Abstract
A mixture of extracts from two common vegetables, red pomegranate and turmeric, when photoexcited at 380 nm, produced almost pure white light emission (WLE) with Commission Internationale d’Eclairage (CIE) chromaticity index (0.35, 0.33) in acidic ethanol. It was also possible to obtain WLE in polyvinyl alcohol film (0.32, 0.25) and in gelatin gel (0.26, 0.33) using the same extract mixture. The colour temperature of the WLE was conveniently tunable by simply adjusting the concentrations of the component emitters. The primary emitting pigments responsible for contributing to WLE were polyphenols and anthocyanins from pomegranate and curcumin from turmeric. It was observed that a cascade of Forster resonance energy transfer involving polyphenolics, curcumin and anthocyanins played a crucial role in obtaining a CIE index close to pure white light. The optimized methods of extraction of the two primary emitting pigments from their corresponding plant sources are simple, cheap and fairly green.
Similar content being viewed by others
Introduction
White light emitting materials have attracted significant attention in recent years as key components in display and lighting devices based on LEDs1. LEDs accounts for almost 20% of the total worldwide energy consumption and have wide applications in backlights, displays, lasers and indicators2,3,4. White light emission has also been used for sensing5. There has been a high level of interest in recent years in looking for white light emitting organic and inorganic molecules and materials, when photoexcited at near UV wavelengths6. A variety of photophysical principles have been used for achieving emission containing the three essential red-green-blue components for effective white light perception. These include FRET7,8, Inter- and intra- molecular charge transfer9, excited state intramolecular proton transfer (ESIPT)10, hydrogen bonding mediated J- aggregation11 and the mixing of monomer and excimer fluorescence12 etc. Although single-molecule WLE is conceptually attractive, it is difficult to adjust the CIE index and colour temperature of such molecules conveniently. In this regard multi component WLE comprising of a mixture of different molecules can, in principle, make it easy to tune the colour temperature by simply adjusting the composition. Not surprisingly, there is significant recent interest for generation of mixed emitter WLE. Such systems comprise of inorganic, organic and hybrid systems like nanomaterials/quantum dots13,14,15, polymer16,17, metal-organic framework18,19,20, inorganic-organic hybrids21, metal complexes22 and lanthanide doped systems23,24. Various systems tried by using mixtures of organic fluorophores include, (i) a simple mixing of three emitting dyes at RGB region25, (ii) donar-acceptor conjugated pairs26, (iii) organic liquids such as π- conjugated oligo (p-phenylenevinylene) (OPV)27, (iv) controlled donor self assembly based organogels28, (v) and a peptide link based two component system29.
Interestingly, generation of white light emission from natural dyes has not been looked at yet. Plant sources have been rich sources of colouring dyes since time immemorial and many of such dyes are strongly fluorescent. Thus, while looking for white light emitters, it makes sense to look for a combination of suitable natural dyes. Literature search did not yield any such report in this regard. We report here the result of our search for a white light emitting natural dye combination based on the following criteria: (i) the sources of the dyes must be easily available and cheap, (ii) the extraction of the dyes must be simple, cost effective and environment friendly, (iii) the dyes must have sufficient high extinction coefficient and fluorescence quantum yield so that the combination has good white light emission efficiency, (iv) the individual dye emissions must be suitably located in the CIE plot so as to give white light emission after mixing and (v) the colour temperature of the emission should be conveniently tunable by simply adjusting the dye composition. In order to achieve facile tuning of CIE index as well as the colour temperature of interest, we followed a strategy in choosing two vegetable extracts containing emitting dyes such that they emit in blue-violet, green and orange regions of visible spectrum.
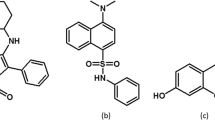
For this purpose, several trials with blue, green and red emitting natural dyes30 eventually converged on to two easily available vegetable extracts, red pomegranate (Pom) seed juice and turmeric (Tur) extract. The essential emitting pigments in Pom seed juice are a group of polyphenolics and anthocyanins31. In general, polyphenols and anthocyanins emit at blue and orange-red region respectively32,33 and are found in many of the fruits and vegetables34. Curcumin is the essential emitting pigment in Tur extract35. The molecular structures of the natural derivatives are shown in Fig. 1a,b respectively.
Pom is rich in antioxidants such as phenolic acids, tannins, flavonols and anthocyanins36. The red colour of Pom is known to be primarily due to six anthocyanin pigments. Hernández et al. have quantitatively and qualitatively analyzed all six anthocyanins by high-performance liquid chromatography and identified them as delphinidin 3- glucoside and 3, 5-diglucoside, cyanidin 3-glucoside and 3, 5-diglucoside and pelargonidin 3-glucoside and 3, 5-diglucoside37. The colour of anthocyanins is pH sensitive; in acidic media it shows a red colour, while in alkaline solutions, it turns blue38.
There are various methods and sources reported in literature for extraction of polyphenols and anthocyanins such as pink petals of rhododendron indicum flowers32, Pom39,40, blackcurrant41, grapes42, red cabbage43,etc. We have adopted a variant of a method reported by Hamidreza et al.37, the details of which are given in the section on Materials and Methods.
Results and discussion
Electronic absorption and fluorescence spectral studies were carried out in the extraction medium containing 1% HCl in ethanol. For fluorescence spectra, the excitation wavelength was fixed at 380 nm. UV-visible electronic spectra (black) of Pom and Tur extracts are shown in Fig. 2a and Fig. 2b respectively. The strong absorption band at 535 nm originates from anthocyanins and at ~295 nm from the polyphenolic components31,32,44. The total anthocyanin concentration was estimated using the known molar extinction coefficient (34300 M−1cm−1) of cyanindin-3-glucoside at 530 nm45. Since the amount of emitting natural dyes can change from source to source, the effective concentration of the dyes can be expressed in terms of molarity calculated with this molar extinction coefficient. This practice has been followed in the rest of this paper.
For the Tur extract, a broad characteristic46 absorption band of curcumin is seen around 350–500 nm with maximum at 420 nm (Fig. 2b (black)). This absorption corresponding to π – π* electronic transition has molar extinction coefficient of 55000 M−1 cm−1 at 420 nm in ethanol43. The effective concentration of the emitting dye in the Tur extract has subsequently been expressed in terms of molarity calculated with this extinction coefficient. Under UV light excitation at 380 nm, Pom extract shows bluish red emission (inset, Fig. 2a) (blue) with two bands at 440 nm and 590 nm corresponding to the phenolic and anthocyanin components respectively32,33,44. For the same excitation Tur extract shows intense green fluorescence emission (green) at around 522 nm47 (Fig. 2b).
The CIE chromaticity48 coordinate for Pom extract appears in the blue-violet region A(0.28, 0.17); and the coordinate for Tur extract appears at green region B(0.31, 0.51) in the CIE diagram (Fig. 3). It is observed that the points A and B appear on the opposite side of the white region in the CIE diagram. This suggested to us that by mixing the two extracts it should be possible to obtain WLE.
Accordingly, a series of solutions were prepared by varying the relative concentrations of Pom and Tur extracts (Supplementary Table S1, Fig 1a and Fig. 1b). Figure 4 shows the trajectory of the colour coordinate of these solutions in the CIE diagram. It is seen that the point C (0.35, 0.33), corresponding to the composition 60.0 μM (anthocyanin equivalent) and 3.0 μM (curcumin equivalent) with a molar ratio of 1:0.05::anthocyanin:curcumin, is extremely close to the pure white coordinate O (0.33, 0.33).
It is interesting to see that the trajectory of the colour coordinate actually bends and passes very close to the pure white coordinate. The emission spectrum corresponding to point C covered the entire visible region (400–700 nm) (Fig. 5a) with two emission maxima at 480 and 590 nm. These two bands, though appear to be similar to polyphenolic and anthocyanin emissions of Pom, have different characteristics. For the extract mixture, there is a red shift of blue emission band from 440 nm to 480 nm and a significant enhancement as well as broadening of the 590 nm band. Clearly, this change in the emission spectral features is due to contribution from Tur emission. The excitation emission matrix fluorescence (Fig. 5b) clearly shows that at 380 nm excitation the emission covers the entire visible spectral range upto about 700 nm.
WLE in acidic ethanol.
(a) Emission spectrum of a mixture of Pom extract (anthocyanin equivalent 60.0 μM) and Tur extract (curcumin equivalent 3.0 μM), excited at 380 nm. Inset shows photograph of corresponding white emitting solution under UV excitation (380 nm) and (b) Excitation emission matrix fluorescence spectrum of white emitting solution at different excitation wavelength (λexc = 360–560 nm).
If the components in the two extracts did not interact photophysically, their emission spectral profile would be additive. Such hypothetical emission spectra (Supplementary Fig. S3) can be created by adding the intensities of component spectral profiles of Pom and Tur extracts (Supplementary Fig. S2). The corresponding colour coordinates of these sum spectra in the CIE diagram are shown in Fig. 6. The reported quantum yield of malvidin 3, 5-diglucoside anthocyanin49 is 4.1 × 10−3 (generally, the anthocyanins are weakly fluorescent) and that of curcumin50 is 6.3 × 10−2. Hence it is seen that the Tur extract fluorescence is much more intense than the Pom extract fluorescence. A comparison of the trajectory, the hypothetical sum spectral coordinates (Fig. 6) and the actual spectral coordinate (Fig. 3) indicates the possible presence of significant photophysical interaction between the components.
The absorption and emission spectra of the two extracts show significant overlap (Supplementary Fig. S4) of (i) curcumin absorption band (420 nm) with polyphenolics emission band (440 nm) and (ii) curcumin emission band (522 nm) with anthocyanin absorption band (535 nm). Thus there is a strong possibility of Forster resonance energy transfer (FRET) cascading from polyphenolics to curcumin to anthocyanins. In ordered to examine the presence of FRET, changes in spectral profiles were monitored with progressive addition of Tur extract (0–6 µM) to a fixed amount of Pom extract (50 µM) (Fig. 7).
As is seen in the Fig. 7, with progressive increase of the Tur extract component, there is progressive (i) decrease of the polyphenolics emission around 440 nm, (ii) increase of curcumin emission around 490 nm and (iii) increase of anthocyanins emission around 585 nm. These spectral changes can be ascribed to a FRET cascade from polyphenolics to curcumin to anthocyanins because of the following observations: (i) Significant spectral overlap of emission band of polyphenolics with the absorption band of curcumin and the emission band of curcumin with the absorption band of anthocyanins, thereby satisfying the essential spectral overlap condition for FRET (Supplementary Fig. S4), (ii) the loss of polyphenolics emission, (iii) the significant loss of curcumin emission intensity in the mixture, (iv) the substantial enhancement of the long wavelength emission at the expense of curcumin emission and (v) the interesting observation that there is a dip of the curcumin emission at 530 nm which is the absorption maximum of anthocyanin absorption.
Observations from a complimentary experiment in which increasing amount Pom extract was added to a fixed Tur extract, given in (Supplementary Fig. S5) shows the progressive loss of curcumin emission intensity with a dip at 530 nm corresponding to maximum spectral overlap of anthocyanin absorption and curcumin emission. This further supports the existence of FRET. The presence of the FRET cascade in the optimized mixture of Pom and Tur extracts thus results in an extended emission spectrum that covers the entire visible spectral range from 400 nm to 700 nm. It also explains the bending of the trajectory towards red thereby making it pass through the pure white CIE. The concept of FRET has been further explored for adjusting the colour temperature. The ‘colour temperature’ of emission from a source refers to the temperature (in Kelvin unit) at which the colour of black body radiation matches the colour of the emissive source. The CIE coordinates and colour temperatures of a few combinations of Pom and Tur in acidic ethanol have been given in table 1. As is seen in table 1, simple variations in relative composition of the components shift the CIE indices and the corresponding colour temperatures in a facile way.
The possibility of WLE using Pom and Tur extracts in media like gelatin gel and polyvinyl alcohol (PVA) film has also been explored. Gelatin is an edible, biodegradable and biocompatible polymer that is produced by the thermal or physical and chemical degradation of collagen. WLE mixture was mixed with prepared gelatin solution and was kept in refrigerator for forming the gel. The resulting solution in acidic ethanol and water (46:54%) forms (Supplementary Fig. 6a) white light emitting gel at 15 °C under UV light illumination. Fluorescence spectrum of gel has been shown in Fig. 8a, which covered the visible region from 400 to 650 nm. This gel shows good white light emission under UV light (inset, Fig. 8a). The CIE colour coordinate for WLE mixed gelatin gel was found to be (0.26, 0.33) (Fig. 8b).
The prepared dry PVA film was kept with WLE mixture in water-ethanol (10:90) for swelling overnight. Subsequently, the swollen film was allowed to dry (Supplementary Fig. 6b). Fluorescence emission spectrum of dye incorporated PVA film shows two bands at 450 and 625 nm (Fig. 9a). A good CIE coordinate value (0.32, 0.25) was obtained from the corresponding emission spectrum (Fig. 9b).
Conclusions
In summary, we have generated white light emission from natural dyes extracted in our laboratory using a green and simple procedure. The optimized mixture of two suitably chosen plant extracts using acidic ethanol, aided by a FRET cascade from polyphenolics to curcumin to anthocyanins, generates almost pure white light, with CIE values of (0.35, 0.33) in solution, (0.26, 0.33) in gelatin gel and (0.33, 0.25) in PVA film. White light emission from such cheap and nature friendly resources could be important in the context of lighting and sensing application. It would be interesting to see if such system can be used as dyes for tunable dye laser applications. To the best of our knowledge this is the first time low cost, biocompatible (edible) natural dyes have been a part of white light emitting system. Given the vast number of excellent natural fluorescent dyes obtainable from renewable biosources, approaches similar to the present could lead to a more extensive range of low-cost and efficient WLE biomaterials with ease of adjusting colour temperature, which will obviate more expensive alternatives currently being pursued.
Materials and methods
Materials and Instruments
Pom, Tur and gelatine gel were purchased from local market of Chennai, India. Spectroscopic grade solvents were purchased from Sigma-Aldrich for extraction of dyes from natural resources. Ploy vinyl alcohol (PVA) (m.wt. approx 150000) was also purchased from Sigma-Aldrich for film preparation. UV-VIS electronic absorption spectra measurements were carried out in a Jasco V-650 UV-Visible Spectrophotometer with a scan rate of 1000 nm s−1. Fluorescence emission spectra of the samples were measured using Fluoromax-4 (Horiba Jobin Yvon) spectrofluorometer, with a xenon lamp of 150 W as excitation source. Excitation and emission monochromator band passes were kept at 5 nm and quartz cell cuvette (1 × 1 cm). CIE colour coordinates have been calculated using freely available online Osram Sylvania software51.
Method for Dye Extraction
Red Pom seeds were squeezed and the extract centrifuged at 5000 rpm. As reported37, the clear supernatant contained polyphenols and anthocyanins as major dye components. The extraction of Curcumin from Curcuma longa root (Tur) was carried out by grinding the rhizome with mortar and pestle using ethanol as extracting solvent. This extract was centrifugated for 5 minutes at 5000 rpm to obtain a clear yellow coloured supernatant that contained Curcumin as the major dye. These optimized methods of extraction are fairly simple and environment friendly.
Polymeric Film and Gel Preparation
Preparation of PVA Film
PVA dry film was prepared by dissolving 1 g in 10 ml of distilled water (10% w/v) at 60 °C. This solution was poured into flat-bottomed dishes in such a manner that a uniform thin layer of liquid covers the surface. This dish was kept in the oven at 80 °C and film was allowed to dry. The dry polymer film could be easily detached from the dish.
Preparation of Gelatin Solution
Gelatin gel was prepared by dissolving 1 g in 20 ml of distilled water (5% w/v) with constant stirring at 65 °C. This solution, when mixed with the dye extracts and set at 15 °C, forms the white emitting gel.
Additional Information
How to cite this article: Singh, V. and Mishra, A. K. White Light Emission from Vegetable Extracts. Sci. Rep. 5, 11118; doi: 10.1038/srep11118 (2015).
References
Gather, M. C., Kohnen, A. & Meerholz, K. White organic light-emitting diodes. Adv. Mater. 23, 233–248 (2011).
Fleetham, T., Ecton, J., Wang, Z., Bakken, N. & Li, J. Single-doped white organic light emitting device with an external quantum efficiency over 20%. Adv. Mater. 25, 2573–2576 (2013).
Reineke, S. et al. White organic light-emitting diodes with fluorescent tube efficiency. Nature 459, 234–238 (2009).
Jang, E. et al. White-light-emitting diodes with quantum dot color converters for display backlights. Adv. Mater. 22, 3076–3080 (2010).
Wang, J., Lin, W. & Li, W. Three-channel fluorescent sensing via organic white light-emitting dyes for detection of hydrogen sulfide in living cells. Biomaterials 34, 7429–7436 (2013).
Mukherjee, S. & Thilagar, P. Organic white-light emitting materials. Dyes pigments 111, 2–27 (2014).
Zhang, X., Gorl, D. & Wurthner, F. White-light emitting dye micelles in aqueous solution. Chem. Commun. 49, 8178–8180 (2013).
Sanju, K. S., Neelakandan, P. P. & Ramaiah, D. DNA-assisted white light emission through FRET. Chem. Commun. 47, 1288–1290 (2011).
Park, Y. I. et al. A new pH sensitive fluorescent and white light emissive material through controlled intermolecular charge transfer. Chem. Sci. DOI: 10.1039/c4sc01911c (2014).
Park, S. et al. A white-light-emitting molecule: frustrated energy transfer between constituent emitting centers. J. Am. Chem. Soc. 131, 14043–14049 (2009).
Molla, R. M. & Ghosh, S. Hydrogen-bonding-mediated J-aggregation and white-light emission from a remarkably simple, single-component, naphthalenediimide chromophore. Chem. Eur. J. 18, 1290–1294 (2012).
Liu, Y. et al. π - Conjugated aromatic enynes as a single-emitting component for white electroluminescence, J. Am. Chem. Soc. 128, 5592–5593 (2006).
Zhang, Y. et al. Employing heavy metal-free colloidal quantum dots in solution- processed white light-emitting diodes. Nano Lett. 11, 329–332 (2011).
Liang, R. et al. Quantum dots-based flexible films and their application as the phosphor in white light-emitting diodes. Chem. Mater. 26, 2595–2600 (2014).
Chen, K. J. et al. White light emitting diodes with enhanced CCT uniformity and luminous flux using ZrO2 nanoparticles. Nanoscale 6, 5378–5383 (2014).
Nicolai, H. T., Hof, A. & Blom, P. W. M. Device physics of white polymer light-emitting diodes. Adv. Funct.Mater. 22, 2040–2047 (2012).
Deng, C., Jiang, P., Shen, X., Ling, J. & Hogen-Esch, T. E. White light emission of multi-chromophore photoluminescent nanoparticles using polyacrylate scaffold copolymers with pendent polyfluorene groups. Polym. Chem. 5, 5109–5115 (2014).
Sun, C. Y. et al. Efficient and tunable white-light emission of metal–organic frameworks by iridium-complex encapsulation. Nat. Commun DOI: 10.1038/ncomms3717 (2013).
Tang, Q. et al. Color tuning and white light emission via in situ doping of luminescent lanthanide metal−organic frameworks. Inorg.Chem. 53, 289–293 (2014).
Yang, Q. Y. et al. Pure white-light and yellow-to-blue emission tuning in single crystals of Dy (III) metal–organic frameworks. Chem. Commun. 50, 7702–7704 (2014).
Wooseok, K., Li, J., Eda, G. & Chhowalla, M. Direct white light emission from inorganic–organic hybrid semiconductor bulk materials. J. Mater. Chem. 20, 10676–10679 (2010).
Sessolo, M., Tordera, D. & Bolink, H. J. Ionic iridium complex and conjugated polymer used to solution- process a bilayer white light-emitting diode. Appl. Mater. Interfaces 5, 630–634 (2013).
Ramya, A. R., Varughese, S. & Reddy, M. L. P. Tunable white-light emission from mixed lanthanide (Eu3+, Gd3+, Tb3+) coordination polymers derived from 4-(dipyridin-2-yl)- aminobenzoate. Dalton Trans. 43, 10940–10946 (2014).
Ledemi, Y. et al. White light and multicolor emission tuning in triply doped Yb3+/Tm3+/Er3+ novel fluoro-phosphate transparent glass-ceramics. J. Mater. Chem. C 2, 5046–5056 (2014).
Sanju, K. S. & Ramaiah, D. White photoluminescence and electroluminescence from a ternary system in solution and a polymer matrix. Chem. Commun. 47, 11626–11628 (2013).
Maiti, D. K., Bhattacharjee, R., Datta, A. & Banerjee, A. Modulation of fluorescence resonance energy transfer efficiency for white light emission from a series of stilbene-perylene based donor−acceptor pair. J. Phys. Chem. C 117, 23178–23189 (2013).
Babu, S. S. et al. Solvent-free luminescent organic liquids. Angew. Chem. Int. Ed. 51, 3391- 3395 (2012).
Vijayakumar, C., Praveen, V. K. & Ajayaghosh, A. RGB Emission through controlled donor self-assembly and modulation of excitation energy transfer: a novel strategy to white-light-emitting organogels. Adv. Mater. 21, 2059–2063 (2009).
Maity, D. K. & Banerjee, A. A peptide based two component white light emitting system. Chem. Commun. 49, 6909–6911 (2013).
Henry, B. S. Natural food colours, 2nd edn, Vol. 187 (eds Hendry, G. A. F. et al. ) Ch. 2, 40–78 (Chapman & Hall, 1996).
Caravaca, A. M. G. et al. Determination of the major phenolic compounds in pomegranate juices by HPLC−DAD−ESI-MS. J. Agric. Food Chem. 61, 5328–5337 (2013).
Iriel, A. & Lagorio, M. G. Biospectroscopy of Rhododendron indicum flowers. Non destructive assessment of anthocyanins in petals using a reflectance-based method. Photochem. Photobiol.Sci. 8, 337–344 (2009).
Agati, G., Matteini, P., Oliveira, J., De Freitas, V. & Mateus, N. Fluorescence approach for measuring anthocyanins and derived pigments in red wine. J. Agric. Food Chem. 61, 10156–10162 (2013).
Ignat, I., Volf, I. & Popa, V. I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chemistry 126, 1821–1835 (2011).
Anderson, A. M., Mitchell, M. S. & Mohan R. S. Isolation of curcumin from turmeric. J. Chem. Ed. 77, 359–360 (2000).
Martos, M. V., López,J. F. & Álvarez J. A. P. Pomegranate and its many functional components as related to human health: a review. Compr. Rev. Food Sci. Food Saf. 9, 635–654 (2010).
Hernández, F., Melgarejo, P. Tomás-Barberán, F. A. & Artés, F., Evolution of juice anthocyanins during ripening of new selected pomegranate (Punica granatum) clones. Eur Food Res Technol. 210, 39–42 (1999).
Willstatter, V. R. & Mallison, H. uber variationen der blutenfarben. Jus. L. Ann. Chem. 408, 147–162 (1915).
Fischer, U. A., Carle, R. & Kammerer, D. R. Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD-ESI/MSn. Food Chem. 127, 807–821 (2011).
1. Alighourchi, H., Barzegar, M. & Abbasi, S. Anthocyanins characterization of 15 Iranian pomegranate (Punica granatum L.) varieties and their variation after cold storage and pasteurization. Eur. Food Res. and Technol. 227, 881–887 (2008).
Cacace, J. E. & Mazza, G. Extraction of anthocyanins and other phenolics from black currants with sulfured water. J.Agric. Food Chem. 50, 5939–5946 (2002).
Revilla, E., Ryan, J.-M. & Martín-Ortega, G. Comparison of Several Procedures Used for the Extraction of Anthocyanins from Red Grapes. J.Agric. Food Chem. 46, 4592–4597 (1998).
Arapitsas, P. & Turner, C. Pressurized solvent extraction and monolithic column-HPLC/DAD analysis of anthocyanins in red cabbage. Talanta 74, 1218–1223 (2008).
Da Silva, P. F., Lima, J. C., Quina, F. H. & Macüanita, A. L. Excited-state electron transfer in anthocyanins and related flavylium salts. J. Phys. Chem. A 108, 10133–10140 (2004).
Giusti, M. M., Saona, L. E. R. & Wrolstad, R. E. Molar absorptivity and color characteristics of acylated and non-acylated pelargonidin-based anthocyanins. J. Agric. Food Chem. 47, 4631–4637 (1999).
Waranyoupalin, R. et al. Studies on complex formation between curcumin and Hg (II) ion by spectrophotometric method: A new approach to overcome peak overlap. Cent. Eur. J. Chem. 7, 388–394 (2009).
Erez, Y., Presiado, I., Gepshtein, R. & Huppert, D. The effect of a mild base on curcumin in methanol and ethanol. J. Phys. Chem. A 116, 2039–2048 (2012).
Smith, T. & Guild, J. The C.I.E. colorimetric standards and their use. Transactions of the Optical Society 33, 73–134 (2002).
Chignell, C. F. et al. Spectral and photochemical properties of curcumin. Photochem. photobiol 59, 295–302 (1994).
Figueiredo, P., Pina, F., Vilas-Boas, L. & Macanita, A. L. Fluorescence spectra and decays of malvidin 3,5-diglucoside in aqueous solutions. J. Photochem. Photobiol. A: Chem 52, 411–424 (1990).
For CIE co-ordinates software, see Osram Sylvania, (2013), available at: https://www.sylvania.com/en-us/tools-and-resources/Pages/LED-ColorCalculator-VersionHistory.aspx (accessed: 17 /11/ 2014).
Acknowledgements
A. K. M. thanks CSIR, New Delhi for financial support for this work through a sponsored project. V.S. is thankful to university grant commission (UGC) New Delhi for senior research fellowship and IIT Madras for providing the research facility.
Author information
Authors and Affiliations
Contributions
A.K.M. designed research; V.S. performed experiments; A.K.M. and V.S. analyzed data; A.K.M. wrote the main manuscript text and V.S. prepared all the figures and table. Both authors discussed the results and reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Singh, V., Mishra, A. White Light Emission from Vegetable Extracts. Sci Rep 5, 11118 (2015). https://doi.org/10.1038/srep11118
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep11118
This article is cited by
-
Green, affordable, and unprecedented photoluminescence investigation on white emission of Y2O3:Clitoria ternatea floral extract complex to replace conventional Dy3+ doping for wLED
Applied Nanoscience (2024)
-
Investigation of properties and applications of ZnO polymer nanocomposites
Polymer Bulletin (2023)
-
Structural and Optical Properties of Nanofibers Prepared with Electrospinning by Using PMMA Integrated with Curcuminoids to Produce White LED
Fibers and Polymers (2020)
-
White Light Emission from a Simple Mixture of Fluorescent Organic Compounds
Scientific Reports (2019)
-
Impedimetric Approach for Estimating the Presence of Metanil Yellow in Turmeric Powder from Tunable Capacitance Measurement
Food Analytical Methods (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.










 Polyphenolics emission band at 440 nm and
Polyphenolics emission band at 440 nm and  anthocyanin emission band at 585 nm,
anthocyanin emission band at 585 nm,  curcumin emission band at 522 nm,
curcumin emission band at 522 nm,  Changes of emission bands (1-6) with progressive addition of Tur extract (1-6 μM).
Changes of emission bands (1-6) with progressive addition of Tur extract (1-6 μM).
