Abstract
In this paper volatile organic compounds (VOCs) from Tuber magnatum fruiting bodies were analyzed using a PTR-TOF-MS instrument. The aim was to characterize the VOC's profile of the fruiting bodies and identify if any VOCs were specific to a season and geographical areas. Multiple factorial analysis (MFA) was carried out on the signals obtained by MS. Experiments using ITS region sequencing proved that the T. magnatum life cycle includes the formation of fruiting bodies at two different times of the year. The VOCs profiles diverge when different seasonal and geographical productions are considered. Using PTR-TOF-MS, compounds present at levels as low pptv were detected. This made it possible to determine both the origin of fruiting bodies (Alba and San Miniato) and the two biological phases of fruiting bodies formation in San Miniato truffles.
Similar content being viewed by others
Introduction
Fungi included in the genus Tuber spp. are ascomycetes belonging to Pezizales, a large group of ectomycorrhizal fungi growing in symbiosis with the roots of several vascular plant species belonging to both Angiosperms and Gymnosperms. The ascoma of this fungus is a hypogeous complex apothecium, commonly known as a truffle. The Tuber genus is one of the 5 genera which make up the Tuberaceae family (subphylum Pezizomycotina) and it is estimated to comprise at least 180 species worldwide. These mainly occur in the temperate zones of the northern hemisphere, with three main regions of genetic differentiation: Europe, South East Asia and North America. The truffles are valued for two important features: they bring benefits to the forest ecosystems and to the host plant, as a result of their colonization by the mycorrhizae and some of the species are edible and have higher economic value1 than other food crops2.
The truffle life cycle, like that of other symbiotic filamentous fungi, begins with a limited extraradical phase of vegetative growth in which the hyphae proliferate before coming into contact with the roots of the host plant (phase 1–2). Once this contact (phase 3) is established, the symbiotic phase begins, leading to the development of the ectomycorrhiza (phase 4), a new organ which is functionally and morphologically distinct from the two original partners. In the final stage, the mycelium is organised into the fruit body (phase 5–6), the role of which is to produce sexual fructifications to be dispersed in the environment at a later date. Vegetative mycelia then develop from these fructifications, originating a new extraradical phase and completing the truffle life cycle. Fruiting bodies are normally collected during the fall/winter (October-December, phase 5–6). The mycelium may produce a further sexual fructification during the summer (June-August), commonly known in Italy as “Marcia” (same developmental phase but different season). Summer fruiting bodies are not described in literature but are well known by field experts3.
Truffle's fruiting bodies release a mixture of volatile compounds which are likely used to communicate with plants, animals and microorganisms4. Other than their biological function, the mixture of volatile compounds (aroma) emitted from the fruiting bodies determines their economic value. The most valued species on the food market are Tuber magnatum Pico or "white truffle” and the Tuber melanosporum Vittadini, or “black truffle”5. Of the two, the T. magnatum is the more expensive one1. This truffle species has limited geographical distribution. It grows in spontaneous colonies in some regions in Italy (Tuscany, Piedmont, Marche, Umbria), as well as in Istria and several Balkan regions1. The mycorrhizal symbiosis is based on a mutual exchange of resources: the fungus brings limiting nutrients to the relationship in return for organic carbon structures that it gets from the plant. The plants and fungi's symbiotic relationship is essential for the fungi in order to be able to complete their life cycle. Unless they form a symbiotic relationships with plant roots and establish ectomycorrhizas, truffles do not form fruiting bodies4. Bacteria are the third component of mycorrhizal associations. They also, as they are loosely or tightly associated with mycorrhizal fungi, are thought to play a role in mycorrhizal function6. Mycorrhizal colonization of the plant roots induces the so-called “mycorrhizosphere effect”, which seems to favour the occurrence of bacteria involved in the mycorrhizal process (mycorrhization helper bacteria, or MHB) and ectomycorrhiza-associated bacteria (EMAB). These bacteria complement the roles of the external mycelium by mobilizing nutrients from minerals7 or through the production of volatile organic compounds8 that could contribute to truffle aroma in association with other Tuber-associated microbes (yeast and other fungi)9.
Truffle aroma is very unique and the complex composition of its volatile compounds has been the object of a number of studies over the past 20 years, which have employed a variety of techniques10 each of which have focused on different goals. For example, one study analysed T. magnatum volatiles11 and compared them to those present in other truffle species12. In another, an attempt was made to link the VOCs profiles to the geographical origin of the truffles using samples collected from different regions in Italy5. A more recent study showed how the effect different post-harvest conditions can have on the quantity and quality of VOCs in T. magnatum10.
Analysis of truffle volatiles has mainly been done using gas chromatography-mass spectrometry (GC-MS) of volatiles concentrated using suitable techniques, e.g. dynamic headspace GC-MS and purge-and-trap GC-MS11,13.
In numerous publications, another technique known as "Headspace Solid-phase Microextraction (HS-SPME) coupled with GC-MS" has been employed as a way to better identify volatile compounds in several truffles species as shown in several papers14,15,16,17.
The benefits of using GC-MS based methods to detect volatiles have also been compared and contrasted with other analytical systems. One such system is the "Proton Transfer Reaction—Mass Spectrometer (PTR-MS)", a soft chemical ionization procedure that allows on-line measurements of trace components with concentrations as low as a few pptv (parts per trillion by volume). This technique constitutes a valid alternative to GC-based methods, as it makes possible fast, accurate and direct measurement of volatile organic compounds, in this case in T. magnatum18. Significant improvements have been made in PTR-MS technology based on time-of-flight (TOF-MS)19. PTR-TOF-MS instruments can generate entire mass spectra (snapshots) of complex trace gas mixtures in short response times with high mass resolution and with virtually no upper mass limit19. This technique is used in the field of food science and technology to obtain a rapid, direct and non-invasive readings of volatiles. For example, it has proved useful to differentiate between specialty coffees20, to identify markers of origin in various protected designation of origin (PDO) of Netherlands cheeses21 and to evaluate the influence of sugar composition on flavor release in a strawberry flavored cereal bar system22. Recently this technique has also been used successfully to rapidly determine the volatile compounds present in the fruits of Capsicum spp23.
Taking full advantage of these recent innovations in analytical systems, this paper presents the results of a study that compared the volatile organic compounds in T. magnatum Pico fruiting bodies gathered from natural colonies in the Tuscany region with the fruiting bodies from the Piedmont region (the two most economically valuable of the Italian sub-species). In addition, fruiting bodies from two different seasons, were both collected in Tuscany: summer fruiting bodies were compared with fruiting bodies picked during the winter. All specimens were analyzed using PTR-MS.
Results
Chemical composition of the Aroma
Analysis of volatiles from T. magnatum fruiting bodies has led to the identification of 111 compounds (Table 1). The quantification of each compound is presented in Fig. 1. The 83% of the identified compounds were detected in the m/z range from 50 to 180 whereas the most abundant compounds were detected below m/z 50. The compounds identified are listed in Table 1 and classified on the base of their m/z ratio (both theoretical and measured), chemical name, molecular formula and the related literature. Citations were divided into four columns: one referring to previous PTR-MS data, the other three were respectively assigned to T. magnatum, other Tuber sp. and other fungi and bacteria. The profile of VOCs were similar for the three populations of samples with a general decrease in signal intensity due to the rise of the m/z ratio (Fig. 1).
To better describe the relationship between specific role of VOCs and the geographic origin as well as the harvesting seasons, the compounds were divided into 6 different chemical classes (Hydrocarbons, H; Aromatic hydrocarbons, AH; Phenols, P; Sulphur compounds, S; ; Terpenes, T; ,Others compounds, O), which could be treated as six groups of variables.
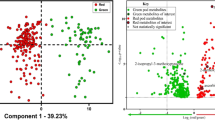
MFA revealed the canonical relationship between the data obtained from PTR-TOF-MS fingerprints for the samples originating from the two geographical regions and different harvest times. The coordinates of the six group of variables were displayed and used to create a map of the group of compounds (Fig. 2a, Groups representation). The coordinates were calculated using the first two dimensions of the MFA (Dim 1 and 2 on the diagram), which included 100% of the total inertia (the inertia is the total variance of a dataset i.e. the trace of the correlation matrix). As to the contribution of individual groups of variables, a general equilibrium can be observed for axis 1 in a range that varies from 14.22% (Phenols) up to 18.12% (Others compounds) (Table 2). Different conclusions can be drawn regarding the contribution of each group of variables to axis 2. The contribution of sulfur compounds appears as to be the most statistically significant (21.46%). The contribution of aromatic hydrocarbons on the other hand, was low (6.14%): this is the least useful group of variables for the purpose of discriminating among the samples on the axis 2 of the MFA. The data provided by MFA was also subjected to further processing to determine how much each class of compounds (Fig. 2b) was useful for discriminating between samples. The same was done to determine the contribution of the individual compounds (Fig. 2c).
Multiple factor analysis (MFA) of transformed PTR-TOF-MS data for the 111 compounds identified in the 3 distinct populations of Tuber magnatum.
(A) Representation of the groups of variables. Key: Darkgreen “HC", hydrocarbons; red “Aromatic HC”, aromatic hydrocarbons; golden “Sulphur”, sulphur compounds; midnightblue “Others”, others compounds; limegreen “Terpenes”, terpenes; lightskyblue “Phenols”, phenols, respectively; (B) Representation of the projection of variables onto the plane defined by the two first principal components of MFA. The coordinates of each variable are the correlation coefficients with the two first principal components. Key: SM = San Miniato Winter; A = Alba; SM* = San Miniato Summer, respectively; (C) Vector representation of the contribution of each compound to the distinction of the populations of samples. The coordinates of each variable are the correlation coefficients with the two first principal components. Key: Darkgreen “H”, 1–11 hydrocarbons; red “AH” 1–6, aromatic hydrocarbons; goldenrod “S”, 1–30 sulphur compounds; midnightblue “O”, 1–35 others compounds; limegreen “T”, 1–24 terpenes; lightskyblue “P”, 1–5 phenols, respectively.
These data can be read in the same way as data in a normal PCA: the individual chemical classes correspond to the correlation coefficients between these variables and the factors. Compounds that significantly correlate (α = 0.05) to the two first dimensions are summarized in Table 3. Of these, 9 compounds were selected for their statistical relevance in the first dimension of MFA, while 8 compounds were chosen for being statistically relevant in the second dimension of MFA. Each truffles has six partial points corresponding to the chemical classes (Table 1). The length and the direction of the vectors are directly correlated to their significance within each population. Factorial axis 1 (57.64 % of the variance) clearly separated the truffles according to the harvest season, whereas the second axis (comprising the 42.36% of the variance) separated the winter samples according to geographic origin (Fig. 2b, individual factor map).
The third plot (“Correlation circle”) represents the normalized vectors of all quantitative variables. The angle between two arrows represents the correlation of the respective variables. There is no linear dependence if the angle is 90 degrees. In Fig. 2c, compounds belonging to the same class are arranged in a uniform manner in a correlation circle though it is not possible to identify any specific accumulation of compounds belonging to the same class. To further understand the differences and similarities between truffles, we next examined the compounds that were used to construct factor maps. The quantitative data are also depicted as a heat-map (Fig. 3) obtaining two dendrograms, one related to the samples and the other to the chemical structures. Both dendrograms were created independently of the heat map using correlation distance and the Ward method of agglomeration. The Ward method24 has a more statistical basis (Fig. 3). Using this method, the distance between groups is defined as the amount of information lost (or error created) by summarizing the objects into n clusters. The hierarchical clustering provided in the heat map confirmed the clustering obtained through MFA (data not shown). The two phylogenetic trees show that the "Marcia" sample is clearly differentiated from the other two samples (Fig. 3, left side of the diagram), while compounds within the same class cannot be grouped on the basis of their intensity signals as shown by the 4 groups of compounds obtained (Fig. 3, above diagram).
Heat-map based on the quantitative data for the three populations of analyzed samples.
Signals were subdivided into classes based on chemical and biochemical features of the compounds. Darkgreen “H”, 1–11 hydrocarbons; red “AH” 1–6, aromatic hydrocarbons; goldenrod “S”, 1–30 sulphur compounds; midnightblue “O”, 1–35 others compounds; limegreen “T”, 1–24 terpenes; lightskyblue “P”, 1–5 phenols, respectively.
The occurrence of a reduced number of quantitatively relevant sulphur compounds can be explained on the base of the results obtained by sequencing the ITS regions of the "Marcia" sample (Fig. 4). The reverse and forward sequencing data of ITS1/ITS4 and ITS5/ITS6 fragments shows as the T. magnatum SM* present a 100% homology with the homologous deposited sequence of T. magnatum.
A phylogenetic tree shows the inferred evolutionary relationship between the SM* sample (obtained from Sanger sequencing) and other truffle species (sequences downloaded from NCBI (http://www.ncbi.nlm.nih.gov/).
Each sequence is listed with by its own name and gene identificator. It can be noted that SM* appears to have the same sequence of the repository sequence of T. magnatum suggesting that both accessions belong to the same species. This tree was generated using phylogeny.fr (www.phylogeny.fr) in “One Click” mode43. The bar represents 0.2 nucleotide substitutions per position. Saccharomyces cerevisiae (gi 31746957)was used as outlier sample.
Discussion
The first group to use PTR-MS to study T. magnatum aroma was Aprea’s group18. Following this initial publication, the use of PTR-MS gained acceptance as a reliable and rapid way to quantitatively analyze volatiles.
The VOCs profiles of Tuber spp. are highly complex and are far from being fully described. Many of the molecules identified in our experiments had previously been found in truffles collected in various european areas. However, 26% (29 out of 111) of them are being reported for the first time as volatiles produced in Tuber spp. (Table 1). Of these, 19 have been identified as BVOC (Biogenic Volatile Organic Compounds) in various other organisms. An additional 7, to our knowledge, have never been associated with any organisms, but have been found (by means of PTR-TOF-MS analysis) in food matrices20,21,25,26. The remaining 3 previously unidentified compounds in Tuber spp. (cyclopentenyl carbenium, 2-ethynylthiophene and 2,5-dimethylthiophene), have never before been cited as BVOC. Although knowledge of which VOCs are present in a given species is useful for identification purposes, it is difficult to distinguish between them based on single compounds. For this reason MFA analysis was used to grouping the VOCs into six most broad categories.
The resulting groups were analyzed to determine their usefulness as markers in distinguishing between samples (Table 3). With regard to axis 1, which is mostly related to the season, we see a general equilibrium in the contribution of each class whereas a different trend is visible along the axis 2, which is mostly related to the geographical origin of the samples. For axis 2, the contribution of each class of compounds is different, ranging from the 6.14% for aromatic hydrocarbons compounds (minimum value) to 21.46% for sulphur compounds (maximum value). This result may be related to the specific volatile profile of T. magnatum in which the sulphur compounds are considered by general consensus to be the main contributors to its unique flavor and are possibly the reason behind the price differences among truffles harvested from different regions.
Of the 17 compounds which best correlated with the two dimensions of the graphical display, 8 belongs to the group “Others”. This was the group that most strongly contributed to the first dimension of MFA (Fig. 2a–c) and includes 4 of the compounds that individually contributed the most to the first dimension (Pearson correlation >0.99, Table 3). Among these are aldehydic and ketonic compounds, which are not clearly assignable to any well-established metabolic pathway. One of these compounds was 2-methyl-1-propanol (previously reported in other species of truffle12,27) and another was acetaldehyde, whose presence in T. magnatum had previously been documented18. A quick examination of the second axis (axis 2) of MFA (42.36% of total variance) illustrates how the class “Others” contains the majority of the most representative compounds in this case too. One of these compounds was: i.e. the 2-methylbutanal, which had previously been found in T. magnatum12. A different scenario unfolds when considering the sulphur compounds which are often considered responsible for the distinctive aroma of the T. magnatum fruiting bodies. In this case only two compounds (one for each dimension of multifactorial analysis, Table 3) are significantly correlated with either of the two dimensions obtained by MFA analysis: diethanol sulfide and methylsulfanyl cyclopentane. Diethanol sulfide (negatively correlated to the first dimension, −1.000 of Pearson coefficient) and has previously been identified in T. magnatum as a compound able to distinguish between samples originating from different locations5. Methylsulfanyl cyclopentane negatively correlated to the second dimension (−1.000 of Pearson coefficient) and has previously been found in Tuber borchii27.
A study from 2008 had identified a series of characteristic compounds able to distinguish among samples of white truffle from seven Italian geographic areas5. In our experiments 6 of those signals (S3, S16, S22, S23, S27, T15, see Table 1 for more detail) were detected and we observed that, for these compounds there was a negative correlation of their Pearson coefficient related in the first dimension of MFA, whereas they generally appeared to correlate positively to second axis (S3, −0.535, 0.845; S16, −0.653, −0.758; S22, −0.788, 0.615; S23, −1.000, 0.027; S27, −0.136, 0.991; T15, −0.490, 0.872 for axis 1 and axis 2 respectively). These compounds did not distinguish between samples originating from our two different geographical locations though one of the compounds, S23, appeared to be specific to the Axis 1 and could possibly differentiate between summer and winter fruiting bodies.
Gioacchini5 found qualitative differences in sulfur compounds and terpenes among truffles originating from seven Italian areas, leading them to suggest that it might be possible to use intra-specific variation of VOC profiles to determine the area of origin of an unknown samples. Our data support this idea. In a series of detailed papers, Splivallo4,17 produced an in-depth description of truffles volatiles, concluding that since GC/MS instrument are less sensitive than the human nose, there is still room for improvement of the identification of truffle volatiles. By focusing their attention on T. uncinatum, Splivallo and coworkers found that C8-VOCs are major players in intraspecific aroma variability and they proved that 1-octen-3-ol does not occur exclusively in fully mature truffles. They also support the idea put forward by Gioacchini5 that if isoprenoids like cedrol and himachalene are to be used as marker for T. magnatum originating from Piedmont and Umbria, intra-specific genetic variability should be factored into the equation.
The aromatic profile of the summer sample “Marcia” (San Miniato Summer or SM*) is strikingly different from that of the other samples, San Miniato and Alba, harvested during the cold season. The data shows how SM*, harvested in summer, produces VOCs which distinguish from both A and SM, both harvested during the period of November-December. The cluster tree (Fig. 3, left side of the diagram) confirmed that the differences detected among the populations of samples are more significant when the fruiting body formation period is considered rather than the geographical origin. The second cluster tree (Fig. 3, above diagram) on the other hand, showed the presence of 4 groups of compounds represented by different signals belonging to different classes and assembled according to their intensity.
To our knowledge, these results include the first set of data published on a "Marcia" fruiting body belonging to the T. magnatum species and reinforce the hypothesis that this species has two distinct biological phases for the production of the fruiting bodies. The fruiting bodies from the different phases present markedly different aromatic profiles.
It has been suggested that this kind of data might be useful for molecular barcoding in fungi because, using it, there is a good chance of successful identification of a very broad range of fungi28. The most clearly defined barcode gap would be between inter- and intraspecific variation. DNA barcoding is the use of a short gene sequence from a standardized region of the genome that can be used to help discover new species, as well as to characterize and distinguish between known species and assign unidentified individuals to species29. Results from the analysis of ITS5/ITS6 confirmed that the SM* sample belonged to the T. magnatum species, as shown in Fig. 4. Consequently, the differences observed between the summer and winter samples might be attributable to the environmental conditions, which vary considerably with the season (summer vs fall-winter) during the growth of fruiting bodies, which logically give rise to altered VOC's production.
In conclusion, although the greatest step forward on truffles volatiles was accomplished with the introduction of mass spectrometry, however the most recent progression is the birth of PTR-TOF-MS technology. Using this technique we were able to detect compounds at extremely low levels; the 111 compounds listed in this paper represent the higher number of VOCs reported in T. magnatum fruiting bodies, even though more work will be needed before a comprehensive picture is available.
Besides, the VOC analysis of the three different fruiting bodies made it possible not only to record the difference between the fruiting bodies of Alba and San Miniato, but also to distinguish between summer ("Marcia") and fall/winter production. VOC analysis proves that the "Marcia" stage of fruiting bodies, although analogous to the fruiting bodies collected during fall/winter develops specific metabolic characteristics as a result of the different season. To the best of author’s knowledge, this has been reported for the first time. It seems that for each season, the resulting truffles are, at least metabolically, quite distinct. On the other hand the fruiting bodies that grow during the summer are not deep in the soil as the winter ones; therefore they grow much faster and rapidly they rot. Consequently their entire metabolism, including the formation of VOCs, is different when compared with the winter fruiting bodies.
Finally, adequate description of truffle aroma requires the use of sophisticated tools due to its complexity, including an accurate in-depth statistical analysis of the data as was done in several figures of this paper. The limited data already collected by various scientists should be assembled with other new results as they become available. Such a methodology will allow for significant advances in knowledge of truffle VOC biology through the implementation of statistical analysis.
Methods
Fruiting bodies and PTR-TOF-MS analysis
VOCs emitted from samples were collected over three harvest seasons (2011–2013), from Piedmont (Alba, A) during the winter and from Tuscany (San Miniato) during winter (SM) and summer (SM*), “Marcia”. For each sample, three carpophores of about 10–15 g were collected and stored at 4 °C in glass vials and analysed within 24 h. Volatiles were analysed with a PTR-TOF-MS 8000 (IoniconAnalytik GmbH, Innsbruck, Austria) using H3O+ as reagent ion for the proton transfer reaction. The reaction takes place between H3O+ions and all the biogenic VOCs having a proton affinity higher than that of water (165.2 kcal mol−1). The separation of the resulting single ions depends on their mass to charge (m/z) ratio. The reaction takes place in a reaction chamber (Drift tube) under controlled conditions of applied voltage (set at 600 V), temperature (at 110 °C) and pressure (at 2.25 mbar). Compounds such as 1,4 dichlorobenzene (m/z = 146.976) and 1,2,3 trichlorobenzene (m/z = 180.937) were continuously used, together with other known low mass ions, for a precise conversion of ‘‘time-of-flight’’ into ‘‘mass-to-charge’’ ratio (m/z) in order to assign the exact mass scale and the sum formula of all ions during VOC analysis23,30. For each sample, about 10 grams of material were placed in a glass jar and covered with a special lid that allowed Teflon connection to a zero-air generator (inlet) and to the PTR-TOF-MS system (outlet). The head space was then measured by direct injection into the PTR-TOF-MS drift tube inlet for 150 seconds, after 10 minutes of exposure. Preliminary measurements on an empty jars were run before every experiment and used for background subtraction. All mass spectra up to m/z = 250 were simultaneously detected and recorded with 1 s as the integration time. Internal calibration was based on m/z = 21.0202 (H318O+), m/z = 29.9974 (NO+) and m/z = 59.0491 (C3H6O+). For a more detailed explanation see references23,30. Data obtained by PTR-TOF analysis were processed as described in reference31. Briefly, raw spectra data (count rate of the analytes recorded were expressed in number of counts per second, cps) were acquired with TOFDaq software (TOFwerk AG, Switzerland) using a dead time of 20 ns for the Poisson correction and peak extraction followed the methodology described in reference31, employing a modified Gaussian peak shape. For peak quantification the resulting data were corrected according to the duty cycle and the signals were normalized to the primary ion signal (cps to ncps) as described in reference32. For each sample, the average data resulting from 20 consecutive seconds of measurement were extracted 3 minutes after the beginning of the experiment. All spectra were corrected for count losses due to the detector dead time, applying Poisson correction in the DAQ settings of TOFDAQ configuration options. External calibration was automatically done by the acquisition program and it achieved a mass accuracy of 0.001Th for the considered mass range, which was in most cases sufficient for formula identification.
Statistical analysis
To identify relationships among the samples (Alba, A; San Miniato winter, SM; San Miniato summer SM*) based on data obtained from PTR-TOF-MS, multiple factorial analyses (MFA) was used33. MFA was performed in two steps. Firstly, a principal component analysis (PCA) was computed on each data set, which was then “normalized” by dividing all its elements, by the square root of the first eigenvalue obtained from of its PCA. Then, the normalized data sets are merged to form a single matrix and a global PCA is performed on this matrix. The individual data sets are then projected onto the global analysis to analyze communalities and discrepancies. Volatile compounds significantly contributing to MFA dimensions were used to explain differences among truffles (normal law adjustment test on compounds correlation coefficients, α = 0.05). A hierarchical clustering on principal components (HCPC) was performed to confirm the product groups observed graphically34. Heat maps method were used for visualizing complex data sets organized as matrices. A heat map does two things to a matrix. First, it reorders the rows and columns so that rows (and columns) with similar profiles are closer to one another, rendering them to be more visible to the eye. Second, each entry in the data matrix is displayed as a color, making it possible to view the patterns graphically. The dendrograms were created using correlation-based distances and the Ward method of agglomeration was used in the present analysis35. All computations were performed with R 3.0.336 language and environment and R packages FactoMineR37 and gplots38 were used.
PCR analysis for species identification
Total genomic DNA was extracted from a sample named as “Marcia” using the CTAB extraction method39,40 with minor modifications. Next, the ITS region was amplified with the ITS5/ITS6 pair of primers41 using a Biorad MyCycler system in a 25 μl of mixture solution containing 100 ng of DNA from fruiting bodies. Amplification was performed using the following protocol to get each sequence. PCR amplification with the pair of primers ITS5/ITS6 was carried using the method described in reference41. Electrophoresis on agarose gel (2 μl of PCR mixture, 2% agarose gel) with ethidium bromide staining confirmed that the PCR products were of the predicted size ITS5/ITS6 (600–650 bp). The amplicons were purified trough Wizard SV Gel and PCR Clean-Up System Kit (Promega) and then sequenced (BMR Genomics, Padova Italy) to get their relative sequences.
The sequences thus obtained were inserted into a multiple sequence alignment program, using the MUSCLE alignment algorithm42. A neighbor-joining tree was constructed based on maximum likelihood (PhyML) using the web resource available on the phylogeny.fr website (http://www.phylogeny.fr), an high performance platform designed to perform phylogenetic analysis based on a multiple alignment43. The phylogenetic tree constructed using this data helped define the species to which the summer fruiting bodies belong.
Additional Information
How to cite this article: Vita, F. et al. Volatile organic compounds in truffle (Tuber magnatum Pico): comparison of samples from different regions of Italy and from different seasons. Sci. Rep. 5, 12629; doi: 10.1038/srep12629 (2015).
Change history
29 April 2016
A correction has been published and is appended to both the HTML and PDF versions of this paper. The error has been fixed in the paper.
References
Mello, A., Murat, C. & Bonfante, P. Truffles: much more than a prized and local fungal delicacy. Fems Microbiol. Lett. 260, 1–8 (2006).
Testa, R., Di Trapani, A. M., Foderà, M., Sgroi, F. & Tudisca, S. Economic evaluation of introduction of poplar as biomass crop in Italy. Renew. Sust. Energ. Rev. 38, 775–780 (2014).
Gori, L. Funghi ipogei della Lucchesia: di altre province italiane e dell'estero: descrizione e illustrazioni di 137 funghi ipogei, di cui 91 provenienti dalla Lucchesia. ( Maria Pacini Fazzi ed., 2005).
Splivallo, R., Ottonello, S., Mello, A. & Karlovsky, P. Truffle volatiles: from chemical ecology to aroma biosynthesis. New Phytol. 189, 688–699 (2011).
Gioacchini, A. M. et al. Geographical traceability of Italian white truffle (Tuber magnatum Pico) by the analysis of volatile organic compounds. Rapid Commun. Mass Spectrom. 22, 3147–3153 (2008).
Bonfante, P. & Anca, I.-A. Plants, mycorrhizal fungi and bacteria: a network of interactions. Annu. Rev. Microbiol. 63, 363–383 (2009).
Frey-Klett, P., Garbaye, J. & Tarkka, M. The mycorrhiza helper bacteria revisited. New Phytol. 176, 22–36 (2007).
Tarkka, M. T. & Piechulla, B. Aromatic weapons: truffles attack plants by the production of volatiles. New Phytol. 175, 381–383 (2007).
Buzzini, P. et al. Production of volatile organic compounds (VOCs) by yeasts isolated from the ascocarps of black (Tuber melanosporum Vitt.) and white (Tuber magnatum Pico) truffles. Arch. Microbiol. 184, 187–193 (2005).
Pennazza, G. et al. Electronic nose and GC–MS analysis of volatile compounds in Tuber magnatum Pico: Evaluation of different storage conditions. Food Chem. 136, 668–674 (2013).
Bellesia, F., Pinetti, A., Bianchi, A. & Tirillini, B. Volatile compounds of the white truffle (Tuber magnatum Pico) from middle Italy. Flavour Frag. J 11, 239–243 (1996).
Mauriello, G., Marino, R., D'Auria, M., Cerone, G. & Rana, G. L. Determination of volatile organic compounds from truffles via SPME-GC-MS. J. Chromatogr. Sci. 42, 299–305 (2004).
Bellesia, F., Pinetti, A., Tirillini, B. & Bianchi, A. Temperature‐dependent evolution of volatile organic compounds in Tuber borchii from Italy. Flavour Fragr. J 16, 1–6 (2001).
March, R. E., Richards, D. S. & Ryan, R. W. Volatile compounds from six species of truffle–head-space analysis and vapor analysis at high mass resolution. Int. J. Mass Spectrom. 249, 60–67 (2006).
Dıaz, P., Ibáñez, E., Senorans, F. & Reglero, G. Truffle aroma characterization by headspace solid-phase microextraction. J. Chromatogr. A 1017, 207–214 (2003).
Culleré, L. et al. Characterisation of aroma active compounds in black truffles (Tuber melanosporum) and summer truffles (Tuber aestivum) by gas chromatography-olfactometry. Food Chem. 122, 300–306 (2010).
Splivallo, R. et al. Intraspecific genotypic variability determines concentrations of key truffle volatiles. New Phytol. 194, 823–835 (2012).
Aprea, E. et al. Rapid white truffle headspace analysis by proton transfer reaction mass spectrometry and comparison with solid‐phase microextraction coupled with gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 21, 2564–2572 (2007).
Jordan, A. et al. A high resolution and high sensitivity proton-transfer-reaction time-of-flight mass spectrometer (PTR-TOF-MS). Int. J. Mass Spectrom. 286, 122–128 (2009).
Özdestan, Ö. et al. Dıfferentıatıon of specıalty coffees by proton transfer reactıon-mass spectrometry. Food Res. Int. 53, 433–439 (2013).
Galle, S. A. et al. Typicality and geographical origin markers of protected origin cheese from the Netherlands revealed by PTR-MS. J. Agric. Food Chem. 59, 2554–2563 (2011).
Heenan, S. et al. PTR-TOF-MS monitoring of in vitro and in vivo flavour release in cereal bars with varying sugar composition. Food Chem. 131, 477–484 (2012).
Taiti, C. et al. Class‐modeling approach to PTR‐TOFMS data: a peppers case study. J. Sci. Food Agric. 10.1002/jsfa.6761 (2014).
Ward Jr, J. H. Hierarchical grouping to optimize an objective function. J. Amer. Statist. Assoc. 58, 236–244 (1963).
Ruuskanen, T. et al. Eddy covariance VOC emission and deposition fluxes above grassland using PTR-TOF. Atmos. Chem. Phys. 11, 611–625 (2011).
Sánchez Del Pulgar, J. et al. Effect of the pig rearing system on the final volatile profile of Iberian dry-cured ham as detected by PTR-ToF-MS. Meat Sci. 93, 420–428 (2013).
Splivallo, R., Bossi, S., Maffei, M. & Bonfante, P. Discrimination of truffle fruiting body versus mycelial aromas by stir bar sorptive extraction. Phytochemistry 68, 2584–2598 (2007).
Schoch, C. L. et al. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 109, 6241–6246 (2012).
Newmaster, S., Fazekas, A. & Ragupathy, S. DNA barcoding in land plants: evaluation of rbcL in a multigene tiered approach. Can. J. Bot. 84, 335–341 (2006).
Brilli, F. et al. Detection of plant volatiles after leaf wounding and darkening by proton transfer reaction “time-of-flight” mass spectrometry (PTR-TOF). PLoS One 6, e20419 (2011).
Cappellin, L. et al. On data analysis in PTR-TOF-MS: From raw spectra to data mining. Sens. Actuators, B 155, 183–190 (2011).
Herbig, J. et al. On-line breath analysis with PTR-TOF. J. Breath Res. 3, 10.1088/1752-7163/3/2/020201 (2009).
Escofier, B. & Pagès, J. Analyses factorielles simples et multiples: objectifs, méthodes et interprétation (Dunod 2008).
Fiches, G., Déléris, I., Saint-Eve, A., Brunerie, P. & Souchon, I. Modifying PTR-MS operating conditions for quantitative headspace analysis of hydro-alcoholic beverages. 2. Brandy characterization and discrimination by PTR-MS. Int. J. Mass Spectrom. 360, 15–23 (2014).
Key, M. A tutorial in displaying mass spectrometry-based proteomic data using heat maps. BMC Bioinformatics 13, 10.1186/1471-2105-13-S16-S10 (2012).
Team, R. C. R : A language and environment for statistical computing. (2014). at http://www.R-project.org/ Date of access: 02/01/2015
Husson, F., Josse, J., Le, S. & Mazet, J. FactoMineR: multivariate exploratory data analysis and data mining with R. R package version 1, 102–123 (2013).
Warnes, G. R. et al. "gplots: Various R programming tools for plotting data. " R package version, 2(1) (2013). at http://CRAN.R-project.org/package=gplots Date of access: 02/01/2015.
Doyle, J. & Doyle, J. CTAB DNA extraction in plants. Phytochem Bull. 19, 11–15 (1987).
Richards, E., Reichardt, M. & Rogers, S. Preparation of genomic DNA from plant tissue. Curr. Protoc. Mol. Biol. 27, 10.1002/0471142727.mb0203s27 (1994).
Bertini, L. et al. A new pair of primers designed for amplification of the ITS region in Tuber species. FEMS Microbiol. Lett. 173, 239–245 (1999).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Dereeper, A. et al. Phylogeny. fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res. 36, W465–W469 (2008).
Du, X., Song, M. & Rouseff, R. Identification of new strawberry sulfur volatiles and changes during maturation. J. Agric. Food Chem. 59, 1293–1300 (2011).
Schäfer, H., Myronova, N. & Boden, R. Microbial degradation of dimethylsulphide and related C1-sulphur compounds: organisms and pathways controlling fluxes of sulphur in the biosphere. J. Exp. Bot. 61, 315–334 (2010).
Stotzky, G., Schenck, S. & Papavizas, G. C. Volatile organic compounds and microorganisms. Crit. Rev. Microbiol. 4, 333–382 (1976).
Bunge, M. et al. On-line monitoring of microbial volatile metabolites by proton transfer reaction-mass spectrometry. Appl. Environ. Microbiol. 74, 2179–2186 (2008).
Chinivasagam, H., Bremner, H., Wood, A. & Nottingham, S. Volatile components associated with bacterial spoilage of tropical prawns. Int. J. Food Microbiol. 42, 45–55 (1998).
Falasconi, M. et al. Study of white truffle aging with SPME-GC-MS and the Pico2-electronic nose. Sens. Actuators, B 106, 88–94 (2005).
Hung, R., Lee, S. & Bennett, J. W. Arabidopsis thaliana as a model system for testing the effect of Trichoderma volatile organic compounds. Fungal Ecol. 6, 19–26 (2013).
Schulz, S. & Dickschat, J. S. Bacterial volatiles: the smell of small organisms. Nat. Prod. Rep. 24, 814–842 (2007).
Strous, M. et al. Missing lithotroph identified as new planctomycete. Nature 400, 446–449 (1999).
Fuerst, J. A. Intracellular compartmentation in planctomycetes. Annu. Rev. Microbiol. 59, 299–328 (2005).
Pillon, N. J. & Soulage, C. O. Lipid peroxidation by-products and the metabolic syndrome. In: Lipid Peroxidation (ed. Catala, A. ) Ch.19, 409–436 (INTEC, 2012).
Ercolini, D., Russo, F., Nasi, A., Ferranti, P. & Villani, F. Mesophilic and psychrotrophic bacteria from meat and their spoilage potential in vitro and in beef. Appl. Environ. Microbiol. 75, 1990–2001 (2009).
Schöller, C. E., Gürtler, H., Pedersen, R., Molin, S. & Wilkins, K. Volatile metabolites from actinomycetes. J. Agric. Food Chem. 50, 2615–2621 (2002).
Gu, Y.-Q., Mo, M.-H., Zhou, J.-P., Zou, C.-S. & Zhang, K.-Q. Evaluation and identification of potential organic nematicidal volatiles from soil bacteria. Soil Biol. Bioc. 39, 2567–2575 (2007).
Kurita-Ochiai, T., Fukushima, K. & Ochiai, K. Volatile fatty acids, metabolic by-products of periodontopathic bacteria, inhibit lymphocyte proliferation and cytokine production. J. Dent. Res. 74, 1367–1373 (1995).
Wiggins, R., Wilks, M. & Tabaqchali, S. Analysis by gas liquid chromatography of production of volatile fatty acids by anaerobic bacteria grown on solid medium. J. Clin. Pathol. 38, 933–936 (1985).
Tracey, R. P. & Britz, T. J. Freon 11 extraction of volatile metabolites formed by certain lactic acid bacteria. Appl. Environ. Microbiol 55, 1617–1623 (1989).
Balistreri, W. F. & Stocker, J. T. Pediatric hepatology. Vol. 3 (CRC Press 1990).
Tirillini, B., Verdelli, G., Paolocci, F., Ciccioli, P. & Frattoni, M. The volatile organic compounds from the mycelium of Tuber borchii Vitt. Phytochemistry 55, 983–985 (2000).
Gajos, M. & Hilszczańska, D. Research on truffles: Scientific journals analysis. Sci. Res. Essays 8, 1837–1847 (2013).
Bruce, A., Verrall, S., Hackett, C. A. & Wheatley, R. E. Identification of volatile organic compounds (VOCs) from bacteria and yeast causing growth inhibition of sapstain fungi. Holzforschung 58, 193–198 (2004).
Long, G. & Meek, M. N. N-Dimethylformamide: Hazard Characterization And Exposure–Response Analysis. J. Environ. Sci. Health C 19, 161–187 (2001).
Bohlmann, F., Zdero, C. & Gronowitz, S. Thiophene and its derivatives. in The Chemistry of Heterocyclic Compounds Vol. 44 (eds Press, J. B. & Gronowitz, S. ) Part 1, 261–324 (Wiley and sons, New York, 1985).
Blom, D. et al. Production of plant growth modulating volatiles is widespread among rhizosphere bacteria and strongly depends on culture conditions. Environ. Microbiol. 13, 3047–3058 (2011).
Moreira, N., Guedes de Pinho, P., Santos, C. & Vasconcelos, I. Volatile sulphur compounds composition of monovarietal white wines. Food Chem. 123, 1198–1203 (2010).
D'Auria, M., Rana, G. L., Racioppi, R. & Laurita, A. Studies on Volatile Organic Compounds of Tuber borchii and T. asa-foetida. J. Chromatogr. Sci. 50, 775–778 (2012).
Groenhagen, U. et al. Production of bioactive volatiles by different Burkholderia ambifaria strains. J. Chem. Ecol. 39, 892–906 (2013).
Dickschat, J. S., Wagner-Döbler, I. & Schulz, S. The chafer pheromone buibuilactone and ant pyrazines are also produced by marine bacteria. J. Chem. Ecol. 31, 925–947 (2005).
Müller, A. et al. Volatile profiles of fungi–chemotyping of species and ecological functions. Fungal Genet. Biol. 54, 25–33 (2013).
Brattoli, M. et al. Gas Chromatography Analysis with Olfactometric Detection (GC-O) as a Useful Methodology for Chemical Characterization of Odorous Compounds. Sensors 13, 16759–16800 (2013).
Jong, S. & Birmingham, J. Mushrooms as a source of natural flavor and aroma compounds. In: Chang, S. T., Buswell, J. A., Chiu, S. W. (eds.) Mushroom biology and mushroom products. The Chinese University Press, Hong Kong, 345–366 (1993).
Li, Y.-Y., Wang, G., Li, H.-M., Zhong, J.-J. & Tang, Y.-J. Volatile organic compounds from a Tuber melanosporum fermentation system. Food Chem. 135, 2628–2637 (2012).
Jeleń, H., Błaszczyk, L., Chełkowski, J., Rogowicz, K. & Strakowska, J. Formation of 6-n-pentyl-2H-pyran-2-one (6-PAP) and other volatiles by different Trichoderma species. Mycol Progr. 13, 1–12 (2013).
Spadone, J.-C., Matthey-Doret, W. & Blank, I. Formation of methyl (methylthio) methyl disulfide in broccoli (Brassica oleracea (L.) var. italica). Dev. Food Sci. 43, 309–314 (2006).
Acknowledgements
This work was financially supported by the Tuscany Region (VOLATOSCA Project).
Author information
Authors and Affiliations
Contributions
F.V., A.A. and V.L. designed the research. F.V., C.T. and A.P. performed the experiments and analysed the data. F.V., A.A., S.M. and N.B. organized and drafted the paper with all authors contributing to the discussion of the data and to the writing.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Vita, F., Taiti, C., Pompeiano, A. et al. Volatile organic compounds in truffle (Tuber magnatum Pico): comparison of samples from different regions of Italy and from different seasons. Sci Rep 5, 12629 (2015). https://doi.org/10.1038/srep12629
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12629
This article is cited by
-
Sustainable cultivation of the white truffle (Tuber magnatum) requires ecological understanding
Mycorrhiza (2023)
-
Mycochemicals in wild and cultivated mushrooms: nutrition and health
Phytochemistry Reviews (2022)
-
Chemical constituents and their bioactivities from truffle Hysterangium inflatum
Journal of Food Measurement and Characterization (2021)
-
Tuberomics: a molecular profiling for the adaption of edible fungi (Tuber magnatum Pico) to different natural environments
BMC Genomics (2020)
-
Potentials of truffles in nutritional and medicinal applications: a review
Fungal Biology and Biotechnology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.