Abstract
The brown planthopper (BPH), Nilaparvata lugens, sugar transporter gene 6 (Nlst6) is a facilitative glucose/fructose transporter (often called a passive carrier) expressed in midgut that mediates sugar transport from the midgut lumen to hemolymph. The influence of down regulating expression of sugar transporter genes on insect growth, development and fecundity is unknown. Nonetheless, it is reasonable to suspect that transporter-mediated uptake of dietary sugar is essential to the biology of phloem-feeding insects. Based on this reasoning, we posed the hypothesis that silencing, or reducing expression, of a BPH sugar transporter gene would be deleterious to the insects. To test our hypothesis, we examined the effects of Nlst6 knockdown on BPH biology. Reducing expression of Nlst6 led to profound effects on BPHs. It significantly prolonged the pre-oviposition period, shortened the oviposition period, decreased the number of eggs deposited and reduced body weight, compared to controls. Nlst6 knockdown also significantly decreased fat body and ovarian (particularly vitellogenin) protein content as well as vitellogenin gene expression. Experimental BPHs accumulated less fat body glucose compared to controls. We infer that Nlst6 acts in BPH growth and fecundity and has potential as a novel target gene for control of phloem-feeding pest insects.
Similar content being viewed by others
Introduction
Hemipteran phloem feeding insects cause persistent yield reductions in many globally important crop species1. The brown planthopper (BPH), Nilaparvata lugens, is the most serious insect rice pest throughout Asia, with outbreaks increasing in intensity1,2. BPHs damage rice crops by removing sugars, reducing photosynthesis, blocking phloem elements3 and transmitting plant viruses4. Control of BPH infestation has depended on deploying broad-spectrum insecticides, however, resistance to commonly used organophosphorous, carbamate and pyrethroid insecticides is now widespread5,6. The global importance of rice, estimated to supply 20% of the world’s calorific intake, drives research designed to develop new BPH control measures.
Sucrose is the principal carbohydrate in rice phloem and the major energy source for phloem feeders, including the BPH7. As a disaccharide, sucrose cannot be transported directly across the hemipteran gut epithelium. It is hydrolyzed into two monosaccharides, glucose and fructose, in the BPH midgut for transport into hemolymph circulation8. Sugar transporters act in carbohydrate transport in most organisms, from bacteria to mammals and are responsible for mediating the movement of sugar into cells9. The BPH glucose transporter, N. lugens hexose transporter 1 (NlHT1), facilitates the uptake of glucose, but not fructose, in the midgut10. BPHs also express a midgut sugar transporter gene, Nlst6, that encodes a facilitative glucose/fructose transporter11. The aphid, Acyrthosiphon pisum, expresses at least two sugar transporters, Ap-ST3 and Ap-ST4, both of which transport glucose and fructose12,13. In mammals, some glucose transporter (GLUT; e.g., GLUT4) proteins are retained within muscle and adipose cells; insulin signaling leads to increased GLUT trafficking, rapid translocation to cell surfaces and increased glucose uptake. A similar mechanism probably occurs in insects, as well. Transgenic Drosophila expressing human GLUT4 in fat body responded to insulin treatments with increased GLUT4 translocation to the surface of fat body cells14. GLUT physiology goes beyond sugar transport, however, because GLUT4 also transports dehydroascorbic acid and glucosamine15. We presume sugar transporters act in multiple actions in membrane transport and energy acquisition. The genes encoding these transporters may serve as potential targets for RNAi based control of insect pests in transgenic plants. We posed this idea as the hypothesis that silencing these genes will lethally reduce BPH fitness in agroecosystems. Here we report on the outcomes of experiments designed to test our hypothesis.
Results
Dietary dsNlst6 reduced gene expression
The dietary treatments (for 8 days) did not influence survival of the synchronous nymph groups, at approximately 78% for the experimental group on media amended with 0.05 μg/μl Nlst6 dsRNA, 80% for the control group on media amended with dsGFP and 81% for controls maintained on untreated media. Relative Nlst6 transcript levels in brachypterous females were down to about 40%, compared to dsGFP and untreated controls through seven days after adult emergence (Fig. 1).
Dietary dsNlst6 treatments reduced Nlst6 expression.
All values were normalized relative to β-actin transcript levels. The data represent the mean values ± SE of three independent biological replicates. Histogram bars annotated with the same letter are not significantly different between control and dsNlst6 treatments (t-test, p < 0.05).
Dietary dsNlst6 led to reduced body weight
Dietary dsRNA treatments led to decreased female body weights, down by about 31% (3.0 mg) compared to untreated controls and by approximately 22% (2.1 mg) compared to dsGFP-treated controls at day 2 (Fig. 2A). At day 3, body weights of experimental females declined by 33% (3.8 mg) compared to untreated controls and by nearly 29% (3.3 mg) compared to dsGFP-treated controls (Fig. 2B).
Dietary dsNlst6 led to reduced ovarian and fat body protein content
Dietary dsNlst6 led to substantially reduced fat body protein content at day 2, down by 49% (by about 5.4 mg/g) and day 3 by 47% (by about 8.1 mg/g) (Fig. 3A-C). We recorded similar reductions in ovarian protein content at day 2, down by circa 58% (by about 2.5 mg/g) and day 3, reduced by about 46% (by 6.7 mg/g) (Fig. 3B–D).
Effects of feeding-based dsRNA treatments on the protein content of brachypterous female fat bodies (Panels A & C) and ovaries (Panels B & D).
Histogram bars report mean protein content (mg/g, n = 3 independent biological experiments) ± SE Histogram bars annotated with the same letter are not significantly different (t-test, p < 0.05).
Dietary dsNlst6 led to reduced fecundity
The dsNlst6 treatments led to reduced fecundity, expressed as numbers of eggs laid, down about 51%, from 430.8 eggs/female in controls to 212.3 eggs/female in experimental BPH (Fig. 4).
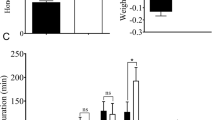
The influence of dietary dsNlst6 on the duration of pre-oviposition and oviposition periods
The pre-oviposition period is the time, in days, from adult emergence to the onset of egg-laying. The dietary dsNlst6 treatments increased this parameter from about 4 to over 6.4 days, compared to untreated controls (up circa 60%) and dsGFP-treated females (up about 45%; Fig. 5A). The oviposition period is the time, in days, of egg-laying, which was substantially reduced, by roughly 31 to 33% from about 33–34 to 22.8 days, compared to controls and dsGFP-treated BPH (Fig. 5B).
Effects of feeding-based dsNlst6 treatments on the duration of preoviposition period (Panel A) and oviposition period (Panel B) of brachypterous females.
Histogram bars report numbers of days ± SE, n = 15 independent biological experiments; bars annotated with the same letter are not significantly different at a p < 0.05.
The influence of dietary dsNlst6 on female longevity
Figure 6 shows the longevity of adult females exposed to dietary dsNlst6 was reduced compared to untreated controls (by around 21%) and dsGFP-treated controls (by about 19%), from about 40–41 days to about 32.4 days.
The influence of dietary dsNlst6 on female fat body glucose content
Fat body glucose content in dsNlst6-treated females was lower, compared to controls and dsGFP-treated controls, decreasing by 29%, from about 28 to 20 μg/g at day 2 (Fig. 7A) and by about 48% (nearly 18 μg/g) at day 3 (Fig. 7B) while no significant differences were found between the control and dsGFP-treated groups.
Effects of feeding-based dsRNA on the glucose content of brachypterous female fat bodies in day 2 (Panel A) and, separately, day 3 (Panel B) adults.
Histogram bars report the mean glucose content (mg/g) ± SE, n = three independent biological experiments; bars annotated with the same letter are not significantly different at a p < 0.05.
Dietary dsNlst6 led to reduced expression of Nlvg mRNA
Figure 8 reports that dietary dsNlst6 led to reduced expression of Nlvg in day 2 (down by 59%) and day 3 (down by 67%) females, compared to controls.
Dietary dsNlst6 treatments decreased Nlvg mRNA expression in day 2 (Panel A) and, separately, day 3 (Panel B) females.
All values were normalized relative to β-actin transcript level. The data represent the mean values ± SE of three replicates. Histogram bars annotated with the same letter are not significantly different (t-test, p < 0.05). Each treatment and control experiment was repeated three times.
Discussion
The data reported in this paper strongly support our hypothesis that Nlst6 silencing lethally reduces BPH fitness in agroecosystems, seen in the following summary. First, dietary dsNlst6 treatments reduced expression of the sugar transporter transcript. Second, the dietary treatments led to decreased female body weight, lowered fat body and ovarian protein contents, diminished fecundity, increased pre-oviposition periods, reduced oviposition periods, shorter female longevity, decreased fat body glucose contents and truncated expression of the BPH vitellogenin gene. Taken together, the uniformly deleterious changes in each of these biological performance parameters amount to a very potent argument that full expression of Nlst6 is absolutely required to produce fully capable and fit adult BPHs. More generally, sugar transporters are essential features of successful feeding and energy metabolism in BPHs and likely all insect species.
Nlst6 is expressed mainly in midgut, along with Nlsts 1, 4, 9, 12, 14 and 1611. Given that midgut expresses seven glucose transporters, the idea that silencing expression of one of the seven genes (by about 60%) could translate into marked deleterious changes at the whole-organism level may seem incongruous. We note that the parameters we measured were reduced to levels significantly, but not catastrophically, lower than control levels. Body weights were down by about 20–30%, ovarian and fat body protein and fecundity down about 50%, longevity down about 25%. It would seem that optimal operation of all seven transporter genes is necessary at the organismal level. We expect that dsRNA constructs that simultaneously silence two or more midgut Nlsts would exert greater impact on BPH biology. We analyzed ovaries and fat body with a focus on reproduction. The reduced protein and sugar contents in these tissues open questions about other tissues, including muscles. Specifically, would protein and sugar contents be similarly reduced on other tissues? Also, do the dietary dsRNA constructs operate solely at the midgut or do they operate systemically to influence sugar transport in individual tissues? While these issues are beyond the scope of this paper, we foresee research to address them in detail.
Body size or weight is, in some animals, a determinant of insect health16. Across butterfly superfamilies Papilionoidea and Hesperioidea, there is a correlation between body size and egg size in which larger eggs tend to develop into larger adults. Within a subset of butterfly species, fecundity is related to adult body size, although this can be influenced by local climate17. Mating with average-sized or heavy black-lyre leafroller females, Cnephasia jactatana, leads to more offspring, which have a higher probability of survival18. However, this broad generalization on the benefits of weight does not apply to all mating systems. For example, the reproductive efficacy of the pyralid moth, Parapediasia teterrella, is positively associated with low to average female weights, but is negatively associated with weights exceeding the average of the population19. Our data show that disruptions in sugar transport led to reduced female weights, which were attended by deleterious outcomes in several parameters of biological performance, particularly fecundity and longevity. In our view, the reduced weight of experimental females is a consequence, rather than cause, of the fitness declines we recorded.
The dietary Nlst6 treatment led to reduced Nlvg mRNA expression levels. Exposure to some insecticides, deltamethrin and triazophos, for examples, similarly induces increases in Nlvg mRNA expression20 and promotes protein synthesis in adult female fat bodies and ovaries21. Alternatively, two other insecticides, chlorantraniliprole and indoxacarb, suppressed Nlvg mRNA expression, decreased fat body and ovarian protein content and attenuated the number of eggs laid22. These insecticides exert their actions through mechanisms unrelated to sugar transporter genes, however, they bring out the idea that the changes in fitness parameters that follow exposure to sub-lethal doses of some insecticides or ingestion of gene-silencing constructs can be quite similar. With respect to deploying dsRNA constructs at the greenhouse or field levels, we surmise that some of the effects of dsRNA can look, in terms of physiology and crop protection, like the effects of insecticides.
Previous investigations discussed relationships between yeast-like symbionts (YLS) and amino acid requirements in BPH23,24,25. Meeting essential amino acid requirements in BPH was associated with the abundance of YLS23. Artificial reduction of the symbiont abundance led to delayed growth and decreased survival rate, adult emergence rate, body weight and fecundity24. Compared to controls, asymbiotic aphids, Acyrthosiphon pisum and BPHs grew more slowly, weighed less and contained less protein25. As to a physiological role, the YLS provide essential amino acids and possibly proteins required for host embryo formation and post-embryonic development26. The symbionts are essential to meeting nutritional amino acid needs for production of a healthy complement of proteins. Although we do not present data on the point, we infer the sugar transporter gene (Nlst6), mediates uptake of sugars necessary for host and symbiont maintenance and reproduction.
Nlst6 silencing led to decreased protein content in the fat bodies and ovaries. The reduced protein contents can disrupt BPH biology, including production of yolk proteins. Vitellogenins are produced in fat bodies and the reduced fat body protein contents may impose a limit on the ability to produce vitellogenin, which can restrict ovarian development and, hence, reproduction.
The dietary dsNlst6 treatments led to decreased female fat body glucose as well as protein. Sugars are also accumulated in fat body cells as glycogen for long-term storage. We did not determine amounts of stored glycogen and the impact of silencing Nlst6 on glycogen stores remains to be determined. Lipid and soluble sugar contents in nymphs and adults developed from nymphs feeding on rice plants treated with triazophos, was significantly increased compared with controls27. Our previous results showed that triazophos significantly stimulated fecundity of BPH females20. We speculated that the YLS populations within individual BPHs are somehow increased following exposure to triazophos at sub-lethal concentrations20. Our working hypothesis holds that dietary Nlst6 led to nutrient deprivation in the symbionts, as well as in BPHs, which entrained a series of conditions. The deprivation led to reduced YLS populations, which led to reduced synthesis of essential amino acids, leading to reduced capability to biosynthesize proteins and, finally, to gaps in the BPH expressed proteome. This partially explains the reduced protein contents in fat bodies and ovaries.
A more complete explanation unfolds with recognition that reducing Nlst6 expression led to restricting the global availability of nutrients within experimental BPHs. Cells respond to nutrient deprivation by inducing autophagy, a catabolic process that breaks down cytosolic components, including proteins and organelles to reallocate their component molecules toward necessary energy-generating metabolism. Once induced, however, autophagy is suppressed by TOR signaling, known in mammals and Drosophila28. Autophagy is thought to protect cells during nutrient deprivation. Hence, we speculate that protein catabolism during the autophagy response is partially responsible for reduced protein contents within fat body and ovarian cells.
BPH and possibly other hemipterans, have 18 sugar transporter genes11. Some of them, Nlst5, 8, 10, 14 and 16, are substantially expressed in most tissues, including head, thorax, abdomen, midgut, ovary, testes, salivary gland, Malpighian tubules and fat body. Seven are expressed in the midgut, where they probably act in moving sugar from the midgut lumen to hemolymph circulation. Nlst5, 8, 10, 11 and 13 are expressed in the fat body, where they may operate in fat body sugar mobilization. Other Nlst genes are expressed during the embryonic stage and facilitate sugar mobilization during embryonic development. Sugar transporters operate throughout the BPH lifecycle. Disruption of these transporters can have far-reaching effects on BPH populations. Recalling the very high mortality of second instar nymphs, which may lead to very efficacious field applications, we propose continued research into nutrient transporters in pest insects, which may yield new target genes for management of phloem-feeding pests through RNAi based transgenic plants.
Materials and Methods
Rice variety and culture
Rice (Oryza sativa L.) variety Ninjing 4 (japonica rice, commonly grown in Jiangu province) was used in all experiments. Seeds were sown outdoors in cement tanks (height 60 cm, width 100 cm and length 200 cm) containing standard rice-growing soil. When seedlings reached the six-leaf stage, they were transplanted into 16 cm diameter plastic pots containing four hills per pot, three plants per hill and used for experiments at the tillering stage.
Insect culture
BPHs were obtained from a laboratory population maintained in a greenhouse under our standard conditions (26 ± 2oC, with 70–80% humidity and a 16L:8D photoperiod at Yangzhou University. The insect colony was originally obtained from the China National Rice Research Institute (Hangzhou, China). Before the experiments started, the colony was allowed to reproduce for two generations in cement tanks (60 × 100 × 200 cm) under natural condition in Yangzhou.
RNAi
We designed gene-specific dsNlst6 primers and amplified a 384-bp (672–1055 bp) Nlst6 cDNA fragment using forward and reverse primers containing the T7 primer sequence at the 5′ ends (Table 1). The amplification program was 35 cycles of 95 °C for 40 s, 58 °C for 40 s and 72 °C for 1 min, with a final extension step of 72 °C for 10 min. The sequence was verified by sequencing (Invitrogen Company, Shanghai, China). We used the GFP gene (ACY56286; was provided by Zhang Chuan-xi, Institute of Insect Sciences, Zhejiang University) as control dsRNA and amplified a 688 bp fragment using primers listed in Table 1. We used the T7 RiboMAX TM Express RNAi System (Promega, Sunnyvale, CA) for dsRNA synthesis, following the Promega instructions. Sense and antisense dsRNAs generated in separate 20 μL total reaction volumes were annealed by mixing both transcription reactions and incubating at 70 °C for 10 min and then cooling to room temperature over 20 min time period. A 2 μL RNase A solution (4 mg/ml) and 2 μL RNase-free DNase (1 u/μL) were added to the reaction tube and incubated in a 37 °C water bath for 30 min. The dsRNA was precipitated by adding 110 μL 95% ethanol and 4.4 μL 3 M sodium acetate (pH5.2), washed with 0.5 mL 70% ethanol and dried at room temperature. The dried product was dissolved in 50 μL nuclease-free water. The purified dsRNAs were quantified by spectroscopy. To deliver dsRNA into BPH, nymphs were reared on an artificial diet29, with some modifications to the rearing protocol, amended with dsRNA. Previous results indicated that dsRNA feeding led to rapid and significant reduction expression levels of BPH genes30. We used glass cylinders (15.0 × 2.5 cm diameter) as feeding chambers, using four concentrations, 0.125 μg/μl, 0.075 μg/μl, 0.05 μg/μl and 0.025 μg/μl. The dsRNA solution (the final concentration, 0.05 μg/μl diet, was determined from the concentrations mentioned just above) was added to the artificial diet (20 μl), held between two layers of stretched Parafilm M membrane that were enclosed at the two open ends of the chamber (the diet capsule). The diet capsule was replaced every second day. The cylinders were covered with a piece of black cotton cloth, but the two ends with the artificial diet were exposed to light. Insects fed on the diets by puncturing the inner Parafilm M membrane of the diet capsule. All insects were transferred into chambers and maintained on artificial diets for one day before initiation of the assays. Twenty 3rd instar individuals were transferred into each chamber and three chambers were used to create three independent biological replicates. The rearing experiments were carried out in a humidified growth cabinet at 26 ± 2 °C, 90% RH and a 16L:8D photoperiod. Mortality was recorded every other day.
The influence of dsRNA on biological performance parameters
We determined the effects of dsNlst6 treatments on several biological performance parameters. We exposed second instar nymphs to the dsRNA construct, which led to over 95% mortality. Ergo, third instar nymphs were transferred to diet capsules containing dsRNA-laced diet; at their fifth (final) instar (8 days), they were collected and a single nymph was transferred into a glass jar (12 cm high x 10 cm ) and reared on tillering rice plants under 26 ± 2 °C and 16L:8D. Eighty adult females were collected separately at days 2 and 3 after emergence and fresh body weight, soluble fat body sugar content, ovary and fat body protein content and Nlst6 expression level were determined. We paired a single newly-emerged adult female with a male. Each pair (♂ × ♀) was maintained in a glass jar (diameter 10 cm, height 12 cm) with rice seedlings under our standard controlled conditions for oviposition. Fifteen copulating pairs were maintained to record duration of the pre-oviposition period, oviposition period, adult female longevity and fecundity for each copulating pair. Rice stems were replaced daily during the pre-oviposition period, at two day intervals during the oviposition period and three day intervals during the female longevity period until the females died. The numbers of eggs laid on each rice stem was recorded under a light microscope. Eggs were scraped from the leaf sheaths and blades using a pin. Fecundity of 15 mated pairs was recorded as the average number of eggs produced by the mated female.
Protein analysis
Protein was extracted from fat bodies and ovaries using a method similar to Gong et al.31 Individual adult females were dissected under a zoom-stereomicroscope (model XTL20, Beijing Tech Instrument Co., Ltd., Beijing, China) in a cooled petri dish. Ovaries and fat bodies of females were removed and placed in separate, pre-weighed, ice-cold centrifuge tubes and then re-weighed using a Mettler-Toledo electronic balance (EC100 model; 1/10,000 g sensitivity). A proportional amount of NaCl solution (0.4 M NaCl: 1M PMSF, v:v at a ratio of 20 ml NaCl solution to 1 g ovary or fat body) was added to the tube, homogenized on ice and centrifuged at 16,000 × g at 4 °C for 20 min. The supernatant was collected after filtering the upper fat layer with glass fibers, placed at 4 °C overnight after adding ddH2O (1 supernatant: 10 ddH2O, v/v) and centrifuged again at 2,800 × g at 4 °C for 20 min. The protein sediment was dissolved with 1.5 ml pre-chilled 0.4 M NaCl solution after removing the supernatant.
We followed the procedure described in Brandford32 to measure protein content using Coomassie Brilliant Blue R250 (Shanghai Chemical Agent Co., Ltd., Shanghai, China). A standard curve was established based on a standard protein (bovine serum albumin, Shanghai Biochemistry Research Institute, Shanghai, China). The absorbance at 595 nm was determined in a UV755 B spectrometer (Shanghai Precision Instrument Co., Ltd., Shanghai, China). The protein content in the sample solution was calculated according to the standard curve
Glucose analysis
Glucose levels were determined using the glucose oxidase method, following Sigma instructions (Sigma, St. Louis, MO). Two ml of 20 mM phosphate buffer (PBS, pH 5.8) were added to 20 mg of whole insect ovaries and fat bodies and then homogenized at 0 °C. The sample solution was centrifuged at 11,200 × g for 10 min at 4 °C. Fifty μl of 0.6 mol.L−1 perchloric acid were added into 50 μl of the supernatant to remove the protein in solution. After centrifugation at 1,008 × g for 5 min at 4 °C, 450 μl of 0.2 mol. L−1 sodium phosphate buffer (pH 7.4) was added into 50 μl of the supernatant. Two hundred μl of chromogen reagent (Covance, New Jersey, USA) and 150 μl of glucose oxidase were added to 50 μl of the sample solution, then incubated at 37 °C for 5 min. The absorbance at 625 nm was determined with a UV755B spectrometer (Shanghai Precision Instrument Co., Ltd., Shanghai, China). The glucose content in the sample solution was calculated based on a standard curve.
Body weights
Five females were used as a replicate at day 2 and day 3. Five adult females were placed in pre-weighed centrifuge tubes and then weighed using a Mettler-Toledo electronic balance (EC100 model; 1/10,000 g sensitivity). Each treatment and each control experiment was replicated six times.
qPCR analysis
We isolated total RNA from the five newly-emerged females, using a SV Total Isolation System Kit (model Z3100, Promega Corporation, Madison, WI, USA). First-strand cDNA was synthesized according to the instructions supplied with the PrimeScript RT reagent Kit (TaKaRa Biotechnology (Dalian) Co., Ltd). First-strand cDNA was synthesized in a 10 μl reaction volume containing 0.5 μg of RNA, 0.5 μl of PrimeScript RT enzyme mix I, 0.5 μl of Oligo dT primer (50 μM), 2 μl of random hexamers (100 μM), 2 μl 5 × PrimeScript Buffer (for real time-PCR) and RNase–free dH2O up to a final volume of 10 μl. The cDNA was reverse transcribed using the following program: 37 °C for 15 min, 85 °C for 5 s and 4 °C for 5 min.
We isolated total RNA from the dsRNA-treated and un-dsRNA (control) females, using a SV Total Isolation System Kit (model Z3100, Promega Corporation, Madison, WI, USA). Portions (2 μl) of the synthesized first-strand cDNA (above synthesis) were amplified by qPCR in 20 μl reaction mixtures using a CFX96 real-time PCR system (Bio-Rad Co. Ltd., California, USA) with the following procedure: 94 °C for 2 min, followed by 40 cycles of 94 °C for 5 s, 61.8 °C (Nlst6) or 58 °C (Nlvg) for 30 s and 72 °C for 30 s. The differences in the Tm values of Nlst6 or Nlvg was 61.8 °C or 58 °C, other procedures were the same. Nlst6 (AB549999) or Nlvg (AB353856) mRNA levels were quantified in relation to constitutive actin-1 expression (EU179846). Primers used for qPCR analysis are listed in Table 1. After amplification, a melting curve analysis was performed in triplicate and the results were averaged. The values were calculated using three independent biological samples and the 2 −△△CT method33 used for the analysis of relative Nlst6 expression level.
Statistical analysis
The results presented in figures are expressed as the means ± S.E. DPS 7.5 software was used to perform t-tests to identify significant differences at a 95% confidence level (p 0.05)34. Biological parameter data were analyzed using an analysis of variance (ANOVA) with one factor and statistical data are presented in Table 2.
Additional Information
How to cite this article: Ge, L.-Q. et al. Silencing a sugar transporter gene reduces growth and fecundity in the brown planthopper, Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). Sci. Rep. 5, 12194; doi: 10.1038/srep12194 (2015).
References
Holt, J., Chancellor, T. C. B., Reynolds, D. R. & Tiongco, E. R. Risk assessment for rice planthopper and tungro disease outbreaks. Crop Prot. 15, 359–368 (1996).
Ali, M. P. et al. Will climate change affect outbreak patterns of planthoppers in Bangladesh? PLoS One 9, e91678 (2014).
Wang, Y. et al. Penetration into rice tissues by brown planthopper and fine structure of the salivary sheaths. Entomol. Exp. App. 129, 295–307 (2008).
Hibino, H. Biology and epidemiology of rice viruses. Annu. Rev. Phytopathol. 34, 249–274 (1996).
Small, G. J. & Hemingway, J. Differential glycosylation produces heterogeneity in elevated esterases associated with insecticide resistance in the brown planthopper Nilaparvata lugens Stål. Insect Biochem. Mol. Biol. 30, 443–453 (2000a).
Small, G. J. & Hemingway, J. Molecular characterization of the amplified carboxylesterase gene associated with organophosphorus insecticide resistance in the brown planthopper, Nilaparvata lugens. Insect Mol. Biol. 9, 647–653 (2000b).
Hayashi, H. & Chino, M. Chemical composition of phloem sap from the uppermost internode of rice plant. Plant Cell Physiol. 31, 247–251 (1990).
Ashford, D. A., Smith, W. A. & Douglas, A. E. Living on a high sugar diet: the fate of sucrose ingested by a phloem-feeding insect, the pea aphid Acyrthosiphon pisum. J. Insect Physiol. 46, 335–341 (2000).
Baldwin, S. A. & Henderson, P. J. F. Homologies between sugar transporters from eukaryotes and prokaryotes. Annu. Rev. Physiol. 51, 459–471 (1989).
Price, D. R. G., Wilkinson, H. S. & Gatehouse, J. A. Functional expression and characterization of a gut facilitative glucose transporter, NlHT1, from the phloem-feeding insect Nilaparvata lugens (rice brown planthopper). Insect Biochem. Mol. Biol. 37, 1138–1148 (2007).
Kikuta, S., Kikawada, T., Hagiwara-Komoda, Y., Nakashima, N. & Noda, H. Sugar transporter genes of the brown planthopper, Nilaparvata lugens: A facilitated glucose/fructose transporter. Insect Biochem. Mol. Biol. 40, 805–813 (2010).
Price, D. R. G. et al. Sugar transporters of the major facilitator superfamily in aphids; from gene prediction to functional characterization. Insect Mol. Biol. 19, 97–112 (2010).
Price, D. R. & Gatehouse, J. A. Genome-wide annotation and functional identification of aphid GLUT-like sugar transporters. BMC Genomics. 15, 647 (2014).
Crivat, G. et al. Insulin stimulates translocation of human GLUT4 to the membrane in fat bodies of transgenic Drosophila melanogaster. PLoS One 8, e77953 (2013).
Uldry, M. & Torens, B. The SLC2 family of facilitated hexose and polyol transporters. Pflugers Arch. European J. Physiol. 447, 480–489 (2004).
Honěk, A. Intrapecific variation in body size and fecundity in insects: A general relationship. Oikos. 66, 483–492 (1993).
García-Barros, E. Body size, egg size and their interspecific relationships with ecological and life history traits in butterflies (Lepidoptera: Papilionoidea, Hesperioidea). Biol. J. linn. Soc. 70, 251–284 (2000).
Jiménez-Pérez, A. & Wang, Q. Effect of body weight on reproductive performance in Cnephasia jactatana (Lepidoptera: Tortricidae). J. Insect Behav. 17, 511–522 (2004).
Marshall, L. D. Intraspecific variation in reproductive effort by female Parapediasia teterrella (Lepidoptera: Pyralidae) and its relation to body size. Can. J. Zool. 68, 44–48 (1990).
Ge, L. Q., Wu, J. C., Zhao, K. F., Chen, Y. & Yang, G. Q. (2010a) Induction of Nlvg and suppression of Nljhe gene expression in Nilaparvata lugens (Stål) (Hemiptera: Delphacidae) adult females and males exposed to two insecticides. Pestic. Biochem. Physiol. 98, 269–278 (2010a).
Ge, L. Q., Wang, L. P., Zhao, K. F., Wu, J. C. & Huang, L. J. Mating pair combinations of insecticide-treated male and female Nilaparvata lugens Stål (Hemiptera: Delphacidae) planthoppers influence protein content in the male accessory glands (MAGs) and vitellin content in both fat bodies and ovaries of adult females. Pesti. Bicohem. Physiol. 98, 279–288 (2010b).
Liu. J. L., Yang, X., Chen, X. & Wu, J. C. Suppression of fecundity, Nlvg gene expression and vitellin content in Nilaparvata lugens Stål (Hemiptera: Delphacidae) adult females exposed to indoxacarb and chlorantraniliprole. Pestic. Biochem. Physiol. 104, 206–211 (2012).
Wang, G. C. et al. Relationship between yeast-like symbiotes and amino acid requirements in the rice brown planthopper, Nilaparvata lugens Stål (Homoptera: Delphacidae). Acta Entomol. Sin. 48, 483–490 (2005b).
Fu, Q., Zhang, Z. T., Hu, C. & Lai, F. X. The effects of high temperature on both yeast-like symbionts and amino acid requirements of Nilaparvata lugens. Acta Entomol. Sin. 44, 534–540 (2001).
Wilkinson, T. L. & Ishikawa, H. On the functional significance of symbiotic microogranisms in the Homoptera: a comparative study of Acyrthosiphon pisum and Nilaparvata lugens. Physiol. Entomol. 26, 86–93 (2001).
Lee, Y. H. & Hou, R. F. Physiological roles of a yeast-like symbiote in reproduction and embryonic development of the brown planthopper. J. Insect Physiol. 33, 852–860 (1987).
Yin, J. L., Xu, H. W., Wu, J. C., Hu, J. H. & Yang, G. Q. Cultivar and insecticide applications affect the physiological development of the brown planthopper, Nilaparvata lugens (Stål) (Homoptera: Delphacidae). Environ. Entomol. 37, 206–212 (2008).
Scott, R., Schuldiner, O. & Neufeld, T. P. Role and regulation of starvation-induced autophagy in the Drosophila fat body. Developmental Cell 7, 167–178 (2004).
Fu, Q., Zhang, Z. T., Lai, F. X. & Hu, C. A. chemically defined diet enables the continuous rearing of the brown planthopper, Nilaparvata lugens (Stål). Appl. Entomol. Zool. 36, 111–1116 (2001).
Chen, J. et al. Feeding-based RNA interference of a trehalose phosphate synthase gene in the brown planthopper Nilaparvata lugens. Insect Mol. Biol. 19, 777–786 (2010).
Gong, H., Zhai, C. H., Wei, D. Y. & Zhang, J. Z. On the vitellogenesis of Coccinella septempunctata L: the occurrence of vitellogenin as influenced by artificial diet. Acta Entomol. Sin. 23, 252–257 (1980).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 –△△CT method. Methods. 25, 402–408 (2001).
Tang, Q. & Feng, M. G. Analysis of Variance. In DPS Data Processing System for Practical Statistics (eds. Tang, Q. & Feng, M. G. ). Ch. 4, 47–71 (Scientific Press, 2002).
Acknowledgements
This research was supported in part by the National Natural Science Foundation of China (31201507), by the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (12KJB210006), by the College Students Practice Innovation Training Program of Jiangsu Province University (201311117033Z), by the major state Basic Research Program of China (973 program, Grant No. 2010CB126200). Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. All programs and services of the U.S. Department of Agriculture are offered on a nondiscriminatory basis without regard to race, color, national origin, religion, sex, age, marital status, or handicap.
Author information
Authors and Affiliations
Contributions
L.Q.G. and J.C.W. designed the study. L.Q.G., Y.P.J., T.X., P.K., X.L.L. and G.Q.Y. performed all the experiments and statistical analysis. L.Q.G. wrote first version. J.C.W. revised the first version, Q.S.S. and D.S. revised the final version. All authors reviewed the manuscript and approved the manuscript for publication.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ge, LQ., Jiang, YP., Xia, T. et al. Silencing a sugar transporter gene reduces growth and fecundity in the brown planthopper, Nilaparvata lugens (Stål) (Hemiptera: Delphacidae). Sci Rep 5, 12194 (2015). https://doi.org/10.1038/srep12194
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12194
This article is cited by
-
Double-stranded RNA mediated knockdown of sucrase gene induced mortality and reduced offspring production in Aphis gossypii
Functional & Integrative Genomics (2023)
-
Integrated analysis of differentially expressed microRNAs and mRNAs to screen miRNAs and genes related to reproduction in Macrobrachium nipponense
3 Biotech (2019)
-
Reference genes for quantitative real-time PCR analysis in symbiont Entomomyces delphacidicola of Nilaparvata lugens (Stål)
Scientific Reports (2017)
-
Suppressing male spermatogenesis-associated protein 5-like gene expression reduces vitellogenin gene expression and fecundity in Nilaparvata lugens Stål
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.