Abstract
The effects of elevated CO2 (E-CO2) on maize and Asian corn borer (ACB), Ostrinia furnacalis, in open-top chambers were studied. The plants were infested with ACB and exposed to ambient and elevated (550 and 750 μl/l) CO2. E-CO2 increased the plant height and kernel number per ear. The plants had lower nitrogen contents and higher TNC: N ratios under E-CO2 than at ambient CO2. The response of plant height to E-CO2 was significantly dampened in plants with ACB infestation. However, the weight gain of the survivors declined in plants grown under E-CO2. Moreover, the plant damage caused by ACB was not different among the treatments. Overwintering larvae developed under E-CO2 had a lower supercooling point than those developed under ambient CO2. The results indicated that there was a positive effect of E-CO2 on the accumulation of maize biomass, i.e., the “air-fertilizer” effect, which led to a nutritional deficiency in the plants. The fitness-related parameters of ACB were adversely affected by the CO2-mediated decreased in plant nutritional quality and ACB might alter its food consumption to compensate for these changes. Larval damage to maize under E-CO2 appears to be offset by this “air-fertilizer” effect, with reductions in larval fitness.
Similar content being viewed by others
Introduction
Since the Industrial Revolution, the huge consumption of fossil fuels and the destruction of natural habitats by the activities of humans have led to a continuous increase in CO2 concentrations in recent decades. The ambient CO2 concentration is expected to double within the 21st century, i.e., it will increase from the current level of ∼379 μl/l to 700 μl/l1.
Because of the “fertilization effect”2, most C3 plants increase the rates of photosynthesis and photosynthate production when exposed to elevated CO2 (E-CO2)3. The phenotype response of plants to E-CO2 is generally an increased rate of growth and biomass accumulation4,5. However, not all plant species respond identically to E-CO2. Exposure to E-CO2 increased the biomass of Agrostis capillaris (L.) and Poa alpine (L.), but the biomass decreased in Festuca vivipara (L.)6. Additionally, the responses of plants may be different in the short term compared with the long term. The biomass of alfalfa, Medicago sativa (L.) and orchard grass, Dactylis glomerata (L.), was unaffected in the final two years of a 3-year experiment7. From the fertilization effect caused by the E-CO2, the increased rate of photosynthesis increases plant metabolism and generates plants with higher carbon-to-nitrogen ratios (C: N)8. Simultaneously, E-CO2 can lead to changes in plant secondary metabolism because of alterations in the plant allocation to defence chemistry and chemical signalling9. The concentrations of phenolics, terpenoids, condensed tannins and gossypol were higher in Bt cotton, Gossypium hirsutum (L.), plants grown under E-CO2, but the concentration of the Bt toxin protein decreased10. In soybean, Glycine max (L.), plants grown under E-CO2 exhibited increased concentrations of quercet in the glycosides11. The plant response induced by the feeding of insect herbivores may be weaker, when grown are grown under E-CO212. Depending on the species, the herbivore-induced response of plants may vary in different growth environments13.
Because of the elevated CO2-mediated dilution of the N content in plants results in a nutritional deficiency for protein-limited insect pests14, many insects compensate for the changes in plant quality by altering their food intake, which results in more severe damage or defoliation to the host plants15,16.
The C3 and C4 pathways of photosynthesis are distinct and C4 plants are less sensitive to E-CO2 than C3 plants17,18. Therefore, an elevation in CO2 theoretically would not influence the rate of photosynthesis in C4 plants19, however, research has found that the rate of photosynthesis and the above ground biomass increased in C4 plants20,21,22. There are several explanations for this phenomenon23. A report showed that C3 grasses were more nutritious and had higher levels of proteins, nonstructural carbohydrates and water and lower levels of fibre and toughness than C4 grasses under E-CO224. Maize is a C4 plant and is the most important food and feed crop in China. The Asian corn borer (ACB), Ostrinia furnacalis (Guenée), is a key pest of maize and causes yield losses of 30% in various agro-climatic regions25. The ACB overwinters as fully developed larvae that are found in the maize stalks, cobs and weed stems, or in a spun-silk covering in the plant debris26.
Numerous studies have been performed to predict the effects of rising CO2 concentrations on C3 crop-pest interactions in agriculture, but few studies have examined the effects on the C4 crops and their insect pests27. Similar to the C3 plants, E-CO2 also changes the interactions between maize and its pests. Yin et al. (2010)16 reported that the growth, development and consumption of Helicoverpa armigera (Hbn.) changed when it was fed maize grains grown under E-CO2. The exposure of maize plants and Chilo partellus (Swinhoe) to E-CO2 levels not only affected the growth and yield of maize, but also affected the development of the insect in the open-top chambers28.
The cold hardiness of an insect species is measured by its supercooling point (SCP), which influences the density of the overwintering population29. The SCP is influenced by various factors, including the host plant species and nutritional quality and the contents of water and of the cryoprotective substances in the larval body30,31,32,33. An understanding of the effects of E-CO2 on the SCP could provide direct evidence for the cold hardiness of the overwintering ACB populations in environments with future climate change.
The objectives of this study were to examine the effects of E-CO2 on the development and abundance of the 1st- and 2nd-generation ACBs and on the damage caused by the ACBs to the maize plants grown in open-top chambers. Additionally, the effect of E-CO2 on the cold hardiness of overwintering larvae was evaluated with tests to determine the supercooling points of diapause larvae. The information in this study on the performance and abundance of insects on plants (insect-plant interactions) under elevated levels of CO2 is as important as understanding the changes in herbivorous damage to agricultural commodities caused by global climate change.
Results
Maize plant chemistry
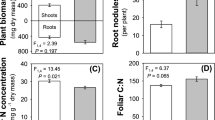
A negative effect of E-CO2 on the N content in maize plants was observed in the experiment (Table 1). Compared with the maize plants grown under ambient CO2, the N content significantly decreased by 8.0% and 17.0% for leaves and stalks, respectively, in maize plant grown under 750 μl/l CO2 (Table 1). The maize plants grown under 750 μl/l CO2 also had a 4.5% decrease in the N content of leaves compared with the maize plants grown under 550 μl/l CO2. There were no significant differences between the ambient and the 550 μl/l CO2 levels for the N content in leaves or stalks. A positive effect of E-CO2 on the C content of the maize plants was observed in the experiment (Table 1). The response of total non-structural carbohydrates (TNCs) including soluble sugars and starch, was consistent, with a significant effect of E-CO2 found only in the 750 μl/l CO2 treatment, for which the increasing was approximately 15.7% for soluble sugars and 8.3% for starch in leaves and 14.2% for soluble sugars and 11.9% for starch in stalks compared with maize plants grown under an ambient CO2 condition. The TNC: N ratio was significantly different among the treatments (Table 1). Compared with the ambient CO2 condition, the TNC: N ratio increased by 8.5 and 18.5% in leaves and 16.6% and 35.3% in stalks under E-CO2 levels (550 and 750 μl/l, respectively). Although the water content declined in the plants grown under elevated levels of CO2, the treatments were not significantly different (Table 1).
Fitness of ACB larvae
For the 1st generation of ACB, the larval survival among the CO2 treatments was not affected (Table 2). However for the 2nd-generation, the survival of the larvae decreased by 16.5% in 2012 and 21.0% in 2013 in the plant grown under elevated CO2 compared with the maize plants grown under ambient CO2. The average weight gain per larva (2nd generatrion, diapause) declined significantly by 13.0% and 16.1%, respectively, when the larvae were fed maize plants grown under the two elevated levels of CO2 (550 and 750 μl/l), compared with the ambient CO2 (Table 3). Additionally, the cold hardiness of the diapause larvae was significantly affected by the CO2 concentrations. The average SCP of the diapause larvae that developed on the maize plants grown under E-CO2 (750 μl/l) was slightly lower (approximately 0.31 °C lower) than that for the larvae that developed on the maize plants grown under ambient CO2.
The fecundity of the moths that developed from the overwintering larvae was marginally affected by the CO2 levels (Table 3). Although the number of eggs laid by the females that developed from the overwintering larvae declined as the atmospheric CO2 concentration increased, the difference was not significant among the treatments.
Maize plant growth and ACB damage ratings
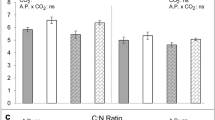
The results revealed that the E-CO2 had a positive effect on the growth of plants, whereas the ACB infestation affected plant growth negatively (Fig. 1). A significant “air fertilizer” effect of E-CO2 was observed on maize plant growth (2012: F2,6 = 4.92, P < 0.05, Fig. 1A; 2013: F2,6 = 5.23, P < 0.05, Fig. 1B). Averaged over 2 years, the height of maize plants significantly increased by 3.85% under 750 μl/l compared with the ambient CO2. By contrast, the ACB damage at the whorl stage significantly suppressed the growth of maize plants (2012: F2,8 = 8.94, P < 0.01, Fig. 1C; 2013: F2,8 = 33.25, P < 0.01, Fig. 1D). Averaged over 2 years, the height of maize plants decreased by 6.54% with an ACB infestation at the whorl stage compared with the control. Overall, the ACB damage at the whorl stage completely dampened the positive height response of the maize plants to the E-CO2 (Fig. 1), which suggested that the larvae altered their food consumption to compensate for the changes in the quality of the maize plants. Additionally, the grain yields of the maize plants were significantly affected by the concentration of CO2 (Table 4). The average number of kernels per ear increased by 4.3% and 4.5% in 2012 and 2013, respectively, under E-CO2 (750 μl/l) compared with the ambient CO2. There was no significant difference in the weight per 100-kernels among the treatments. These results demonstrated the positive effect of E-CO2 on maize grain production. The damage ratings of the ACB were unaffected by the concentration of CO2 during the two years (Table 5). There were no significant differences in the number of tunnels per plant or in the length of cavities per plant among the treatments.
Discussion
The effects of E-CO2 on maize plants have been assessed in a number of places with variable conditions. A few studies have suggested that maize plant are insensitive or less sensitive to elevated levels of CO2 in the absence of drought and heat34,35. Moreover, most studies have revealed that E-CO2 has a positive effect on the maize plant, i.e., maize plant are likely to have greater rates of photosynthesis and above ground biomass accumulation in addition to reduced transpirational water losses and increased water-use efficiencies36,37,38,39. The number of seed is also greater with E-CO2 than that of plants grown under ambient CO2 level (+5.0%)40. The stimulation of the photosynthesis and growth of maize plants under E-CO2 typically results in a reduction in leaf N content, or an increased in the TNC: protein ratio of maize grains16. The results of the present study were in accordance with previous studies that showed positive effects of E-CO2 on plant biomass, i.e. the heights of plant and the kernels per ear increased in the E-CO2 environment. Additionally, the chemical changes in the maize plants followed the general pattern of plants in responses to E-CO2, such as a decrease in the nitrogen content and an increase in the total nonstructural carbohydrates41. The TNC: N ratio significantly increased in response to E-CO2 in the present study, which suggested that the nutritional quality of the maize plant was reduced when the maize was grown under an E-CO2 condition. Similar research found that C4 grasses were poor host plants primarily because of their lower level of nutrient, higher level of fibre and greater toughness42.
Despite the direct response of many insects to E-CO2, the changes in the performance of herbivorous insects are intimately correlated with changes in the quality of food plants grown under E-CO2 conditions. The nitrogen (protein) content of the host plant is only one limiting nutrient for insect herbivores43 and a number of chemical compositions affect the nutritional quality of host plants44. A decrease in the foliar nitrogen content of host plants can affect the rates of development and survival of insect herbivores45. Moreover, insects that can compensate for the CO2-mediated dilution of foliar nitrogen by increasing the rate of feeding will experience retarded growth and will be subject to predation for a longer period of time (the slow-growth-high-mortality hypothesis)46. In present study, the survival decreased and the average weight gain per larva declined significantly when the larvae fed on the maize plants grown under E-CO2, possibly because of the CO2-mediated declines in the nitrogen content of the maize plants. These results indicated that the ACB is a protein-limited insect; the development and survival of the larvae were adversely affected by the CO2-mediated reduced suitability and nutritional quality of the host maize plants. Prominent among the many factors that affect the amount of plant tissues consumed by insect herbivores is that of the suitability and nutritional quality of their host plants. Studies have found that some leaf-chewing herbivores perform compensatory feeding and by increase the intake of foliage with a lower nitrogen content to meet their nutritional requirements under an E-CO2 environment27,45. As a consequence, levels of damage or defoliation increase. By contrast, the plants may be damaged less and have more undamaged foliar area when the E-CO2 causes an increase in plant biomass and reduces the plant fitness-mediated population density of insect herbivores47. In the present study, the survival of the ACB larvae declined in the maize plants exposed to E-CO2 compared with the ambient treatment level. The E-CO2 also reduced the suppression of maize plant height caused by the ACB infestation. Therefore, the damage caused by the ACB to maize will be offset by the “air-fertilizer” effect for the plant and the reduced fitness of the insect herbivores on the host plants.
The ACB overwinters as fully developed larvae in maize stalks in northern China. The overwintering larval population (2nd-generation) has an important role in the overwinter survival of the ACB and in the regulation of the population for the subsequent year48. The SCP of diapause larvae is largely related to the larval cold hardiness. In the present study, the diapause larvae that developed on the maize plants grown under 750 μl/l CO2 weighed less and had a slightly lower SCP than the larvae that developed on the maize plants grown under ambient CO2. These results suggested that there was a positive effect of E-CO2 on the cold hardiness of diapausing larvae and many reports have shown that the host plants play a pivotal role in the coldhardiness of insect herbivore. For example, the average SCP was significantly lower for the 3rd instar larvae of beet armyworm, Spodoptera exigua (Hbn.), that developed from cabbage than those that developed from pakehoi, shallot and spinach49. Similar evidence was found for the hemlock looper, Lambdina fiscellaria (Guenée)50. The host plant quality affected the overwintering success of the leaf beetle, Chrysomela lapponica (L.)32, the hypothesis to explain this result was that the high water content in the high-weight beetles of C. lapponica might be the primary cause of the increased winter mortality51. In the present study, the larval body weight was lower and the increase in the SCP occurred under E-CO2 conditions; however, these results might also be associated with the nutritional quality and the lower water content of the maize plants that were grown under E-CO2 than those that were grown in ambient conditions.
In this study, the survival rate of the overwintering larvae (data not shown) was not influenced by the E-CO2 although there was a small decrease in the SCPs of the larvae. Additionally, the number of eggs laid per female that developed from the overwintering larvae declined at E-CO2 treatments although the difference was not statistically significant compared with the ambient condition. Taken together, there was insufficient evidence to conclude that the ACB exhibited a direct response to the elevated levels of CO2.
Methods
Open-top chambers
This experiment was conducted in regular octagonal open-top chambers (4.20 m in diameter by 3.0 m in height) located at the Gongzhuling Experimental Station of the Institute of Plant Protection, Chinese Academy of Agricultural Sciences, Gongzhuling, Jilin Province, China (43°30′ N, 124°47′ E; 224.9 m above sea level). Three levels of CO2 were applied continuously, i.e., ambient CO2 (∼390 μl/l) and E-CO2 (550 μl/l and 750 μl/l), which represented the current and predicted levels of CO2 in future years, respectively52,53, each treatment was replicated four times, for a total of twelve chambers in the experiment. The air was continuously distributed from the blowers into the chambers through a water curtain cooling system into perforated polyethylene ducts inside the chamber base-wall at 10 cm above the level of the soil. The CO2 was added to the inlet airstreams in the chambers of the elevated treatments to reach the target CO2 concentrations (550 μl/l and 750 μl/l). The concentrations were monitored and adjusted with a CO2 sensor (JQAW-8VACD, ColliHigh Company, Beijing, China) once every 60 s to ensure relatively stable levels of CO2. The actual mean CO2 concentrations in the chambers were 542 ± 14 μl/l and 746 ± 15 μl/l for the two E-CO2 levels, whereas in the ambient chambers, the concentration was ∼390 μl/l. The concentrations in the ambient chambers were monitored but were not controlled. The automatic-control system for adjusting the CO2 concentration was similar to that described by Chen et al. (2005)54, which also included the specifications for the open-top chambers. The open tops of the OTCs were covered with nylon netting to prevent insect immigration. The detailed descriptions of the chamber design and the experimental set-up will be published separately.
Maize variety and growth conditions
The maize (Zea mays L., Poaceae) (XY335, DuPont Pioneer Hi-Bred, Beijing, China) was planted with 50 cm row spacing (a total of 60 plants per chamber) in the open-top chambers on 10 May, 2012 and 15 May, 2013. This hybrid is typical of those grown for commercial production and the field management was consistent with the common cultural practices used in local farming.
Insect stocks and plant infestation
The ACB neonates used in this study were obtained from a laboratory colony that originated from a field population collected every year, which was maintained on a regular artificial diet for ACBs26 for 3–4 generations in the Institute of Plant Protection, Chinese Academy of Agricultural Science.
The plants were infested when they developed to the whorl and silking stages, which represent the 1st- and the 2nd-generation infestations in nature, respectively. Before the first infestation, each chamber was separated into two plots with screen to prevent the larvae from transferring between the two plots. The two plots in a chamber were used as treatments with either the 1st-generation infestation or the 2nd-generation infestation. Each plant was infested with 50 neonates (<24 h) of the ACB with traditional artificial infestation techniques similar to those described by He et al. (2000)55 on 29 and 28 June (the 1st-generation) and on 8 and 7August (the 2nd-generation) in 2012 and 2013. To avoid exposing the neonates to high temperature and direct sunlight, the infestations were applied during the late afternoon to evening. Nine chambers were used for the ACB infestation, which included three for each level of CO2 and another three chambers were used for the assays on chemical composition of the maize leaves.
Larval survival sampling and maize plant damage rating
For the 1st-generation, when the fifth instar larvae of the ACB were observed (24 July, 2012 and 25 July, 2013) in the maize field around the open-top chambers, the maize plants were dissected and the plant height and the number of larvae and tunnels and the tunnel lengths were recorded or measured. For the 2nd-generation, the maize plants were dissected on 22 September, 2012 and 20 September, 2013 (ready to harvest), the supercooling points and the overwinter status for 2012 were determined for the larvae from the dissected maize plants.
Supercooling points and overwinter status
The larvae were collected from the dissected maize plants during the autumn harvest, introduced into plastic centrifuge tubes with an air hole punched through the bottom (1.5 ml) and placed into cartons maintained, in the open-top chambers during the winter. The supercooling points of the larvae after overwintering were determined on 18 January 2012. A tube was constructed by removing the bottom part from a micro-centrifuge tube (0.5 ml), which was used to position larvae that were connected to a multichannel temperature recorder (TMC-40A, designed by the Institute of Agro-meteorology, Chinese Academy of Agricultural Sciences, Beijing). These tubes were then placed in the temperature test chamber (Heraeus-Votsch VM 04/100) at 0 °C to equilibrate for 24 h before cooling at a rate of 1 °C/min until a temperature of −40 °C was reached. The lowest temperature reached before the release of the latent heat of fusion was recorded as the supercooling point (SCP). Twenty-four larvae were tested for each treatment and each treatment was replicated three times. The larvae that were not unsupercooled with the same origin were then placed individually into modified 5-ml centrifuge tube (two holes 1 mm in diameter were made in each lid; sterilized) with a piece of wet cotton as a moisture source and a piece of corrugated paper as a cryptic habitat. Finally, these larvae were reared to pupation at 26 °C, 70% RH and with a 16:8 h (L:D) photoperiod. The newly emerged moths from each treatment were transferred in pairs to an oviposition cage (11 cm × 8 cm × 8 cm), which was covered with a piece of waxed paper as an oviposition substrate26. The number of eggs laid per female was recorded daily.
Chemical compositions of maize leaves
One entire unfolded ear leaf and stalk was collected from each maize plant in the OTCs for tissue samples for the chemical composition assays on the 75th day after sowing. Ten maize plants were selected at random from each of the three CO2 treatments, on three separate occasions, for a total of 30 leaves and 30 stalks per treatment. The water content, as a proportion of fresh weight, was calculated after the maize leaves and stalks were dried at 80 °C for 72 h. The total non-structural carbohydrates (primarily soluble sugars and starch) were analysed using the method of Tissue and Wright (1995)56. The nitrogen content was assayed using a Kjeltec N analyser (Model KDY-9830; Foss automated Kjeltec instruments, Beijing, China).
Data analyses
One-way analyses of variance (ANOVAs) were used to analyse the effects of elevated CO2 on the chemical compositions of maize leaves and stalks, larval survival per maize plant, larval body weights, supercooling points, kernel numbers, 100-kernel weights, maize plant heights, the ACB damage ratings and the number of eggs. All data were analysed with a general linear model procedure (PROC GLM) (SAS Institute 2001). Differences in the treatments were compared using CONTRASTS test. The significance level was set at P < 0.05. Before the analyses, the data were subjected to standard transformations to improve their normality and the homogeneity of variance. The percentage data were arcsine transformed to meet the assumptions of homogeneity of variance.
Additional Information
How to cite this article: Xie, H. et al. A field experiment with elevated atmospheric CO2-mediated changes to C4 crop-herbivore interactions. Sci. Rep. 5, 13923; doi: 10.1038/srep13923 (2015).
References
Rossi, A. M., Stiling, P., Moon, D. C., Cattell, M. V. & Drake, B. G. Induced defensive response of myrtleoak to foliar insect herbivory in ambient and elevated CO2 . J. Chem. Ecol. 30, 1143–1152 (2004).
Allen, L. H. Jr., Baker J. T. & Boote K. J. The CO2 fertilization effect: higher carbohydrate production and retention as biomass and seed yield. In FAO Corporate Document Repository: Global climate change and agricultural production. Direct and indirect effects of changing hydrological, pedological and plant physiological processes. (1996) Available at: http://www.fao.org/docrep/w5183e/w5183e06.htm (Accessed: 19th November 2014).
Drake, B. G., Gonzalez-Meler, M. A. & Long, S. P. More efficient plants: a consequence of rising atmospheric CO2? Ann. Rev. Plant Physiol. Mol. Biol. 48, 609–639 (1997).
Saxe, H., Ellsworth, D. S. & Heath, J. Tansley Review No. 98. Tree and forest functioning in an enriched CO2 atmosphere. New Phytol. 139, 395–436 (1998).
Owensby, C. E., Ham, J. M., Knapp, A. K. & Allen, L. M. Biomass production and species composition change in a tall grass prairie ecosystem after long-term exposure to elevated atmospheric CO2 . Global Change Biol. 5, 497–506 (1999).
Baxter, R., Ashenden, T. W., Sparks, T. H. & Farar, J. F. Effects of elevated carbon dioxide on three montane grass species: I. Growth and dry matter partitioning. J. Exp. Bot. 45, 305–315 (1994).
Bunce, J. A. Long-term growth of alfalfa and orchard grassplots at elevated carbon dioxide. J. Biogeogr. 22, 341–348 (1995).
Wilsey, B. J. Plant responses to elevated atmospheric CO2 among terrestrial biomes. Oikos 76, 201–205 (1996).
Bidart-Bouzat, M. & Imeth-Nathaniel, A. Global change effects on plant chemical defenses against insect herbivores. J. Integr. Plant Biol. 50, 1339–1354 (2008).
Coviella, C. E., Stipanovic, R. D. & Trumble, J. T. Plant allocation to defensive compounds: interactions between elevated CO2 and nitrogen in transgenic cotton plants. J. Exp. Bot. 53, 323–331 (2002).
O’Neill, B. F. et al. Impact of elevated levels of atmospheric CO2 and herbivory on flavonoids of soybean (Glycine max Linnaeus). J. Chem. Ecol. 36, 35–45 (2010).
Vuorinen, T., Nerg, A. M., Ibrahim, M. A., Reddy, G. V. P. & Holopainen, J. K. Emission of Plutella xylostella-induced compounds from cabbages grown at elevated CO2 and orientation behavior of the natural enemies. Plant Physiol. 135, 1984–1992 (2004).
Agrell, J., Anderson, P., Oleszek, W., Stochmal, A. & Agrell, C. Combined effects of elevated CO2 and herbivore damage on alfalfa and cotton. J. Chem. Ecol. 30, 2309–2324 (2004).
Coviella, C. E. & Trumble, J. T. Effects of elevated atmospheric carbon dioxide on insect-plant interactions. Conserv. Biol. 13, 700–712 (1999).
Salt, D. T., Brooks, G. L. & Whittaker, J. B. Elevated carbon dioxide affects leaf-miner performance and plant growth in docks (Rumex spp.). Global Change Biol. 1, 153–156 (1995).
Yin, J., Sun, Y. C., Wu, G. & Ge, F. Effects of elevated CO2 associated with maize on multiple generations of the cotton bollworm, Helicoverpa armigera. Entomol. Exp. Appl. 136, 12–20 (2010).
Lawlor, D. W. & Mitchell, R. A. C. The effects of increasing CO2 on crop photosynthesis and Productivity, a review of field studies. Plant Cell Environ. 14, 807–818 (1991).
Leakey, A. D. B. Rising atmospheric carbon dioxide concentration and the future of C4 crops for food and fuel. P. Roy. Soc. B 276, 2333–2343 (2009).
Caemmerer, S. Von & Furbank, R. T. The C4 pathway: an efficient CO2 pump. Photosynth. Res. 77, 191–207 (2003).
Wand, S. J. E., Midgley, G. F., Jones, M. H. & Curtis, P. S. Responses of wild C4 and C3 grass (Poaceae) species to elevated atmospheric CO2 concentration: a meta-analytic test of current theories and perceptions. Global Change Biol. 5, 723–741 (1999).
Ziska, L. H., Sicher, R. C. & Bunce, J. A. The impact of elevated carbon dioxide on the growth and gas exchange of three C4 species differing in CO2 leak rates. Physiol. Plant. 105, 74–80 (1999).
Watling, J. R., Press, M. C. & Quick, W. P. Elevated CO2 induces biochemical and ultrastructural changes in leaves of the C4 cereal sorghum. Plant Physiol. 123, 1143–1152 (2000).
Wang, Y. X., Yang, L. X., Manderscheid, R. & Wang, Y. L. Progresses of free-air CO2 enrichment (FACE) researches on C4 crops: a review. Acta Ecol. Sin. 31, 1450–1459 (2011).
Barbehenn, R. V., Chen, Z., Karowe, D. N. & Spickard, A. C3 grasses have higher nutritional quality than C4 grasses under ambient and elevated atmospheric CO2 . Global Change Biol. 10, 1565–1575 (2004).
Wang, Z. Y., Lu, X., He, K. L. & Zhou, D. R. Review of history, present situation and prospect of the Asian maize borer research in China. J. Shenyang Agric. Univ. 31, 402–412 (2000).
Zhou, D. R. et al. Asian corn borer and its integrated management. Golden Shield Press, Beijing, China (1995).
Chen, F. J., Wu, G., Parajulee, M. N. & Ge, F. Long-term impacts of elevated carbon dioxide and transgenic Bt cotton on performance and feeding of three generations of cotton bollworm. Entomol. Exp. Appl. 124, 27–35 (2007).
Mina, U., Bhatia, A. & Kumar, U. Response of maize and its pest Chilo partellus to ozone and carbon dioxide exposure. Maydica 57, 183–187 (2012).
Sømme, L. Supercooling and winter survival in terrestrial arthropods. Comp. Biochem. Physiol. A-mol. 73, 519–543 (1982).
Worland, M. P. The relationship between water content and cold tolerance in the arctic collembolan Onychiurus arcticus (Collemola: Onychiuridae). Eur. J. Entomol. 93, 341–348 (1996).
Fields, P. G. et al. The effect of cold acclimation and deacclimation on cold tolerance, trehalose and free amino acid levels in Sitophilus granaries and Cryptoleste ferrugineus (Coleoptera). J. Insect Physiol. 44, 955–965 (1998).
Zvereva, E. L. Effects of host plant quality on overwintering success of the leaf beetle Chrysomela lapponica (Coleoptera: Chrysomelidae). Eur. J. Entomol. 99, 189–195 (2002).
Xu, L. R., He, K. L. & Wang, Z. Y. Studies on variation in cold hardiness in relation to the in vivo water, lipid and sugar content of Conogethes punctiferalis (Guenée) larvae living on three different host plant species. Chin. J. Appl. Entomol. 49, 197–204 (2012).
Leakey, A. D. B. et al. Photosynthesis, productivity and yield of maize are not affected by open-air elevation of CO2 concentration in the absence of drought. Plant Physiol. 140, 779–790 (2006).
Twine, T. E. et al. Impacts of elevated CO2 concentration on the productivity and surface energy budget of the soybean and maize agroecosystem in the Midwest USA. Global Change Biol. 19, 2838–2852 (2013).
Maroco, J. P., Edwards, G. E. & Ku, M. S. B. Photosynthetic acclimation of maize to growth under elevated levels of carbon dioxide. Planta 210, 115–125 (1999).
Leakey, A. D. B., Bermacchi, C. J., Dohleman, F. G., Ort, D. R. & Long, S. P. Will photosynthesis of maize (Zea mays) in the US Corn Belt increase in future [CO2] rich atmosphere? An analysis of diurnal courses of CO2 uptake under free-air concentration enrichment (FACE). Global Change Biol. 10, 951–962 (2004).
Bootsma, A., Gameda, S. & McKenney, D. W. Potential impacts of climate change on corn, soybeans and barley yields in Atlantic Canada. Can. J. Plant Sci. 85, 345–357 (2005).
Hussain, M. Z. et al. Future carbon dioxide concentration decreases canopy evapotranspiration and soil water depletion by field-grown maize. Global Change Biol. 19, 1572–1584 (2013).
Jablonski, L. M., Wang, X. Z. & Curtis, P. Rapid report: plant reproduction under elevated CO2 conditions: a meta-analysis of reports on 79 crop and wild species. New Phytol. 156, 9–26 (2002).
Zvereva, E. L. & Kozlov, M. V. Consequences of simultaneous elevation of carbon dioxide and temperature for plant-herbivore interactions: a meta analysis. Global Change Biol. 12, 27–41 (2006).
Barbehenn, R. V., Karowe, D. N. & Spickard, A. Effects of elevated atmospheric CO2 on the nutritional ecology of C3 and C4 grass-feeding caterpillars. Oecologia 140, 86–95 (2004).
Mattson, W. J. Jr. Herbivory in relation to plant nitrogen content. Ann. Rev. Ecol. Syst. 11, 119–161 (1980).
Scriber, J. M. & Slansky, F. The nutritional ecology of immature insects. Ann. Rev. Entom. 26, 183–211 (1981).
Rao, M. S. et al. Impact of elevated CO2 on tobacco caterpillar, Spodoptera litura on peanut, Arachis hypogea. J. Insect Sci. 12, 103 (2012).
Lill, J. T. & Marquis, R. J. The effects of leaf quality on herbivore performance and attack from natural enemies. Oecologia 126, 418: 428 (2001).
Caulfield, F. & Bunce, J. A. Elevated atmospheric carbon dioxide concentration affects interactions between Spodoptera exigua (Lepidoptera: Noctuidae) larvae and two host plant species outdoors. Environ. Entomol. 23, 999–1005 (1994).
Lu, X. Factors affecting Asian corn borer emergency size and its forecast. J. Jilin Agric. Sci. 1, 44–48 (1997).
Zheng, X. L., Quan, W. L., Chen, W. J., Lei, C. L. & Wang, X. P. Effect of host plant on cold-hardiness of 3rd instar larvae of the beet armyworm, Spodoptera exigua. Chin. J. Appl. Entomol. 49, 1461–1467 (2012).
Rochehort, S., Berthiaume, R., Hébert, C., Charest, M. & Bauce, E. Effect of temperature and host tree on cold hardiness hemlock looper eggs along a latitudinal gradient. J. Insect Physiol. 57, 751–759 (2011).
Zvereva E. L. Effects of host plant quality on overwintering success of the leaf beetle Chrysomela lapponica (Coleoptera: Chrysomelidae). Eur. J. Entomol. 99, 189–195 (2002).
Intergovernmental Panel on Climate Change. Climate Change1995: Impacts, Adaptations and Mitigation of Climate Change: Scientific-Technical Analysis. [ Watson, R. T., Zinyowera, M. C. & Moss, R. H. (ed.)]. [3–18] (Cambridge University Press, Cambridge, UK 1996).
Intergovernmental Panel on Climate Change. Climate Change 2001: the Scientific Basis. [ Houghton, J. T., Ding Y., Griggs, D. J., Noguer, M., Linden, P. J. van der, Dai X. et al. (ed.)] [1–21] (Cambridge University Press, Cambridge, UK 2001).
Chen, F. J., Ge, F. & Su, J. W. An improved open-top chamber for research on the effects of elevated CO2 on agricultural pests in the field. Chin. J. Ecol. 24, 585–590 (2005).
He, K. L. et al. Efficacy of transgenic Bt cotton for resistance to the Asian corn borer (Lepidoptera; Crabidae). Crop Prot. 25, 167–173 (2000).
Tissue, D. T. & Wright, S. J. Effects of seasonal water availability on phenology and the annual shoot carbohydrate cycle of tropical forest shrubs. Funct. Ecol. 9, 518–527 (1995).
Acknowledgements
This research was supported by the Special Fund for Agro-scientific Research in the Public Interest (201303026), by the National Basic Research Program of China (973 Program) (2010CB951503) and by Science and Technology Research Project for Colleges and Universities in Hebei province (QN2015076).
Author information
Authors and Affiliations
Contributions
K.H. and H.X. designed the experiment. H.X., K.L., D.S. and X.L. performed the experiment. Z.W. and K.L. provided the insect, reagents and materials. H.X. and K.H. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Xie, H., Liu, K., Sun, D. et al. A field experiment with elevated atmospheric CO2-mediated changes to C4 crop-herbivore interactions. Sci Rep 5, 13923 (2015). https://doi.org/10.1038/srep13923
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep13923
This article is cited by
-
Effects of elevated CO2 on resistant and susceptible rice cultivar and its primary host, brown planthopper (BPH), Nilaparvata lugens (Stål)
Scientific Reports (2021)
-
Different adaptability of the brown planthopper, Nilaparvata lugens (Stål), to gradual and abrupt increases in atmospheric CO2
Journal of Pest Science (2020)
-
Elevated atmospheric CO2 concentrations alter grapevine (Vitis vinifera) systemic transcriptional response to European grapevine moth (Lobesia botrana) herbivory
Scientific Reports (2019)
-
CO2 fixation in above-ground biomass of summer maize under different tillage and straw management treatments
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.