Abstract
The continuous increase in the intensity and extent of anthropogenic artificial light has significantly shaped Earth’s nighttime environment. This environmental change has effects across the natural world, with consequences for organismal physiology and behaviour and the abundances and distributions of species. Here, we evaluate for the first time the relations between the spatio-temporal patterns of anthropogenic nighttime light and the distribution of terrestrial mammals, one of the most endangered species groups and one that expresses varying time partitioning strategies. Using descriptive statistics, trend tests and spatial prioritization analysis we show that in most places on earth there is a terrestrial mammal species whose range is experiencing detectable artificial light. For most species this tends only to be for small parts of their range and those affected across large parts are typically rare. Over time (1992–2012), an increase in mean light intensity was found for the ranges of the majority of species, with very few experiencing a decrease. Moreover, nocturnal species are more likely to experience an increase in light within their ranges. This is of conservation concern as many terrestrial mammals are nocturnal and therefore often particularly vulnerable to a pressure such as artificial light at night.
Similar content being viewed by others
Introduction
Natural light regimes are being disrupted over an increasingly large extent of the Earth’s surface. This has resulted in part from the direct effects of artificial light at night (ALAN), predominantly produced by electric lighting. However, it is also caused by the skyglow that results from the diffuse scattering of these light emissions in the atmosphere. Indeed, by 2001, 19% of the global land surface was already estimated to be above a threshold of light set for polluted status1.
ALAN has a wide diversity of biological impacts, affecting both diurnal and nocturnal species2,3,4,5. These can be characterised as comprising influences on natural light regimes as a resource and as a source of information to organisms6. The former include effects on photosynthesis, the partitioning of activity between day and night and dark repair and recovery and the latter effects on circadian clocks and photoperiodism, visual perception and spatial orientation (for review of empirical examples see6). This array of influences has given rise to substantial concerns and growing evidence, about the consequences for the abundance and distribution of species, community structure and ecosystem processes and dynamics7,8,9,10 and as to the ways in which these can be mitigated11,12.
Despite these concerns, there are a lack of estimates regarding how the occurrence of and trends in, ALAN are distributed with respect to the geographic ranges of the species in particular taxonomic groups. Key questions concern how many species have ranges that are experiencing ALAN, what proportions of their ranges are influenced and how this is changing and in which regions those species which are most extensively influenced reside.
Here we address these issues using terrestrial mammals as a case study, investigating the extent and change in ALAN within their ranges. These provide an interesting study group because they are globally distributed, occupy a broad range of environments and exhibit a wide diversity of time partitioning behaviour that can in substantial part be predicted based on patterns of natural light and darkness13. Moreover, ALAN has already been shown to have a wide diversity of impacts on mammals including on their circadian rhythms and photoperiodism14, immune responses15, foraging16, movements17,18,19 and reproduction20.
Results
In very few places was there a terrestrial mammal species whose geographic range was not experiencing some detectable ALAN (Fig. 1(c)). Examples of these areas include small pockets of land found in Madagascar, central Australia, Baja California in Mexico, the Amazon rainforest, parts of large islands and several small islands in south-east Asia. For many species lighting occurred in only a small proportion of their range, with 3594 experiencing ALAN in less than 10% of their range during 1992–1995 (Fig. 2a). As the proportion of the geographic range experiencing light increased, the geographical focus shifted mainly to the Northern Hemisphere, specifically, N. America, Europe and Japan (Fig. 1(d–e)). Few species were experiencing ALAN in more than 60% of their range, with those with ranges bound to small islands predominantly falling into this group (Fig. 1f). Indeed, those species experiencing ALAN over high proportions of their geographic ranges were typically rare (small range sizes), with the more widely distributed species almost invariably occurring in many places with no ALAN (Fig. 2b).
The global distributions of (a) nighttime lights, shown using a 2012 DMSP-OLS nighttime lights image (downloaded from http://ngdc.noaa.gov/eog/dmsp/downloadV4composites.html); (b) mammalian species richness; the numbers of species with given percentages of their geographic range experiencing detectable ALAN (DN > 5.5) for (c) >10%; (d) >20%; (e) >40% and (f) >60%. Richness maps created using range maps from the IUCN30 in R (R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria) using the ‘raster’ package (Hijmans, R.J. & van Etten, J. raster: Geographic data analysis and modeling. Version 2.1–49). Final display made in ArcGIS (ESRI. ArcGIS Desktop: Version 10, Environmental System Research Institute, Redlands, CA).
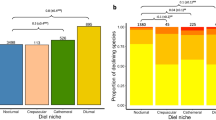
The magnitude and trend of conflict between ALAN and mammals.
(a) The difference in the spread of light between the first and last four years for all species, (b) A comparison of the average intensity of light between the first and last four years for affected species (those that have a mean DN higher than 0), (c) The strength of trend in ALAN over the 21 year study period by tau value (Mann-Kendall trend test). Grey shaded areas indicate significantly negative and positive results (p < 0.05) respectively, for affected species.
Of the 4370 mammal species studied, many have seen change in the ALAN within their range between 1992–2012. The majority (n = 3624) experienced a significant increase in the mean nighttime light (Mann-Kendall trend test, Τ > 0, p < 0.05) (Fig. 2c). Nocturnal species were most prominent in this group (62.4% of species), whereas 18.4% were diurnal, 7% cathemeral, 2.5% crepuscular and the remainder lacked information on time partitioning strategy. Forty-one species experienced significant decreases in ALAN within their ranges (Mann-Kendall trend test, Τ < 0, p < 0.05) (Fig. 2c). Nineteen of these species were nocturnal, 9 diurnal, 5 cathemeral, 1 crepuscular and 7 lacked information. Nocturnal species were significantly more likely to have experienced an increase in ALAN and less likely to experience no change than species with other time partitioning behaviours (χ-squared=84.45, p < 0.001).
We found no significant difference between species in different Red List categories with regards to the strength of the trend (for significantly positive trending species) in ALAN, the change in the proportion of lit pixels (see methods section for description), or the change in mean DN values (see Supplementary Information) within their range over time.
For two periods, three spatial prioritization analyses were performed with one representing mammals (Only Mammals), one representing ALAN (Only Light) and a third representing mammals and, when possible, avoiding ALAN areas (Balanced) (see Methods section for more details). Using the resulting priority values from these analyses, we used Spearman rank coefficients to assess the extent of conflict between different priority sites. In the period 1992–1995, the correlation between Only Mammals and Only Light priority areas was low (0.22, p < 0.01), indicating that a low relative proportion of mammals’ ranges experienced ALAN. The strength of this correlation slightly increased (0.29, p < 0.01) for the period 2009–2012. However, the decrease in the correlation between Only Mammals and Balanced priority areas from the period 1992–1995 (0.80, p < 0.01) to 2009–2012 (0.73, p < 0.01) suggests that avoiding ALAN within areas that over-represent the occurrence of mammals became more difficult with time. This is also reflected in the increase over time of the correlation between Only Light and Balanced (−0.03, p < 0.01 [1992–1995]; −0.06, p < 0.01 [2009–2012]), showing that balancing a set of areas that represent mammals and ALAN without overlapping has tended to become harder.
Discussion
Much concern has been expressed with regards to the potential impacts of ALAN on mammals and many studies have documented significant effects, particularly on foraging and movement patterns17,20,21,22,23. Whilst the effects of ALAN can be highly species specific and critically influenced by sensory (e.g. visual acuity) and environmental (e.g. habitat cover) variables24, mammals are especially vulnerable because a high proportion of species in the group are nocturnal13. Here we show that, in addition, the majority of species experience ALAN in some portion of their geographic range, that in most cases this ALAN is increasing and that for some rare species this can be occurring over most of their range (Fig 2b). These increases could have both positive and negative effects on mammals. ALAN can effectively increase the length of available activity time for diurnal species, reduce it for nocturnal species and cause more complex changes to the activity cycles of crepuscular and cathemeral species. While some species may be able to utilise the additional light for foraging or other behaviours, others may suffer from increased predation risk25, or altered patterns of time partitioning through competition for resources26,27.
As often with other global studies of anthropogenic impacts, these results are only indicative of what might be occurring. First, albeit being the best that are freely available, the species range data are relatively coarse in resolution compared to the ALAN data and therefore the levels of overlap may be somewhat over or under estimated. However, equally, the ALAN data do not capture the full extent of skyglow, which may propagate emissions even hundreds of kilometres from the source28, suggesting that the overlap between species ranges and ALAN is underestimated. Second, ALAN co-occurs with and is arguably an important component of, urbanisation and thus rarely acts as an anthropogenic pressure in isolation. Moreover, it is likely to act in additive and synergistic ways with an array of such pressures, including habitat fragmentation, climate change and chemical pollution29.
Given their high conservation profile and the predominance of nocturnal species, mammals are of particular concern with regard to the impacts of ALAN. However, there is little reason to believe that the high proportion of species shown here with geographic ranges experiencing ALAN and the growth in this effect, are atypical of many other groups of organisms.
Material and Methods
Data Sources
Analyses were based on global mammal species range maps downloaded from IUCN (International Union for the Conservation of Nature)30. The terrestrial and non-fossorial (following13) subset of species was used (including bats), excluding Melomys rubicola, which has a range too small for analysis. All data were projected to a Behrmann equal-area projection using ArcGIS (ESRI. ArcGIS Desktop: Version 10, Environmental System Research Institute, Redlands, CA). Species were classified as (i) diurnal, nocturnal, crepuscular, cathemeral or unknown, following13; and (ii) according to their IUCN threat category30, excluding those listed as ‘Extinct in the Wild’ or ‘Extinct’, leaving 4370 species. Species classed as ‘Data Deficient’ by the IUCN lack spatial information and were therefore not included in this analysis.
Nighttime stable lights composite images were downloaded from the National Oceanic and Atmospheric Administration archives (1992–2012, n = 21), created with data from the Defense Meteorological Satellite Program’s Operational Linescan System (DMSP/OLS). The images are at 1 km resolution and each pixel is represented by a digital number (DN) between zero and 63. Zero represents darkness, while brightly lit areas saturate at values of 63. Images were intercalibrated and drift-corrected according to31.
Extraction & Analysis
The DMSP data for all years were extracted for each species using GDAL utility tools (GDAL: Geospatial Data Abstraction Library: Version 1.10, Open Source Geospatial Foundation). Pixels were associated with a mammalian range if their centre point fell within the polygon boundary. This extraction resulted in a raster file per species per year that could then be further analysed in R (R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria), using the ‘raster’ (Hijmans, R.J. & van Etten, J. raster: Geographic data analysis and modeling. Version 2.1–49) package. Using the package ‘Kendall’ (McLeod, A.I. Kendall: Kendall rank correlation and Mann-Kendall trend test. Version 2.2), Mann-Kendall trend tests identified significant trends in yearly-mean nighttime light values for each species. Chi-squared tests were also performed in R to explore the relationship between time-partitioning behaviour and changes in ALAN within species ranges.
It has been shown that over 94% of observed increases in DN of more than 3 units and over 93% of observed decreases of the same magnitude could be attributed to a known change on the ground consistent with the direction of change (i.e. growth in urban areas, deindustrialisation)31. We defined a threshold of darkness of <5.5 DN. Lit pixels were those with any value above this threshold. By using a threshold effectively twice the detection limit for change, we defined a conservative estimate of lit area and limited the extent to which dark sites may be classified as lit due to noise in the data or calibration errors.
Zonation analysis
The spatial prioritization software Zonation32 was used to investigate spatial conflicts between mammal occurrence and ALAN and their changes over time. Data were aggregated to 10 km. Due to the non-additive nature of the DMSP data, the images containing average DN values for the periods 1992–1995 and 2009–2012 were converted to binary, with a 1 representing a positive DN and a 0 for 0 DN. For these periods we generated three hierarchical prioritization maps, in which the representation of mammals and ALAN on the Earth’s surface was optimized according to the following rules: i) Only Mammals, spatial prioritization aiming to optimize the representation of mammals (i.e. relative proportion of mammals’ ranges). Light raster was weighted as 0 (hence ignored in the prioritization selection) and each of the species as 1; ii) Only Light, spatial prioritization aiming to optimize the representation of lit areas. Here the light raster was weighted as 1 and each of the mammal species weighted as 0; and iii) Balanced, spatial prioritization aiming to optimize the representation of mammals while simultaneously excluding lit areas, thus reducing potential conservation conflicts. Light raster was weighted as −1 (hence ALAN is avoided within mammal priority areas) and all the species were weighted equally ω = 1/4370. This implies that species were jointly equal to the ALAN value 1.
Additional Information
How to cite this article: Duffy, J. P. et al. Mammalian ranges are experiencing erosion of natural darkness. Sci. Rep. 5, 12042; doi: 10.1038/srep12042 (2015).
References
Cinzano, P., Falchi, F. & Elvidge, C. D. The first world atlas of the artificial night sky brightness. Mon Not R Astron Soc. 328, 689–707 (2001).
Longcore, T. & Rich, C. Ecological light pollution. Front Ecol Environ 2, 191–198 (2004).
Rich, C. & Longcore, T. (eds.) Ecological consequences of artificial night lighting (Island Press, Washington DC 2013).
Perkin, E. K. et al. The influence of artificial light on stream and riparian ecosystems: questions, challenges and perspectives. Ecosphere. 2, 122 (2011).
Davies, T. W., Duffy, J. P., Bennie, J. & Gaston, K. J. The nature, extent and ecological implications of marine light pollution. Front Ecol Environ. 12, 347–355 (2014).
Gaston, K. J., Bennie, J., Davies, T. W. & Hopkins, J. The ecological impacts of nighttime light pollution: a mechanistic appraisal. Biol Rev. 88, 912–927 (2013).
Davies, T. W., Bennie, J. & Gaston, K. J. Street lighting changes the composition of invertebrate communities. Biol Letters 8, 764–767 (2012).
Mazor, T. et al. Can satellite-based night lights be used for conservation? The case of nesting sea turtles in the Mediterranean. Biol Conserv. 159, 63–72 (2013).
Meyer, L. A. & Sullivan, S. M. P. Bright lights, big city: influences of ecological light pollution on reciprocal stream-riparian invertebrate fluxes. Ecol Appl. 23, 1322–1330 (2013).
Gaston, K. J. & Bennie, J. Demographic effects of artificial nighttime lighting on animal populations. Environ Rev. 22, 323–330 (2014).
Falchi, F., Cinzano, P., Elvidge, C. D., Keith, D. M. & Haim, A. Limiting the impact of light pollution on human health, environment and stellar visibility. J Environ Manage 92, 2714–2722 (2011).
Gaston, K. J., Davies, T. W., Bennie, J. & Hopkins, J. Reducing the ecological consequences of night-time light pollution: options and developments. J Appl Ecol. 49, 1256–1266 (2012).
Bennie, J., Duffy, J. P., Inger, R. & Gaston, K. J. The biogeography of time partitioning in mammals. P Natl Acad Sci USA 111, 13727–13732 (2014).
Haim, A., Shanas, U., Zubidad, A. E. S. & Scantelbury, M. Seasonality and seasons out of time – The thermoregulatory effects of light interference. Chronobiol Int. 22, 59–66 (2005).
Bedrosian, T. A., Fonken, L. K., Walton, J. C. & Nelson, R. J. Chronic exposure to dim light at night suppresses immune responses in Siberian hamsters. Biol Letters 7, 468–471 (2011).
Bird, B. I., Branch, L. C. & Miller, D. L. Effects of coastal lighting on foraging behaviour of beach mice. Conserv Biol. 18, 1435–1439 (2004).
Beier, P. Dispersal of juvenile cougars in fragmented habitat. J Wildlife Manage. 59, 228–237 (1995).
Stone, E. L., Jones, G. & Harris, S. Conserving energy at a cost to biodiversity? Impacts of LED lighting on bats. Glob Change Biol. 18, 2458–2465 (2012).
Threlfall, C. G., Law, B. & Banks, P. B. The urban matrix and artificial light restricts the nightly ranging behaviour of Gould’s long-eared bat (Nyctophilus gouldi). Austral Ecol. 38, 921–930 (2013).
LeTallec, T., Théry, M. & Perret, M. Effects of light pollution on seasonal estrus and daily rhythms in a nocturnal primate. J Mammal. 96, 438–445 (2015).
Beier, P. Effects of artificial night lighting on terrestrial mammals. Ecological consequences of artificial night lights ( Rich, C. & Longcore, T. ) [19–42] (Island Press, Washington DC, 2006).
Stone, E. L., Jones, G. & Harris, S. Street lighting disturbs commuting bats. Curr Biol. 19, 1123–1127 (2009).
Rotics, S., Dayan, T. & Kronfeld-Schor, N. Effect of artificial night lighting on temporally partitioned spiny mice. J Mammal. 92, 159–168 (2011).
Prugh, L. R. & Golden, C. D. Does moonlight increase predation risk? Meta-analyss reveals divergent responses of nocturnal mammals to lunar cycles. J Anim Ecol. 83, 504–514 (2014).
Daly, M., Behrends, P. R., Wilson, M. I. & Jacobs, L. F. Behavioural modulation of predation risk: moonlight avoidance and crepuscular compensation in a nocturnal desert rodent, Dipodomys merriami. Anim Behav. 44, 1–9 (1992).
Gutman, R. & Dayan, T. Temporal partitioning: an experiment with two species of spiny mice. Ecology. 86, 164–173 (2005).
Kronfeld-Schor, N. & Dayan, T. Partitioning of time as an ecological resource. Annu Rev Ecol Evol S. 34, 153–181 (2003).
Luginbuhl, C. B., Bodey, P. A. & Davis, D. R. The impact of light source spectral power distribution on sky glow. J Quant Spectrosc Ra. 139, 21–26 (2014).
Gaston, K. J., Duffy, J. P., Gaston, S., Bennie, J. & Davies, T. W. Human alteration of natural light cycles: causes and ecological consequences. Oecologia 176, 917–931 (2014).
IUCN, IUCN Red List of Threatened Species - http://www.iucnredlist.org (2013) (Date of access: 02/02/2014).
Bennie, J., Davies, T. W., Duffy, J. P., Inger, R. & Gaston, K. J. Contrasting trends in light pollution across Europe based on satellite observed night time lights. Sci Rep 4, 3789 (2014).
Moilanen, A. et al. Prioritizing multiple-use landscapes for conservation: methods for large multi-species planning problems. P R Soc B. 272, 1885–1891 (2005).
Acknowledgements
The research leading to this paper has received funding from the European Research Council under the European Union’s Seventh Framework Programme (FP7/2007–2013)/ERC grant agreement no. 268504 to K.J.G.
Author information
Authors and Affiliations
Contributions
J.P.D. and K.J.G. designed the study; J.P.D., J.B. and A.P.D. analysed the data; J.P.D. and K.J.G. wrote the first draft of the manuscript and all authors contributed to subsequent versions.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Duffy, J., Bennie, J., Durán, A. et al. Mammalian ranges are experiencing erosion of natural darkness. Sci Rep 5, 12042 (2015). https://doi.org/10.1038/srep12042
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12042
This article is cited by
-
The dark side of raptors’ distribution ranges under climate change
Landscape Ecology (2020)
-
Assessing coastal artificial light and potential exposure of wildlife at a national scale: the case of marine turtles in Brazil
Biodiversity and Conservation (2020)
-
Erosion of natural darkness in the geographic ranges of cacti
Scientific Reports (2018)
-
Light pollution is greatest within migration passage areas for nocturnally-migrating birds around the world
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.