type of tissue. Statistical significance was assessed by the Student’s t-test (*P < 0.05; **P < 0.01; ***P < 0.001).
Discussion
The PKM2 gene, located on chromosome 15q22, encodes a pyruvate kinase that catalyzes the transfer of a phosphoryl group from phosphoenolpyruvate to ADP, thereby generating ATP and pyruvate22. Universally, PKM2 was believed to be highly expressed in malignant carcinoma to accommodate aerobic glycolysis, and PKM2 upregulation was believed to meet the needs of malignant tumor transformation23. These findings prompted us to determine the clinico-pathological relationship and prognostic significance of PKM2 overexpression in human gallbladder adenocarcinoma. In the present study, we demonstrated that PKM2 was frequently highly expressed in a majority of GBC patients (55.7%), and the high expression was an independent poor prognostic factor. A combined PKM2 and TNM score was used to classify the extremely different prognoses of GBC patients and showed the potential value of the combined score in predicting the relapse and survival interval after surgical resection.
A relevant study of the clinical significance of PKM2 for gallbladder squamous cell/adeonosquamous carcinomas and adenocarcinoma has been reported recently24. In a study based on 2 years of follow up of a population from western mainland China, the authors found that PKM2 and ACVR1C were highly expressed in approximately 56% of malignant gallbladder tumors, and this expression was correlated with cell differentiation, invasion, TNM stage and poor prognosis. The patients in our study were mainly from Eastern China, and the follow-up duration was 10 years (between 2004 and 2014). Although PKM2 was not associated with GBC cell differentiation or with neurovascular infiltration in the present study, we showed that high PKM2 expression was a poor independent prognosticator, and positively related to an unfavorable disease-free interval after surgical dissection. The longest recorded OS was 72 months, which was much longer than that reported in Li’s study. Moreover, we evaluated the efficiency of the combined risk factor, which reflected both TNM stage and PKM2 expression level, to predict relapse and survival risk in GBC patients. The results showed that the combined risk factor were much more effective in the trichotomic stratification of GBC patients according to the OS and in the dichotomous classification of patients according to DFS. Importantly, we found that PKM2 had outstanding predictive value in early stage GBC. This was not discussed in previously reports. We speculate that the strong predictive value of PKM2 in the early stage GBC patients was due to their relatively favorable clinical outcomes. It was known that surgical resection that was the most effective treatment for GBC was more suitable for early stage patient. However, part of the patients with early stage GBC had short DFS and poor OS clinically. In this condition, detection of PKM2 expression could help to distinguish high risk population which highlight the strong predictive value of PKM2 as a biomarker especially in early stage GBC.
There are 4 pyruvate kinase isotypes in mammals: PKL, expressed in liver and kidney; PKR, expressed in erythrocytes; PKM1, predominantly expressed in adult muscle, brain, and bladder cells and in adult fibroblasts; and PKM2, expressed in most cells except adult muscle, brain, and liver15. However, the isotypes expression of PK in gallbladder tissue had not been reported in literature so far. The PKM1 and PKM2 isoforms result from mutually exclusive alternative splicing of the PKM pre-mRNA that results in inclusion of either exon 9 (PKM1) or exon 10 (PKM2)25. It is reported that the PKM1 to PKM2 shift was widely observed in distinct malignancies26. Therefore we further investigated the different PK isotypes expression in gallbladder tissue and gallbladder cancer samples. In this study, we found that M1, M2 and L type of PK mRNA were expressed in gallbladder cancer tissue as well as in noncancerous gallbladder tissues. PKM2 were significantly high expressed in GBC tissues than in adjacent noncancerous tissues, while PKM1 decreased in GBC tissues, therefore a PKM1 to PKM2 shift were suggested in this small cohort of GBC.
To date, PKM2 had been shown to play diverse and different roles in the malignant transformation of multiple carcinomas; these roles include regulation of the Warburg effect, promotion of proliferation, prevention of apoptosis, and even promotion of cancer angiogenesis8,27,28,29,30. However, the bio-function of PKM2 in GBC cells has not been investigated. The high expression of PKM2 in GBC tissue and its significance for poor OS prompted the idea that PKM2 may be involved in GBC carcinogenesis. PKM isoforms are considered as the key factors involved in the metabolic homeostasis. Although the PKM proteins were reported to correlate with multiple cellular processes, including proliferation and progression, the metabolic regulation of PKM2 in GBC had not been investigated in the present study. It is well accepted that PKM2 is a glycolytic enzyme that participate in glycolysis, which renders energy and building blocks for malignant cancer cells. The role of PKM2 in glycolysis and in regulates glucose utilization of gallbladder carcinoma cells, glucose uptake, lactate production, and change in expression of glycolysis genes in the PKM2 modulating cells are worth for further explorer. Moreover, mitochondria-related properties need to be further examined by using metabolism analyzer.
In summary, we demonstrated that the PKM were the dominant isotype of pyruvate kinase expressed in gallbladder tissue. The expression of type 2 PKM was frequently up-regulated in GBC, indicating its prognostic value in patients with GBC. The aberrant high expression of PKM2 is associated with aggressive tumor features and poor prognosis. Although our results support the possibility that PKM2 as an important molecular marker for determining malignant properties, studies of larger cohorts are required to validate these findings before moving to a clinical setting.
Additional Information
How to cite this article: Lu, W. et al. Up-regulation of PKM2 promote malignancy and related to adverse prognostic risk factor in human gallbladder cancer. Sci. Rep. 6, 26351; doi: 10.1038/srep26351 (2016).
References
Gourgiotis, S. et al. Gallbladder cancer. Am J Surg 196, 252–264, 10.1016/j.amjsurg.2007.11.011 (2008).
Reddy, S. K. & Clary, B. M. Surgical management of gallbladder cancer. Surg Oncol Clin N Am 18, 307–324, ix, 10.1016/j.soc.2008.12.004 (2009).
Andren-Sandberg, A. & Deng, Y. Aspects on gallbladder cancer in 2014. Curr Opin Gastroenterol 30, 326–331, 10.1097/MOG.0000000000000068 (2014).
Jayaraman, S. & Jarnagin, W. R. Management of gallbladder cancer. Gastroenterol Clin North Am 39, 331–342, x, 10.1016/j.gtc.2010.02.006 (2010).
Bizama, C. et al. Targeting specific molecular pathways holds promise for advanced gallbladder cancer therapy. Cancer Treat Rev 41, 222–234, 10.1016/j.ctrv.2015.01.003 (2015).
Wang, C. et al. Clinicopathological significance of SOX4 expression in primary gallbladder carcinoma. Diagn Pathol 7, 41, 10.1186/1746-1596-7-41 (2012).
Tan, Z. et al. NLK is a key regulator of proliferation and migration in gallbladder carcinoma cells. Mol Cell Biochem 369, 27–33, 10.1007/s11010-012-1365-0 (2012).
Li, W., Liu, J. & Zhao, Y. PKM2 inhibitor shikonin suppresses TPA-induced mitochondrial malfunction and proliferation of skin epidermal JB6 cells. Mol Carcinog 53, 403–412, 10.1002/mc.21988 (2014).
Kwon, O. H. et al. Pyruvate kinase M2 promotes the growth of gastric cancer cells via regulation of Bcl-xL expression at transcriptional level. Biochem Biophys Res Commun 423, 38–44, 10.1016/j.bbrc.2012.05.063 (2012).
Wong, T. S. et al. Identification of pyruvate kinase type M2 as potential oncoprotein in squamous cell carcinoma of tongue through microRNA profiling. Int J Cancer 123, 251–257, 10.1002/ijc.23583 (2008).
Li, W., Xu, Z., Hong, J. & Xu, Y. Expression patterns of three regulation enzymes in glycolysis in esophageal squamous cell carcinoma: association with survival. Med Oncol 31, 118, 10.1007/s12032-014-0118-1 (2014).
Nemazanyy, I., Espeillac, C., Pende, M. & Panasyuk, G. Role of PI3K, mTOR and Akt2 signalling in hepatic tumorigenesis via the control of PKM2 expression. Biochem Soc Trans 41, 917–922, 10.1042/BST20130034 (2013).
Wang, H. J. et al. JMJD5 regulates PKM2 nuclear translocation and reprograms HIF-1alpha-mediated glucose metabolism. Proc Natl Acad Sci USA 111, 279–284, 10.1073/pnas.1311249111 (2014).
Wong, N., De Melo, J. & Tang, D. PKM2, a Central Point of Regulation in Cancer Metabolism. Int J Cell Biol 2013, 242513, 10.1155/2013/242513 (2013).
Yang, W. & Lu, Z. Nuclear PKM2 regulates the Warburg effect. Cell Cycle 12, 3154–3158, 10.4161/cc.26182 (2013).
Edge, S. B. & Compton, C. C. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17, 1471–1474, 10.1245/s10434-010-0985-4 (2010).
Lee, H. S., Lee, H. K., Kim, H. S., Yang, H. K. & Kim, W. H. Tumour suppressor gene expression correlates with gastric cancer prognosis. J Pathol 200, 39–46, 10.1002/path.1288 (2003).
Drachenberg, C. B., Papadimitriou, J. C., Balaton, A. J. & Vaury, P. The total test approach to standardization of immunohistochemistry. Arch Pathol Lab Med 125, 471, 10.1043/1543-2165(2001)125<471b:IR>2.0.CO;2 (2001).
Taylor, C. R. The total test approach to standardization of immunohistochemistry. Arch Pathol Lab Med 124, 945–951, 10.1043/0003-9985(2000)124<0945:TTTATS>2.0.CO;2 (2000).
O’Leary, T. J. Standardization in immunohistochemistry. Appl Immunohistochem Mol Morphol 9, 3–8 (2001).
Li, M. et al. Prognostic significance of nemo-like kinase (NLK) expression in patients with gallbladder cancer. Tumour Biol 34, 3995–4000, 10.1007/s13277-013-0988-4 (2013).
Dang, C. V. PKM2 tyrosine phosphorylation and glutamine metabolism signal a different view of the Warburg effect. Sci Signal 2, pe75, 10.1126/scisignal.297pe75 (2009).
Hitosugi, T. et al. Tyrosine phosphorylation inhibits PKM2 to promote the Warburg effect and tumor growth. Sci Signal 2, ra73, 10.1126/scisignal.2000431 (2009).
Li, J. et al. PKM2 and ACVR 1C are prognostic markers for poor prognosis of gallbladder cancer. Clin Transl Oncol 16, 200–207, 10.1007/s12094-013-1063-8 (2014).
David, C. J., Chen, M., Assanah, M., Canoll, P. & Manley, J. L. HnRNP proteins controlled by c-Myc deregulate pyruvate kinase mRNA splicing in cancer. Nature 463, 364–368, 10.1038/nature08697 (2010).
Desai, S. et al. Tissue-specific isoform switch and DNA hypomethylation of the pyruvate kinase PKM gene in human cancers. Oncotarget 5, 8202–8210, 10.18632/oncotarget.1159 (2014).
Warner, S. L., Carpenter, K. J. & Bearss, D. J. Activators of PKM2 in cancer metabolism. Future Med Chem 6, 1167–1178, 10.4155/fmc.14.70 (2014).
Li, L., Zhang, Y., Qiao, J., Yang, J. J. & Liu, Z. R. Pyruvate kinase M2 in blood circulation facilitates tumor growth by promoting angiogenesis. J Biol Chem 289, 25812–25821, 10.1074/jbc.M114.576934 (2014).
Goldberg, M. S. & Sharp, P. A. Pyruvate kinase M2-specific siRNA induces apoptosis and tumor regression. J Exp Med 209, 217–224, 10.1084/jem.20111487 (2012).
Li, Z., Yang, P. & Li, Z. The multifaceted regulation and functions of PKM2 in tumor progression. Biochim Biophys Acta 1846, 285–296, 10.1016/j.bbcan.2014.07.008 (2014).
Acknowledgements
This work was supported by China National High Technology Research and Development Program (863 Program) (No. 2012AA022606); National Natural Science Foundation of China (Nos 81170457, 91440203, 81172026, 81272402, 81301816 and 81172029); International S&T Cooperation Program of China (2013DFA11150); China Postdoctoral Science Foundation (No. 2014M561487) and Interdisciplinary Program of Shanghai Jiao Tong University (No. 14JCRY05).
Author information
Authors and Affiliations
Contributions
W.L. and Y.L. was the funding financial support owner and provide the conception of research. W.L., J.M., X.-A.W. and W.G. collected and assembled the clinical and pathological data. Y.Z., Y.H., P.D. and L.J. carried out the experimental studies. W.L., J.G. and S.L. performed the statistical analysis and interpreted the results. W.L. and Y.C. wrote the manuscript majorly. All authors had proved study materials, reviewed and finally approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Lu, W., Cao, Y., Zhang, Y. et al. Up-regulation of PKM2 promote malignancy and related to adverse prognostic risk factor in human gallbladder cancer. Sci Rep 6, 26351 (2016). https://doi.org/10.1038/srep26351
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep26351
This article is cited by
-
Identification and subsequent validation of transcriptomic signature associated with metabolic status in endometrial cancer
Scientific Reports (2023)
-
Pyruvate kinase M2 (PKM-2) expression and prognostic significance in glioblastoma patients
Journal of Neuro-Oncology (2023)
-
Circular RNA FOXP1 promotes tumor progression and Warburg effect in gallbladder cancer by regulating PKLR expression
Molecular Cancer (2019)
-
Pyruvate kinase M2 fuels multiple aspects of cancer cells: from cellular metabolism, transcriptional regulation to extracellular signaling
Molecular Cancer (2018)
-
Long noncoding RNA MEG3 suppresses liver cancer cells growth through inhibiting β-catenin by activating PKM2 and inactivating PTEN
Cell Death & Disease (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.