Abstract
Understanding woolly mammoth ecology is key to understanding Pleistocene community dynamics and evaluating the roles of human hunting and climate change in late Quaternary megafaunal extinctions. Previous isotopic studies of mammoths’ diet and physiology have been hampered by the ‘mammoth conundrum’: woolly mammoths have anomalously high collagen δ15N values, which are more similar to coeval carnivores than herbivores and which could imply a distinct diet and (or) habitat, or a physiological adaptation. We analyzed individual amino acids from collagen of adult woolly mammoths and coeval species and discovered greater 15N enrichment in source amino acids of woolly mammoths than in most other herbivores or carnivores. Woolly mammoths consumed an isotopically distinct food source, reflective of extreme aridity, dung fertilization and (or) plant selection. This dietary signal suggests that woolly mammoths occupied a distinct habitat or forage niche relative to other Pleistocene herbivores.
Similar content being viewed by others
Introduction
Woolly mammoths (Mammuthus primigenius) were keystone herbivores in the Pleistocene mammoth steppe1,2. This megacontinental biome was inhabited by a now-extinct community of mammals, dominated by woolly mammoth, horse and bison. The mammoth steppe reached from north-western Canada, across the exposed Bering Isthmus, to Western Europe3. The ecological role of woolly mammoths within this ecosystem has been a subject of vigorous investigation3,4,5. Reconstructions of woolly mammoth behaviour and physiology have been largely based on morphology4. Isotopic studies of bulk tissues have provided independent tests of morphology-based hypotheses, as well as suggesting new ones3,6,7,8,9. Compound-specific isotopic studies can provide a further level of understanding of ecosystem functioning within the mammoth steppe.
Bulk collagen nitrogen isotopic compositions (δ15NBulk) are commonly used in ecological studies to reveal the diet and trophic level of a species, as these values typically reflect the isotopic compositions of the plants at the base of the food web plus a 2–5‰ increase with each trophic level10. As a result, the δ15NBulk values of mammoth-steppe herbivore collagen are commonly ~+6‰ where the values of carnivores (~+9‰) are higher, reflective of this trophic enrichment3. The carnivore- rather than herbivore-like δ15NBulk values of woolly mammoth collagen (~+8‰3) are seemingly problematic and therefore require examination. The various hypotheses to explain this phenomenon (unique diet, niche feeding in a special habitat or distinct metabolic processes3,6,7,8,9,11,12,13,14) have different implications for our understanding of the now-vanished mammoth steppe ecosystem, woolly mammoth ecology and related factors that contributed to extirpation of the woolly mammoth in this region.
Woolly mammoths may have consumed plants with higher δ15N values, such as graminoids and herbs rather than woody vegetation7,12,13, as suggested by the morphology of their enamel plates4. However, an herbaceous diet alone is not sufficient to fully explain the woolly mammoth’s high δ15N values; some further form of habitat or plant selection is also required15. While modern Arctic graminoids and forbs from some sites have a δ15N range of –0.3 to +10‰, the average value of these species ranges from ~+1 to ~+4‰16 and still other studies have reported maximum δ15N values for modern sedges of +2‰17 and for modern herbs of +5.3‰18. The majority of these plants, therefore, are not sufficiently enriched in 15N to explain the woolly mammoth δ15NBulk values. Plants growing in drier habitats, however, have higher δ15N values than plants from a more mesic environment7 and woolly mammoths may have eaten plants experiencing water-stress3,8. Water stress can also cause 13C-enrichment of plants19. This enrichment, however, is unlikely to be directly observable in woolly mammoth collagen, because its carbon isotopic composition is likely dominated by the low δ13C values of fat reserves used to survive the winter7.
Several other factors could also have contributed to high δ15N values for woolly mammoth collagen. Woolly mammoths that had small ranges, or repeatedly travelled the same routes, could have deposited significant quantities of faeces in those areas12, causing 15N-enrichment in plants arising from this dung fertilization20. Partially decayed plant material can also have higher δ15N values than the original living plant21. Woolly mammoths may have removed snow and ice cover by trampling and (or) with their tusks5, allowing them to forage on winter-killed plants generally not utilized by other large herbivores that did not share the mammoth ecological niche. It has also been proposed that woolly mammoths had distinct metabolic processes, such as increased levels of nitrogen recycling associated with winter starvation22,23 or poor quality food with low protein levels3,7,8,9,11,13,14,24, or that woolly mammoths engaged in coprophagy12.
The nitrogen isotopic compositions of the individual amino acids in collagen, as opposed to bulk collagen, enable discrimination between 15N-enrichment occurring at the base of the food chain prior to consumption (source amino acids) versus that associated with metabolic processes (trophic amino acids) (Fig. 1). Phenylalanine (Phe) and glutamate (Glu) have been identified as characteristic of source and trophic amino acids, respectively25,26. The δ15NPhe value reflects the isotopic composition of those amino acids in plants at the base of the food web, while the Δ15NGlu-Phe spacing (δ15NGlu – δ15NPhe) serves as a proxy for metabolic enrichment of 15N in the consumer’s body25.
Simplified pathway for nitrogen incorporation from soil to animal protein.
Arrows represent: uptake (dashed line), chemical transformations (solid line) and metabolic processes (solid curves). (a) Plant NO3– uptake. (b) NO3– converted to glutamine31. (c) NH4+ uptake. (d) NH4+ converted to glutamine by attachment to glutamate31. (e) Glutamine supplies nitrogen for synthesis of other amino acids. The associated shift in δ15N values depends on the specific amino acid, plant part and plant type31. (f) Consumption of amino acids by the animal. (g) Source amino acids are minimally involved in metabolic processes, undergoing small changes in δ15N values (e.g. increases in δ15NPhe values from diet to consumer tissue are commonly ≤ 2‰20). (h) Trophic amino acids are heavily involved in metabolic processes, undergoing enrichment in 15N (e.g. increases in δ15NGlu values from diet to consumer tissue are commonly 6–7‰20). Katherine Allan drew the images of grass and mammoth in Figure 1.
Results
Eight Pleistocene megafauna species were analyzed in this study. These included four herbivore species: woolly mammoth (Mammuthus primigenius), mastodon (Mammut americanum), horse (Equus sp.) and giant beaver (Castoroides ohioensis) and four carnivore species: brown bear (Ursus arctos), scimitar cat (Homotherium serum), wolf (Canis lupus) and short-faced bear (Arctodus simus) (see Supplementary Table S1). All samples were obtained from specimens collected near Old Crow, Yukon, Canada (latitude: 67°34’N; longitude: 139°48’W). A subset of these specimens was dated, including both herbivores and carnivores. Two horse specimens were dated to 18,370 and 27,180 14C years BP. The rest of the specimens yielded effectively non-finite radiocarbon dates ≥37,200 14C yr BP and one specimen was dated by context to ~140,000 years BP (see Supplementary Table S1)27,28. All collagen samples were considered well preserved based on their collagen yields, C/N ratios and carbon and nitrogen contents29 (see Supplementary Table S2).
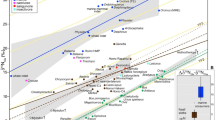
The δ15NBulk values for the Old Crow samples follow the pattern previously observed for Pleistocene megafauna3,8,11; woolly mammoth collagen generally has δ15NBulk values similar to the carnivores and higher than the other herbivores, with some overlap with horses (Fig. 2a). The δ15NPhe values of woolly mammoth collagen, however, are higher than those of the carnivores and most of the other herbivores (Fig. 2b); horses with high δ15NBulk values for collagen show the most overlap with the δ15NPhe values of woolly mammoths. Woolly mammoth Δ15NGlu-Phe spacings overlap those of the other herbivores but are lower than the Δ15NGlu-Phe spacings of the carnivores, extending to negative values for most samples (Fig. 2c). Negative Δ15NGlu-Phe spacings have observed in terrestrial herbivores previously25,30 and may be the result of relatively high δ15NPhe values in vascular plants31.
Nitrogen isotopic compositions of Pleistocene Old Crow megafauna.
(a) Bulk collagen nitrogen isotopic compositions (δ15NBulk). Results for woolly mammoths are displayed in blue, other herbivores in purple and carnivores in red. (b) Phenyalanine (source) amino acid nitrogen isotopic compositions (δ15NPhe) of each species. (c) Difference between the nitrogen isotopic composition of glutamate and phenylalanine (Δ15NGlu-Phe) for each species.
Discussion
The high δ15NPhe values of the woolly mammoth imply that they selectively consumed plants more enriched in 15N than forage consumed by most of the other herbivores. The fact that the δ15NPhe values of woolly mammoths are higher than those of carnivores suggests that the latter consumed herbivores subsisting on less 15N-rich forage than consumed by woolly mammoths. In short, the carnivores did not consume significant quantities of woolly mammoth. Partial overlap between horse and woolly mammoth δ15NBulk and δ15NPhe values likely indicates that horses exploited a similar niche to the woolly mammoth in some cases. The low Δ15NGlu-Phe spacings of woolly mammoths indicate that their δ15NBulk values arise from the higher δ15N values of the plants they consumed and not from a specialized metabolic process.
It seems that woolly mammoths occupied a specialized dietary or habitat niche. A dietary niche implies that woolly mammoths selected particular herbaceous plants or consumed large quantities of decayed plants in winter, while a habitat niche suggests that woolly mammoths occupied more arid habitats, or lived in distinct ranges where they left considerable quantities of dung that fertilized the plants growing there. While some Old Crow horses appear also to have exploited such a niche, it was not generally shared by other mammoth steppe megafauna. The Old Crow samples likely represent various time intervals through the late Pleistocene and potentially varied climate regimes. The fact that the relative differences in average δ15NBulk values among herbivore species are generally consistent across the mammoth steppe3 suggests that most herbivore species ate the same forage types regardless of climatic differences or time period. This implies that mammoth steppe herbivores targeted specific forage types.
Two significant conclusions arise from these observations. First, the woolly mammoth occupied a distinct niche from other contemporaneous herbivores. This unique habitat or forage existed across the entirety of the mammoth steppe, although its size may have varied with changing climate across geographic or temporal zones. Other evidence of the woolly mammoth’s dependence on a specialized niche may be provided by the retraction of woolly mammoth populations into small, isolated refugia during the last interglacial warm period (MIS 5e, 130-116 kyr BP) and the subsequent re-expansion upon return to glacial conditions32. An investigation of the isotopic compositions of woolly mammoths from a variety of time periods and sites across the mammoth steppe could reveal the extent of adaptability of the woolly mammoth to disruptions in its niche, such as may have occurred with the onset of climatic shifts at the end of the Pleistocene33.
Second, the horse δ15NBulk and δ15NPhe values overlap those of the woolly mammoth and the other herbivores. This implies that horses fed from a wide diversity of habitats or forage types, including the woolly mammoths’ niche. Such behaviour would suggest that the mammoth steppe ecosystem supported herbivores with a variety of ecological adaptations and that even in the Pleistocene Arctic, resources were sufficiently abundant to support both specialist and generalist strategies.
Methods
Radiocarbon dating
Radiocarbon dates for six woolly mammoths discussed here have been published previously27,28. A further subset of samples was dated for this study; these included two other herbivores and two carnivores (see Supplementary Table S1). Collagen was extracted, combusted, graphitized and radiocarbon dated at the University of Arizona Accelerator Mass Spectrometry (AMS) Laboratory. All dates are presented as uncalibrated radiocarbon years before present (mean ±1SD).
Bulk collagen nitrogen isotope (δ 15NBulk) analysis
Collagen for δ15NBulk analysis was extracted at the Laboratory for Stable Isotope Science (LSIS) following previously published methods28, or was previously extracted and analyzed for another study27,28 (see Supplementary Table S2). The δ15NBulk values were obtained using a Costech Elemental Combustion System (ECS 4010) attached to a ThermoFisher Delta Plus XL IRMS or to a ThermoFisher Delta V Plus IRMS. The δ15NBulk values were measured over three analytical sessions. In the first two analytical sessions, the δ15N values were calibrated to AIR using USGS40 (L-glumatic acid; accepted value –4.52‰34) and IAEA-N2 (ammonium sulfate; accepted value +20.3‰35), while the third analytical session substituted USGS41 (L-glumatic acid; accepted value +47.57‰34) for IAEA-N2. The same standards were used to create calibration curves for determining carbon and nitrogen contents (wt%) of each sample, from which C/N ratios were calculated. Keratin (MP Biomedicals Inc., Cat No. 90211, Lot No. 9966H) was used as an internal standard in each analytical session. For a total of 18 keratin measurements over the three analytical sessions, average values (mean ±1 SD) were δ15N = +6.4 ± 0.2‰ (accepted value = +6.4‰) and C/N = 3.6 ± 0.4 (accepted value = 3.7). The standard deviation of a sample analyzed as an instrumental duplicate was δ15NBulk = ±0.0‰ and C/N = ±0.1 (1 SD). The standard deviations (1 SD) for method duplicates of δ15NBulk values ranged from 0.0 to 0.2‰ and for C/N ratios, from 0.0 to 0.3. All samples were considered to be well preserved based on their extraction yields, C/N ratios and carbon and nitrogen contents29,36. Eight samples had high carbon and/or nitrogen contents, but this anomaly likely arises from a weighing error as they were well preserved by other measures.
Amino acid nitrogen isotope (δ 15NAmino Acid) Analysis
Using collagen first extracted for δ15NBulk measurements, amino acids were hydrolysed, derivatized to their N-acetyl-methyl ester derivative and their individual δ15NAmino Acid values measured using an Aligent 6890N-ThermoFisher Gas Chromatograph-Combustion 3-ThermoFisher Delta Plus XL IRMS. An Agilent Technologies VF-23MS column was used in the GC. We followed published methods25,37 with only slight modifications: (i) the quantity of collagen hydrolysed was increased from 2 to 6 mg and the quantity derivatized was increased from 0.25 to 1.5 mg; and (ii) the initial GC column temperature was set at 60 °C instead of 40 °C and its final temperature of 250 °C was held for 15 min instead of 20 min. A representative chromatogram is shown in Supplementary Fig. S1. Three reference gas pulses were introduced into the IRMS at the beginning of each analytical session and one pulse was introduced at the end of each session. The isotopic composition of the reference gas was calibrated using four amino acid standards. Three of these standards, alanine, leucine and phenylalanine, were purchased as their NACME derivative from Sigma Aldrich. The fourth, proline, was purchased as an amino acid and derivatized in-house. The nitrogen isotopic compositions of the derivatized standards were established by multiple measurements performed in the same manner as used for isotopic analysis of bulk collagen and calibration to AIR using international standards. The amino acid reference standards were injected every three to five runs. All samples were analyzed a minimum of three times and the average variation was ±0.7‰ (1 SD) for phenyalanine and ±0.7‰ (1 SD) for glutamate, with a range of 0.0–3.9‰. An internal standard, norleucine, was also analyzed. Its nitrogen isotopic compositions were offset from expected values by an average of +1.3‰.
Additional Information
How to cite this article: Schwartz-Narbonne, R. et al. Solving the woolly mammoth conundrum: amino acid 15N-enrichment suggests a distinct forage or habitat. Sci. Rep. 5, 09791; doi: 10.1038/srep09791 (2015).
References
Owen-Smith, N. Pleistocene extinctions: the pivotal role of megaherbivores. Paleobiology 13, 351–362 (1987).
Zimov, S. et al. Steppe-tundra transition: a herbivore-driven biome shift at the end of the Pleistocene. Am. Nat. 146, 765–794 (1995).
Bocherens, H. Isotopic biogeochemistry and the paleoecology of the mammoth steppe fauna. Deinsea 9, 57–76 (2003).
Haynes, G. Mammoths, Mastodonts and Elephants. 413 Cambridge University Press, 1991).
Putshkov, P. The impact of mammoths in their biome: clash of two paradigms. Deinsea 9, 365–379 (2003).
Bocherens, H., Fizet, M. & Mariotti, A. Diet, physiology and ecology of fossil mammals as inferred from stable carbon and nitrogen isotope biogeochemistry: implications for Pleistocene bears. Palaeogeogr. Palaeoclimatol. Palaeoecol. 107, 213–225 (1994).
Szpak, P. et al. Regional differences in bone collagen δ13C and δ15N of Pleistocene mammoths: implications for paleoecology of the mammoth steppe. Palaeogeogr. Palaeoclimatol. Palaeoecol. 286, 88–96 (2010).
Iacumin, P., Nikolaev, V. & Ramigni, M. C and N stable isotope measurements on Eurasian fossil mammals, 40 000 to 10 000 years BP: herbivore physiologies and palaeoenvironmental reconstruction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 163, 33–47 (2000).
Iacumin, P., Di Matteo, A., Nikolaev, V. & Kuznetsova, T. Climate information from C, N and O stable isotope analyses of mammoth bones from northern Siberia. Quat. Int. 212, 206–212 (2010).
Koch, P., Fogel, M. & Tuross, N. in Stable Isotopes in Ecology and Environmental Science (eds Lajtha, K. & Michener, R. ) Ch. 4, 63–92 Blackwell Scientific Publications, 1994).
Fox-Dobbs, K., Leonard, J. & Koch, P. Pleistocene megafauna from eastern Beringia: paleoecological and paleoenvironmental interpretations of stable carbon and nitrogen isotope and radiocarbon records. Palaeogeogr. Palaeoclimatol. Palaeoecol. 261, 30–46 (2008).
Metcalfe, J. Z., Longstaffe, F. J. & Hodgins, G. Proboscideans and paleoenvironments of the Pleistocene Great Lakes: landscape, vegetation and stable isotopes. Quat. Sci. Rev. 76, 102–113 (2013).
Koch, P. The isotopic ecology of Pleistocene proboscideans. J. Vertebr. Paleontol. 11, 40A (1991).
Bocherens, H., Pacaud, G., Lazarev, P. A. & Mariotti, A. Stable isotope abundances (13C, 15N) in collagen and soft tissues from Pleistocene mammals from Yakutia: implications for the palaeobiology of the Mammoth Steppe. Palaeogeogr. Palaeoclimatol. Palaeoecol. 126, 31–44 (1996).
Matheus, P., Guthrie, R. & Kunz, M. Isotope ecology of late Quaternary megafauna in eastern Beringia. Paper presented at Third International Mammoth Conference, Whitehorse, Yukon, Canada. Whitehorse: Government of Yukon, Heritage Branch, . 2003, May 18-20.
Barnett, B. Carbon and nitrogen isotope ratios of caribou tissues, vascular plants and lichens from northern Alaska. Master’s Thesis. University of Alaska Fairbanks. 171 pp. (1994).
Nadelhoffer, K., Shaver, G., Fry, B. & Giblin, A. 15N natural abundances and N use by tundra plants. Oecologia 107, 386–394 (1996).
Ben-David, M., Shochat, E. & Adams, L. Utility of stable isotope analysis in studying foraging ecology of herbivores: examples from moose and caribou. Alces 37, 421–434 (2001).
Wooller, M. et al. Stable carbon isotope compositions of Eastern Beringian grasses and sedges: investigating their potential as paleoenvironmental indicators. Arctic, Antarct. Alp. Res. 39, 318–331 (2007).
Szpak, P., Millaire, J., White, C. & Longstaffe, F. J. Influence of seabird guano and camelid dung fertilization on the nitrogen isotopic composition of field-grown maize (Zea mays). J. Archaeol. Sci. 39, 3721–3740 (2012).
Tremblay, L. & Benner, R. Microbial contributions to N-immobilization and organic matter preservation in decaying plant detritus. Geochim. Cosmochim. Acta 70, 133–146 (2006).
Polischuk, S., Hobson, K. & Ramsay, M. Use of stable-carbon and -nitrogen isotopes to assess weaning and fasting in female polar bears and their cubs. Can. J. Zool. 79, 499–511 (2011).
Hobson, K. A., Alisauskas, R. T. & Clark, R. G. Stable-nitrogen isotope enrichment in avian tissues due to fasting and nutritional stress: implications for isotopic analyses of diet. Condor 95, 388–394 (1993).
Bocherens, H., Fizet, M., Mariotti, A., Gangloff, R. & Burns, J. Contribution of isotopic biogeochemistry (13C, 15N, 18O) to the paleoecology of mammoths (Mammuthus primigenius). Hist. Biol. 7, 187–202 (1994).
Styring, A. K., Sealy, J. C. & Evershed, R. P. Resolving the bulk δ15N values of ancient human and animal bone collagen via compound-specific nitrogen isotope analysis of constituent amino acids. Geochim. Cosmochim. Acta 74, 241–251 (2010).
McClelland, J. & Montoya, J. Trophic relationships and the nitrogen isotopic composition of amino acids in plankton. Ecology 83, 2173–2180 (2002).
Metcalfe, J. Z. Late Pleistocene climate and proboscidean paleoecology in North America: insights from stable isotope compositions of skeletal remains. Doctoral Thesis. The University of Western Ontario. 277 pp. (2011).
Metcalfe, J. Z., Longstaffe, F. J. & Zazula, G. D. Nursing, weaning and tooth development in woolly mammoths from Old Crow, Yukon, Canada: implications for Pleistocene extinctions. Palaeogeogr. Palaeoclimatol. Palaeoecol. 298, 257–270 (2010).
Van Klinken, G. Bone collagen quality indicators for palaeodietary and radiocarbon measurements. J. Archaeol. Sci. 26, 687–695 (1999).
Chikaraishi, Y., Ogawa, N. O., Doi, H. & Ohkouchi, N. 15N/14N ratios of amino acids as a tool for studying terrestrial food webs: a case study of terrestrial insects (bees, wasps and hornets). Ecol. Res. 26, 835–844 (2011).
Styring, A. K., Fraser, R. A., Bogaard, A. & Evershed, R. P. Cereal grain, rachis and pulse seed amino acid δ15N values as indicators of plant nitrogen metabolism. Phytochemistry 97, 20–29 (2014).
Palkopoulou, E. et al. Holarctic genetic structure and range dynamics in the woolly mammoth. Proc. R. Soc. B. 208, 20131910 (2013).
Willerslev, E. et al. Fifty thousand years of Arctic vegetation and megafaunal diet. Nature 506, 47–51 (2014).
Qi, H., Coplen, T. B., Geilmann, H., Brand, W. & Böhlke, J. K. Two new organic reference materials for δ13C and δ15N measurements and a new value for the δ13C of NBS 22 oil. Rapid Commun. Mass Spectrom. 17, 2483–2487 (2003).
Brand, W., Coplen, T., Vogl, J., Rosner, M. & Prohaska, T. Assessment of international reference materials for isotope-ratio analysis (IUPAC Technical Report). Pure Appl. Chem. 86, 425–467 (2014).
Ambrose, S. Preparation and characterization of bone and tooth collagen for isotopic analysis. J. Archaeol. Sci. 17, 431–451 (1990).
Corr, L., Berstan, R. & Evershed, R. Development of N-acetyl methyl ester derivatives for the determination of δ13C values of amino acids using gas chromatography-combustion- isotope ratio mass spectrometry. Anal. Chem. 79, 9082–90 (2007).
Acknowledgements
We thank Kim Law, Li Huang, Nadia Dildar, Joanne Potter and Karyn Olsen for technical advice. Samples were collected with the permission of the American Museum of Natural History and the Yukon Government Palaeontology Program. Greg Hodgins and the staff at the NSF Arizona AMS Laboratory assisted with the radiocarbon dating. Funding was provided by a Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant (F.J.L.) and an NSERC Canada Graduate Scholarship (R.S.N., J.Z.M), an NSERC Postgraduate Scholarship (R.S.N.) and an Ontario Graduate Scholarship (R.S.N.). Laboratory infrastructure was funded by the Canada Foundation for Innovation (F.J.L.) and the Ontario Research Fund (F.J.L.). Additional time for research was funded through the Canada Research Chairs program (F.J.L.). This is Laboratory for Stable Isotope Science Contribution #322.
Author information
Authors and Affiliations
Contributions
R.S.N. conducted the amino acid isotopic analyses, interpreted the data and wrote the initial manuscript; R.S.N. and F.J.L. conceived the study, completed the data reduction and revised the manuscript; F.J.L. and J.Z.M. participated in interpretation and discussion; R.S.N., J.Z.M. and F.J.L. conducted the sampling; J.Z.M. and G.Z. provided substantive comments on the manuscript; G.Z. provided access to samples; F.J.L. provided funding to support the research.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Schwartz-Narbonne, R., Longstaffe, F., Metcalfe, J. et al. Solving the woolly mammoth conundrum: amino acid 15N-enrichment suggests a distinct forage or habitat. Sci Rep 5, 9791 (2015). https://doi.org/10.1038/srep09791
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep09791
This article is cited by
-
Giant beaver palaeoecology inferred from stable isotopes
Scientific Reports (2019)
-
Isotopic analyses suggest mammoth and plant in the diet of the oldest anatomically modern humans from far southeast Europe
Scientific Reports (2017)
-
Central European Woolly Mammoth Population Dynamics: Insights from Late Pleistocene Mitochondrial Genomes
Scientific Reports (2017)
-
‘Trophic’ and ‘source’ amino acids in trophic estimation: a likely metabolic explanation
Oecologia (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.