Abstract
Well-acclimatized nitrifiers in high-nitrate agricultural soils can quickly nitrify NH4+ into NO3− subject to leaching and denitrifying loss. A 120-day incubation experiment was conducted using a greenhouse soil to explore the fates of applied fertilizer N entering into seven soil N pools and to examine if green manure (as ryegrass) co-application can increase immobilization of the applied N into relatively stable N pools and thereby reduce NO3− accumulation and loss. We found that 87–92% of the applied 15N-labelled NH4+ was rapidly recovered as NO3− since day 3 and only 2–4% as microbial biomass and soil organic matter (SOM), while ryegrass co-application significantly decreased its recovery as NO3− but enhanced its recovery as SOM (17%) at the end of incubation. The trade-off relationship between 15N recoveries in microbial biomass and SOM indicated that ryegrass co-application stabilized newly immobilized N via initial microbial uptake and later breakdown. Nevertheless, ryegrass application didn’t decrease soil total NO3− accumulation due to its own decay. Our results suggest that green manure co-application can increase immobilization of applied N into stable organic N via microbial turnover, but the quantity and quality of green manure should be well considered to reduce N release from itself.
Similar content being viewed by others
Introduction
Human-induced nitrogen (N) input increased over tenfold with respect to one century ago1,2. Excessive N fertilization caused accumulation of significant amounts of N beyond crop absorption in soils, largely in the form of nitrate (NO3−)3,4. Surplus NO3− in soil is problematic because it is susceptible to loss by leaching or denitrification2,5, which is both economically and environmentally undesirable4,6. High nitrification potential and low NO3− immobilization are responsible for NO3− accumulation in agricultural soils7. Long-term N fertilization and tillage greatly increase the population and alter the community structure of ammonia oxidizing bacteria, as well as the nitrification capacity4,8,9,10. This is confirmed by the significant positively relationship between nitrification potential and soil NO3− content7,11.
Co-application of N fertilizer with organic materials is especially common in high-input cropping systems and it is considered to be a good agronomic practice as it is thought to elevate microbial bioavailability of the applied N and reduce N loss to the environment12. Much of immobilized N was speculated to be rapidly transformed to microbial residues or necromass as the average life cycle of N in microbial biomass was just several days13,14. While a recent study shows that soil newly synthesized amino acids are relatively easier to decompose in contrast with original soil amino acids15, the fate of newly immobilized N in various soil N pools is generally not well quantified.
In recent years, large areas of conventional cereal cultivation in China have been transferred to intensive greenhouse cultivation due to fast economic development and increased consumer demand3. Greenhouse soil cultivated with vegetables represents typical high NO3− soil in agricultural system3,16. Constant monoculture, excessive fertilization and high-intensity anthropogenic interference during greenhouse cultivation change the process of soil N transformation and accelerate the accumulation of NO3− 10,16. Thus, special attention is required to pay on N transformation in the NO3− polluted soil (e.g., greenhouse soil) after the application of ammonium-based fertilizers.
Isotope 15N tracing technology is widely used to study the fate of chemical fertilizers and other N amendments in ecosystems. Previous studies on the fate of soil added 15N mainly focused on its transformation to extractable NH4+ and NO3− pools, as well as N retention during a certain period (either short-term or long-term mostly)7,9,17,18. Due to the difficulties in experimental operation, dynamic and systematic studies on the fates of soil added 15N entering into various N pools (e.g. microbial biomass N, extractable organic N, mineral fixed N, non-extractable organic N) are rare, while these forms of N may play significant roles as important intermediates in biogeochemical N transformation processes in soil15,19.
Altering the fate of chemical fertilizer N to lessen NO3− loss by manure co-application largely relies on the scale and the stability of manure-enhanced immobilization of fertilizer N. In this study, we conducted a 120-day incubation experiment to trace the fates of the inorganic fertilizer N alone or in combination with a green manure (as ryegrass in this study) to a greenhouse soil, using a 15N trace technique. The purpose of this study was to examine the effect of co-application of green manure on the fates of applied N entering into seven different N pools. Specifically, we examined if co-application of green manure promoted the incorporation of soil newly immobilized N into relatively stable organic N pools instead of remineralization and in a long run, reduced NO3− accumulation and loss risk in high-input greenhouse soil. We expected that green manure co-application would increase the stable immobilization of fertilizer N in the studied soil.
Results
Dynamics of soil extractable N (NH4+-N, NO3−-N, EON)
In the control treatment, NH4+ concentration was 0.6 to 4.9 mg N kg−1 and did not change much over the 120-day incubation, while NO3− concentration increased somewhat from 152 mg N kg−1 at the beginning of incubation to 199 mg N kg−1 at the end of incubation (Fig. 1a,c). In the N addition treatment, NH4+ decreased by 93–97% within 3 days from >80 mg N kg−1 to background level of 2.7–4.5 mg N kg−1. In contrast, NO3− concentration increased rapidly to 225–244 mg N kg−1 from 152 mg N kg−1 in the first 3 days (Fig. 1b,d). From the 7 to 120 days, NO3− concentration slightly increased and 15N recovery in NO3− pool, however, was relatively constant. Co-application of ryegrass significantly decreased the proportion of applied fertilizer NH4+ conversion to NO3−. During the days from day 3 to day 120, 56–70% of the applied fertilizer NH4+ was recovered as NO3− in the +15NH4++Ryegrass treatment, being significantly lower than that (87–92%) in the +15NH4+ treatment (Fig. 1d).
Concentrations (left) of extractable N speciation (NH4+-N, NO3−-N, EON), microbial biomass N (MBN) and mineral fixed N (MFN) and their 15N recoveries (right) after the addition of (15NH4)2SO4 alone (+15NH4+) or combined with ryegrass (+15NH4++Ryegrass) to the tested soil.
Error bars are smaller than the symbol when they are not visible. Separate bars in a line indicate the range of LSD (P = 0.05) for different treatments in the same incubation time. Confidence intervals (95%) of 15N recoveries in different pools were expressed as ±1.96 times of standard error.
Extractable organic N (EON) was 8.4 to 25.3 mg N kg−1 and accounted for 3–11% of total extractable nitrogen (TEN, NH4+-N + NO3−-N + EON) (Fig. 1e). Ryegrass addition slightly increased EON concentrations. Less than 2% of the applied fertilizer NH4+ was recovered as EON and there was no significant difference in the 15N recovery in EON between the two 15NH4+ addition treatments (Fig. 1f).
Dynamics of soil microbial biomass N (MBN) and mineral fixed N (MFN)
In the control treatment, MBN concentrations was relatively constant, being 32.9 to 52.0 mg N kg−1 (Fig. 1g). NH4+ addition increased MBN concentration slightly only in the first 3 day of incubation. However, co-application of ryegrass with NH4+ increased MBN significantly in the first 30 days. After 60 days, there was no significant difference in MBN concentrations among the three treatments. In the +15NH4+ treatment, 1.5–2.9% of the applied fertilizer NH4+ was recovered to microbial biomass (Fig. 1h). In the +15NH4++Ryegrass treatment, the proportion to microbial biomass changed over the incubation, from 5% in the 0.1 day increased to a maximum of 22% in the 1 day and decreased to 4% at the end of incubation.
MFN concentrations were comparable to NO3− concentration, but were not different among the three treatments (Fig. 1i). The 15N recovery in MFN pool did not change and stabilize at 2–3% in the two N addition treatments during day 1–120 (Fig. 1j).
Incorporation into soil organic matter (SOM) and N loss
The two 15N addition treatments yielded different 15N recoveries in TEN and non-extractable N (NEN) pools (Fig. 2b,d). The 15N recoveries in TEN pool were 88–99% for the +15NH4+ treatment and 57–81% for the +15NH4++Ryegrass treatment during the 120-day incubation and showed a slight increase after an initial sharp drop. A reverse trend was found for the 15N recovery in NEN pool (Fig. 2).
Concentrations (left) of total extractable N (TEN, = NH4+-N + NO3−-N + EON) and non-extractable N (NEN) and their 15N recoveries (right) after the addition of (15NH4)2SO4 alone (+15NH4+) or combined with ryegrass (+15NH4++Ryegrass) to the tested soil.
Error bars are smaller than the symbol when they are not visible. Separate bars in a line indicate the range of LSD (P = 0.05) for different treatments in the same incubation time.
We defined the 15N recovery in NEN pool minus in MFN and MBN pools (NEN-MFN-MBN) in the unfumigated post-extracted soil as incorporation into SOM and defined the unrecovered 15N (neither in TEN nor NEN pools) as loss (Fig. 3). During day 3–120 in the +15NH4+ treatment, only 0.1–2% and 3–6% of the applied fertilizer N was incorporated into SOM and lost, respectively. When ryegrass was co-applied, 8–17% of the applied fertilizer N was incorporated into SOM and 6–15% of applied N was lost, respectively (Fig. 3).
Changes in recoveries of 15N in the soil as NH4+-N, NO3−-N, extractable organic N (EON), microbial biomass N, incorporated into SOM, mineral fixed N fractions and loss during the 120-day lab incubation after the addition of (15NH4)2SO4 alone (+15NH4+) or combined with ryegrass (+15NH4++Ryegrass) to the tested soil.
15N recovery in SOM pool was calculated as the difference of recoveries in “non-extractable N” and “microbial biomass N + mineral fixed N”. Nitrogen loss is the unrecovered 15N neither in extractable nor non-extractable pools. Numbers in the areas are the corresponding 15N recoveries in relevant N pool at the related incubation time.
Discussion
Nitrification
Our results show that the studied greenhouse soil is characterized by high NO3− accumulation (>150 mg N kg−1) and very fast nitrification. The net nitrification rates (net NO3− change) of the applied fertilizer NH4+ were 28.7 and 27.7 mg N kg−1 d−1 for the +15NH4+ and +15NH4++Ryegrass treatments respectively during day 0.1–1 and 17.9 and 8.3 mg N kg−1 d−1 during day 1–3 (Fig. 4), much higher than the reported values for many other ecosystems4,11,20. Nitrifiers were acclimatized with high quantity and improved function in the tested soils due to high-rate fertilization and long-term vegetable cultivation but limited C input20,21. In contrast, in N-limited forest soil, low N input and sufficient available C to fuel high denitrification and immobilization, prevents the NO3− accumulation7,22,23.
The relative strength of N mineralization and immobilization mainly depends on the C/N of added organic material as well as the incubated soil24,25,26. The total rate of nitrification (including 14N and 15N) in the ryegrass co-applied soil was not reduced in this study (Figs 1c and 4), primarily due to the supply of non-labelled NH4+ from relatively fast mineralization of the ryegrass with a C/N ratio only 12.4 (soil C/N ratios were 10.5, 10.0 and 10.2 respectively after the addition for Control, +15NH4+ and +15NH4++Ryegrass treatments). The total nitrification rate and N release would get reduced if organic material with a higher C/N than ryegrass was applied27. But the use of organic material with a wider C/N ratio (e.g. crop straw) may not really favor transformation of NH4+ into organic N pool instead of NO3− pool, due to the desynchrony between slow release of available C from crop straw and fast nitrification of applied NH4+ in greenhouse soil. Therefore, reducing nitrification of applied fertilizer N by co-application of organic material in high-input agricultural soils should well consider the quality (e.g. C/N, carbon availability) of added organic material22,26,27.
Incorporation into MBN and SOM
Our study dynamically evaluated the stability and further transformation of microbially assimilated N using the indicator “non-extractable organic 15N”. The results suggest that co-application of green manure led to effective NH4+ immobilization and stabilization in soil (Fig. 3). During day 1–120, 23–28% of the applied fertilizer N was incorporated into non-extractable organic N, initially primarily as MBN and eventually primarily as SOM. According to the low soil NH4+ concentration after day 3 and the low microbial assimilation to NO3− in agricultural soils7,28, we propose that the slow decline of 15N recovery in non-extractable organic N pool (15N incorporated into MBN and SOM) after day 3 was dominated by the remineralization of newly immobilized organic 15N (Fig. 3). Our results also show that newly immobilized organic 15N is a relatively stable N pool that can accumulate rapidly but is not readily accessible to microbial mineralization.
Studies of soil organic matter after 15NH4+ or 15N-urea addition by nuclear magnetic resonance technique (NMR) confirm that the immobilized organic 15N is concentrated as the form of peptide or amide17,29,30, which is regarded as a microbial source19,31. In this study, the ryegrass-induced rapid up-down change of 15N recovery in MBN (Fig. 1h), as well as the trade-off changes of 15N recoveries in MBN and “incorporated into SOM” pools (Fig. 3), indicate that the initial immobilization of 15NH4+ and its subsequent transformation in the soil should be mainly microbially mediated32,33,34 and the disappeared MB15N was mainly further incorporated into SOM, a stable soil organic N pool that hard to decompose35. Appel et al.36 confirmed that almost no extractable 15N was extracted from soils when 15N-labelled bacterial biomass was added prior to extraction. This goes against usual understanding that the newly immobilized N will release to bioavailable N after the death and breakdown of live microbes. It is expected that the soil N immobilized by microbes may contribute a lot to the low but long availability of residual fertilizer-N (residual effect) to subsequent crops37,38.
The biotic immobilization of added NH4+ was mainly initiated and regulated by active microorganisms39. MB15N was too variable within a short period26 as also evidenced by this study to reliably reflect the scale of 15NH4+ immobilization, especially in a relatively long period. In contrast, newly immobilized organic 15N, composed of 15N incorporation into both MBN and SOM, can be fast accumulated but slowly mineralized in the long-term process (Fig. 3) and may be a better indicator versus MB15N to more reliably quantify the microbial assimilation of added 15NH4+ in a long period.
Immobilization of N in soils might also be a result of abiotic reaction40,41, but the proportion is generally low in agricultural soil42 and in our study soil (Fig. 1j). Nevertheless, no matter how much the immobilization of applied 15NH4+ is biotically or abiotically regulated, the evidence provided by the present study suggested that applying green manures (e.g. ryegrass) to soil could significantly enhance the stable immobilization of applied NH4+. This is very significant for N management in intensive cultivation systems (e.g. greenhouse cultivation, orchard, vineyard, etc.), where large amounts of N fertilizer are applied.
Incorporation into EON, MFN and N loss
Compared to NO3− and MBN, the proportions of 15N entering into EON and MFN pools were low in this study (Figs 1 and 3), which was consistent with previous studies26,43,44. Because most of EON in soils is resistant to decomposition, rapid turnover of easily degradable EON released from fresh organic matter will lead to the low contribution of added organic material to the total EON pool44. With the decline of the recovery of MB15N, the EO15N recovery kept continuously low (Fig. 3), reflecting indirectly a low contribution of the disappeared MBN to EON during the 120-day incubation. The content of clay-fixed ammonium depends on the degree of K saturation of the interlayers of 2:1 clay minerals45,46. Low 15N recovery in MFN pool in this study may be due to the block of sufficient K+ and NH4+ resulting from high rate fertilization of K and N in greenhouse vegetable cultivation process. Available K in the greenhouse soil is up to 201.7 ± 2.8 mg kg−1, being much higher than that in nearby cropland soil (74.4–127.0 mg kg−1)35,47 Still, low moisture change during the first three days of the incubation may also contribute a part to the low fixation as soil dry-wet cycle is also thought to be an important factor controlling NH4+ fixation45.
Co-application of ryegrass might increase the loss of applied fertilizer NH4+ (Fig. 3), in the forms of nitrogenous gases (NH3, N2, N2O, NO, HONO, etc.). The quantity of loss via ammonia volatilization in the soil was low (measured by absorption with dilute H2SO4, Kjeldahl distillation and titration) and the difference was not significant among treatments (<0.5 mg N kg−1 during the 120-day incubation period, detailed data was not shown here). Compared to the treatment with only NH4+ addition, we suggest that denitrification was fueled by available C from ryegrass and thereby increased the loss of gaseous N. Also, fast ryegrass mineralization rapidly stimulated aerobic microbial activity, likely reducing the permeability and availability of oxygen in soil pores to increase denitrifying loss of 15NO3− in the very initial period, which was verified by the observation of lower water perviousness (need more time to completely infiltrate) in the soil with ryegrass addition than that without ryegrass addition when we added deionized water at regular intervals.
Methods
Soil and ryegrass samples preparation
In the present study, Fluvo-aquic soil (silty loam, Hapli-Udic Cambisols in Chinese soil taxonomy) samples were collected from the surface layer (0–20 cm) of a greenhouse field planted with pepper (Capsicum annuum L.) in Damintun town, Xinmin county, Liaoning province (122°50´E, 41°59´N). The greenhouse soil had a 7-yr history of vegetable cropping (cucumber, tomato, pepper, etc.) and before that it was under maize cultivation for decades. The soil had a low C/N ratio of 10.5 and a high NO3− accumulation (152 mg N kg−1, Table 1).
In this study, ryegrass (Lolium multiflorum Lam.) samples was applied as a green manure in the greenhouse soil, which was obtained by culturing ryegrass in pots (21 cm in diameter and 20 cm in height filled with 3.5 kg soil) until it matured. Each pot was applied with 1 g of non-labelled urea.
Experimental design
Three treatments were set with 4 replicates for each treatment: (1) Control, soil without additions; (2) +15NH4+, adding as 15N-labelled ammonium sulfate ((NH4)2SO4); (3) +15NH4+ + Ryegrass, adding as 15N-labelled (NH4)2SO4 and non-labelled ryegrass. 15N abundance of (NH4)2SO4 was 50.17 atom% and (NH4)2SO4 was added at a rate of 80 mg N kg−1 dry soil, being equivalent to the addition of 200 kg N ha−1 into the 0–20 cm soil layer in the field. The ryegrass was smashed to powder and sieved through 0.5 mm before use and added at a rate of 4.21 g kg−1 dry soil. Selected properties of the soil and ryegrass were listed in Table 1.
For the laboratory incubation, soil was sieved through 2 mm and mixed homogeneously after gravimetric soil moisture was lowered to approximately 15% and then pre-incubated at 25 °C for 7 days. Thereafter, 25 g soil (on an oven-dried basis) was weighed into polyethylene plastic centrifuge cups (60 mm in diameter and 81 mm high, n = 64 for each treatment). After mixing the soil thoroughly with ryegrass for the +15NH4++Ryegrass treatment, aqueous solution of (15NH4)2SO4 or deionized water was added over the soil surface with a pipette to bring the gravimetric soil moisture uniformly to 25.3% (corresponded to 60% water filled pore space). The application was made drop-wise to the soil in the cup and the soil then mixed to ensure the application was even. Finally, the soil was mechanically compressed to field bulk density (1.25 g cm−3) using a small disc with a handle. All cups were covered with parafilm punctured with a needle to maintain aerobic conditions and incubated in an automatically controlled incubator (25 °C). Distilled H2O was added at regular intervals (4 days) to maintain the soil moisture and prevent from decreasing the microbial activity due to water limitation during the 120-day incubation.
Sample processing and chemical analysis
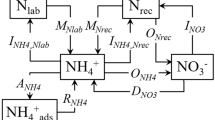
Approximately 0.1 (2.5 h), 1, 3, 7, 15, 30, 60 and 120 days after adding (15NH4)2SO4, eight replicates of each treatment were destructively sampled, extracted directly (four cups) or after fumigation (four cups) with 100 ml 0.5 mol l−1 K2SO4 for 30 minute on a shaker (220 rev min−1) (Fig. 5). The fumigation was carried out with alcohol-free chloroform in a desiccator lined with wet filter paper at 25 °C for 24 h in the dark48. After centrifugation (5000×g, 10 min), the supernatant was filtered through cellulose acetate membranes (0.45 μm, Xinya, Shanghai) that had been prewashed with 0.5 mol l−1 K2SO4. The extracts were determined immediately or stored at −20 °C until future analysis. The soil residue was extracted two more times with K2SO4 solution (100 ml each, supernatant was discarded) to remove extractable N thoroughly from soil particles. All residual soil was carefully recovered, air-dried, ground and sieved (<0.15 mm).
Soil extracts (20 ml) were distilled after the addition of MgO and Devarda’s alloy respectively for quantifying the content of NH4+-N and NO3−-N49. A modified micro-diffusion procedure using Teflon strips was performed to prepare samples for analysis of 15N abundance in the NH4+ and NO3− pools50. After distillation above, the remaining samples were digested with H2SO4 and H2O2 to convert all organic N to NH4+ and subsequently distilled with excess NaOH solution to determine extractable organic N (EON) concentration. Similarly, after conversion of EON (after diffusion above) to NH4+, the digested solution was transferred to plastic container (resistant to acid and alkaline solutions), kept frozen at −20 °C overnight and then diffused with excess NaOH solution (4 °C) to prepare samples for analysis of 15N abundance in the EON pool51. The 15N abundances in the prepared samples were obtained by an elemental analyzer (Flash EA1112, Thermo Finnigan, USA) coupled with an Isotope-Ratio Mass Spectrometer (Delta plus XP, Thermo Finnigan, USA) (EA-IRMS). The standard deviation of 15N abundance measurements (δ15N) for the standard sample is less than 0.3‰ (8 replicates).
Concentrations of non-extractable N (NEN) and mineral fixed NH4+-N (MFN) and their 15N abundances in residual soil after extraction mentioned above were determined by the EA-IRMS. Organic N in residual soil was removed by excessive fresh prepared alkaline KOBr solution before determining MFN52,53. Soil and ryegrass total organic carbon (TOC) was measured using the solid module of TOC/TN analyzer (Multi N/C 3100, Analytikjena, Germany). Soil and ryegrass total N were determined by the Kjeldahl method. Soil available potassium (K) was determined by a flame photometer (6400A, Shanghai) after extraction with 1 mol l−1 ammonium acetate (v/w = 10:1).
Calculation
The soil microbial biomass nitrogen (MBN) was quantified using the following formula with a KEN = 0.45 54.

The 15N recovery in each N pool was calculated based on the simple mass balance and a mixing model in each pool, as follows41:



where, TEN (total extractable N) = NH4+-N + NO3−-N + EON. Letters of “f” and “uf” respectively mean “fumigated” and “unfumigated”; N_x is the amount of soil N components (NH4+-N, NO3−-N, EON, MFN or NEN, mg kg−1); 15N_x is the amount of 15N (mg kg−1) from labelled ammonium sulfate added in related N pool;  ,
,  and
and  are the atom percent excesses (%) of N component in related N pool, background and ammonium sulfate added, respectively; Recoveryx is the percentage of the 15N tracer recovered in the labelled N pool; Input15N is the amount of 15N in ammonium sulfate added (mg kg−1).
are the atom percent excesses (%) of N component in related N pool, background and ammonium sulfate added, respectively; Recoveryx is the percentage of the 15N tracer recovered in the labelled N pool; Input15N is the amount of 15N in ammonium sulfate added (mg kg−1).
Net nitrification rates were calculated as the difference between final and initial NO3−-N concentrations for three time intervals, days 0.1–1, days 1–3 and days 3–7 .
Statistical analyses
Data are expressed on oven-dried soil basis and subjected to analyses of variance using SPSS for Windows v16.0 software package (SPSS Inc., Chicago, IL, USA). All figures were obtained from SigmaPlot 12.5. Error bars in figures represented standard errors. The differences between means of variables for different treatments were statistically tested by ANOVA procedure with least significant difference (LSD) test. The standard errors of composite variables (the concentration of MBN; the recoveries of 15NH4+, 15NO3−, EO15N, MB15N, MF15N, TE15N, NE15N and the net change of NO3−) were calculated through the formula of Gaussian error propagation55. Confidence intervals (95%) of 15N recoveries in different pools were expressed as ±1.96 times of standard error.
Additional Information
How to cite this article: Quan, Z. et al. The fate of fertilizer nitrogen in a high nitrate accumulated agricultural soil. Sci. Rep. 6, 21539; doi: 10.1038/srep21539 (2016).
References
Galloway, J. N., Townsend, A. R. & Erisman, J. W. Transformation of the nitrogen cycle: recent trends, questions and potential solutions. Science 320, 889–892 (2008).
Schlesinger, W. H. On the fate of anthropogenic nitrogen. Proc. Natl. Acad. Sci. USA 106, 203–208 (2009).
Ju, X., Liu, X., Zhang, F. & Roelcke, M. Nitrogen fertilization, soil nitrate accumulation and policy recommendations in several agricultural regions of China. Ambio 33, 300–305 (2004).
Subbarao, G. V. et al. Scope and strategies for regulation of nitrification in agricultural systems-challenges and opportunities. Crit. Rev. Plant Sci. 25, 303–335 (2006).
Stark, J. M. & Hart, S. C. High rates of nitrification and nitrate turnover in undisturbed coniferous forests. Nature 385, 61–64 (1997).
Ju, X. et al. Reducing environmental risk by improving N management in intensive Chinese agricultural systems. Proc. Natl. Acad. Sci. USA. 106, 3041–3046 (2009).
Zhang, J. et al. Agricultural land use affects nitrate production and conservation in humid subtropical soils in China. Soil Biol. Biochem. 62, 107–114 (2013).
Chu, H., Fujii, T., Morimoto, S., Lin, X. & Yagi, K. Population size and specific nitrification potential of soil ammonia-oxidizing bacteria under long-term fertilizer management. Soil Biol. Biochem. 40, 1960–1963 (2008).
Zhu, T., Dang, Q., Zhang, J., Müller, C. & Cai, Z. Reductive soil disinfestation (RSD) alters gross N transformation rates and reduces NO and N2O emissions in degraded vegetable soils. Plant Soil 382, 269–280 (2014).
Li, S., Jiang, X., Wang, X. & Wright, A. L. Tillage effects on soil nitrification and the dynamic changes in nitrifying microorganisms in a subtropical rice-based ecosystem: A long-term field study. Soil Till. Res. 150, 132–138 (2015).
Guo, Z. & Jia, Z. Meta-analysis of soil nitrification activity in ecosystems typical of China. Acta Pedologica Sinica 5, 138–145 (2014). (in Chinese with English abstract).
Xie, H. et al. Long-term manure amendments enhance neutral sugar accumulation in bulk soil and particulate organic matter in a Mollisol. Soil Biol. Biochem. 78, 45–53 (2014).
Nannipieri, P. & Eldor, P. The chemical and functional characterization of soil N and its biotic components. Soil Biol. Biochem. 41, 2357–2369 (2009).
Kuzyakov, Y. & Xu, X. Competition between roots and microorganisms for nitrogen: mechanisms and ecological relevance. New Phytol. 198, 656–669 (2013).
Zhang, W. et al. Differentiating the mineralization dynamics of the originally present and newly synthesized amino acids in soil amended with available carbon and nitrogen substrates. Soil Biol. Biochem. 85, 162–169 (2015).
Shen, W. et al. Higher rates of nitrogen fertilization decrease soil enzyme activities, microbial functional diversity and nitrification capacity in a Chinese polytunnel greenhouse vegetable land. Plant Soil 337, 137–150 (2010).
Morier, I., Schleppi, P., Siegwolf, R., Knicker, H. & Guenat, C. 15N immobilization in forest soil: a sterilization experiment coupled with 15CPMAS NMR spectroscopy. Eur. J. Soil Sci. 59, 467–475 (2008).
Sørensen, P. Short-term nitrogen transformations in soil amended with animal manure. Soil Biol. Biochem. 33, 1211–1216 (2001).
Miltner, A., Kindler, R., Knicker, H., Richnow, H. H. & Kästner, M. Fate of microbial biomass-derived amino acids in soil and their contribution to soil organic matter. Org. Geochem. 40, 978–985 (2009).
Qin, Z., Zhang, Y., Zhou, Z., Shi, X. & Guo, T. Characteristics of mineralization and nitrification in neutral purple paddy soil from a long-term fertilization experiment. Scientia Agricultura Sinica. 46, 3392–3400 (2013). (in Chinese with English abstract)
Ste-Marie, C. & Paré, D. Soil, pH and N availability effects on net nitrification in the forest floors of a range of boreal forest stands. Soil Biol. Biochem. 31, 1579–1589 (1999).
Tahovská, K. et al. Microbial N immobilization is of great importance in acidified mountain spruce forest soils. Soil Biol. Biochem. 59, 58–71 (2013).
Fang, Y. et al. Microbial denitrification dominates nitrate losses from forest ecosystems. Proc. Natl. Acad. Sci. USA. 112, 1470–1474 (2015).
Nicolardot, B., Recous, S. & Mary, B. Simulation of C and N mineralisation during crop residue decomposition: a simple dynamic model based on the C: N ratio of the residues. Plant Soil 228, 83–103 (2001).
Probert, M. E., Delve, R. J., Kimani, S. K. & Dimes, J. P. Modelling nitrogen mineralization from manures: representing quality aspects by varying C:N ratio of sub-pools. Soil Biol. Biochem. 37, 279–287 (2005).
Luce, M. S., Whalen, J. K., Ziadi, N., Zebarth, B. J. & Chantigny, M. H. Labile organic nitrogen transformations in clay and sandy-loam soils amended with 15N-labelled faba bean and wheat residues. Soil Biol. Biochem. 68, 208–218 (2014).
Farrell, M., Prendergast-Miller, M., Jones, D. L., Hill, P. W. & Condron, L. M. Soil microbial organic nitrogen uptake is regulated by carbon availability. Soil Biol. Biochem. 77, 261–267 (2014).
Shi, W. & Norton, J. M. Microbial control of nitrate concentrations in an agricultural soil treated with dairy waste compost or ammonium fertilizer. Soil Biol. Biochem. 32, 1453–1457 (2000).
Knicker, H., Lüdemann, H. D. & Haider, K. Incorporation studies of NH4+ during incubation of organic residues by 15N-CPMAS-NMR spectroscopy. Eur. J. Soil Sci. 48, 431–441 (1997).
Abe, T., Katayama, A. & Watanabe, A. Temporal changes in distribution and composition of N from labeled fertilizer in soil organic matter fractions. Bio. Fert. Soils 43, 427–435 (2007).
Miltner, A., Bombach, P., Schmidt-Brücken, B. & Kästner, M. SOM genesis: microbial biomass as a significant source. Biogeochemistry 111, 41–55 (2012).
Simpson, A. J., Simpson, M. J., Smith, E. & Kelleher, B. P. Microbially derived inputs to soil organic matter: are current estimates too low? Environ. Sci. Technol. 41, 8070–8076 (2007).
Tremblay, L. & Benner, R. Microbial contributions to N-immobilization and organic matter preservation in decaying plant detritus. Geochim Cosmochim Ac. 70, 133–146 (2006).
Malik, M. A. Khan, K. S. & Marschner, P. Microbial biomass, nutrient availability and nutrient uptake by wheat in two soils with organic amendments. J. Soil Sci Plant Nutr. 13, 955–966 (2013).
Ma, Q. et al. Responses of biotic and abiotic effects on conservation and supply of fertilizer N to inhibitors and glucose inputs. Soil Biol. Biochem. 89, 72–81 (2015).
Appel, T., Schneide, B. & Kosegarten, H. Extractability of labelled microbial biomass N by electro-ultrafiltration and CaCl2 extraction. Bio. Fert. Soils 23, 314–320 (1996).
Macdonald, A. J., Poulton, P. R., Stockdale, E. A., Powlson, D. S. & Jenkinson, D. S. The fate of residual 15N-labelled fertilizer in arable soils: its availability to subsequent crops and retention in soil. Plant Soil 246, 123–137 (2002).
Sebilo, M., Mayer, B., Nicolardot, B., Pinay, G. & Mariotti, A. Long-term fate of nitrate fertilizer in agricultural soils. Proc. Natl. Acad. Sci. USA. 110, 18185–18189 (2013).
Blagodatskaya, E. & Kuzyakov, Y. Active microorganisms in soil: critical review of estimation criteria and approaches. Soil Biol. Biochem. 67, 192–211 (2013).
Micks, P., Aber, J. D., Boone, R. D. & Davidson, E. A. Short-term soil respiration and nitrogen immobilization response to nitrogen applications in control and nitrogen-enriched temperate forests. Forest Ecol. Manag. 196, 57–70 (2004).
Zhu, W. & Wang, W. Does soil organic matter variation affect the retention of 15NH4+ and 15NO3- in forest ecosystems? Forest Ecol. Manag. 261, 675–682 (2011).
Frey, S. D., Elliott, E. T., Paustian, K. & Peterson, G. A. Fungal translocation as a mechanism for soil nitrogen inputs to surface residue decomposition in a no-tillage agroecosystem. Soil Biol. Biochem. 32, 689–698 (2000).
Lin, S., Dittert, K., Wu, W. & Sattelmacher, B. Added nitrogen interaction as affected by soil nitrogen pool size and fertilization–significance of displacement of fixed ammonium. J. Plant Nutr. Soil Sci. 167, 138–146 (2004).
Ros, G. H., Hoffland, E. & Temminghoff, E. J. M. Dynamics of dissolved and extractable organic nitrogen upon soil amendment with crop residues. Soil Biol. Biochem. 42, 2094–2101 (2010).
Nieder, R., Benbi, D. K. & Scherer, H. W. Fixation and defixation of ammonium in soils: a review. Bio. Fert. Soils 47, 1–14 (2011).
Scherer, H. W., Feils, E. & Beuters, P. Ammonium fixation and release by clay minerals as influenced by potassium. Plant Soil Environ. 60, 325–331 (2014).
Lu, C., Chen, X., Shen, S., Shi, Y., Ma, J. & Zhao, M. Use efficiency and residual effect of 15N-labelled ryegrass green manure over a 9-year field micro-plot experiment. J. Soil Sci Plant Nutr. 13, 544–555 (2013).
Brookes, P. C., Landman, A., Pruden, G. & Jenkinson, D. S. Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method for measuring microbial biomass nitrogen in soil. Soil Biol. Biochem. 17, 837–842 (1985).
Keeney, D. R. & Nelson, D. W. Nitrogen-inorganic forms. Page, A. L. Eds.; Methods of soil analysis. Part 2. Chemical and microbiological properties, Madison, pp 643–698 (1982).
Stark, J. M. & Hart, S. C. Diffusion technique for preparing salt solutions kjeldahl digests and persulfate digests for Nitrogen-15 analysis. Soil Sci. Soc. Am. J. 60, 1846–1855 (1996).
Chen, R. & Dittert, K. Diffusion technique for 15N and inorganic N analysis of low-N aqueous solutions and Kjeldahl digests. Rapid Commun. Mass Spectrom. 22, 1727–34 (2008).
Silva, J. A. & Bremner, J. M. Determination and isotope-ratio analysis of different forms of nitrogen in soils: 5. Fixed ammonium. Soil Sci. Soc. Am. J. 30, 587–594 (1966).
Beuters, P. & Scherer, H. W. Modification of the standard method for determination of non-exchangeable NH4-N in soil. Plant Soil Environ. 58, 557–560 (2012).
Jenkinson, D. S. The determination of microbial biomass carbon and nitrogen in soil. In: Wilson, J. R. Eds.; Advances in nitrogen cycling in agricultural ecosystems. C.A.B. International, Wallingford, pp. 368–386 (1988).
Ku H. Notes on the use of propagation of error formulas. Journal of Research of the National Bureau of Standards (National Bureau of Standards) 70, 263–273 (1966).
Acknowledgements
This research was supported by grant from the National Natural Science Foundation of China [grant number 41001176], the Strategic Priority Research Program of the Chinese Academy of Sciences [grant number XDB15020200] and the Liaoning Science and Technology Project Program [grant number 2011215003; 2014215016]. We thank Ci Sun from the test center of Institute of Applied Ecology, Chinese Academy of Sciences for assistance with N stable isotope analysis.
Author information
Authors and Affiliations
Contributions
Z.Q. and C.L. designed the experiment. Z.Q. conducted the measurements, data analyses. Z.Q., B.H. and Y.F. wrote the manuscript. Y.S., X.C. and H.Z. assisted with the experiments. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Quan, Z., Huang, B., Lu, C. et al. The fate of fertilizer nitrogen in a high nitrate accumulated agricultural soil. Sci Rep 6, 21539 (2016). https://doi.org/10.1038/srep21539
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep21539
This article is cited by
-
Straw retention and inhibitor application reduce the leaching risk of mineral N in no-tillage systems of Northeast China
Plant and Soil (2024)
-
Animal manure rhizobacteria co-fertilization suppresses phytonematodes and enhances plant production: evidence from field and greenhouse
Journal of Plant Diseases and Protection (2022)
-
Effects of N Application Rate and Dicyandiamide on the Fate of 15N Fertilizer and the Abundance of Microbial Genes in a Sandy Soil Amended with Sugarcane Litter
Journal of Soil Science and Plant Nutrition (2022)
-
Soil microbial biomass and extracellular enzymes regulate nitrogen mineralization in a wheat-maize cropping system after three decades of fertilization in a Chinese Ferrosol
Journal of Soils and Sediments (2021)
-
Quantifying soil N pools and N2O emissions after application of chemical fertilizer and straw to a typical chernozem soil
Biology and Fertility of Soils (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.