Abstract
The radioactive fission product 90Sr has a long biological half-life (˜18 y) in the human body. Due to its chemical similarity to calcium it accumulates in bones and irradiates the bone marrow, causing its high radio-toxicity. Assessing 90Sr is therefore extremely important in case of a nuclear disaster. In this work 16 soil samples were collected from the exclusion zone (<30 km) of the earthquake-damaged Fukushima Daiichi nuclear power plant, to measure 90Sr activity concentration using liquid scintillation counting. 137Cs activity concentration was also measured with gamma-spectroscopy in order to investigate correlation with 90Sr. The 90Sr activity concentrations ranged from 3.0 ± 0.3 to 23.3 ± 1.5 Bq kg−1 while the 137Cs from 0.7 ± 0.1 to 110.8 ± 0.3 kBq kg−1. The fact that radioactive contamination originated from the Fukushima nuclear accident was obvious due to the presence of 134Cs. However, 90Sr contamination was not confirmed in all samples although detectable amounts of 90Sr can be expected in Japanese soils, as a background, stemming from global fallout due to the atmospheric nuclear weapon tests. Correlation analysis between 90Sr and 137Cs activity concentrations provides a potentially powerful tool to discriminate background 90Sr level from its Fukushima contribution.
Similar content being viewed by others
Introduction
Radioactive strontium isotopes in the atomic mass number range of 89 to 92 are generated with high cumulative fission yield (5–6%) during thermal neutron fission in a nuclear reactor1,2. Nuclear accidents can cause radioactive contamination of the environment, as it happened when the Fukushima Daiichi Nuclear Power Plant (FDNPP) was severely damaged by earthquake and tsunami on 11 March 2011. Such event can include radioactive strontium isotopes to disperse into the environment. In this situation 91Sr and 92Sr have little radiological significance owing to their short physical half-life (a few hours) but this is different for 89Sr (half-life 51 days) and 90Sr (29 years). Nuclear accidents are not the only source of 89Sr and 90Sr isotopes: atmospheric nuclear weapon tests3, misconducted underground nuclear weapon tests4, improper handling of by-products of nuclear weapon production5,6 or normal operation of nuclear facilities (e.g. reprocessing plants)3 can also result in contamination. As far as known only 90Sr was present in the environment of Japan before the Fukushima accident, derived from global fallout from large-scale bomb tests conducted from 19457,8 and perhaps from the Chernobyl accident in 1986, the latter however a very small contribution, in comparison, if any. 89Sr has decayed since, thus its presence is an indicator of recent contamination. However, one year after emission by a nuclear event, 89Sr cannot be determined in environmental samples owing to its short half-life, thus the identification of the origin of 90Sr could be a challenging task. In our case, it consists of discrimination between Fukushima-borne and global 90Sr after decay of 89Sr.
Compared to volatile radioisotopes of caesium released into the atmosphere (137Cs, ˜15 PBq), non-volatile 90Sr (˜0.14 PBq) emission has been estimated two orders lower. 90Sr contamination is also expected in environmental samples due to Fukushima accident9. Moreover, ˜80% of the 90Sr emission was deposited over the Pacific Ocean, significantly decreasing the terrestrial pollution10, whereas no 90Sr concentration that would be of radiological concern (i.e. over 100 Bq kg−1) in soil samples have been reported11,12 outside the damaged FDNPP by the authorities.
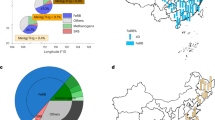
In June 2011, Ministry of Education, Culture, Sports, Science and Technology (MEXT) and Nuclear Regulation Authority (NRA) Japan conducted a radiation survey that included 89Sr and 90Sr analysis in the vicinity of the damaged reactors of FDNPP in Fukushima prefecture12. Figure 1 shows the radiostrontium concentration levels in sampling points with detection of 89Sr and 90Sr over 10 Bq kg−1 along north-west direction. It was reported that 89Sr and 90Sr has been detected in 45% and 80% of samples, respectively, with average background level of 90Sr about 3 Bq kg−1 (n = 100)12,13.
89Sr and 90Sr detection in soil samples collected in 2011 after FDNPP accident.
Map source: Geospatial Information Authority of Japan (GSI) http://maps.gsi.go.jp/#10/37.357059/140.787048. The original map was modified using Adobe Photoshop Elements 9 software.
Gamma emitter radionuclides such as iodine, tellurium and caesium isotopes14,15,16,17,18,19,20 have been published and studied much more extensively after the Fukushima accident than the pure beta emitter radiostrontium isotopes21,22,23,24. The explanation of this phenomenon is in the measurement technique: it is much simpler to measure gamma than pure beta emitters. Gamma rays are emitted in discrete energies, so qualitative identification of radioisotopes can be achieved easily by analysing the characteristic of energy spectra of gamma rays without element specific separation and complex sample preparation25. On the contrary, the energy distribution of the emitted electrons in the beta decay is continuous, therefore element specific separation from the interfering beta emitters is necessary for qualitative radioisotopes identification and subsequent measurement, making 90Sr determination complicated, time-consuming procedure with corrosive and aggressive chemicals during sample preparation1,26.
To increase the database of 90Sr, a new separation laboratory was established in the National Institute of Radiological Sciences, Japan (NIRS) in 2012. Sixteen soil samples were collected from the exclusion zone in the north-western direction of the FDNPP in 2013 to determine 90Sr and radiocaesium concentrations. The measurements of 90Sr concentration were duplicated, at the laboratories of NIRS and the Department of Nuclear Physical Chemistry, Institute of Nuclear Physics, Polish Academy of Sciences, Poland (IFJ-PAN).
Attempts were made to establish a procedure to distinguish 90Sr contamination caused by the Fukushima accident and atmospheric nuclear weapon tests by analysing the frequency distribution of the 90Sr activity concentration and obtaining a correlation of 90Sr and 137Cs. Finally, the measured data were compared to other data obtained by MEXT, NRA and other scientific research laboratories.
Results
In our study 16 top layer (up to a 10 cm depth) soil grab samples were collected from the north-western direction of the FDNPP (Namie district) in the exclusion zone (<30 km from the FDNPP). This area was chosen because the highest radiation doses have been reported in this district due to wet deposition of radionuclides from the radioactive plumes released on 15 March 201119,20,27. Locations and other details of the sampling points are shown in Fig. 2 13 and Table 1. The 90Sr and radiocaesium data of seven points (No. 1–4 and No. 7–9) were recently published elsewhere as preliminary results of this study21,22 and discussed here again to establish an extended database on 90Sr. A little contaminated area (No. 3), along north direction, was also monitored to compare with background 90Sr concentration.
Locations of sampling points in the exclusion zone of FDNPP.
Map source: Geospatial Information Authority of Japan (GSI) http://maps.gsi.go.jp/#10/37.357059/140.787048. The original map was modified using Adobe Photoshop Elements 9 software.
Fukushima radioactive contamination was confirmed by the presence of 134Cs in all collected samples. The 134Cs/137Cs activity ratio was 1.0 at the time of the accident17. In this study, the average value of this ratio was the same, 1.0 (SD 0.02, n = 16) (decay correction date: 15 March 2011).
The results of the activity concentrations of 90Sr and 137Cs in this study are presented in Table 1 and as it can be seen, no cases of 90Sr concentrations exceeding 100 Bq kg−1 were detected. As expected, lowest activity concentrations of 90Sr and 137Cs were found at the little contaminated site (No. 3). 90Sr activity above 20 Bq kg−1 was revealed in two locations, No. 2 and No. 9, respectively. No. 2 was the closest sampling point to the FDNPP with the second highest 137Cs activity (97.9 ± 0.3 kBq kg−1), however No. 9 was a distant point over 20 km with somewhat lower 137Cs activity (58.2 ± 0.2 kBq kg−1).
Frequency distribution
Figure 3 shows the frequency distribution of 90Sr activity concentration in the 16 analysed soil samples. The 90Sr activity concentrations were below 5 Bq kg−1 in 12.5% of samples analysed while in the major part, 62.5% were between 5 and 15 Bq kg−1 and above 15 Bq kg−1, 12.5% were detected.
Correlation
The correlation between the 90Sr and 137Cs concentration in the soil samples collected from the exclusion zone of the FDNPP is moderate as can be seen in Fig. 4. Thus 137Cs concentration (or ambient gamma dose rate) does not predict 90Sr concentration accurately. The activity concentrations of 90Sr were very similar (˜11 Bq kg−1) at sampling points No. 4, 7, 8, 13 and 15 but 137Cs concentrations fluctuated between 27.4 ± 0.1 and 86.2 ± 0.3 kBq kg−1 at these sites.
Discussion
In 2005, i.e. before the Fukushima accident, MEXT had conducted a radiation monitoring survey at 55 sampling sites around Fukushima prefecture11. Low level 90Sr contamination likely originating from atmospheric nuclear weapons tests with an average of 3.0 Bq kg−1 were reported, with minimum and maximum <0.2 and 17.7 Bq kg−1, respectively. The maximum value of this baseline study shows that 90Sr concentrations of 20 Bq kg−1 cannot confirm contamination from Fukushima if the site specific background is not known exactly. The same 55 sampling points were also monitored in 2011 after the Fukushima nuclear accident and a slight increment of 90Sr level was found11. The reported average was 5.6 Bq kg−1 with minimum and maximum <0.9 and 80.8 Bq kg−1, respectively. The high maximum value found in this study clearly points to Fukushima contamination. Comparing the values detected before and after the Fukushima accident using Z score test (k = 2), a significant increase was found at 54.5% of the 55 sampling points, although the increment was marginal (<3 Bq kg−1) in most cases (83.3%). Significant increment was not verified at the maximum background point (17.7 Bq kg−1). (Decay correction date of this database was June to July 2011. Correction to 15 March 2011 was not possible hence the exact date was not published but the estimated decay could be around 1%, which is negligible).
Figure 5 presents the frequency distributions of 90Sr concentrations measured before (2005) and after (2011) the FDNPP accident. The frequency distribution before the accident denotes that the dominating background level is below 5 Bq kg−1 (85.5%), while about 11% of the measured values were between 5 and 10 Bq kg−1 and about 4% were between 10 and 20 Bq kg−1.
Analysing the frequency distribution of the detected 90Sr values after the accident, slight increments can be seen in the ranges above 5 Bq kg−1. However, the dominating 90Sr level remained below 5 Bq kg−1 (72.7%). The frequencies were duplicated in the range of 5 to 10 Bq kg−1 and above 10 Bq kg−1. The differences in the frequency distribution before and after the accident verify 90Sr contamination from the Fukushima accident in Fukushima prefecture. Relatively low decrement (˜10%) in the range below 5 Bq kg−1 indicates that the 90Sr dispersion over the Fukushima prefecture was limited and the major part of the atmosphere released 90Sr was deposited in the proximity of the FDNPP. The theory of limited 90Sr dispersion was also supported by result of two-sample Kolmogorov-Smirnov statistical test. The cumulative distributions of the before and after results did not show significant difference; p value was 0.06 which is greater than 0.05 that is commonly considered as threshold of significance.
In our study, information about background 90Sr is not available. In order to decide whether a contribution of Fukushima 90Sr is present, we performed two analyses, one based on frequency distribution analysis of the measured 90Sr concentrations, the second on correlation between 90Sr and 137Cs.
Comparison of frequency distribution
The database of our study was small (n = 16) as compared to MEXT’s (n = 55) and the sample collection was focused on one specific area instead of the whole prefecture. We assume that the frequency distribution of pre-Fukushima (background) 90Sr in our samples is similar to the one in MEXT’s study conducted in 2005, although the geographical areas of their origins are different (entire Fukushima prefecture for the old vs. Namie district for the new samples). Comparing Fig. 5, red bars (samples before Fukushima), with Fig. 3 (our samples), very different shapes of the distributions were noticed. The distribution of our samples is symmetric, centred around 5–15 Bq kg−1 while the background samples are positively skewed, concentrated in the class <5 Bq kg−1. Comparing the cumulative distribution of the two datasets by two-sample Kolmogorov-Smirnov statistical test significant difference was proved; p value was 1.6 × 10−7, i.e. highly significant at 0.05 level.
The results of the frequency distribution analysis emphasize the importance of environmental baseline studies in countries with nuclear energy reactors or other nuclear facilities.
Analysis of correlation coefficients
The second method to identify Fukushima contribution to observed 90Sr concentration relies on correlation with other quantities. In sampling radiometric survey, the simplest way to reveal significant radioactive contamination is measuring ambient dose rate, which is produced by the gamma-rays emitted by the deposited radiocaesium isotopes (after the decay of iodine and tellurium isotopes). However, due to the different physical and chemical properties of alkali metals (Cs) and alkali earth metals (Sr), the presence of radiocaesium contamination cannot quantify 90Sr contamination with precision. The ratio between 137Cs and 90Sr in the fallout may not be preserved in the samples because of different environmental behaviours of the elements. The ratio observed in the samples is therefore different from the one in fallout, in general. The degree of difference is in addition different between sites (i.e. between soil types). Therefore, the correlation suffers not only because of the different behaviours of Sr and Cs, but additionally, because even these differences are not constant between sample points.
In spite of this fact, the data analysis of the MEXT and NRA’s database12 demonstrates that the correlation between the 137Cs and 90Sr deposition rate was strong in June 2011 after the accident as can be seen in Fig. 6. According to correlation coefficient of linear regression analysis (R2), about 70% of the 90Sr deposition is predicted from the 137Cs deposition at each sampling point in the vicinity of the FDNPP wherein 90Sr contamination from Fukushima was confirmed by the detection of 89Sr.
Figure 7 shows the correlation between 137Cs and 90Sr where 89Sr was not detected, which means that 90Sr contamination by the Fukushima accident was very small, if present at all. One can see visually that correlation has practically disappeared and the 90Sr in these samples can be assumed to be almost exclusively pre-Fukushima 90Sr. As a consequence, can be concluded that 137Cs (or gamma dose rate) indicates the presence of 90Sr, although prediction is uncertain because of the imperfect correlation.
On the other hand, in the presence of background contamination only of both 137Cs and 90Sr, as is the case in the pre-Fukushima samples of 2005, one can observe a moderate correlation (R2 = 0.45), Fig. 8. Presence of 134Cs may be due to recent contamination by FDNPP than global fallout and was not detected in these samples.
In our study, the samples were collected approximately two years after the Fukushima accident. The R2 was 0.45 (Fig. 4) which is lower than the R2 (0.67) calculated from database in which the Fukushima 90Sr contamination was confirmed by the presence of 89Sr (Fig. 6). Reasons for the lower R2 may be seen in the smaller number of samples, the longer time elapsed between fallout and sampling which gave more time for ecological processes which distort the original ratio and a higher contribution of 90Sr pre-Fukushima background. However, the R2 is high enough to reject the null hypothesis, there is no correlation, with p < 0.005 error probability, which confirms the presence of Fukushima 90Sr contamination in the monitored area.
There was a good agreement between the database of our 90Sr results with MEXT and NRA’s database12. In the MEXT and NRA’s database, noticeable 90Sr concentrations (>˜5 kBq kg−1) and 89Sr concentrations could not be determined if the 137Cs level was below ˜12 kBq kg−1. In the database of our study, 90Sr concentrations below 5 Bq kg−1 were related to ˜15 kBq kg−1 or lower 137Cs concentrations.
Table 2 shows 90Sr results in soil samples from various references, including, in some cases, sampling depth. The soil samples were collected from different depths between 2.5 cm and 10 cm and on different dates from 30 March 2011 to 02 July 2012. In case of nuclear accidents, the depth of sampling is one of the most important factors to understand migration of radionuclides due to contamination. Most of the deposited 90Sr could be found on the soil surface (1–2 cm) shortly after the accident. A few years later it could be expected to have migrated into deeper layers (˜10 cm) due to different redistribution processes in soil. The velocity of the migration is site-specific and particularly depends on soil type, the amount of precipitation and permeability28,29. Therefore, 74 Bq kg−1 of 90Sr measured in 2.5 cm soil layer directly after the accident (No. 3) could have migrated into deeper layers during the following years29. Assuming 90Sr concentration migration velocity is a few cm per year, we can obtain a value for 90Sr about 20 Bq kg−1 approximately up to 10 cm depth. Accepting this theoretical approach, our results are in good agreement with the result of Maekawa et al. (No. 1 and 2), Takagai et al. (No. 3–5) and Mishra et al. (No. 6–9)24,30,31. On the other hand, Steinhauser et al.23 (No. 10–18) have reported very high 90Sr concentrations (up to 1,000 Bq kg−1) as well as extremely high 137Cs concentration (up to 4,600 kBq kg−1). The possible explanation of the high contamination could be that soil samples were collected from very thin surface layers (organic horizons) at highly contaminated spots within 4 km of FDNPP accident, even though the sampling was carried out after six months of the accident.
Also in Table 2, quite high radionuclide depositions were reported from the north-western direction of the FDNPP which is not surprising because this is the direction of the main contamination trace; however noticeable 90Sr contaminations (˜20 and ˜270 Bq kg−1) were also detected in the south direction (No. 8, 9 and 15) about 15 km from FDNPP23,24. These results emphasize the importance of radionuclides monitoring beyond the Namie district. It has been recognized that the 137Cs contamination due to Fukushima was quite low (˜1 kBq kg−1) at these southern points (No. 8 and 9) giving a contradiction to our theory stated above that considerable 90Sr contamination is not expected if the 137Cs contamination level is around 10 kBq kg−1 or lower. However, this contradiction could be accepted as result of rarely occurring anomaly as it can be observed in Fig. 8 where 90Sr concentrations above 20 Bq kg−1 are related to 137Cs below 100 Bq kg−1 but further study is necessary to prove this theory.
One more discrepancy can be noticed in results of Maekawa et al., samples No 1 and 2 in Table 2. Considering the fact that date of the ambient gamma dose rate measurement was shortly (a few weeks difference) after the accidental radionuclides were released (12–31 March 2011) and the radioactive contamination was considerable (137Cs concentration was 262 kBq kg−1), the measured ambient radiation dose rate was quite low (˜20 μSv h−1) at these points. During the accident many other volatile and gamma-ray emitter radionuclides (iodine and tellurium isotopes) with shorter physical half-life (max. 33.6 d) were also released into the atmosphere and deposited in the terrestrial environment generating extremely high dose rates in the early days of the accident in the exclusion zone32, therefore much higher dose rate should have been measured than the reported ˜20 μSv h−1 in 18 April 2011. Higher dose rate (˜70 μSv h−1) was measured (No 6 and 7) with lower 137Cs concentration by Mishra et al.24 and similar dose rate was determined in our study after two years of the accident (Table 1, No 2).
Methods
Sample collection and preparation
The detailed information about sample collection and preparation for gamma spectroscopy measurement are given elsewhere21,33. In brief, at each sampling point, five grab samples (up to 10 cm depth) were collected from an area of about 1 m2, of which four from four vertices and one from the centre point. The five samples were mixed and placed in a plastic bag. During sample collection, the ambient gamma radiation dose rate was measured on a triplicate basis at a height of 1 m using a Radeye PRD-ER (Thermo Scientific, USA) personal radiation detector (with a micro-photomultiplier NaI(Tl)-detector). In the laboratory, the soil samples were dried at 105 °C for 24 hours. The dried soil samples were homogenized and sieved (<2 mm), stones and plant remains were removed. After this procedure, U8 standard cylindrical containers were filled up to 1–3 cm height with the soil samples to measure the specific activity of radiocaesium isotopes for 1800 sec.
Reagents and materials
Tamapure AA-100 ultrapure hydrochloric, hydrofluoric, nitric and perchloric acids were used for the mass spectrometric measurement. Wako high pure hydrochloric, hydrofluoric and nitric acids were used for preparing the radiometric measurement. Standard solutions were prepared using high-purity water (>18 MΩ cm−1) produced with the Milli-Q2TM water purification system. The detailed procedure for the separation of strontium is discussed elsewhere22,34. To summarize, the chemically digested soil samples (10–15 g) were dissolved in 8M HNO3 to ensure the highest strontium retention on Sr resin. These solutions were loaded into L-size polypropylene gravity columns (the flow rate was 1–2 ml min−1) attached with 50 ml syringes. The columns were packed with approximately 3 g (˜10 ml) Sr extraction chromatography resin (part number SR-B50-A) of 100–150 μm particle size (Eichrom Technologies, Inc., USA). The loaded Sr resins were first washed with 8M HNO3 and then with 3M HNO3 + 0.05M oxalic acid. Strontium was stripped from the column with 0.05M HNO3. The recovered amount of 90Sr was measured by a liquid scintillation counter. The chemical yields of strontium separations were between 25% and 70%.
Instruments
The instruments used are described elsewhere21,33,34. Briefly, for strontium recovery calculation, stable strontium (88Sr) was analysed applying inductively coupled plasma mass spectrometer Agilent 8800 (Agilent Technologies, USA) at NIRS and 85Sr spike solution was used at IFJ-PAN. Gamma-spectrometer ORTEC GEM-100210 (ORTEC, USA) was used mounted with high-purity germanium detector and multichannel analyser for radiocesium determination. After the separation procedure, liquid scintillation counter TriCarb-3100 and Wallac 1414-003 (Perkin Elmer, USA) was used with Ultima Gold AB scintillation cocktail for 90Sr determination. The full analytical procedure was verified using soil reference sample produced by the International Atomic Energy Agency (IAEA 375) and tested using water samples in a world-wide open proficiency test, the IAEA-TEL-2014-03. The accuracy and precision of the applied procedure received accepted status.
Statistical analysis
OriginPro 9 data analysis and graphing software (OriginLab Corp., USA) was used for running two-sample Kolmogorov-Smirnov statistical test.
Additional Information
How to cite this article: Sahoo, S. K. et al. Strontium-90 activity concentration in soil samples from the exclusion zone of the Fukushima daiichi nuclear power plant. Sci. Rep. 6, 23925; doi: 10.1038/srep23925 (2016).
References
Vajda, N. & Kim, C. K. Determination of radiostrontium isotopes: a review of analytical methodology. Appl. Radiat. Isot. 68, 2306–2326, doi: 10.1016/j.apradiso.2010.05.013 (2010).
JAEA. Nuclear Data Center. (2015) Available at: http://www.ndc.jaea.go.jp/cgi-bin/nucltab10?38. (Accessed: 12th September 2015).
UNSCEAR. Sources, Effects and Risks of Ionizing Radiation. Scientific Annex C: Exposures to the public from man-made sources of radiation. United Nation (2002).
Ramzaev, V. et al. Radioecological studies at the Kraton-3 underground nuclear explosion site in 1978–2007: a review. J. Environ. Radioact. 100, 1092–1099, doi: 10.1016/j.jenvrad.2009.04.002 (2009).
Friday, G. P., Cummins, C. L. & Schwartzman, A. L. Radiological bioconcentration factors for aquatic, terrestrial and wetland ecosystems at the Savannah River site. Westinghouse Savannah River Company, doi: 10.2172/573692 (1996).
Shutov, V. N. et al. Current contamination by 137Cs and 90Sr of the inhabited part of the Techa river basin in the Urals. J. Environ. Radioact. 61, 91–109, doi: 10.1016/s0265-931x(01)00117-5 (2002).
DOENV. United States Nuclear Tests July 1945 through September 1992. (2000) Available at: http://www.nv.doe.gov/library/publications/historical/DOENV_209_REV15.pdf. (Accessed: 14th September 2015).
Mangano, J. J., Gould, J. M., Sternglass, E. J., Sherman, J. D. & McDonnell, W. An unexpected rise in strontium-90 in US deciduous teeth in the 1990s. Sci. Total Environ. 317, 37–51, doi: 10.1016/S0048-9697(03)00439-X (2003).
Povinec, P., Hirose, K. & Aoyama, M. Fukushima Accident Radioactivity Impact on the Environment. (Elsevier, 2013).
Ten Hoeve, J. E. & Jacobson, M. Z. Worldwide health effects of the Fukushima Daiichi nuclear accident. Energy Environ. Sci. 5, 8743, doi: 10.1039/c2ee22019a (2012).
Ministry of Education, Culture, Sports, Science and Technology-Japan. Results of the Radiation Monitoring of Soil in Fukushima Prefecture (2012) Available at: http://radioactivity.nsr.go.jp/en/contents/6000/5025/24/232_e_0409.pdf. (Accessed: 30th June 2015).
Ministry of Education, Culture, Sports, Science and Technology and the Secretariat of the Nuclear Regulation Authority-Japan. Results of the Nuclide Analysis of Soil Sampling at 2,200 Locations in Fukushima Prefecture and Neighboring Prefectures. (2011) Available at: http://emdb.jaea.go.jp/emdb/en/portals/b211/. (Accessed: 14th September 2015).
Geospatial Information Authority of Japan. Map of Japan. (2015) Available at: http://maps.gsi.go.jp/#10/37.357059/140.787048. (Accessed: 15th December 2015).
Yamaguchi, M., Kitamura, A., Oda, Y. & Onishi, Y. Predicting the long-term Cs distribution in Fukushima after the Fukushima Dai-ichi nuclear power plant accident: a parameter sensitivity analysis. J. Environ. Radioact. 135C, 135–146, doi: 10.1016/j.jenvrad.2014.04.011 (2014).
Hosoda, M. et al. Estimation of internal exposure of the thyroid to (131)I on the basis of (134)Cs accumulated in the body among evacuees of the Fukushima Daiichi Nuclear Power Station accident. Environ. Int. 61, 73–76, doi: 10.1016/j.envint.2013.09.013 (2013).
Kanai, Y. Monitoring of aerosols in Tsukuba after Fukushima Nuclear Power Plant incident in 2011. J. Environ. Radioact. 111, 33–37, doi: 10.1016/j.jenvrad.2011.10.011 (2012).
Hirose, K. 2011 Fukushima Dai-ichi nuclear power plant accident: summary of regional radioactive deposition monitoring results. J. Environ. Radioact. 111, 13–17, doi: 10.1016/j.jenvrad.2011.09.003 (2012).
Nakanishi, T. M., Kobayashi, N. I. & Tanoi, K. Radioactive cesium deposition on rice, wheat, peach tree and soil after nuclear accident in Fukushima. J. Radioanal. Nucl. Chem. 296, 985–989, doi: 10.1007/s10967-012-2154-7 (2012).
Hosoda, M. et al. Activity concentrations of environmental samples collected in Fukushima Prefecture immediately after the Fukushima nuclear accident. Sci. Rep. 3, 2283, doi: 10.1038/srep02283 (2013).
Hosoda, M. et al. The time variation of dose rate artificially increased by the Fukushima nuclear crisis. Sci. Rep. 1, 87, doi: 10.1038/srep00087 (2011).
Kavasi, N. et al. Measurement of 90Sr in soil samples affected by the Fukushima Daiichi Nuclear Power Plant accident. J. Radioanal. Nucl. Chem. 303, 2565–2570, doi: 10.1007/s10967-014-3649-1 (2014).
Kavasi, N. et al. Measurment of 90Sr in contaminated Fukushima soils using liquid scintillation counter. Radiat. Prot. Dosim. doi: 10.1093/rpd/ncv282 (2015).
Steinhauser, G., Schauer, V. & Shozugawa, K. Concentration of strontium-90 at selected hot spots in Japan. PloS one 8, e57760, doi: 10.1371/journal.pone.0057760 (2013).
Mishra, S., Sahoo, S. K., Arae, H., Watanabe, Y. & Mietelski, J. W. Activity Ratio of Caesium, Strontium and Uranium with Site Specific Distribution Coefficients in Contaminated Soil near Vicinity of Fukushima Daiichi Nuclear Power Plant. J. Chromatogr. Sep. Tech. 5, 1000250, doi: 10.4172/2157-7064.1000250 (2014).
L’Annunziata, M. F. in Handbook of radioactivity analysis (ed Michael F. L’Annunziata ) Ch. 1, 60–61 (Academic Press, 2003).
Cook, G. T., Passo, C. J. & Carter, B. In Handbook of radioactivity analysis (ed Michael F. L’Annunziata ) Ch. 6, 577–580 (Academic Press, 2003).
Katata, G., Terada, H., Nagai, H. & Chino, M. Numerical reconstruction of high dose rate zones due to the Fukushima Dai-ichi Nuclear Power Plant accident. Journal of environmental radioactivity 111, 2–12, doi: 10.1016/j.jenvrad.2011.09.011 (2012).
Watanabe, T. et al. Distribution of artificial radionuclides (110mAg, 129mTe, 134Cs, 137Cs) in surface soils from Miyagi Prefecture, northeast Japan, following the 2011 Fukushima Dai-ichi nuclear power plant accident. Geochem. J. 46, 279–285, doi: 10.2343/geochemj.2.0205 (2012).
Solecki, J. & Chibowski, S. Studies on Horizontal and Vertical Migration of 90Sr in Soil Systems. Pol. J. Environ. Stud. 11, 157–163 (2002).
Takagai, Y., Furukawa, M., Kameo, Y. & Suzuki, K. Sequential inductively coupled plasma quadrupole mass-spectrometric quantification of radioactive strontium-90 incorporating cascade separation steps for radioactive contamination rapid survey. Anal. Methods 6, 355–362, doi: 10.1039/C3ay41067f (2014).
Maekawa, A., Momoshima, N., Sugihara, S. & Tamari, T. Determination of radiostrontium released from Fukushima Daiichi Nuclear Power Plant through extraction chromatography and liquid scintillation counting. Paper presented at International Symposium on Environmental monitoring and dose estimation of residents after accident of TEPCO’s Fukushima Daiichi Nuclear Power Stations: Shiran Hall, Kyoto, Japan. Kyoto, Japan: Office KUR Research Program for Scientific Basis of Nuclear Safety, Research Reactor Institute, Kyoto University. (2012, December 14).
Ministry of Education, Culture, Sports, Science and Technology-Japan. Distribution Map for Radiation Dose. (2011) Available at: http://ramap.jmc.or.jp/map/engpdf/pdf/air/20110828/dr/5640-D.pdf. (Accessed: 14th September 2015).
Sahoo, S. K. et al. Naturally occurring radionuclides and rare earth elements in weathered Japanese soil samples. Acta. Geophys. 61, 876–885, doi: 10.2478/s11600-013-0131-3 (2013).
Łokas, E., Mietelski, J. W., Kleszcz, K. & Tomankiewicz, E. A sequential procedure for determining 238Pu, 239+240Pu, 241Am, 90Sr, U and Th activities in soils and peats from Spitsbergen. Nukleonika 55, 195−199 (2010).
Acknowledgements
This research was supported partially by a Grant-in-Aid for Scientific Research (15F14801) from the Japan Society for the Promotion of Science and Fukushima Prefecture related to Research and Development in Radiological Sciences.
Author information
Authors and Affiliations
Contributions
S.K.S. designed the study. S.T. and A.S. arranged the official permission to collect samples from FDNPP exclusion zone with S.K.S. and N.K. N.K. prepared the soil samples for analysis. H.A. carried out the gamma-spectroscopy and mass spectrometry measurement. J.W.M., E.L., S.K.S. and N.K. accomplished the 90Sr analysis. N.K. and S.K.S. wrote the manuscript. S.K.S. and S.Y. contributed to discussions about the results and commented on the manuscript. S.K.S. supervised the whole study. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sahoo, S., Kavasi, N., Sorimachi, A. et al. Strontium-90 activity concentration in soil samples from the exclusion zone of the Fukushima daiichi nuclear power plant. Sci Rep 6, 23925 (2016). https://doi.org/10.1038/srep23925
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep23925
This article is cited by
-
Simultaneous isotopic analysis of fission product Sr, Mo, and Ru in spent nuclear fuel particles by resonance ionization mass spectrometry
Scientific Reports (2023)
-
LARCalc, a tool to estimate sex- and age-specific lifetime attributable risk in populations after nuclear power plant fallout
Scientific Reports (2023)
-
Nanostructure of humic acid adsorption layer in the presence of Cs and Sr ions on the surface of waste material obtained from residue after supercritical extraction of hops
Applied Nanoscience (2023)
-
A part per trillion isotope ratio analysis of 90Sr/88Sr using energy-filtered thermal ionization mass spectrometry
Scientific Reports (2022)
-
90Sr and stable element levels in bones of brown bears: long-term trends in bear populations from Croatia and Poland
Environmental Science and Pollution Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.