Abstract
The level of emotional timing deficit is a critical determinant of daily functions and social interactions in people with schizophrenia. This study demonstrated that people with schizophrenia have significant deficits in emotional time perception. Behaviorally, while the healthy controls overestimated the duration of happy and fearful faces, the patients underestimated the duration of emotional and neutral faces. Accordingly, an online ERP index of timing—the contingent negative variation (CNV) displayed larger amplitudes for emotional faces in the controls, whereas the CNV in the patients only showed overall smaller amplitudes when compared with the controls. In addition, the results of the N170 and the CNV suggest that the emotional processing and timing for facial expressions in schizophrenia might have a pattern of two-stage deterioration. Findings from the present work point to the importance of considering the time dimension of emotional processing in schizophrenia, based on which we are likely to discover aspects of emotional deficits that would be unnoticed in other studies. Furthermore, the perception deviation of the duration of emotional faces in schizophrenia suggests us to consider the magnitude of this temporal deviation as a quantitative biomarker for specific emotional/social dysfunctions in schizophrenia.
Similar content being viewed by others
Introduction
The past two decades of research on emotional processing in schizophrenic populations has revealed that the impaired processing of emotion, which is evident at the onset of psychosis and persists during acutely symptomatic and remitted states, represents a trait susceptibility marker and a unique endophenotype for schizophrenia1,2. Compared to people without schizophrenia, patients with schizophrenia display impaired emotional perception, diminished emotional expression and a relatively normal level of emotional experience3,4,5.
However, when considering the temporal course of emotion, the function of emotional processing in schizophrenic patients may display a more refined or even distinctively different pattern5. For instance, studies have demonstrated that the timing of emotional expression/experience is different between people with and without schizophrenia: people with schizophrenia are able to exhibit emotionally congruent, albeit less observable, expressions to facial pictures, but only during the first 500 ms of picture presentation6; people with and without schizophrenia show comparable regions of brain activation in the presence of emotional pictures, but patients cannot maintain their emotional experience responses after the offset of stimuli7. Furthermore, many researchers are currently stressing the importance of characterizing the temporal course of emotional experience to distinguish anticipatory from consummatory responses in schizophrenia, since accumulating evidences reveal that people with schizophrenia do not have a deficit in consummatory pleasure but instead have a deficit in anticipatory pleasure8,9,10; and that it is the deficit in anticipatory pleasure that results in a symptom of anhedonia9. (Note: consummatory pleasure is the pleasure being received when individuals are directly engaged in an enjoyable activity; anticipatory pleasure is the experience of pleasure related to future activities9).
Whereas these results consistently support the notion that people with schizophrenia have difficulty in anticipating emotional events and maintaining their emotional responses and experiences5, analysis of the complex interplay between emotion and timing remains relatively rare in this population. Impaired time perception of emotional stimuli, such as faces and voices, is associated with problems in synchronizing our activity with other individuals, adopting others’ rhythms and understanding the intentions of others11. In addition, the core manifestations of schizophrenia such as thought disorder and contextually inappropriate responses may be partly due to the time perception dysfunction for emotional events/cues12. Thus, the level of emotional timing deficit appears to be a critical determinant of daily functioning and social interactions in schizophrenia.
The present study investigated and compared the behavioral and the event-related potential (ERP) responses of emotional time perception in individuals with and without schizophrenia. Since emotional facial expressions play an essential role in social communication, pictures of emotional faces were used as the target of time perception11. Accordingly, we investigated ERP components of P1, N170, the vertex positive potential (VPP) and the contingent negative variation (CNV) in this study. The P1 reflects bottom-up visual processing and is sensitive to attention and stimulus parameters13. The N170 and its positive counterpart VPP are face-sensitive components; both are frequently modulated by emotional faces, with larger amplitudes in response to emotional, compared to neutral, facial expressions14,15. The CNV is a slow negative potential which has been shown in numerous studies of time perception as an online index of timing16,17; its amplitude correlates positively with the length of the estimated duration18,19,20. It is expected that the emotion-sensitive N170/VPP would show different patterns across emotional conditions between schizophrenic patients and healthy controls; and that the timing-sensitive CNV displays not only group differences but also a significant interaction of emotion by group, reflecting that general time perception and emotion-modulated time perception are both impaired in individuals with schizophrenia.
This study also aims to investigate the potential relationship between the deficits in emotional time perception and the emotional/social impairments in schizophrenic patients. Clinically, the severity of emotional/social impairments is often assessed using the Positive and Negative Syndrome Scale (PANSS)21. An accumulating literature suggests that the severity of negative, rather than positive, symptoms, reflects poor performances on emotion-related tasks4,22 and severe social dysfunctions in schizophrenic patients23,24. Factor analytic studies suggest that negative symptoms consistently have two factors linked to emotional/social dysfunction24,25. Among the seven items of negative symptoms, three (“blunted affect”, “poor rapport” and “lack of spontaneity and flow of conversation”) are grouped into the dimension of emotional expressive deficit (or diminished expression) and two (“emotional withdrawal” and “passive social withdrawal”) are grouped into the dimension of social amotivation (or anhedonia-asociality)23,26. Accordingly, these five negative items are our major concerns in this study. It is expected that the ERP abnormity found in the emotional time perception task would correlate with specific negative items in the PANSS.
Methods
Participants
Forty-seven inpatients (27 males) of Beijing Huilongguan Hospital and forty-six (26 males) normal controls were recruited as paid participants.
Patients were diagnosed according to the criteria for Schizophrenia in Diagnostic and Statistical Manual (DSM-IV)27. The diagnosis was based on structured clinical interview for DSM (SCID)28. Patients with schizoaffective disorder, schizotypal or schizoid personality disorder were excluded. None of the patients were in a major depressive or manic episode at the time of testing. Additional exclusion criteria included: 1) history of significant brain trauma, 2) neurological disorder, 3) substance abuse or dependence in the past six months, 4) IQ < 70 and 5) who had received electroconvulsive therapy in the past six months. All patients were receiving stable medication treatments (no medication changes) for at least one month before the experiment. Patients’ symptom severity was assessed using the PANSS within ten days of ERP assessment.
Healthy control participants were screened with the SCID-I/NP29 and SCID-II30. Exclusion criteria were 1) any lifetime Axis I psychotic or mood disorders, 2) recurrent depression, 3) paranoid, schizotypal or schizoid personality disorder, 4) seizure disorder, 5) history of head injury with possible neurological sequelae, 6) the presence of a first-degree relative with schizophrenia and 7) substance abuse or dependence in the past six months.
There was no significant difference between the two groups with respect to age, handedness, IQ, education and sex (Table 1). The interview and clinical symptom rating were based on consensus of two senior psychiatrists who were trained with a high reliability (κ = 0.83). The experimental protocol was approved by the local ethics committee (Beijing Huilongguan Hospital) and this study was performed strictly in accordance with the approved guidelines. Written informed consent was obtained from participants prior to the experiment.
Stimuli and procedure
The stimuli used for the representation of duration were a grey oval and three types of facial pictures, namely, happy, fearful and neutral faces. Facial pictures were black and white photographs selected from the Chinese Facial Affective Picture System31, with equal number of facial pictures between males and females. A total of 72 faces were used (24 happy, 24 fearful and 24 neutral ones). All stimuli were presented with the same contrast and brightness on the black background (4.0° × 4.6° visual angle).
The 72 facial pictures had been assessed for its valence and arousal on a 9-point scale by another 100 Chinese adults; and the demographic data between the 100 adults and the control participants of this study were very similar (refer to31). The ANOVA performed on the average scores of the 72 faces showed that the three categories of faces have significantly different emotional valence scores (F(2,69) = 219, p < 0.001,  = 0.864; happy = 6.24 ± 0.12, fear = 2.70 ± 0.12, neutral = 4.20 ± 0.12) as well as arousal scores (F(2,69) = 51.6, p < 0.001,
= 0.864; happy = 6.24 ± 0.12, fear = 2.70 ± 0.12, neutral = 4.20 ± 0.12) as well as arousal scores (F(2,69) = 51.6, p < 0.001,  = 0.600; happy = 5.78 ± 0.21, fear = 6.58 ± 0.21, neutral = 3.64 ± 0.21).
= 0.600; happy = 5.78 ± 0.21, fear = 6.58 ± 0.21, neutral = 3.64 ± 0.21).
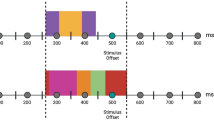
The temporal judgment task was composed of two phases-a training phase and a testing phase (Fig. 1). In the training phase, participants watched the “standard” stimulus duration (700 ms) ten times, represented by the grey oval. In the testing phase, participants watched three comparison durations (490, 700 and 910 ms)19,32 presented in the form of happy, fearful or neutral faces. Participants were required to judge whether the face was presented in shorter, longer or equal durations compared with the standard stimulus they had studied by pressing the “left”, “right” or “up” buttons, respectively, on the joystick with their right thumb.
The testing phase consisted of 9 blocks (each containing 48 testing trials). Each face was presented twice in each of the three duration conditions. The emotion category and the presentation duration of faces varied randomly across trials. The standard stimulus was presented five times at the beginning of each block to prevent the participants from forgetting it.
EEG recording and analysis
Brain electrical activity was recorded referentially against left mastoid and off-line re-referenced to the average of bilateral mastoids, by a 64-channel amplifier with a sampling frequency of 250 Hz (Brain Products, Gilching, Germany). The recorded data were band-pass filtered online within 0.01–80 Hz. Ocular artifacts were removed from EEGs using a regression procedure implemented in NeuroScan software (Scan 4.3). The EEG data (after ocular artifacts removed) were filtered (0.01–30 Hz), segmented and baseline-corrected (−200 to 0 ms), followed by averaging in association with experimental conditions irrespective of response19,33.
This study focused on the peak amplitudes and the peak latencies of P1, N170 and VPP, as well as the area amplitude of CNV across different sets of electrodes according to grand-mean ERP topographies and relevant literatures13,14,19. The CNV was estimated using an area measurement which was calculated based on the integral under the ERP waveforms between two zero crossing points on the time axis20. The measures of P1 were calculated as the mean value of the electrode sites of O1 and PO3 (for left hemisphere) and the mean value of O2 and PO4 (for right hemisphere), within a time window for peak detection of 90–150 ms. Similarly, the measures of N170 were calculated as the mean value of P7 and PO7 (for left hemisphere) and the mean value of P8 and PO8 (for right hemisphere), within a time window for peak detection of 140–200 ms. The components of VPP and CNV were measured irrespective of hemisphere and were calculated as the mean value of FC1, FCz and FC2 (time window for VPP peak detection = 140–200 ms).
Statistics
Descriptive data were presented as mean ± standard error. Repeated-measures ANOVA was performed on measurements of ERP and behavioral data, with emotion (happiness, fear and neutral) as the within-subject factor and group (patient and control) as the between-subject factor. When analyzing the P1 and the N170, another within-subject factor of hemisphere (left and right) was included. When analyzing the CNV and behavioral measures, the within-subject factor of stimulus duration (490, 700 and 910 ms) was added. In addition, the response count was compared between different conditions, with emotion, stimulus duration and response (short, equal and long) as the within-subject factors and group as the between-subject factor. Greenhouse-Geisser and Bonferroni corrections were used whenever appropriate. Two-tailed Pearson’s r correlation was performed between negative symptoms and the ERP measurements of patients (Holm’s stepwise correction).
Results
For the sake of brevity, this section only reports the most important results. Please refer to the supplementary material for the other significant findings.
Behaviors
Accuracy rate
The main effect of group was significant (F(1,91) = 98.8; p < 0.001;  = 0.521). The ACC was lower in the patients (0.474 ± 0.013) than in the controls (0.656 ± 0.013).
= 0.521). The ACC was lower in the patients (0.474 ± 0.013) than in the controls (0.656 ± 0.013).
The “short”, “equal” and “long” responses
The interaction effect of emotion by response by group was significant (F(4,364) = 5.11; p = 0.001;  = 0.053; Fig. 2A). The controls tended to give more “long” responses (happy = 20.5 ± 1.14 counts; fearful = 20.5 ± 1.07 counts) compared with “equal” (happy = 14.6 ± 1.36 counts, p = 0.003; fearful = 15.1 ± 1.34 counts, p = 0.013) and “short” responses (happy = 13.4 ± 0.77 counts, p < 0.001; fearful = 14.2 ± 1.10 counts, p < 0.001) in the happy (F(2,184) = 7.30; p = 0.001) and the fearful conditions (F(2,184) = 5.12; p = 0.007), while they gave the three responses almost equally in the neutral condition (F(2,184) < 1). However, the patients consistently gave more “short” responses (happy = 19.5 ± 0.76 counts; fearful = 20.3 ± 1.09 counts; neutral = 20.0 ± 0.89 counts) compared with “long” responses (happy = 11.4 ± 1.13 counts; fearful = 11.2 ± 1.06 counts; neutral = 10.5 ± 1.10 counts; ps < 0.001) in the three emotional conditions (F(2,184) = 8.62 to 12.3; ps < 0.001). In addition, the interaction effect of stimulus duration by response by group was significant (Fig. 2B; refer to supplementary materials for details).
= 0.053; Fig. 2A). The controls tended to give more “long” responses (happy = 20.5 ± 1.14 counts; fearful = 20.5 ± 1.07 counts) compared with “equal” (happy = 14.6 ± 1.36 counts, p = 0.003; fearful = 15.1 ± 1.34 counts, p = 0.013) and “short” responses (happy = 13.4 ± 0.77 counts, p < 0.001; fearful = 14.2 ± 1.10 counts, p < 0.001) in the happy (F(2,184) = 7.30; p = 0.001) and the fearful conditions (F(2,184) = 5.12; p = 0.007), while they gave the three responses almost equally in the neutral condition (F(2,184) < 1). However, the patients consistently gave more “short” responses (happy = 19.5 ± 0.76 counts; fearful = 20.3 ± 1.09 counts; neutral = 20.0 ± 0.89 counts) compared with “long” responses (happy = 11.4 ± 1.13 counts; fearful = 11.2 ± 1.06 counts; neutral = 10.5 ± 1.10 counts; ps < 0.001) in the three emotional conditions (F(2,184) = 8.62 to 12.3; ps < 0.001). In addition, the interaction effect of stimulus duration by response by group was significant (Fig. 2B; refer to supplementary materials for details).
There were 48 trials in each experimental condition. Responses given within 1500 ms were considered valid data. The main effect of group was significant (F(1,91) = 28.0; p < 0.001;  = 0.235). Compared with the controls (16.3 ± 0.08 counts), the patients gave less valid responses (15.7 ± 0.08 counts).
= 0.235). Compared with the controls (16.3 ± 0.08 counts), the patients gave less valid responses (15.7 ± 0.08 counts).
Response time
The interaction effect of emotion by group was significant (F(2,182) = 9.20; p < 0.001;  = 0.092; Fig. 3). The response of the patients (F(2,182) = 5.16; p = 0.007) was slower in the fearful condition (735 ± 14.4 ms) compared with that in the neutral condition (694 ± 15.3 ms; p = 0.008). In contrast, the response of the controls (F(2,182) = 4.28; p = 0.015) was quicker in the fearful condition (606 ± 11.0 ms) compared with that in the neutral condition (646 ± 12.8 ms; p = 0.003).
= 0.092; Fig. 3). The response of the patients (F(2,182) = 5.16; p = 0.007) was slower in the fearful condition (735 ± 14.4 ms) compared with that in the neutral condition (694 ± 15.3 ms; p = 0.008). In contrast, the response of the controls (F(2,182) = 4.28; p = 0.015) was quicker in the fearful condition (606 ± 11.0 ms) compared with that in the neutral condition (646 ± 12.8 ms; p = 0.003).
The main effect of group was significant (F(1,91) = 34.9; p < 0.001;  = 0.277). The response was slower in the patients (718 ± 10.9 ms) than in the controls (627 ± 11.0 ms).
= 0.277). The response was slower in the patients (718 ± 10.9 ms) than in the controls (627 ± 11.0 ms).
ERPs
The P1
Peak amplitude
The main effect of group was significant (F(1,91) = 77.8; p < 0.001;  = 0.461) (Fig. 4). The P1 amplitude evoked in the patients (2.34 ± 0.09 μV) was smaller than that evoked in the controls (3.48 ± 0.09 μV).
= 0.461) (Fig. 4). The P1 amplitude evoked in the patients (2.34 ± 0.09 μV) was smaller than that evoked in the controls (3.48 ± 0.09 μV).
The N170
Peak amplitude
The interaction effect of emotion by group was significant (F(2,182) = 19.9; p < 0.001;  = 0.179; Fig. 5). The N170 amplitude evoked in the patients (F(2,182) = 15.2; p < 0.001) was larger in the happy condition (−3.76 ± 0.12 μV) compared with that in the fearful (−3.00 ± 0.11 μV; p < 0.001) and the neutral conditions (−3.26 ± 0.13 μV; p = 0.012). However, the emotion effect showed a different pattern in the controls (F(2,182) = 19.3; p < 0.001): the N170 amplitude was smaller in the neutral condition (−3.82 ± 0.13 μV) compared with that in the happy (−4.38 ± 0.13 μV; p < 0.001) and the fearful conditions (−4.68 ± 0.11 μV; p < 0.001).
= 0.179; Fig. 5). The N170 amplitude evoked in the patients (F(2,182) = 15.2; p < 0.001) was larger in the happy condition (−3.76 ± 0.12 μV) compared with that in the fearful (−3.00 ± 0.11 μV; p < 0.001) and the neutral conditions (−3.26 ± 0.13 μV; p = 0.012). However, the emotion effect showed a different pattern in the controls (F(2,182) = 19.3; p < 0.001): the N170 amplitude was smaller in the neutral condition (−3.82 ± 0.13 μV) compared with that in the happy (−4.38 ± 0.13 μV; p < 0.001) and the fearful conditions (−4.68 ± 0.11 μV; p < 0.001).
The main effect of group was significant (F(1,91) = 56.6; p < 0.001;  = 0.383). The N170 amplitude evoked in the patients (−3.34 ± 0.09 μV) was smaller than that evoked in the controls (−4.29 ± 0.09 μV).
= 0.383). The N170 amplitude evoked in the patients (−3.34 ± 0.09 μV) was smaller than that evoked in the controls (−4.29 ± 0.09 μV).
Correlation with the PANSS
According to the N170 results reported above, correlations were performed between negative items and N170 amplitude (left and right hemispheres) in the fearful condition. The severity of blunted affect was found to be correlated significantly with the N170 amplitude at the left hemisphere (r = 0.53; corrected p = 0.018; Fig. 6).
The CNV
Area amplitude
The interaction effect of emotion by group was significant (F(2,182) = 23.0; p < 0.001;  = 0.202; Figs 7 and 8). The CNV area evoked in the controls (F(2,184) = 57.2; p < 0.001) was smaller in the neutral condition (−1.11 ± 0.12 μV•s) compared with that in the happy (−1.83 ± 0.14 μV•s; p < 0.001) and the fearful conditions (−2.14 ± 0.12 μV•s; p < 0.001); and it was larger in the fearful than in the happy condition (p = 0.006). However, the emotion effect did not show any significant difference in the patients (F(2,184) < 1). Furthermore, it is found that the CNV area was larger in the controls than that in the patients both in the fearful (F(1,91) = 22.4; p < 0.001; patient = −1.20 ± 0.14 μV•s, control = −2.14 ± 0.14 μV•s) and happy conditions (F(1,91) = 12.1; p = 0.001; patient = −1.13 ± 0.14 μV•s, control = −1.83 ± 0.14 μV•s); however, the CNV did not show any significant difference between the two groups in the neutral condition (F(1,91) < 1; patient = −1.08 ± 0.15 μV•s, control = −1.11 ± 0.15 μV•s).
= 0.202; Figs 7 and 8). The CNV area evoked in the controls (F(2,184) = 57.2; p < 0.001) was smaller in the neutral condition (−1.11 ± 0.12 μV•s) compared with that in the happy (−1.83 ± 0.14 μV•s; p < 0.001) and the fearful conditions (−2.14 ± 0.12 μV•s; p < 0.001); and it was larger in the fearful than in the happy condition (p = 0.006). However, the emotion effect did not show any significant difference in the patients (F(2,184) < 1). Furthermore, it is found that the CNV area was larger in the controls than that in the patients both in the fearful (F(1,91) = 22.4; p < 0.001; patient = −1.20 ± 0.14 μV•s, control = −2.14 ± 0.14 μV•s) and happy conditions (F(1,91) = 12.1; p = 0.001; patient = −1.13 ± 0.14 μV•s, control = −1.83 ± 0.14 μV•s); however, the CNV did not show any significant difference between the two groups in the neutral condition (F(1,91) < 1; patient = −1.08 ± 0.15 μV•s, control = −1.11 ± 0.15 μV•s).
The grand-mean ERP waveforms of the VPP and the CNV components in neutral, fearful and happy conditions in the patients and controls.
The CNV was estimated using an area measurement which was calculated based on the integral under the ERP waveforms between two zero crossing points on the time axis (refer to the light black region in the lower-left corner of the figure).
The main effect of group was significant (F(1,91) = 8.93; p = 0.004;  = 0.089). The CNV area evoked in the patients (−1.13 ± 0.13 μV•s) was smaller than that evoked in the controls (−1.70 ± 0.13 μV•s).
= 0.089). The CNV area evoked in the patients (−1.13 ± 0.13 μV•s) was smaller than that evoked in the controls (−1.70 ± 0.13 μV•s).
Correlation with the PANSS
According to the CNV results reported above, correlations were performed between negative items and the area amplitude of the CNV in the fearful/happy conditions. The severity of passive/apathetic social withdrawal was found to be correlated significantly with the 490-ms CNV area in the fearful and the happy conditions (fearful: r = 0.59, corrected p = 0.012; happy: r = 0.50, corrected p = 0.035) (Fig. 6).
Discussion
ERP components index impairments in different psychological procedures
The present study investigated the temporal perception of emotional faces in people with and without schizophrenia in order to better understand the nature of emotion deficits in schizophrenia. Three ERP components showed different patterns between the two groups. First, the attention-related P1 component34 displayed a significant group effect, with larger amplitudes in the controls than in the patients; at the same time, no interaction effect of emotion by group was found (Fig. 4). Considering that the occipital P1 can reflect early attentional modulation by emotion35,36, the current P1 result suggested that the bottom-up visual processing and its associated attention allocation were impaired for both emotional and neutral stimuli in schizophrenic patients.
Second, significant interaction effect of emotion by group was found in the N170, which is a face-sensitive ERP component and is often modulated by emotional faces14,15. Although there was also a significant group effect (larger amplitudes in the controls than in the patients), the N170 result emphasized that compared with happy and neutral faces, schizophrenic patients were more impaired in fearful face processing (Fig. 5).
Similar with the statistical result of N170, both interaction effect and main effect of group were significant on the CNV, a sensitive index for online timing16,17,18,19,20. The observed smaller CNV in response to fearful and happy faces in schizophrenia indicated that compared with the controls, the patients not only showed general dysfunction in temporal perception, but, more important, had a larger damage when they timing emotional, compared with neutral, stimuli (Fig. 7).
Unlike the ERP data, whose components reflected separate psychological procedures, the behavioral data only provided an overall characterization of the deficits in schizophrenia. All the three behavioral measures showed a main effect of group, indicating the patients had a general impairment when performing the task of time perception, irrespective the emotional content (fear, happiness or neutral) of stimuli. Furthermore, response data suggested that while the controls overestimated the duration of emotional (i.e. happy and fearful) faces, the patients underestimated the duration of emotional and neutral faces (Fig. 2A) and exhibited less temporal precision than the controls (Fig. 2B). In addition, the interaction effect of emotion by group on response time was significant: the shorter RT in fearful than in neutral condition for healthy participants may indicate the well-known “negativity bias” in emotion processing37; the longer RT in fearful than in neutral conditions for the patients was consistent with the result of N170, suggesting that schizophrenic patients were more severely impaired for fearful rather than happy facial processing.
Since the focus of the current study was the impairment in time perception of emotional stimuli, the later part of the Discussion mainly concerns the time- and emotion-related CNV and N170 components.
Impaired emotional time perception
The present result in the healthy subjects is consistent with previous studies in non-psychotic people reporting that the duration of happy and fearful faces is always overestimated compared to neutral ones38,39. This phenomenon is usually thought to be linked to the increase of the physiological arousal level in response to emotional stimuli11,40. The increased arousal accelerates the pacemaker in the pacemaker-accumulator model41, leading to a greater number of accumulated pulses, thus resulting in overestimation of time42,43. (Note: the psychological mechanism of timing is usually explained by the pacemaker-accumulator model. In brief, the model includes an internal pacemaker that sends pulses to an accumulator. The longer the stimulus duration, the more pulses are accumulated and the longer the subjective duration is judged to be). Since the accumulation of pulses is reflected by the CNV on the brain activity level, the increased arousal caused by happy and fearful faces also leads to larger CNV amplitudes18. This ability of unconscious time distortion under the influence of emotion facilitates people to optimize their responses to various emotional events in the environment11. For example, people tend to overestimate the duration of fearful pictures, which enlarges the danger delivered by the picture and prompts individuals to fight or flight44. Thus, the effect of emotion on time perception has an evolutionary and adaptive significance and is essential for everyday activities40.
Unfortunately, this emotional effect on timing totally disappeared in schizophrenic patients, as shown in our behavioral and CNV results. Here we preliminarily propose a possible mechanism underlying this impairment in schizophrenia. According to the theory of embodying emotion, individuals automatically imitate the perceived facial expressions and this imitation process produces an actual experience of the same emotion45. The effect of embodied emotion on time perception suggests that people synchronize activities with others and that sharing other individuals’ time implies a desire to empathize with them11,46. Consistent with this inference, studies showed that the overestimation of the duration of angry and happy faces was not observed when the imitation of facial expressions was inhibited39; and that the low-empathy subjects showed a reduced temporal overestimation for angry faces in compared with the high-empathy subjects47. Therefore we might assume that in the current task, the empathy deficit in schizophrenia48 prevented emotional faces from inducing appropriate arousal level in patients, resulting in a slower pacemaker rate in them, which in turn gave rise to a temporal underestimation. However, since we mistakenly did not require the patients/controls to explicitly assess the valence and the arousal of facial expression pictures, this data interpretation needs to be proved in future studies.
An alternative interpretation for the convergent CNVs in the patients is that insufficient attention was allocated to emotional faces, due to negative symptoms such as passive/apathetic social withdrawal. It has been proposed that the supplementary motor area (SMA) is the neural substrate of the timing function reflected by the CNV17,18,49. Since increasing attention to timing enhances the activation of the SMA50, we can infer that the level of neural activation contributing to time perception (i.e. the CNV area amplitude) depends on the amount of attention paid to the target. As a result, the observed smaller CNV area in response to fearful and happy faces in schizophrenia reflected that compared with the control subjects, the patients could not keep sustained attention on emotional facial expressions.
In addition, it was also found in Fig. 6 that the CNV amplitudes increased with temporal durations. This pattern is inconsistent with some earlier CNV studies20,32, which indicated that the CNV peaks at the end of stimulus duration and then declines when the perceived stimulus is shorter than the “standard” duration (700 ms in our study); while the stimulus longer than the standard duration evokes the CNV with a peak around the end of the standard duration. This discrepancy may be due to two reasons. First, the involved cognitive systems during time estimation may be slightly different: the testing stimuli were emotional faces in this study while they were simple stimuli without emotion (e.g. the illumination of a diode) in earlier CNV studies. Second, the CNV pattern may be different across different electrodes32. Nevertheless, the correlation between the amplitude of the CNV and the estimated duration is still in debate51 and is not the focus of the current study.
Preserved early response to positive emotion
Another interesting finding of the present study was that the early emotional processing of happy faces was preserved to a large extent when patients performing the time perception task. However, this happy vs. neutral difference in the N170/VPP disappeared in the later stage of emotional time perception, as reflected by the CNV.
The finding that early emotional processing of happy, rather than fearful, faces was largely preserved in schizophrenic subjects is in line with previous observations suggesting that abnormalities in negative, but not positive emotional processing may be core to affective disturbance in schizophrenia52,53. Studies of emotional processing have shown that patients are often associated with decreased activation and accuracy in responses to facial expressions of fear, anger and disgust, compared with happy faces54. Furthermore, it was found that people with schizophrenia tend to show greater deficits for the recognition of negative compared with positive emotions and of the negative emotions, patients show the greatest impairment for the perception of fear55,56.
Taken together, the results of N170 and CNV suggested that patients may adequately perceive and experience the emotion of happy in the moment, but their temporal judgment of happy faces is largely impaired. Consistently, as we mentioned in the introduction, many studies in schizophrenia showed that patients and controls usually report experiencing similar levels of pleasure in response to a pleasant mood induction3,57; the patients, however, have an anticipatory pleasure deficit and do not expect to experience positive affect when engaging in future goal-directed activities3,9,58. As a complementary study to the previous anticipatory investigations, the current work directly examined the timing function for emotional stimuli in schizophrenia. Our results further underlined the importance of taking time dimension into consideration when examining the emotional deficits in schizophrenia.
Correlation between ERPs and emotional/social impairments
One of the important contributions of this study was that we presented a preliminary argument for the need to understand the relationships between blunted affect, passive social withdrawal and the function of emotional time perception. It was found that the N170 amplitude in the fearful condition was correlated with the severity of blunted affect, the latter of which has been suggested as the most important negative symptom associated with emotion deficits and could independently predict the performance in emotion-related tasks59. In addition, the CNV in the happy and the fearful conditions was correlated with the severity of passive/apathetic social withdrawal, the latter of which is a primary negative symptom that assesses diminished interest and initiative in social interactions due to passivity or apathy60.
Our result highlights the potential value of ERP measures as direct predictors of emotional/social impairments in patients. Furthermore, as numerous studies have documented the difficulties of improving negative symptoms61,62, this study presents emotional time perception as a promising target for treatment efforts and suggests that remediation of emotional timing deficits may results in improved functional outcome.
Conclusion
The current study examined the interaction of emotion and time perception in schizophrenia. Using neutral faces to provide a putative baseline, we demonstrated in the patients that the emotional processing and timing for facial expressions might have a pattern of two-stage deterioration: while the early brain response to happy, but not fearful, faces was largely preserved, the function of emotional timing was drastically impaired for both fearful and happy faces. Findings from the present work point to the importance of considering the time dimension of emotional processing in schizophrenia, based on which we are likely to discover aspects of emotional deficits that would be unnoticed in other studies. It was observed that schizophrenic patients were not only less accurate in time estimation, but also showed a particular kind of perception deviation in emotional timing (underestimation compared with healthy subjects). In light of the result, we suggest that the direction and magnitude of the temporal deviation for emotional stimuli may provide novel insights into the core pathophysiology and offer quantitative biomarkers for specific emotional/social dysfunctions in schizophrenia.
Additional Information
How to cite this article: Zhang, D. et al. Perception of the duration of emotional faces in schizophrenic patients. Sci. Rep. 6, 22280; doi: 10.1038/srep22280 (2016).
References
Eack, S. M. et al. Social cognition deficits among individuals at familial high risk for schizophrenia. Schizophr Bull 36, 1081–1088 (2010).
Barkl, S. J., Lah, S., Harris, A. W. & Williams, L. M. Facial emotion identification in early-onset and first-episode psychosis: a systematic review with meta-analysis. Schizophr Res 159, 62–69 (2014).
Cohen, A. S. & Minor, K. S. Emotional experience in patients with schizophrenia revisited: meta-analysis of laboratory studies. Schizophr Bull 36, 143–150 (2010).
Kohler, C. G., Bilker, W., Hagendoorn, M., Gur, R. E. & Gur, R. C. Emotion recognition deficit in schizophrenia: association with symptomatology and cognition. Biol Psychiatry 48, 127–136 (2000).
Kring, A. M. & Elis, O. Emotion deficits in people with schizophrenia. Annu Rev Clin Psychol. 9, 409–433 (2013).
Varcin, K., Bailey, P. & Henry, J. Empathic deficits in schizophrenia: the potential role of rapid facial mimicry. J Int Neuropsychol Soc 16, 621–629 (2010).
Ursu, S. et al. Prefrontal cortical deficits and impaired cognition-emotion interactions in schizophrenia. Am J Psychiatry 168, 276–285 (2011).
Juckel, G. et al. Dysfunction of ventral striatal reward prediction in schizophrenia. NeuroImage 29, 409–416 (2006).
Gard, D. E., Kring, A. M., Germans Gard, M., Horan, W. P. & Green, M. F. Anhedonia in schizophrenia: distinctions between anticipatory and consummatory pleasure. Schizophr Res 93, 253–260 (2007).
Trémeau, F. et al. Anticipated, on-line and remembered positive experience in schizophrenia. Schizophr Res 122, 199–205 (2010).
Droit-Volet, S. & Gil, S. The time-emotion paradox. Philos Trans R Soc Lond B Biol Sci. 364, 1943–1953 (2009).
Andreasen, N. C., Paradiso, S. & O’Leary, D. S. “Cognitive dysmetria” as an integrative theory of schizophrenia: a dysfunction in cortical–subcortical–cerebellar circuitry? Schizophr Bull 24, 203–218 (1998).
Luck, S. J. An introduction to the event-related potential technique. London: MIT press (2014).
Schyns, P. G., Petro, L. S. & Smith, M. L. Dynamics of visual information integration in the brain for categorizing facial expressions. Curr Biol 17, 1580–1585 (2007).
Zhang, D., Luo, W. & Luo, Y. Single-trial ERP analysis reveals facial expression category in a three-stage scheme. Brain Res 1512, 78–88 (2013).
Walter, W., Cooper, R., Aldridge, V. J., McCallum, W. C. & Winter, A. L. Contingent negative variation: an electric sign of sensori-motor association and expectancy in the human brain. Nature. 203, 380–384 (1964).
Macar, F. & Vidal, F. Event-related potentials as indices of time processing: a review. J Psychophysiol 18, 89–104 (2004).
Macar, F., Vidal, F. & Casini, L. The supplementary motor area in motor and sensory timing: Evidence from slow brain potential changes. Exp Brain Res 135, 271–280 (1999).
Pouthas, V., Garnero, L., Ferrandez, A. M. & Renault, B. ERPs and PET analysis of time perception: Spatial and temporal brain mapping during visual discrimination tasks. Hum Brain Mapp 10, 49–60 (2000).
Macar, F. & Vidal, F. The CNV peak: an index of decision making and temporal memory. Psychophysiology 40, 950–954 (2003).
Kay, S. R., Flszbein, A. & Opfer, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13, 261–276 (1987).
Sachs, G., Steger-Wuchse, D., Kryspin-Exner, I., Gur, R. C. & Katschnig, H. Facial recognition deficits and cognition in schizophrenia. Schizophr Res 68, 27–35 (2004).
Blanchard, J. J. & Cohen, A. S. The structure of negative symptoms within schizophrenia: implications for assessment. Schizophr Bull 32, 238–245 (2006).
Foussias, G. & Remington, G. Negative symptoms in schizophrenia: avolition and Occam’s razor. Schizophr Bull 36, 359–369 (2010).
Messinger, J. W. et al. Avolition and expressive deficits capture negative symptom phenomenology: implications for DSM-5 and schizophrenia research. Clin Psychol Rev 31, 161–168 (2011).
Liemburg, E. et al. Two subdomains of negative symptoms in psychotic disorders: established and confirmed in two large cohorts. J Psychiatr Res 47, 718–725 (2013).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders : Fourth Edition. Washington, DC: American Psychiatric Press (1994).
First, M. B., Spitzer, R., Gibbon, M. & Williams, J. Structured Clinical Interview for DSM-IV Axis I Disorders: Patient Edition. SCID-I/P. New York: New York State Psychiatric Institute (1996).
First, M. B., Gibbon, M., Spitzer, R. L. & Williams, J. B. W. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Non-Patient Edition (SCID-I/NP). New York: New York State Psychiatric Institute (2002).
First, M. B., Spitzer, R. L., Gibbon, M., Williams, J. B. W. & Benjamin, L. Structured Clinical Interview for DSM-IV Axis II Personality Disorders (SCID-II). New York : New York: State Psychiatric Institute, (1996).
Gong, X., Huang, Y., Wang, Y. & Luo, Y. Revision of the Chinese facial affective picture system. Chinese Mental Health Journal 25, 40–46 (2011).
Pfeuty, M., Ragot, R. & Pouthas, V. When time is up: CNV time course differentiates the roles of the hemispheres in the discrimination of short tone durations. Exp Brain Res 151, 372–379 (2003).
Gibbons, H., Brandler, S. & Rammsayer, T. H. Dissociating aspects of temporal and frequency processing: a functional ERP study in humans. Cortex 39, 947–965 (2003).
Hillyard, S. A., Vogel, E. K. & Luck, S. J. Sensory gain control (amplification) as a mechanism of selective attention: electrophysiological and neuroimaging evidence. Philos Trans R Soc Lond B Biol Sci. 353, 1257–1270 (1998).
Krusemark, E. A. & Li, W. Do all threats work the same way? Divergent effects of fear and disgust on sensory perception and attention. J Neurosci. 31, 3429–3434 (2011).
Liu, Y., Zhang, D. & Luo, Y. How disgust facilitates avoidance: an ERP study on ateention modulation by threats. Soc Cogn Affect Neurosci. 10, 598–604 (2015).
Cacioppo, J. T. & Gardner, W. L. Emotion. Annu Rev Psychol. 50, 191–214 (1999).
Droit-Volet, S., Brunot, S. & Niedenthal, P. Perception of the duration of emotional events. Cogn Emot 18, 849–858 (2004).
Effron, D. A., Niedenthal, P. M., Gil, S. & Droit-Volet, S. Embodied temporal perception of emotion. Emotion 6, 1–12 (2006).
Droit-Volet, S. & Meck, W. H. How emotions colour our perception of time. Trends Cogn Sci. 11, 504–513 (2007).
Gibbon, J., Church, R. M. & Meck, W. H. Scalar timing in memory. Ann N Y Acad Sci. 423, 52–77 (1984).
Angrilli, A., Cherubini, P., Pavese, A. & Manfredini, S. The influence of affective factors on time perception. Percept Psychophys 59, 972–982 (1997).
Noulhiane, M., Mella, N., Samson, S., Ragot, R. & Pouthas, V. How emotional auditory stimuli modulate time perception. Emotion 7, 697–674 (2007).
Grommet, E. K. et al. Time estimation of fear cues in human observers. Behav Processes 86, 88–93 (2011).
Niedenthal, P. M. Embodying emotion. Science 316, 1002–1005 (2007).
Wittmann, M. & van Wassenhove, V. The experience of time: neural mechanisms and the interplay of emotion, cognition and embodiment. Philos Trans R Soc Lond B Biol Sci. 364, 1809–1813 (2009).
Mondillon, L., Niedenthal, P. M., Gil, S. & Droit-Volet, S. Imitation of in-group versus out-group member’s facial expression of anger. Soc Neurosci. 2, 223–237 (2007).
Derntl, B. et al. Generalized deficit in all core components of empathy in schizophrenia. Schizophr Res 108, 197–206 (2009).
Pfeuty, M., Ragot, R. & Pouthas, V. Relationship between CNV and timing of an upcoming event. Neurosci. Lett 382, 106–111 (2005).
Coull, J. T., Vidal, F., Nazarian, B. & Macar, F. Functional anatomy of the attentional modulation of time estimation. Science 303, 1506–1508 (2004).
van Rijn, H., Kononowicz, T. W., Meck, W. H., Ng, K. K. & Penney, T. B. Contingent negative variation and its relation to time estimation: a theoretical evaluation. Front Integr Neurosci. 5, 91 (2011).
Jung, H. T., Kim, D. W., Kim, S., Im, C. H. & Lee, S. H. Reduced source activity of event-related potentials for affective facial pictures in schizophrenia patients. Schizophr Res 136, 150–159 (2012).
Strauss, G. P. et al. Emotion regulation abnormalities in schizophrenia: cognitive change strategies fail to decrease the neural response to unpleasant stimuli. Schizophr Bull 39, 872–883 (2013).
Phillips, M. L. et al. A differential neural response to threatening and non-threatening negative facial expressions in paranoid and non-paranoid schizophrenics. Psychiatry Res 92, 11–31 (1999).
Edwards, J., Pattison, P. E., Jackson, H. J. & Wales, R. J. Facial affect and affective prosody recognition in first-episode schizophrenia. Schizophr Res 48, 235–253 (2001).
Kohler, C. G. et al. Facial emotion recognition in schizophrenia: intensity effect and error pattern. Am J Psychiatry 160, 1768–1774 (2003).
Kring, A. M., Germans Gard, M. & Gard, D. E. Emotion deficits in schizophrenia: timing matters. J Abnorm Psychol. 120, 79–87 (2011).
Kring, A. M. & Moran, E. K. Emotional response deficits in schizophrenia: Insights from affective science. Schizophr Bull 34, 819–834 (2008).
Gur, R. E. et al. Flat affect in schizophrenia: relation to emotion processing and neurocognitive measures. Schizophr Bull 32, 279–287 (2006).
Hansen, C. F., Torgalsbøen, A. K., Melle, I. & Bell, M. D. Passive/apathetic social withdrawal and active social avoidance in schizophrenia: difference in underlying psychological processes. J Nerv Ment Dis 197, 274–247 (2009).
Erhart, S. M., Marder, S. R. & Carpenter, W. T. Treatment of schizophrenia negative symptoms: future prospects. Schizophr Bull 32, 234–237 (2006).
Kirkpatrick, B., Fenton, W. S., Carpenter, W. T. & Marder, S. R. The NIMH-MATRICS consensus statement on negative symptomes. Schizophr Bull 32, 214–219 (2006).
Wechsler, D. Wechsler Adult Intelligence Scale (4th ed.) Administration and Scoring Manual. San Antonio: Psychological Corporation (2008).
Wechsler, D. Wechsler Adult Intelligence Scale (4th ed.) Technical and Interpretive Manual. San Antonio: Psychological Corporation (2008).
Wang, J. et al. Reliability and construct validity of Chinese version of Wechsler Adult Intelligence Scale-Fourth Edition. Chinese Mental Health Journal 27, 62–67 (2013).
Woods, S. W. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry 64, 663–667 (2003).
Acknowledgements
This study was funded by the National Natural Science Foundation of China (31300867; 31571120), the National Key Basic Research Program of China (2014CB744600), Beijing Municipal Science & Technology Commission Grant (Z141107002514016) and Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding (XMLX201609; zy201409).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: D.Z. and Y.L.; performed the experiments: Y.Z.; analyzed the data: D.Z.; wrote the manuscript: D.Z. and Y.Z.; contributed materials and analysis tools: S.T.; provided lab equipment for running the study: S.T.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, D., Zhao, Y., Liu, Y. et al. Perception of the duration of emotional faces in schizophrenic patients. Sci Rep 6, 22280 (2016). https://doi.org/10.1038/srep22280
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep22280
This article is cited by
-
Electrophysiological Studies of Reception of Facial Communication in Autism Spectrum Disorder and Schizophrenia
Review Journal of Autism and Developmental Disorders (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.