Abstract
Increase in atmospheric CO2 concentration ([CO2]) and associated soil warming along with global climate change are expected to have large impacts on grain mineral nutrition in wheat. The effects of CO2 elevation (700 μmol l−1) and soil warming (+2.4 °C) on K, Ca and Mg concentrations in the xylem sap and their partitioning in different organs of wheat plant during grain filling were investigated. Results showed that the combination of elevated [CO2] and soil warming improved wheat grain yield, but decreased plant K, Ca and Mg accumulation and their concentrations in the leaves, stems, roots and grains. The reduced grain mineral concentration was attributed to the lowered mineral uptake as exemplified by both the decreased stomatal conductance and mineral concentration in the xylem sap. These findings suggest that future higher atmospheric [CO2] and warmer soil conditions may decrease the dietary availability of minerals from wheat crops. Breeding wheat cultivars possessing higher ability of mineral uptake at reduced xylem flux in exposure to climate change should be a target.
Similar content being viewed by others
Introduction
Wheat (Triticum aestivum L.) is one of the most important agricultural food crops worldwide, providing around 20% of protein in the human diet. It is also an important source for minerals1. However, the human nutrition depending on wheat grains might be threatened by the climate change2. The increasing atmospheric CO2 concentration (hereafter abbreviated to [CO2]) has been reported to decrease the concentrations of minerals, including the macro-elements, such as magnesium (Mg), calcium (Ca) and potassium (K), micro-elements and trace elements in wheat grains3,4.
Several reasons may account for the reduced mineral nutrition of wheat grains under CO2 elevation. Dilution by increased accumulation of carbohydrates under high [CO2] is a likely cause, but it cannot explain all micronutrient reductions well5. For instance, grain mass was not influenced by CO2 elevation, whereas cadmium concentration was lowered4. On the other hand, limited uptake of nutrients by roots under elevated [CO2] could be another possible reason6,7. This reduced root uptake could be related to factors influencing plant transpiration, since most of minerals are transported from root to shoot in the transpiration stream via the xylem5. The nutrient transport rate in the xylem is mainly determined by two independent factors: the sap flow rate and the concentration of nutrients in the xylem sap6,8. In exposure to elevated [CO2], wheat increases photosynthesis but reduces the stomatal conductance resulting in lower transpiration rate9,10. The lowered transpiration rate could limit the transport of minerals from the root to shoot, hereby reduce their concentration in the grains5. However, the effect of elevated [CO2] on xylem concentrations of minerals and their relations to nutrient loading to the grains in wheat remain largely elusive.
The possibility of changed mineral uptake by root should also be directly related to the changes in whole-plant growth including roots and the allocation of minerals to the different organs under elevated [CO2]11,12. In particular, elevated [CO2] and the associated global warming have been reported to have significant effects on physiological processes and grain yield in wheat13,14. The temperature trend under climate change is indicated as not only air temperature increase but also rise in soil temperature, which has been reported to limit wheat crop development and yield15,16, especially the root growth17. In barley, the concentrations of K and Ca in grain were increased with soil warming (+2.5 °C), while concentration of Mg was slightly reduced15. The elevated [CO2] induced changes in grain mineral concentration have been well studied and lowered nutritional value of wheat by raising atmospheric [CO2] is significant1,18,19,20. Yet, to date the combined effects of elevated [CO2] and soil warming on wheat grain mineral status have not been well illustrated.
Therefore, the objective of this experiment was to explore the mechanisms by which elevated [CO2] and soil warming, individually and in combination, affect the mineral nutrition in wheat grain. It was hypothesized that: (i) combination of elevated [CO2] and soil warming would improve grain yield while reduce grain mineral concentration; (ii) the xylem transport of nutrients would be decreased by this combination due to lowered stomatal conductance and decreased mineral concentration in the xylem sap, both are responsible for the reduced mineral concentration in wheat grains.
Results
Grain yield and phenological dates
Grain yield per pot (PC = 0.003) and kernel per spike (PC < 0.001) were significantly increased by elevated [CO2] (Table 1). Soil temperature had no significant effects on grain yield, kernel number per spike, spike number per pot and thousand-kernel weight, neither did the interaction between [CO2] and soil temperature treatments. In addition, the dates of anthesis and maturity were not significantly affected by elevated [CO2] and soil warming (Data not shown).
Mineral concentrations in wheat organs
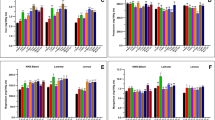
The concentrations of K, Ca and Mg in leaves were the highest, while those in the grains were the lowest among the organs (Fig. 1). Elevated [CO2] and soil warming alone decreased K concentration in leaves and roots and this effect was much stronger with their combination (PC×T < 0.001). In both soil warmed and unwarmed pots, elevated [CO2] decreased K concentration in the stems and grains, while soil warming alone had no significant effect. Higher Ca concentration in roots was found in the soil warming treatments (i.e., AW and EW), but no significant difference was found between the control (AN) and soil warming treatments in stem Ca concentration. Soil warming reduced Ca concentration in leaves and grains under elevated [CO2], but had no effect on the leaf and grain Ca concentrations under ambient [CO2]. In addition, the combination of elevated [CO2] and soil warming strongly decreased Ca concentration in the leaves and grains (PC×T < 0.001). Root Mg concentration was higher in plants grown under ambient [CO2] than those grown under elevated [CO2]. In the stems and grains, Mg concentration was decreased by either elevated [CO2] or the combination of elevated [CO2] and soil warming. In addition, leaf Mg concentration was also decreased by elevated [CO2] and soil warming treatments.
Effects of CO2 elevation and soil warming on concentrations of K, Ca and Mg among organs in wheat.
Mean values ± SE for each combination of CO2 level (A = ambient, E = elevated) and soil temperature (N = non-warmed, W = warmed) are shown (n = 4). Different small letters indicate significant difference at P < 0.05 level.
Plant K, Ca and Mg accumulation
Compared to the control (AN), K accumulation was decreased by 11.5, 9.0 and 11.3% in EN, AW and EW plants, respectively (Fig. 2). In relation to the AN plants, Ca accumulation was decreased by 10.7 and 10.9% in EN and EW plants, respectively; but was not affected by soil warming alone (Fig. 2). Plant Mg accumulation was influenced by the treatments in a similar pattern as for K; EN, AW and EW treatments decreased Mg accumulation by 18.5, 15.3 and 30.1%, respectively, as compared to the AN control (Fig. 2).
Effects of CO2 elevation and soil warming on K, Ca and Mg accumulation in wheat plants.
Mean values ± SE for each combination of CO2 level (A = ambient, E = elevated) and soil temperature (N = non-warmed, W = warmed) are shown (n = 4). Different small letters indicate significant difference at P < 0.05 level.
Changes in K, Ca, Mg concentrations during grain filling
Changes of the concentrations of K, Ca and Mg in the developing grains, measured five times during grain filling as affected by elevated [CO2] and soil warming are shown in Fig. 3. Elevated [CO2] significantly decreased K concentration in the developing grains on 17 and 31 DAA and the mature grains. The soil warming induced reduction of K concentration was significant from 31 DAA to maturity. However, the interactive effect of elevated [CO2] and soil warming was not statistically significant. Elevated [CO2] significantly decreased Ca concentration in the developing grains from 23 DAA onwards, whereas the decrease in Ca concentration caused by soil warming was not significant during grain filling. Mg concentration was significantly decreased by the combination of elevated [CO2] and soil warming from 17 DAA to maturity and which was mainly ascribed to the negative effect of elevated [CO2] on grain Mg concentration. Compared to the control plants, the reduction in Mg concentration under elevated [CO2] become larger in both warmed and unwarmed pots from 17 DAA onwards.
Effects of CO2 elevation and soil warming on concentrations of K, Ca and Mg in wheat grain during grain filling.
Mean values ± SE for each combination of CO2 level (A = ambient, E = elevated) and soil temperature (N = non-warmed, W = warmed) are shown (n = 4). *,***indicate significant at P < 0.05 and P < 0.001, respectively; NS, not significant.
Neither [CO2] nor soil temperature had significant effect on grain dry weight resulting in similar dry weight of mature grains among these treatments (Table 1). However, the grain filling rate was seemingly greater in plants grown under elevated [CO2] than those grown under ambient [CO2], causing an earlier cessation of dry weight accumulation (Data not shown). Grain moisture content declined steadily from 10 DAA onwards and dropped rapidly after 31 DAA (Data not shown). The grain moisture content was approximately 20% at maturity in all treatments. It was interesting that elevated [CO2] increased the grain moisture content on 24 and 31 DAA. Whilst, soil warming decreased the grain moisture content significantly on 17 and 31 DAA.
Stomatal pore aperture and stomatal conductance
Figure 4 shows the SA remained unchanged till 17 DAA, thereafter it decreased gradually in all treatments. Three-way ANOVA shows that SA was only affected significantly by [CO2] treatment. The SA was the highest in AN and followed by AW and the lowest in EN and EW. Under ambient [CO2], gs increased initially, peaking around 5 DAA and tapering off gradually afterward; while for plants grown under elevated [CO2] the decrease in gs occurred after 10 DAA. Elevated [CO2] significantly reduced the gs of flag leaves, while soil warming had no significant effect on the gs: the gs of AN plants was the highest followed by AW and the lowest in EN and EW.
Effects of CO2 elevation and soil warming on stomatal pore aperture (SA) and stomatal conductance (gs) of wheat leaves during grain filling.
Mean values ± SE for each combination of CO2 level (A = ambient, E = elevated) and soil temperature (N = non-warmed, W = warmed) are shown (n = 4). *,*** indicate significant at P < 0.05 and P < 0.001, respectively; NS, not significant.
Correlations between stomatal conductance and stomatal pore aperture
Significant positive linear relationship between gs and SA was found across the four treatments during grain filling (Fig. 5).
Ionic concentrations in the xylem sap
The concentrations of K+, Ca2+ and Mg2+ in the xylem sap of wheat plants were all significantly decreased by elevated [CO2] and soil warming (Table 2). In relation to the AN controls, the reductions of K+, Ca2+ and Mg2+ concentrations in the xylem sap were 6.5, 12.6 and 20.6%, respectively, in the EN plants. The lowest concentration of these ions was found in plant grown under the combination of elevated [CO2] and soil warming. Compared to the AN treatment, EW treatment decreased the concentrations of K+, Ca2+ and Mg2+ by 28.2%, 28.6% and 32.2%, respectively.
Discussion
Effects of [CO2] elevation on wheat
Compared to the AN plants, the grain yield increased by 6% in plants grown under elevated [CO2], which was mainly due to an increase in kernel number per spike. Improved photosynthate availability under elevated [CO2] resulting in an increased source activity could have contributed to the greater kernel number in plants grown under elevated [CO2]21. It has been demonstrated that the potential for increase of grain yield in cereals grown at elevated [CO2] is temperature dependent9. Recent studies have shown that grain yield of wheat increases in a climate scenario that combines elevated [CO2] (700 μmol l−1, Tunnel houses) and increased air temperatures (+2 °C, 4 °C or 6 °C) moderately above the current ambient temperature22.
In the present study, elevated [CO2] did not result in greater grain size, in line with previous findings in wheat21,22,23,24. However, it was noticed that the grain filling rate was slightly greater in plants grown under elevated [CO2], causing an earlier cessation of grain weight accumulation (Data not shown). It is well known that the grain moisture content is closely related to the grain filling process and thus the final grain weight25,26. Here, the grain moisture content was higher in the plants grown under elevated [CO2] than those grown under ambient [CO2] on 24 and 31 DAA, which could be due to a greater osmotic potential gradient for water deposition into to the grain25 caused by higher concentration of sucrose under elevated [CO2]. Also, higher grain moisture content may indicate a greater sink capacity (i.e., more endosperm cells) of the grains26 and might result in a bigger grain size at harvest. However, this was apparently not the case. On the contrary, it was found that soil warming treatment decreased grain moisture content on 17 and 31 DAA, which may be associated with a relatively smaller sink of the grains and thus a lower grain size. Again, this was not true as the final grain weight was identical among all treatments. The reasons behind these inconsistencies remain unknown.
It has frequently been observed that [CO2] elevation reduces mineral concentration in plant organs, while increasing the total mineral uptake due to greater plant biomass accumulation26. This has led to the notion that the lowered mineral concentration in plant grown under elevated [CO2] was ascribed to the dilution by the enhanced plant growth5. In the present study, both the concentrations of K, Ca and Mg in the wheat organs and the total accumulations of K, Ca and Mg in the plants were significantly decreased by [CO2] elevation, indicating that the reduced mineral concentrations was not due to dilution by an enhanced growth. This was particularly the case for the grain, as [CO2] elevation had not influenced on the final grain weight, but the K, Ca and Mg concentrations of the grain were significantly lowered. Thus, it is apparent that dilution by greater grain biomass is not the primary reason for reduction in grain mineral concentration under elevated [CO2]. In addition, in the developing grains, the concentrations of K, Ca and Mg in plant grown under elevated [CO2] become lower than those of the AN plants since 10, 23 and 23 DAA, respectively, indicating the translocation of K to the grain during grain filling was more sensitive to [CO2] elevation.
Plant mineral nutrition is determined by both nutrient acquisition by roots and nutrient transport and partitioning among plant organs8. In the present study, the accumulations of K, Ca and Mg in wheat were decreased by elevated [CO2] and the reductions were more profound for Mg, followed by K and Ca the least. It is widely recognized that the elevated [CO2]-induced decrease in plant mineral uptake might be related to the decreased mass flow or diffusion of mineral ions from the soil solution to the root surface due to lower transpiration rate and/or the reduced ability of mineral acquisition by the roots6,26. In literature, there is evidence, although variable, that elevated [CO2] enhances the root growth and root length density and which should enable the plants to acquire more nutrients11,27,28. A positive effect of elevated [CO2] on root growth was also noticed in the present study (Data not shown), which however did not result in higher mineral uptake in the plants as exemplified by the lower mineral concentration in the xylem sap and the reduced total mineral accumulation in the plants. Therefore, lowered transpiration rate and/or root nutrient acquisition ability could have been the main reasons for the decreased mineral uptake in wheat plants grown under elevated [CO2]. In the present study, although the plant transpiration rate was not determined, the lowered gs due to stomatal closure in the wheat plants grown under elevated [CO2] was evident. It is well known that elevated [CO2] reduces SA via depolarizing the guard cell membrane potential, hence leading to stomatal closure29,30. In this study, elevated [CO2] significantly decreased the SA and had no significant effect on stomatal density (data not shown) during grain filling in wheat. It has been reported that the short-term changes in SA likely determines most of the long-term response of gs to elevated [CO2]31. Also, it was found that along with the progress of senescence of the flag leaf, both gs and SA were declined and the differences between the treatments were diminished on 24 DAA. Accordingly, elevated [CO2] induced partial stomatal closure might have reduced the transpiration rate of the wheat plants resulting in decreased mass flow of the minerals to the root surface6, which in turn reduced the root nutrient uptake leading to significant lower concentrations of K+, Ca2+ and Mg2+ in the xylem sap. In good agreement with this, a recent study on wheat reported that the decrease of uptake of Ca and Mg under [CO2] elevation is not only by a reduced transpiration flow, but is further reduced by lowered Ca2+ and Mg2+ concentrations in the xylem6. However, it has been proposed that the effect of transpiration rate on mineral uptake is more pronounced with those primarily transported via mass flow (e.g., Ca and Mg) than those via diffusion (e.g., P and K)5. Consistent with this, here the reduction of xylem sap K+ concentration (6.4%) caused by elevated [CO2] was much less compared to those for Ca2+ (12.6%) and Mg2+ (20.6%). The lowered transpiration rates (as exemplified by the reduced gs) and in combination with the decreased mineral concentrations in the transpiration flux (i.e., the xylem sap) could have caused reductions in mineral accumulation in the wheat plants in response to [CO2] elevation. In addition, here the effect of [CO2] elevation on plant Ca (10.6% reduction) accumulation was found to be less profound than that on Mg (18.6% reduction), which coincided with the reductions in the concentrations of those ions in the xylem sap. However, the 6.4% reduction of K+ concentration in the xylem could not fully explain the 10.7% reduction of K accumulation in the plant caused by elevated [CO2]. Thus, other factors relating to K metabolisms in the plant might have been involved. We proposed that the relative small reduction of K concentration in the xylem sap of wheat could be a result of shoot-derived K recycled in the phloem and reloaded into the xylem in the roots32.
Effects of soil warming on wheat
In the present study, a slight reduction in grain yield was found with soil warming, where the difference was statistically insignificant. Consistent with this, Weldearegay et al.23 also found that soil warming only decreased seed set but had no significant effect on grain yield in spring wheat cultivars. Soil warming effect on mineral uptake could be attributed to its effects on root and shoot growth17, on the bioavailability of soil nutrients under higher soil temperature15, or on the root nutrients uptake capacity33. Studies have shown that an increase in the root temperature results in an increase in K concentration but a decrease in Ca concentration of the xylem sap34. Compared with Ca and Mg, uptake of K is often more affected by root zone temperatures35. Here, an increase of soil temperature by 2.4 °C significantly decreased K+, Ca2+ and Mg2+ concentrations in the xylem sap and the reduction was greater for Mg2+ (22.9%) than for K and Ca (both ca. 15%), contrasting to earlier findings34. It has been reported that the optimal temperature for root growth of wheat is lower than 20 °C36. In the present study, the day/night temperature in the greenhouse cells were 25/16 ± 2 °C, if the soil temperature was the same, then 2.4 °C increase of soil temperature in the AW treatment would result in soil temperature much higher than the optimal, i.e., 20 °C, thus might have negatively affected the root growth of the wheat plants causing a reduction of acquisition area for mineral nutrients. This may partially account for the reduced mineral concentration in the xylem sap. Besides, the total K, Ca and Mg accumulated in the AW plants was 9.0, −0.2 and 15.3%, respectively, lower than those of the AN plants, indicating that plant Ca accumulation was unaffected by soil warming alone. However, the reasons for the different effect of elevated [CO2] on wheat K, Ca and Mg accumulation remain unknown. In the developing grains, in relation to the AN treatment, only Mg concentration was significantly lowered by soil warming since 24 DAA; while the concentrations of K and Ca were identical between the two treatments.
Combined effect of [CO2] elevation and soil warming on wheat
Results of this study have clearly shown that the mineral concentrations in the organs (except for Ca in root) and in the xylem sap and the total mineral accumulation of wheat plants were all significantly reduced by the combined [CO2] elevation and soil warming treatment. For most of the variables observed, the combined effects were more pronounced than the individual effect of [CO2] elevation or soil warming, especially for the mineral concentrations in the leaf, in the xylem sap and the total K and Mg accumulation in the plants, where soil warming had exacerbated the negative effect of elevated [CO2] on those variables. Although neither of the grain yields components was affected by the combination of [CO2] elevation and soil warming, the dramatic effect on mineral concentrations of the wheat plants particularly those in the grains will reduce the grain quality in terms of human nutrition. Thus, selection of wheat cultivars with greater mineral uptake efficiency will of strategic importance to sustain wheat production and grain quality in future climate change scenarios.
Conclusion
The elevated [CO2] increased wheat grain yield due to higher grain number per spike, but reduced the concentrations of K, Ca and Mg in the grain and plant organs. The decreased grain mineral concentration was caused primarily by decreased mineral uptake and was not due to a dilution effect. Elevated [CO2]-driven stomatal closure resulted in lower gs, exerted an important role in reduction of mineral uptake, especially for Ca and Mg, in wheat plant. On the other hand, the decreased mineral concentration in xylem sap which could be partially resulted from a reduced capacity of root nutrients uptake might also be responsible for the reduced mineral nutrition in wheat plants. These results documented that future higher [CO2] and warmer soil conditions would decrease the dietary availability of K, Ca and Mg from wheat crops.
Methods
Experimental setup
A pot experiment was conducted from February to July 2014 in CO2 controlled greenhouses (Agrotech, Taastrup, Denmark) in Faculty of Science, University of Copenhagen, Taastrup, Denmark. Eight selected seeds of winter wheat (cv. Lianmai6) were sown per pot (25 cm in height and 15.2 cm in diameter) filled with 2.4-kg peat material (Sphagnum, 32% organic matter, pH = 5.6–6.4 and EC = 0.45 ms cm−1). After sowing, half of the pots in each greenhouse were placed on a heating carpet as a sub-group and the soil temperature was increased by 2.4 °C compared with another sub-group22. Four seedlings were retained after thinning at the 3rd leaf stage. The pots in the same sub-group were rotated on a daily basis to avoid pseudo replication and heterogeneity of environment inside the greenhouse. The pots were watered daily to keep the soil at an optimal moisture condition (i.e., 80–85% soil relative water content). At jointing stage, all plants were drip irrigated with nutrient solution (in total 2 g N, 1 g P and 1.4 g K were applied to each pot) to prevent any deficiency of nutrients. The photosynthetic active radiation (15 h photoperiod and >500 μmol m−2 s−1) was supplied by sunlight plus metal-halide lamps. The air temperature and relative humidity were 25/16 ± 2 °C (day/night) and 60 ± 5% in the cells, respectively.
Treatments
Each sub-group served as a combination of atmospheric [CO2] and soil temperature regime, the four treatments are: (i) Ambient [CO2] (380 μmol l−1) + normal soil temperature (AN); (ii) Elevated [CO2] (700 μmol l−1) + normal soil temperature (EN); (iii) Ambient [CO2] (380 μmol l−1) + soil warming (+2.4 °C) (AW); (iv) Elevated [CO2] (700 μmol l−1) + soil warming (+2.4 °C) (EW).
Sampling and mineral concentration analysis
On 10, 17, 24 and 31 days after anthesis (DAA), wheat grains were harvested and weighed (g fresh weight, g FW) and then dried at 70 °C in an oven to constant weight for determining the dry weight (g dry weight, g DW). Grain moisture content was calculated according to grain fresh weight and dry weight. At each sampling date, four independent samples, each composed of four plants, were collected. At maturity, the plants were separated into root, stem, leaf and grain for mineral analysis. Concentrations of K, Ca and Mg were analyzed after high-pressure digestion with nitric acid (UltraClave III, MLS, Leutkirch, Germany) using inductively-coupled plasma optical emission spectrometry (ICP-OES 720, Varian, Palo Alto, CA, USA). At maturity, grain was harvested and spike number, kernel per spike and thousand-kernel weight were determined on 4 pots from each treatment.
Xylem sap collection and ionic concentration analysis
For xylem sap collection, the entire pot was sealed into a Scholander-type pressure chamber and all stems were faggoted and detopped at 10 cm from the stem base. Xylem sap was collected on 10 DAA using a pipette from the cutting surface into an Eppendorf vial wrapped with aluminium foil. All sap samples were frozen immediately in liquid nitrogen after sampling and stored at −80 °C until analysis. Concentrations of K+, Ca2+ and Mg2+ in the xylem sap were determined by ion chromatography (Metrohm AG, Herisau, Switzerland) using a Metrosep C4-100 analytical column (4 × 125 mm, 1.7 mM nitric acid/0.7 mM dipicolinic acid eluent)32.
Stomatal conductance
Stomatal conductance (gs) of flag leaves was measured using a leaf porometer (Decegon Devices, Pullman, WA, USA) at anthesis and on 5, 10, 17 and 24 DAA.
Stomatal pore aperture
Epidermal impressions of both adaxial and abaxial leaf surfaces were taken from three flag leaves in each treatment at anthesis and on 5, 10, 17 and 24 DAA. Fingernail polish imprints were obtained halfway from the leaf tip to the base of each leaf and using clear cellophane tape to transfer the ‘impression’ to a microscope slide37. The imprints were observed under a LEITZ DMRD microscope camera system (Leica Microscope and System GmbH, D35530, Wetzlar, Germany) equipped with a digital camera and the images were presented using image-editing software (Leica Microsystems, version 2.5.0, CMS GmbH, Switzerland) on a computer screen. Stomatal pore aperture length (La) and stomatal pore aperture width (Wa) were measured with the images using UTHSCSA ImageTool software (UTHSCSA Image Tool for Windows version 3.00). Stomatal pore aperture (SA) was calculated according to Doheny-Adams et al.38 as the following equations:

where Wa is pore aperture width, La is pore aperture length.
Statistical analysis
For SA and concentrations of K+, Ca2+ and Mg2+ in the xylem sap and in different organs and plant total K, Ca and Mg accumulation amount, the interactive effect between [CO2] and soil temperature was analyzed by two-way ANOVA using the Duncan test (P < 0.05) (SigmaSATA, V3.5, Systat Software, CA, USA). For grain dry weight, moisture content, gs and grain concentrations of K, Ca2+ and Mg2+ during grain filling, post hoc ANOVAs were used to illustrate significant CO2 × soil temperature, CO2 × date or soil temperature × date interactions (SPSS version 20.0 for windows, IBM SPSS Statistics, Chicago, IL, USA).
Additional Information
How to cite this article: Li, X. et al. Soil warming enhances the hidden shift of elemental stoichiometry by elevated CO2 in wheat. Sci. Rep. 6, 23313; doi: 10.1038/srep23313 (2016).
References
Shewry, P. R. Wheat. J. Exp. Bot. 60, 1537–1553 (2009).
Myers, S. S. et al. Increasing CO2 threatens human nutrition. Nature 510, 139–142 (2014).
Fernando, N. et al. Intra-specific variation of wheat grain quality in response to elevated [CO2] at two sowing times under rain-fed and irrigation treatments. J. Cereal Sci. 59, 137–144 (2014).
Pleijel, H. & Högy, P. CO2 dose–response functions for wheat grain, protein and mineral yield based on FACE and open-top chamber experiments. Environ. Pollut. 198, 70–77 (2015).
McGrath, J. M. & Lobell, D. B. Reduction of transpiration and altered nutrient allocation contribute to nutrient decline of crops grown in elevated CO2 concentrations. Plant Cell Environ. 36, 697–705 (2013).
Houshmandfar, A., Fitzgerald, G. J. & Tausz, M. Elevated CO2 decreases both transpiration flow and concentrations of Ca and Mg in the xylem sap of wheat. J. Plant Physiol. 174, 157–160 (2015).
Taub, D. R. & Wang, X. Why are nitrogen concentrations in plant tissues lower under elevated CO2? A critical examination of the hypotheses. J. Integr. Plant Biol. 50, 1365–1374 (2008).
White, P. J. Marschner H, Marschner P. Mineral Nutrition, Yield and Source–Sink Relationships. In: Marschner’s mineral nutrition of higher plants, Amsterdam: Elsevier, 85–133.(2012).
Dahal, K., Knowles, V. L., Plaxton, W. C. & Hüner, N. P. A. Enhancement of photosynthetic performance, water use efficiency and grain yield during long-term growth under elevated CO2 in wheat and rye is growth temperature and cultivar dependent. Environ. Exp. Bot. 106, 207–220 (2014).
Rubio-Asensio, J. S., Rachmilevitch, S. & Bloom, A. J. Responses of Arabidopsis and wheat to rising CO2 depend on nitrogen source and nighttime CO2 levels. Plant Physiol. 168, 156–163 (2015).
Benlloch-Gonzalez, M., Berger, J., Bramley, H., Rebetzke, G. & Palta, J. A. The plasticity of the growth and proliferation of wheat root system under elevated CO2 . Plant Soil 374, 963–976 (2013).
Berntson, G. M. & Bazzaz, F. A. Belowground positive and negative feedbacks on CO2 growth enhancement. Plant Soil 187, 119–131 (1995).
Gutierrez, D. et al. Acclimation to future atmospheric CO2 levels increases photochemical efficiency and mitigates photochemistry inhibition by warm temperatures in wheat under field chambers. Physiol. Plantarum 137, 86–100 (2009).
Wang, L., Feng, Z. & Schjoerring, J. K. Effects of elevated atmospheric CO2 on physiology and yield of wheat (Triticum aestivum L.): A meta-analytic test of current hypotheses. Agr. Ecosyst. Environ. 178, 57–63 (2013).
Högy, P., Poll, C., Marhan, S., Kandeler, E. & Fangmeier, A. Impacts of temperature increase and change in precipitation pattern on crop yield and yield quality of barley. Food Chem. 136, 1470–1477 (2013).
Patil, R. H., Laegdsmand, M., Olesen, J. E. & Porter, J. R. Growth and yield response of winter wheat to soil warming and rainfall patterns. J. Agr. Sci. 148, 553–566 (2010).
Gavito, M. E., Curtis, P. S., Mikkelsen, T. N. & Jakobsen, I. Interactive effects of soil temperature, atmospheric carbon dioxide and soil N on root development, biomass and nutrient uptake of winter wheat during vegetative growth. J. Exp. Bot. 52, 1913–1923 (2001).
Erbs, M. et al. Effects of free-air CO2 enrichment and nitrogen supply on grain quality parameters and elemental composition of wheat and barley grown in a crop rotation. Agr. Ecosyst. Environ. 136, 59–68 (2010).
Sanchez de la Puente, L., Pérez Pérez, P., Martinez-Carrasco, R., Morcuende Morcuende, R. & Martín del Molino, I. Action of elevated CO2 and high temperatures on the mineral chemical composition of two varieties of wheat. Agrochimica 44, 221–230 (2000).
Högy, P. et al. Effects of elevated CO2 on grain yield and quality of wheat: results from a 3-year free-air CO2 enrichment experiment. Plant Biol. 11, 60–69 (2009).
Dias de Oliveira, E. et al. Can elevated CO2 combined with high temperature ameliorate the effect of terminal drought in wheat? Funct. Plant Biol. 40, 160 (2013).
Weldearegay, D. F., Yan, F., Jiang, D. & Liu, F. Independent and combined effects of soil warming and drought stress during anthesis on seed set and grain yield in two spring wheat varieties. J. Agron. Crop Sci. 198, 245–253 (2012).
Uddling, J., Gelang-Alfredsson, J., Karlsson, P. E., Selldén, G. & Pleijel, H. Source–sink balance of wheat determines responsiveness of grain production to increased CO2 and water supply. Agr. Ecosyst. Environ. 127, 215–222 (2008).
Jenner, C. F., Ugalde, T. D. & Aspinall, D. The physiology of starch and protein deposition in the endosperm of wheat. Funct. Plant Biol. 18, 211–226 (1991).
Schnyder, H. & Baum, U. Growth of the grain of wheat (Triticum aestivum L.). The relationship between water content and dry matter accumulation. Eur. J. Agron. 1, 51–57 (1992).
Conroy, J. P. Influence of elevated atmospheric CO2 concentrations on plant nutrition. Aust. J. Bot. 40, 445–456 (1992).
Pacholski, A., Manderscheid, R. & Weigel, H.-J. Effects of free air CO2 enrichment on root growth of barley, sugar beet and wheat grown in a rotation under different nitrogen supply. Eur. J. Agron. 63, 36–46 (2015).
Stulen, I. & Den Hertog, J. Root growth and functioning under atmospheric CO2 enrichment. Vegetatio 104, 99–115 (1993).
Leakey, A. D. et al. Elevated CO2 effects on plant carbon, nitrogen and water relations: six important lessons from FACE. J. Exp. Bot. 60, 2859–2876 (2009).
Assmann, S. The cellular basis of guard cell sensing of rising CO2 . Plant Cell Environ. 22, 629–637 (1999).
Ainsworth, E. A. & Rogers, A. The response of photosynthesis and stomatal conductance to rising [CO2]: mechanisms and environmental interactions. Plant Cell Environ. 30, 258–270 (2007).
Wang, Y., Liu, F. & Jensen, C. R. Comparative effects of deficit irrigation and alternate partial root-zone irrigation on xylem pH, ABA and ionic concentrations in tomatoes. J. Exp. Bot. 63, 1907–1917 (2012).
Clarkson, D. T. The uptake and translocation of manganese by plant roots. In: Manganese in soils and plants, Berlin: Springer, 101–111.
Engels, C., Munkle, I. & Marschner, H. Effect of root zone temperature and shoot demand on uptake and xylem transport of macronutrients in maize (Zea mays L.). J. Exp. Bot. 43, 537–547 (1992).
Miyasaka, S. C. & Grunes, D. L. Root temperature and calcium level effects on winter wheat forage: I. Shoot and root growth. Agron. J. 82, 236–242 (1990).
Porter, J. R. & Gawith, M. Temperatures and the growth and development of wheat: a review. Eur. J. Agron. 10, 23–36 (1999).
Yan, F., Sun, Y., Song, F. & Liu, F. Differential responses of stomatal morphology to partial root-zone drying and deficit irrigation in potato leaves under varied nitrogen rates. Sci. Hortic. 145, 76–83 (2012).
Doheny-Adam, T., Hunt, L., Franks, P. J., Beerling, D. J. & Gray, J. E. Genetic manipulation of stomatal density influences stomatal size, plant growth and tolerance to restricted water supply across a growth carbon dioxide gradient. Phil. Trans. Royal Soc. B: Biol. Sci. 367, 547–555 (2012).
Acknowledgements
The research was partly support by the Villum Foundation Block Stipend (341/300-123012). Technical assistance by Lene Korsholm Jørgensen, Rene Hvidberg Petersen and Jens Bertelsen is gratefully acknowledged. The authors have declared no conflict of interest.
Author information
Authors and Affiliations
Contributions
D.J. and F.L. conceived the idea and led the study design. X.L. carried out the experiment, performed analyses and wrote the paper. All authors contributed to the editing of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Li, X., Jiang, D. & Liu, F. Soil warming enhances the hidden shift of elemental stoichiometry by elevated CO2 in wheat. Sci Rep 6, 23313 (2016). https://doi.org/10.1038/srep23313
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep23313
This article is cited by
-
Combined Effect of Alternate Partial Root-Zone N-Fertigation and Elevated CO2 on Grain Quality Characteristics of Barley
Journal of Soil Science and Plant Nutrition (2023)
-
Biomass, chemical composition, and microbial decomposability of rice root and straw produced under co-elevated CO2 and temperature
Biology and Fertility of Soils (2020)
-
Changes in plant C, N and P ratios under elevated [CO2] and canopy warming in a rice-winter wheat rotation system
Scientific Reports (2019)
-
ABA-mediated regulation of leaf and root hydraulic conductance in tomato grown at elevated CO2 is associated with altered gene expression of aquaporins
Horticulture Research (2019)
-
Effect of elevated temperature on soil hydrothermal regimes and growth of wheat crop
Environmental Monitoring and Assessment (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.