Abstract
In this work, microscopic and histological studies suggest that Strobilanthes crispus ethanol extract reduce azoxymethane (AOM)-induced colonic aberrant crypt foci (ACF) in rats. S. crispus is considered a traditional medicine and used as an antioxidant. Its leaf contains a large amount of phenolic compounds to which its radical scavenging role is attributed and enhance its ability to eradicate oxidative stress reactions. The study was designed to determine the chemopreventive effect of S. crispus ethanol extract in vivo and in vitro by elucidating the effect of the extract on intermediate biomarkers which can be used as effective predictors of colon cancer. S. crispus was analyzed for DPPH free radical scavenging, nitric oxide (NO) and ferric acid reduction. The results indicated that S. crispus oral administration significantly inhibited colorectal carcinogenesis induced by AOM as revealed by the reduction in the number of ACF. S. crispus down-regulated the expression of PCNA, Bcl2 and β-catenin. Additionally, it exerted a pronounced inhibitory effect on MDA and NO levels and stimulatory effect on CAT and GPx activities. These results demonstrate that S. crispus is a chemopreventive agent for colorectal cancer through the suppression of early and intermediate carcinogenic phases that may be related to its flavonoid content.
Similar content being viewed by others
Introduction
Colorectal cancer is quite common and undoubtedly a major health problem among the Asian population1. The development of colorectal cancer is directly attributed to the activation of oxidative stress pathways in the intestinal lumen, which is manifested by the increased number and size of aberrant crypt foci. Aberrant crypt foci are benign adenomatous polyps that develop into an advanced adenoma with high-grade dysplasia and then progress to an invasive cancer2. The abundant appearance of aberrant crypt foci (ACF) as a precursor lesion and their increase in number are considered early abnormalities that occur during colorectal cancer induction3. Elevation of biochemical markers and consequent pathological signs are considered late markers associated with the imbalance between cellular growth and death and, hence, the progressive alteration of the intestinal homeostasis.
Oxidative stress plays a crucial role in the molecular mechanism of cancer development and progression4. Many studies have shown that oxidative radicals are among the contributing factors involved in cancer formation, invasion and metastatic spread. Exposure to oxidants may lead to enhanced expression of the enzyme nitric oxide synthase, resulting in an increase in the cellular metabolic rate. The oxidative radicals react with protein tyrosine and kinase residues and cause a second wave of lipid peroxidation. Lipid peroxidation activates a programmed death cascade and has been identified as a source of erroneous DNA replication, leading to gene mutation, mitochondrial membrane leakage, cell cycle transition and apoptosis (programmed cell death)5.
People are increasingly becoming aware that healthy nutrients cleanse their bodies from free radicals and improve their well-being. Nutrients with calcium, folate, fiber, omega-3 fatty acids, vitamin D and vitamin A are highly appreciated by practitioners and healers as chemopreventive and chemotherapeutic mediators6,7. Dietary phytochemicals are proving to be a relatively simple and practical approach to a healthy life and the daily consumption of flavonoid-rich vegetables or fruits has been recommended to reduce cancer risk. The wide range of chemical components that are naturally present in these dietary products has been associated with oxidative radical scavenging effects8,9. Flavonoid-rich phytochemicals from green tea are emerging as therapeutic agents for cancer cases in preclinical and clinical studies10. Their pharmacological effects relevant to cancer control have also been reported in the American Journal of Clinical Nutrition in which Lorenz 201311 highlighted the active constituents of plant ethanol extracts and pinpointed the relevant mechanism of action. This research has been conducted in the context of chemotherapeutic treatments12 and the ethanol extracts have not consistently been shown to reduce the mortality rate of colorectal cancer cases, although tumor size control has been suggested. For instance, the five-year survival rate is only 10% in patients with metastatic colorectal cancer, even with the heavy use of the most common chemotherapy, fluoropyrimidine 5-fluorouracil (5-FU). Nevertheless, fluoropyrimidine 5-fluorouracil (5-FU) has some side effects and has shown cytotoxicity to normal cells13, as it irreversibly inhibits thymidylate synthase (TS).
In the present study, we wanted to extend the potential benefits of diets based on flavonoids for cancer prevention. In particular, we evaluated the chemopreventive effects of Strobilanthes crispus, with an emphasis on an azoxymethane (AOM)-induced ACF model. S. crispus (Family of Acanthaceae) is native to countries such as Malaysia, Madagascar and Indonesia. S. crispus is commonly known as “pecah beling” or “jin batu” in the Malay language. The consumption of nutraceutical S. crispus herbal tea is increasing due to the additional nutrients and antioxidants it provides. The recommended daily level of consumption of the leafy ethanol extract is 5 g. S. crispus herbal tea is known for its diuretic, laxative and antidiabetic properties14. It contains highly bioactive compounds15, which are reported to be responsible for its antioxidant16 and antibacterial effects17 and wound healing18; its anticancer19 and specifically anti-breast and anti-prostate cancer properties have also been assessed20. The phenolic components have been identified as one of the main S. crispus bioactive compounds, in addition to the catechins, alkaloids, caffeine and tannins, which further contribute to the total defense mechanism. Wasman and Mahmood reported the protective effect of S. crispus on gastric ulcers21,22, but the potential effect of S. crispus for preventing colorectal cancer formation has not been studied.
Results
S. crispus exhibited DPPH free radical scavenging activity
The ability of S. crispus to reduce the free radical scavenging activity was tested by assaying DPPH. The DPPH absorption is attributed to the donation of an electron or hydrogen to a stable free radical that is observed in vitro as fading of the purple coloration of the absorption band. Our results demonstrated a significant difference between the obtained values of S. crispus-treated samples (approximately 86% at 25 μg/ml) and control samples. S. crispus exhibited dose-dependent free radical scavenging activity (Figure S1). S. crispus showed an IC50 value of 5.44 ± 1.76 μmol/l, which was closer to that of vitamin C (3.882 ± 0.628 μmol/l) (Figure S1, Table 1). S. crispus ethanol extract eradicated the overproduction of oxygen free radicals and significantly recovered the DPPH free radical scavenging activity.
S. crispus reduced ferric oxidation and decreased the level of nitric oxide radicals
Nitric oxide and ferric acids are very unstable radical species that produce stable products i.e. nitrate, nitrite, ferrous when reacting with oxygen molecules. The oxidation effect of S. crispus was tested in vitro under specific emission and excitation range and relative to the reported IC50 value. Figures S2,S3 and Table 1 illustrate the variation of the scavenging activity with maximum effect in samples treated with S. crispus compared to gallic acid (GA), whereas vitamin C exhibited less effect at the same concentration. The ethanol extract of S. crispus exhibited a scavenging activity against nitric oxide radicals that expressed an IC50 of 0.39 ± 0.11 μmol/l compared to vitamin C and GA (IC50 of 0.529 ± 0.009 and 2.78 ± 0.13 μmol/l, respectively). Supplementation with the ethanolic extract of S. crispus decreased the reactive oxygen species profiles by decreasing the nitric oxide parameters.
A two-fold higher effect on the reduction of iron from the ferric (Fe3+) to ferrous (Fe2+) form was observed with S. crispus-treated samples relative to BHT and vitamin C (12085.7 ± 0.006, 5228.6 ± 0.01 and 1181.4 ± 0.006 mmol/g, respectively) (Figure S2, Table 1). The cytotoxic effect of reactive oxygen species and the intercellular liberation of nitric oxide directly or indirectly affected the balance between the ferric (Fe3+) and ferrous (Fe2+) derivatives. Thus, in the S. crispus-treated model, the reduction of the ferric (Fe3+) to ferrous (Fe2+) form was remarkably increased compared to that observed with other conventional antioxidant agents.
Phenol and flavonoid contents were identified from the plant ethanolic extract
S. crispus ethanol extract was evaluated for its total phenolic and total flavonoid content. The TPC was 737.67 ± 0.024 mg (Figure S4-a) and was expressed as gallic acid equivalent in mg per g of plant ethanol extract. The TFC was 262.86 ± 0.005 mg (Figure S4-b) and was expressed as quercetin equivalent in mg per g of plant ethanol extract (Table 1).
Sublethal dose of S. crispus showed no toxicity when orally administered
S. crispus at a dose of 2500 mg/kg was nontoxic. No mortality or visible manifestations of hepatotoxic and nephrotoxic effects were observed within the 14 days of observations. Furthermore, there were no significant differences in blood parameters (urea, creatinine, total protein, albumin, glucose and enzymes (ALT, AST, ALP)) between the control and treated groups. Oral administration to rats was therefore considered safe. This gives a wide safety margin for the dosage of S. crispus extracts in the tested animals as there were no notable side effects associated to higher doses.
S. crispus supplementation affected body and colon weights
The initial body weights of rats were measured at the start of the experiment and thereafter weekly, spanning the two weeks of induction and eight weeks of treatment. Apart from the weight gain in the animal group treated with 250 mg/kg S. crispus, the average weights of 500 mg/kg S. crispus-treated and 5-FU drug-treated rats were similar to those of the AOM induced group (Table 2). The colon weights of S. crispus-treated groups (both doses) were significantly increased compared to the AOM induced group. In general, there were no significant differences in the liver, spleen and kidney weights among all groups. S. crispus induced an increase in the colon and body weight, which might have enhanced the stability of the intestinal wall and therefore improved the removal of superoxide radicals. The integrity of the lumen displayed catalase decomposition of hydrogen peroxide to water and oxygen; thus, allowing the intestinal enzymes to modulate the redox state of lysate and plasma.
S. crispus ethanol extract induced a decrease in ACF number in rat colon
Aberrant crypt foci (ACF) were visualized as slightly elevated connective tissue above the surrounding mucosa and demonstrated characteristic oval or slit-like orifices. The intestinal lumen had irregular luminal, elongated protruded crypts. The ACF was observed predominantly in the middle and distal colon in AOM-treated animals and to a lesser extent in the proximal colon (Fig. 1). AOM induction in the animal models leads to mitochondrial dysfunction and inhibits the release of protective intestinal acids. This was accompanied by the increase of the reactive oxygen species content in the intestinal lumen, contributing to hypertrophy of the cells and deformity of the glands followed by the ACF formation.
(a) Effect of S. crispus on macroscopically apparence of ACF in rats’ colon induced by AOM: (A) Normal group with normal crypts, (B) AOM group (ACF with multiple crypts >five), (C) 5-FU group, (D) S. crispus 250 mg/kg plant treated group (ACF with crypts <five), (E) S. crispus 500 mg/kg plant treated group (ACF with crypts <five) (methylene blue staining) The arrows indicated the crypts that were more than five foci in AOM control group and less than five foci in all treated groups. (b) Effect of S. crispus on AOM-induced ACF number in rat colon.
All rat groups treated with AOM developed ACF. Table 3 summarizes the effect of S. crispus on AOM-induced ACF formation. S. crispus produced a marked decrease in the number of ACF (approximately 71–74% of the AOM-induced values) (P < 0.01). S. crispus supplementation attenuated the distribution of ACF throughout the colon segments. ACF was mainly found in the middle and distal parts of the colon in the ethanol extract-treated groups and in the middle and proximal areas of the colon in the AOM-induced and 5-FU-treated groups. S. crispus treatment resulted in restoring the low number of crypts per focus compared to the AOM-induced rats, as shown in (Table 4). The decrease in the number and size of ACF in S. crispus-treated animals indicated that the extract restored the anatomical integrity and decreased the free radical generation, which may be due to the increased antioxidant defense. The average number of ACF in total and per foci decreased remarkably in the group of animals treated with S. crispus. The decline in the crypt formation was dominant in animals given S. crispus compared to positive and negative control. The above findings may constitute another evidence on how S. crispus addition contributes towards the cellular defense mechanisms against free radicals.
S. crispus-treated rats recovered normal intestinal anatomy and histopathological features
The histology of ACF and normal colon cells was observed using H&E staining. The colon sections showed normal architecture with intact mucosal and submucosal features. Proliferating mucosal glands and the presence of stained cells with elongated stratified nuclei and depletion of mucus were observed in the colon sections of AOM-induced rats (Fig. 2). It may be relevant that the increase in colon weight was consistent with the reaction of S. crispus towards the free radicals, suggesting that S. crispus played a role in intestinal homeostasis. The long term oral administration of S. crispus revealed no histopathological changes of liver and kidney in plant treated groups with respect to the normal (vehicle) group (Figure S5).
Cross-section of the rat colon stained with hematoxylin and eosin.
(A) Normal group with normal crypts, (B) AOM group (C) 5-FU group, (D) S. crispus 250 mg/kg plant treated group, (E) S. crispus 500 mg/kg plant treated group (100× magnification). Arrow is indicating to the normal colon cells with round nuclei in normal and treated groups while it is indicating the ACF cell with elongated nuclei and depletion of mucus in AOM induced group.
S. crispus ethanol extract decreased the level of MDA and NO and increased the GPx and CAT activities
Radical species play a crucial role in oxidative stress and their excessive generation is believed to react at the mitochondrial system and produce up- and down-regulation of key proteins that have toxic effects in the biological tissues. We tested the indirect suppressive effect of S. crispus on the radical species and the secondary expression of critical cellular proteins. For example, MDA is an aldehydic product of lipid peroxidation that reacts quickly with biomolecules, such as proteins, lipids and nucleic acids and leads to cellular dysfunction. On the other hand, the enhanced interaction of superoxide with NO in the oxidant environment increases the formation of peroxynitrite, which in turn oxidizes tetrahydrobiopterin (BH4, a cofactor for eNOS), leading to eNOS uncoupling. The uncoupling of eNOS, increase in nitric oxide synthase (iNOS) expression and diminished NO bioavailability play major roles in the onset of colon cancer formation. S. crispus and 5-FU caused a remarkable decrease in the level of MDA and lipid peroxidation. The group treated with S. crispus supplementation was found to normalize the formation of oxygen-derived free radicals. The levels of nitric oxide were significantly decreased in S. crispus-treated groups compared with the AOM induced group and the 5-FU reference group. Elevation of GPx and CAT activities was observed in both the AOM induced group and reference 5-FU treated group more than the S. crispus-treated groups (Table 5). The decline in MDA and NO activities accompanied by a significant increase in GPx and CAT and the overall change of the antioxidant markers indicate a positive effect of S. crispus against the oxidative stress that may also occur in all rat tissues.
S. crispus ethanol extract resulted in faint methylene blue staining of ACF
Aberrant crypt foci (ACF) were visualized as slightly elevated connective tissue above the surrounding mucosa and demonstrated characteristic oval or slit-like orifices. The intestinal lumen had irregular luminal, elongated protruded crypts. The ACF was observed predominantly in the middle and distal colon in AOM-treated animals and to a lesser extent in the proximal colon (Fig. 1). AOM induction in the animal models leads to mitochondrial dysfunction and inhibits the release of protective intestinal acids. The increase in the reactive oxygen species content in the intestinal lumen contributes to cell hypertrophy and deformity of the glands followed by ACF formation. The above findings may constitute additional evidence of how S. crispus participates in cellular defense mechanisms against free radicals.
Low expression of PCNA and Bcl2 staining in the S. crispus-treated groups
Representative photographs of the immunohistochemical staining of PCNA and Bc12 positive cells in AOM-treated groups alone, 5 FU-induced AOM-treated groups and S. crispus-induced AOM-treated groups clearly show that S. crispus suppress the proteins expression. Immunohistochemical results showed that the AOM-induced group had a higher expression of PCNA and Bcl2 protein, seen as heavy brownish staining (Figs 3 and 4), compared to the 5 FU and S. crispus-treated groups. The obvious bluish appearance indicated down-regulation of the PCNA protein. The supplementation with S. crispus modulated antioxidant gene expression and down-regulated intracellular genes, leading to lower mitochondrial oxygen radical generation.
(a) Effect of S. crispus on regulation of PCNA protein in the colon tissue of rats (A) Normal group with normal crypts, (B) AOM group, (C) 5-FU group, (D) S. crispus 250 mg/kg plant treated group, (E) S. crispus 500 mg/kg plant treated group (100× magnification). Arrow indicates cells with PCN protein. (Immunohistochemical stain, PCNA stain). (b) Effect of S. crispus on % regulation of PCNA protein in AOM-induced rats’ colon tissue.
(a) Effect of S. crispus on regulation of Bcl2 protein in the colon tissue of rats (A) Normal group with normal crypts, (B) AOM group, (C) 5-FU group, (D) S. crispus 250 mg/kg plant treated group, (E) S. crispus 500 mg/kg plant treated group (100× magnification). Arrow indicate cells with Bcl2 protein. (Immunohistochemical stain, Bcl2 stain). (b) Effect of S. crispus on % regulation of Bcl2 protein in AOM-induced rats’ colon tissue.
Suppression of beta catenin staining in the S. crispus-treated groups
Beta catenin is a multifunctional protein complex that is a component of cell-to-cell adhesion and part of the WNT signaling pathway. Beta catenin is identified also as one protein of DNA polymerase and its high expression suggests either cellular proliferation (turn over) or DNA mutation. Accumulation of beta-catenin in the nucleus was detected in almost all the AOM-treated groups (Fig. 5). Beta-catenin is a key factor in colorectal carcinogenesis and it is mostly expressed in the membrane at an early stage of the cancer, while the nucleus translocation occurs at an advanced stage illustrating the stages of genetic mutations. The labeling index of beta catenin in the colonic mucosa is presented in Fig. 5. Qualitative analysis of the stained sections following AOM- treatment showed a clear elevation and translocation of the protein to the colonic cancer cells compared with groups treated with 5 FU and S. crispus. Microscopic examination of the colonic tissue sections stained for beta catenin clearly showed a faint or no staining in S. crispus-induced AOM-treated groups.
Effect of S. crispus on the regulation of β-catenin protein in the colon tissue (A) Normal group (B) AOM control group (C) FU treated group, (D) 250 mg/kg S. crispus treated group and (E) 500 mg/kg S. crispus treated group (100× magnification). In the AOM and treated groups the arrows indicated the cells expressed the β-catenin protein. The β-catenin showed to be more expressed in AOM control group than that of treated groups. (Immunohistochemical stain, Bcl2 stain). (b) Effect of S. crispus on % regulation of β-catenin in AOM-induced rats’ colon tissue.
Discussion
We investigated the chemopreventive effect of S. crispus ethanol extracts on AOM-induced colorectal lesions by in vivo and the scavenging activity for free radicals by in vitro assays. AOM has been commonly used as a carcinogenic method to determine the chemopreventive effects of food ingredients such as indigestible sugars, red meat and green tea in rodent models23. We used the AOM experimental approach24 to initiate colon cell outgrowth that mimics the cryptal foci lesions in cancers. The AOM carcinogenic effect is largely reported in the literature by other groups of researchers25, as well as in our work9. In fact, we had already validated this approach from histopathological and biochemical perspectives in a previous study26. The dose safety and efficacy had been evaluated previously18 and in the present study and in accordance with the highest recommended dose and the animals were healthy and showed no hepatotoxicity and/or nephrotoxicity. Moreover, because an objective of the present work was to study the pharmacological effects of S. crispus, a simple acute model was useful.
S. crispus ethanol extract reduced the expected aberrant cryptal foci histological deficit, restoring the anatomical appearance of the wall as well as the physiological performance of mucus production. Histologically, the AOM-induced lesions showed proliferating mucosal glands with ACF characterized by elongated stratified nuclei, depletion of mucin and damage of submucosal glands, which is characteristic of benign aberrant focal crypti. In contrast, our observations showed a decrease in the number and size of aberrant cryptal foci with the S. crispus treatment. Immunohistochemical staining showed increased binding activity of PCNA and Bcl2 proteins and nuclear translocation with a concomitant deformity of the mucosal and submucosal features. Enhancement of PCNA and Bcl2 expression after AOM application, but not with S. crispus administration, was correlated with the proliferation process and the variation of mucosal thickness. PCNA and Bcl2 play an important role in DNA synthesis and DNA repair. PCNA and Bcl2 antibodies are commonly used for grading neoplasms27,28. It is well known that AOM produces loss of mucosal integrity and decreases the power of releasing mucin, accompanied by deficits in the submucosal glands29. Introduction of S. crispus ethanol extracts before AOM seems beneficial in retarding colorectal cancer formation by preventing cell proliferation and inhibiting the activation of PCNA and Bcl2. In conclusion, the integrity of the intraluminal wall structure is fundamental from both a protection and absorption prospective, as it contributes to the encoding of physiological and anatomical balance, hinders the transmission of harmful chemical signals and is essential for maintaining the flow in the mucous layer on the intraluminal surface. Therefore, it is not surprising that S. crispus supplementation increased the colon and total animal weights compared to other treatment groups.
Colorectal cancer, among other serious cancers, is correlated with oxidative insults. In vitro evidence shows that AOM administration generates a toxic cascade that mediates the apoptotic pathway in culture models30. Our data indicated the scavenging effect of S. crispus in the treated samples. S. crispus inhibits oxidative stress via attenuation of apoptotic signaling pathways in the cell. The high potent activities of S. crispus were due to the deactivation of the metabolic redox correlated directly with its suppressing effect on mitochondrial and nuclear levels. The S. crispus ethanol extract was shown to attenuate the toxic action of oxidative free radicals induced by the demonstration of AOM. The effect was higher than that of the anti-oxidative agents (i.e., vitamin C, gallic acid, quercetin and butylated hydroxytoluen) as assessed by DPPH, ferric reducing and nitric oxide scavenging assays. AOM supplementation induced the accumulation of free radicals and increased the intracellular level of apoptotic-sensitive proteins. The free radical scavenging mechanism involves oxidative-mediated apoptosis, which requires ferric acid reduction; therefore, the production of NO and iNOS is regulated31. S. crispus at an IC50 of 5.44 + 1.76 effectively inhibited DPPH free radicals, nitric oxide and ferric reduction sequences. These findings are supported by other studies that have shown decreased MDA31 and NO levels32 in rats treated with Strobilanthes asperrimus. Moreover, the measurements of serum biomarkers following oxidative stress showed that the levels were within the normal range and suggested a possible role of S. crispus to normalize the level of serum biomarkers (i.e., urea, creatinine, total protein, albumin, glucose and enzymes (ALT, AST and ALP)). The apoptotic signaling pathway of AOM induced a deleterious increase in the apoptosis-sensitive proteins (specifically MDA, GPx and CAT, among others) in the tissue, which were attenuated by S. crispus.
The intestinal microenvironment after chemical erosions by AOM seems to produce the second messenger nitric oxide (NO) following the excessive release of inducible nitric oxide synthase (iNOS). The production of iNOS is Ca2+-independent and known to participate in and inhibit the initiation of certain diseases. This process altered specific genes expression as well as a wide range of proteins and signaling cascades31,32. For instance, ROS release causes lipid peroxidation of polyunsaturated fatty acids and damage to specific mitochondrial proteins and transport systems by direct inhibition of enzymes resulting in loss of mitochondrial integrity. Malondialdehyde (MDA), one of the final products of polyunsaturated fatty acids peroxidation, is commonly known as a marker of oxidative stress and reflects the antioxidant status. In the present study, the data showed that MDA levels significantly decreased, indicating that S. crispus stopped the lipid peroxidation that is the prominent player in the membranes damage. Previous studies have demonstrated that the primary antioxidant enzymes which include SOD, GPx and CAT generate the antioxidant defense systems. The antioxidant enzymes provide the first line of cellular defense against ROS. In the present study, the data showed that SOD, GPx and CAT significantly increased leading to reduction of the free radical damage. Additionally, PCNA and Bc12 genes are produced by enzymatic cleavage and considered as a biomarker of oxidative DNA damage. Increased levels of DNA damage can cause the synthesis of a variety of incorrect proteins and therefore mutations and cancer formation.
At least two models for genetic alterations can be characterized in the development of colorectal cancer. In the presence of β-catenin staining, the scenario can be that the transcriptional effects were initiated by Rac1-dependent nuclear translocation of β-catenin followed by mutation of the Wnt ligand and deleted in colorectal cancer (DCC). The WNT signal transduction can also transcriptionally activate both NOS2 and COX2 by TCF4-β-catenin binding to their promoters in a regulatory feedback loop. Under these conditions and secondary to the release of iNOS, K1 or 2 kinases and GSK3beta phosphorylated β-catenin and activated threonine and serine residues in the N-terminal domain. Conversely, the NO-related DNA damage leads to p53 accumulation and p53-mediated apoptosis. NO can activate the WNT pathway by enhancing the enzymatic activity of cyclo-oxygenase 2 (COX2 and the release of prostaglandins 2E (PGE2))25. Beside these two pathways there is a possibility that less characterized co-activators/co-repressors displayed the transcription with a high degree of overlap among the main pathways.
Finally, recent studies have opened a new perspective for the possible use of S. crispus in medicine18,22. In fact, experimental data clearly indicated a significant protective role of S. crispus in breast cancer20. These findings are consistent with previous studies reporting the phenolic content and antioxidant activity of this plant15,16,33. Moreover, a recent study demonstrated the phenolic content and cytotoxic potential role of S. crispus16,34. Accordingly and consistent with this research, our laboratory recently reported, in vivo, using rats, a significant inhibition of colorectal cancer formation after similar treatment with medicinal plants (Andrographis paniculata and Gynura procumbens)9,25. Our results demonstrated the chemoprotective efficiency of S. crispus ethanol extracts, which can be attributed to its anti-proliferative, antioxidant and anti-radical scavenging properties. Accordingly, this natural agent might provide a potential template for the design of drug targets for treating colorectal cancers.
Materials and Method
Chemicals
Azoxymethane (Sigma Chemical Co., St Louis, MO, USA) was dissolved in sterile 0.9% normal saline and was given subcutaneously at 15 mg/kg body weight once a week for 2 weeks to induce ACF35. Fluorouracil (5-FU) (Sigma Chemical Co., St. Louis, MO, USA) was dissolved in 0.9% normal saline and was administered at a concentration of 35 mg/kg body weight intraperitoneally twice a week36.
Plant materials
The fresh plant leaves were obtained from Ethno Resources Sdn. Bhd., Selangor, Malaysiy and identified by a voucher specimen placed at the Herbarium of Rimba Ilmu, Institute of Biological Sciences, University of Malaya, Kuala Lumpur. The leaves were washed, shade-dried and grounded. The powdered plant material (100 g) was extracted with ethanol 95% (1000 ml) at room temperature three times, three days per each crop with occasional stirring and filtered. Three extracts were combined and evaporated in vacuo to give 11.5 g of crude ethanol extract37 which was kept dry at 45 °C and the clear semisolid ethanol extract was then dissolved in the vehicle solution.
In vitro study of the antioxidant properties of S. crispus ethanol extract
The 2, 2-diphenyl-1-picryl-hydrazyl free radical scavenging assay
The free radical scavenging activity of the plant ethanol extract was determined spectrophotometrically using 2, 2-diphenyl-1-picryl-hydrazyl (DPPH, Sigma-Aldrich, UK). DPPH, which is violet, produces a yellow complex when it is reduced in the presence of antioxidants38. The DPPH working solution was prepared using 95% ethanol at a concentration of 3.94 mg/100 ml and ascorbic acid was used as the standard. The sample and DPPH reagent were mixed well and incubated before the absorbance was measured using an ELISA plate reader at 517 nm in triplicate39. The scavenging activity of the ethanol extract was calculated based on the percentage of scavenged DPPH. (BIO-TEK instruments, Winooski, VT, USA).
Ferric-reducing antioxidant potential (FRAP) assay
The ferric-reducing antioxidant potential (FRAP, Thermo Fisher Scientific, USA) assay was performed as previously described40. FRAP reagent was prepared by mixing 16.7 mM FeCl3.6H2O and 8.3 mM 2,4,6-tripyridyl-s-triazine (TPTZ) with 250 mM acetate buffer, pH 3.6. The ethanol extract absorbance was read at zero minutes and at 4 minutes at 593 nm relative to FeSO4.7H2O as the standard negative control at 400–1000 mM by using a microplate reader. Vitamin C, gallic acid (GA), butylated hydroxytoluene and quercetin were used as positive controls39.
Nitric oxide scavenging activity
Nitrite (NO2,) is the stable product of nitric oxide conversion as assessed using Griess reagent (Sigma-Aldrich, UK). A series of dilutions were prepared from a 1 mg/ml stock of S. crispus ethanol extract. For every concentration, 50 μl of the ethanol extract was transferred into a 96-well microplate and an equal volume of sodium nitroprusside was added prior to the addition of Griess reagent (100 μl). After 5 minutes, the absorbance was read using a microplate reader at 550 nm under visible polychromatic light41 using sodium nitrite as the standard. The percentage of inhibition was obtained by calculating the ability of the plant ethanol extracts to inhibit nitric oxide formation relative to 0% inhibition of the control42. This ability was defined as (Ao − As/Ao)*100, where Ao is the absorption of control and As is the absorption of the test samples.
Total phenolic and total flavonoid content of the plant ethanol extract
Total phenolic content (TPC) was determined according to the Folin-Ciocalteu (Thermo Fisher Scientific, USA) spectrophotometric method with slight modifications using a microplate reader. The measurement was compared to a standard curve of GA solution and the value was expressed as milligrams of GA equivalent (GAE) per gram of dry plant ethanol extract (mg GAE/g db)43. The total flavonoid (TFC) content was measured using the aluminum chloride colorimetric method. The measurement was expressed as a quercetin equivalent in mg (QE)/g of ethanol extract44. The assays were performed in triplicate.
In vivo study
Ethical approval
The animal studies were approved by the Ethical Committee for Animal Experimentation, Faculty of Medicine, University of Malaya, Malaysia; [Ethic No. PM/07/05/2011/MMA (a) (R) for acute toxicity study and PM/07/05/2012/MMA (b) (R) for chemopreventive study]. Throughout the experiments, all animals received care according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” (2011).
Experimental animals
Healthy adult SD female rats were used for the acute toxicity study, whereas male rats were used for the chemopreventive study. Rats (6–8 weeks old), weighing between 150–180 g, were placed individually in separate cages. The animals were maintained on a standard diet and tap water ad libitum, under controlled conditions of humidity (50–60%), lighting (12-h light/dark cycle) and temperature (22–24 °C) and they were weighed weekly.
Acute toxicity study
A total of 16 healthy SD female rats were obtained from the Experimental Animal House, Faculty of Medicine, University of Malaya. The animals were divided equally into two groups: a) treated with vehicle (10% Tween-20, Thermo Fisher Scientific, USA) and treated with 2500 mg/kg ethanol extract in vehicle, respectively. The animals were fasted overnight (food but not water) prior to dosing. Food was withdrawn for a further 3 to 4 hours and the animals were observed for 30 min and at 2, 4, 24 and 48 h for the onset of clinical or toxicological symptoms. Mortality, if any, was noted over a period of 2 weeks; then, the animals were sacrificed on the 15th day. The colon, liver and spleen weights were reported. Serum biochemical and histological (liver and kidney) parameters were determined following standard methods45.
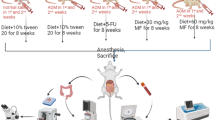
Experimental design
Following the safety/toxicity test and within the range of the chosen safe dose, we divided the adult male SD rats randomly into the following five groups:-
Group 1: (Saline group) injected with 0.9% sterile normal saline once a week for two weeks and fed daily with 10% Tween-20 (Thermo Fisher Scientific, USA) (5 ml/kg) for 60 days.
Group 2: (AOM group) injected with (15 mg/kg/ml) of AOM once a week for two weeks and fed daily with 10% Tween-20 (5 ml/kg) for 60 days.
Group 3: (5-FU group) injected with (15 mg/kg/ml) of AOM once a week for two weeks and injected intraperitoneally with (35 mg/kg/ml) of 5-FU twice weekly eight weeks.
Groups 4 & 5: (Plant treated groups) injected with (15 mg/kg/ml) of AOM once a week for two weeks and fed daily with 250 mg/kg and 500 mg/kg (5 ml/kg) of ethanol extract for 60 days36,46.
Histopathological examination
All rats were sacrificed by cervical dislocation after receiving an overdose of ketamine and xylazine (50 and 5 mg/kg body weight, respectively). Colons were removed and washed with cold phosphate buffered saline (PBS, Thermo Fisher Scientific, USA). They were then dissected longitudinally and cut into three equal segments (proximal, middle and distal). The segments were fixed flat between filter paper in 10% formalin for 5–10 minutes and stained with methylene blue (Sigma-Aldrich, UK) (0.2% in PBS solution) for 15–20 minutes to visualize the internal surface of the lumen furnished with the crypts. Each segment was further sliced into 2 cm sections and placed on microscope slides with the mucosa facing upwards. The aberrant crypts were recognized by their abnormal sizes and scored under the light microscope at 4× magnification. The number of ACF per colon, the number of aberrant crypts (ACs) in each optical focus and the location of each focus were recorded46,47. The paraffin blocks were processed (Leica Microsystem, Nussloch, Germany) and sectioned at 4 μm thickness using a microtome and stained with hematoxylin-eosin (H&E). Sections were examined and imaged with a Nikon camera connected to an Olympus light microscope (Tokyo, Japan).
Biochemical markers
Blood samples were collected in gel activating tubes and centrifuged at 3000 rpm for 10 minutes. The serum was separated and sent for determination of creatinine, total protein, glucose, albumin, alkaline phosphatase (ALP), alanine aminotransferase (ALT), aspartate aminotransferase (AST) and urea levels. Biochemical parameters were determined using the standard automated analyzer at the Central Diagnostic Laboratory, University of Malaya Medical Center.
Determination of antioxidants in colon homogenate
The colon tissue was washed with ice-cold PBS solution, pH 7.4, to remove other cells or debris. The colon tissue was homogenized on ice and centrifuged at 4500 rpm for 15 minutes at 4 °C. Tissue homogenate supernatant was used to measure the antioxidants48,49. Protease, a mixture of protease inhibitors with broad specificity for the inhibition of serine, cysteine, aspartic proteases and amino peptidases, was used to preserve enzymes from being degraded. Malonaldehyde (MDA) (cat #10009055) and nitric oxide (NO) (cat #D2NO-100) levels were measured. Glutathione peroxidase (GPx) (cat #703102) and catalase (CAT) (cat #707002) activities were also measured. All measurements were performed using commercial kits (Cayman Chemical Company, U.S.A) on a microplate reader.
Immunohistochemical staining
Colon sections were mounted on poly-lysine coated slides, dried overnight in an oven at 60 °C and then deparaffinized and rehydrated in graded alcohols. For retrieval of the antigen target, a retrieval solution containing 10 mM sodium citrate buffer was used. Immunohistochemical staining was done according to the manufacturer’s protocol (Dako ARK™ USA), based on avidin, biotin and peroxidase methodologies. Biotinylated primary antibody of diluted mouse PCNA and Bcl2 monoclonal antibody were applied at 1:100 and 1:50 dilution, respectively, followed by applying streptavidin-peroxidase (streptavidin conjugated to horseradish peroxidase in PBS containing the anti-microbial agent). Proliferating cell nuclear antigen (PCNA) is a well-known indicator of proliferation and Bcl2 is a well-known anti-apoptotic protein indicator. Staining was completed by a 5-minute incubation with 3, 3’-diaminobenzidine (DAB)-substrate-chromogen. Negative control sections were processed similarly but with the omission of the primary antibodies. Slides were rinsed in a bath of distilled or deionized water for 2–5 min, mounted and cover-slipped with an aqueous-based mounting medium or a non-aqueous permanent mounting.
Beta catenin immunostaining
Briefly, sections were deparaffinized and rehydrated through graded alcohol. Endogenous peroxidase activity was blocked with 0.5% hydrogen peroxide in methanol for 20 minutes. Antigen retrieval was used by microwave treatment; dilution was 1:100 for beta catenin (Novocastra, United Kingdom). Biotinylated universal antiserum (Starr Trek HRP Universal, Biocare Medical, CA) was used as the secondary antibody. After washing, the slides were incubated for 20 minutes at room temperature with Trekavidin HRP label (Starr Trek HRP Universal Biomedical CA) and developed for 10 minutes using 3- 3- diaminobenzidine as chromogen. After rinsing in water, the sections were counterstained with Meyer haematoxylin, dehydrated and mounted. Appropriate positive and negative controls were included in each run of immunohistochemistry.
Statistical analysis
Values were expressed as the mean ± SEM. Variation between groups was investigated employing one-way ANOVA followed by Tukey’s post-hoc test using SPSS version 20 (SPSS Inc. Chicago, IL, USA). P values less than 0.05 were statistically significant when compared to the control group.
Additional Information
How to cite this article: Al-Henhena, N. et al. Chemopreventive effects of Strobilanthes crispus leaf extract on azoxymethane-induced aberrant crypt foci in rat colon. Sci. Rep. 5, 13312; doi: 10.1038/srep13312 (2015).
References
Su, T. T. et al. Level of colorectal cancer awareness: a cross sectional exploratory study among multi-ethnic rural population in Malaysia. BMC Cancer. 13, 1–8 (2013).
Traverso, G. et al. Detection of proximal colorectal cancers through analysis of faecal DNA. The Lancet. 359, 403–404 (2002).
Kubben, F. et al. Proliferating cell nuclear antigen (PCNA): a new marker to study human colonic cell proliferation. Gut. 35, 530–535 (1994).
Seril, D. N. et al. Oxidative stress and ulcerative colitis-associated carcinogenesis: studies in humans and animal models. Carcinogenesis. 24, 353–362 (2003).
El-Seedi, H. R. et al. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties and biological activities of hydroxycinnamic acids. Journal of Agricultural and Food Chemistry. 60, 10877–10895 (2012).
Peters, R. et al. Diet and colon cancer in Los Angeles County, California. Cancer Causes Control. 3, 457–473 (1992).
Bingham, S. et al. Dietary fibre in food and protection against colorectal cancer in the European prospective investigation into cancer and nutriction (epic): an observational study. Lancet. 361, 1496–1501 (2003).
Bagchi, D. et al. Free radicals and grape seed proanthocyanidin ethanol extract: importance in human health and disease prevention. Toxicology. 148, 187–197 (2000).
Al-Henhena, N. et al. Chemopreventive Efficacy of Andrographis paniculata on Azoxymethane-Induced Aberrant Colon Crypt Foci In Vivo. PloS one. 9, e111118 (2014).
Boehm, K. et al. Green tea (Camellia sinensis) for the prevention of cancer. Cochrane Database Syst Rev. 3 (2009).
Lorenz, M. Cellular targets for the beneficial actions of tea polyphenols. The American Journal of Clinical Nutrition. 98, 1642S–1650S (2013).
Greenlee, R. T. et al. Cancer statistics, 2001. CA: a Cancer Journal for Clinicians. 51, 15–36 (2009).
Akhdar, H. et al. Involvement of Nrf2 activation in resistance to 5-fluorouracil in human colon cancer HT-29 cells. European Journal of Cancer. 45, 2219–2227 (2009).
Fadzelly, A. M. et al. Effects of Strobilanthes crispus tea aqueous ethanol extracts on glucose and lipid profile in normal and streptozotocin-induced hyperglycemic rats. Plant Foods for Human Nutrition. 61, 6–11 (2006).
Liza, M. et al. Supercritical carbon dioxide ethanol extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). Food and Bioproducts Processing. 88, 319–326 (2010).
Qader, S. W. et al. Antioxidant, total phenolic content and cytotoxicity evaluation of selected Malaysian plants. Molecules. 16, 3433–3443 (2011).
Nurshaira, C. K. Antibacterial Activity of Methanolic Crude Ethanol extracts from Selected Plant Against Bacillus cereus. Selected Articles from the 7th National Genetics Congress. 2, 175–183 (2007).
Al-Henhena, N. et al. Histological study of wound healing potential by ethanol leaf ethanol extract of Strobilanthes crispus in rats. Journal of Medicinal Plants Research. 5, 3660–6 (2011).
Bakar, M. F. A. et al. Antiproliferative properties and antioxidant activity of various types of Strobilanthes crispus tea. International Journal of Cancer Research. 2, 152–158 (2006).
Yaacob, N. S. et al. Anticancer activity of a sub-fraction of dichloromethane ethanol extract of Strobilanthes crispus on human breast and prostate cancer cells in vitro. BMC Complementary and Alternative Medicine. 10, 42 (2014).
Wasman, S. et al. Gastric protection ability of some medicinal Malaysian plants against ethanol induction model in sprague dawley rats. Jurnal Teknologi. 57, 199–209 (2012).
Mahmood, A. et al. Evaluation of gastroprotective effects of Strobianthes crispus leaf ethanol extract on ethanol-induced gastric mucosal injury in rats. Scientific Research and Essays. 6, 2306–2314 (2011).
Marotta, F. et al. Chemopreventive effect of a probiotic preparation on the development of preneoplastic and neoplastic colonic lesions: an experimental study. Hepato-gastroenterology. 50, 1914 (2003).
Lijinsky, W. et al. Organ-specific carcinogenesis in rats by methyl-and ethylazoxyalkanes. Cancer research. 45, 76–79 (1985).
Robertis, M. et al. The AOM/DSS murine model for the study of colon carcinogenesis: From pathways to diagnosis and therapy studies. Journal of carcinogenesis. 10, 9 (2011).
Shwter, A. N. et al. Chemoprevention of colonic aberrant crypt foci by Gynura procumbens in rats. Journal of Ethnopharmacology. 151, 1194–1201 (2014).
Garcia, R. L. et al. Analysis of proliferative grade using anti-PCNA/cyclin monoclonal antibodies in fixed, embedded tissues. Comparison with flow cytometric analysis. The American journal of pathology. 134, 733 (1989).
Zhang, W. et al. Roles of sequential ubiquitination of PCNA in DNA-damage tolerance. FEBS Letters. 585, 2786–2794 (2011).
Lee, H.-N. et al. The Azoxymethane Plus Dextran Sulfate Sodium-Induced Mouse Colon Cancer Model for the Study of Dietary Chemoprevention of Inflammation-Associated Carcinogenesis. Cancer Prevention, 10.1007/978-1-4614-9227-6_7, 155–172 (2014).
Visalli, G. et al. Bergamot Juice Ethanol extract Inhibits Proliferation by Inducing Apoptosis in Human Colon Cancer Cells. Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents). 14, 1402–1413 (2014).
Rus, A. et al. Is endothelial-nitric-oxide-synthase-derived nitric oxide involved in cardiac hypoxia/reoxygenation-related damage? Journal of biosciences. 36, 69–78 (2011).
Samal, P. K. Antioxidant activity of Strobilanthes asperrimus in albino rats. Asian J. Pharm. Res. 3, 71–74 (2013).
Liza, M. et al. Supercritical carbon dioxide extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). Food and Bioproducts Processing 88, 319–326 (2010).
Nurraihana, H. & Norfarizan-Hanoon, N. Phytochemistry, pharmacology and toxicology properties of Strobilanthes crispus. International Food Research Journal 20, 2045–2056 (2013).
Ohishi, T. et al. Synergistic effects of (-)-epigallocatechin gallate with sulindac against colon carcinogenesis of rats treated with azoxymethane. Cancer letters. 177, 49–56 (2002).
Tomimatsu, T. & Horie, T. Enhanced glucose absorption in the rat small intestine following repeated doses of 5-fluorouracil. Chemico-Biological Interactions. 155, 129–139 (2005).
El-Seedi, H. R. Antimicrobial arylcoumarins from Asphodelu smicrocarpus. Journal of Natural Products. 70, 118–120 (2007).
Boldootar, D. et al. Genotoxicity and antioxidant of flavonoid glycoside from the Mongolian medicinal plant Leptopyrum fumarioides. Journal of Ethnopharmacology. 155, 559–606 (2014).
Ikram et al. Antioxidant capacity and total phenolic content of Malaysian underutilized fruits. Journal of Food Composition and Analysis. 22, 388–393 (2009).
Benzie, I. F. & Strain, J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical biochemistry. 239, 70–76 (1996).
Saha, K. et al. Evaluation of antioxidant and nitric oxide inhibitory activities of selected Malaysian medicinal plants. Journal of Ethnopharmacology. 92, 263–267 (2004).
Yen, G.-C. et al. Nitric oxide-scavenging and antioxidant effects of Uraria crinita root. Food Chemistry. 74, 471–478 (2001).
Gan, R. Y., F. L., Kuang, L. & Li, H. B. Antioxidant activity and total phenolic content of medicinal plants associated with prevention and treatment of cardiovascular and cerebrovascular diseases. J. Med. Plants Res. 4, 2438–2444 (2010).
Sathuvan, M. et al. In Vitro Antioxidant and Anticancer potential of Bark of Costus pictus D.DON. Asian Pacific Journal of Tropical Biomedicine. 2, S741–S749 (2012).
OECD, D. OECD Guideline for testing of chemicals. Proposal for revised introduction to the oecd guidelines for testing of chemicals. http://dx.doi.org/10.1787/9789264070684-en. (2005).
Takahashi, M. et al. Suppressive effect of an inducible nitric oxide inhibitor, ONO-1714, on AOM-induced rat colon carcinogenesis. Nitric Oxide. 14, 130–136 (2006).
Bird, R. P. Role of aberrant crypt foci in understanding the pathogenesis of colon cancer. Cancer letters. 93, 55–71 (1995).
Nirmala, P. & Ramanathan, M. Effect of kaempferol on lipid peroxidation and antioxidant status in 1,2-dimethyl hydrazine induced colorectal carcinoma in rats. European Journal of Pharmacology. 654, 75–79 (2011).
Busserolles, J. et al. Substituting honey for refined carbohydrates protects rats from hypertriglyceridemic and prooxidative effects of fructose. The Journal of nutrition. 132, 3379–3382 (2002).
Acknowledgements
This research was supported by a University of Malaya Grant (PS246-2010B), UMRG (RG373/11HTM) and University Malaya High Impact Research Grant (UM/MOHE/HIR Grant (F000009-21001) from Ministry of High Education.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: N.A.-H., M.A.A., R.P.Y.Y. and P.H. Performed the experiments: N.A.-H., N.S.A., M.A.A. and P.H. Analyzed the data: N.A.-H., R.P.Y.Y., M.A.A. and H.E.-S. Contributed reagents/materials/analysis tools: N.A.-H., R.P.Y.Y., H.M.A., M.A.A. and E.R. Wrote the paper: N.A.-H., R.P.Y.Y., M.A.A., S.A.M.K. and H.E.-S.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Al-Henhena, N., Khalifa, S., Ying, R. et al. Chemopreventive effects of Strobilanthes crispus leaf extract on azoxymethane-induced aberrant crypt foci in rat colon. Sci Rep 5, 13312 (2015). https://doi.org/10.1038/srep13312
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep13312
This article is cited by
-
Mangiferin (mango) attenuates AOM-induced colorectal cancer in rat’s colon by augmentation of apoptotic proteins and antioxidant mechanisms
Scientific Reports (2024)
-
Boric Acid (Boron) Attenuates AOM-Induced Colorectal Cancer in Rats by Augmentation of Apoptotic and Antioxidant Mechanisms
Biological Trace Element Research (2023)
-
Streptomyces malaysiense sp. nov.: A novel Malaysian mangrove soil actinobacterium with antioxidative activity and cytotoxic potential against human cancer cell lines
Scientific Reports (2016)
-
Evaluation of chemopreventive potential of Strobilanthes crispus against colon cancer formation in vitro and in vivo
BMC Complementary and Alternative Medicine (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.