Abstract
Selective actuation of a single microswimmer from within a diverse group would be afirst step toward collaborative guided action by a group of swimmers. Here wedescribe a new class of microswimmer that accomplishes this goal. Our swimmer designovercomes the commonly-held design paradigm that microswimmers must usenon-reciprocal motion to achieve propulsion; instead, the swimmer ispropelled by oscillatory motion of an air bubble trapped within theswimmer's polymer body. This oscillatory motion is driven by theapplication of a low-power acoustic field, which is biocompatible with biologicalsamples and with the ambient liquid. This acoustically-powered microswimmeraccomplishes controllable and rapid translational and rotational motion, even inhighly viscous liquids (with viscosity 6,000 times higher than that of water). Andby using a group of swimmers each with a unique bubble size (and resulting uniqueresonance frequencies), selective actuation of a single swimmer from among the groupcan be readily achieved.
Similar content being viewed by others
Introduction
The physics of swimming at the microscale1, where viscous forces dominateover inertial effects, is distinct from that at the macroscale2,3.Devices capable of finely controlled motion at the microscale could enable bold ideassuch as targeted drug delivery4,5, non-invasive microsurgery6,7 and precise materials assembly8,9,10,11. Artificialmicroswimmers and nanomotors have been intensively developed over the past decade in anattempt to achieve controlled, powered, autonomous motion at the micro- andnanoscales12,13,14. Autonomous swimmers need to harvest energy fromtheir environment and transduce it to mechanical form. For example, chemical energy canbe harvested by bimetallic nanomotors that move by self-electrophoresis, decomposing afuel such as hydrogen peroxide asymmetrically over their surfaces15,16,17,18,19. Motion can be modulated by chemical or opticalgradients20, local analyte concentrations21, or localelectrochemical control22. Micron-scale swimmers also can be powered,assembled, or steered by magnetic fields23,24,25,26,27, electricfields28, optical excitation29, acousticscattering30,31,32,33,34, or thermal gradients35.Catalytically driven propulsion within more complex, confined reaction geometries isalso possible36,37, as is generation of motion from the reorganizationof hydrophobic/hydrophilic interactions within mobile microporous hosts38, or through quasi-oscillatory bursting at smaller length scales39.
Autonomous microswimmers are particularly compelling in biological or biomedicalcontexts. However, propulsion in a complex fluid medium (i.e., highly viscous ornon-Newtonian fluids) and motion against high flow rates has remained a significantchallenge. In addition, many of the existing swimmers that use electric fields orchemical/electrochemical fuels cannot be used in biological environments such as thehuman body. More importantly, the prevailing goal of selective actuation of asingle microswimmer from within a group—the first step towards collaborativeaction by a group of swimmers—has so far not been achieved. Here wedemonstrate a new class of acoustic microswimmers that move through aqueous solutiondriven by ultrasonically powered bubble oscillation engines. These acousticmicroswimmers respond to ambient acoustic energy and do not need to gather chemicalfuel from their environment. The means of implementing this propulsion mechanism areextremely simple and the applied acoustic fields are in the similar power intensityrange as those used in ultrasonic imaging, which has proven to be a highlybiocompatible, gentle technique. Furthermore, by creating bubbles of different sizes(and different resonance frequencies), selective actuation of a single swimmer fromamong the group can readily be achieved—a first in the field.
Results
Fabrication of the acoustic microswimmer
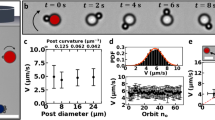
We fabricate the microswimmer using straightforward ultravioletphotopolymerization followed by chemical treatment to make the polymer surfaceshydrophobic (see Methods). The PEG/photo-initiator mixture was sandwichedbetween two glass slides; these slides were coated with PDMS to enable easyremoval of the swimmer bodies once cured. The two slides were separated by 150or 250 µm spacers, which determined the length of the swimmer body.Photomasks of different geometries (designed with AutoCAD software) were printedat 20,000 dpi resolution (CAD/Art Services, California). The mask was theninserted to the field stop of an inverted microscope (Nikon TE-2000U). A mercurylamp was used as the UV light source. A filter cube (11000v2: UV, Chroma)selected light of 373 µm wavelength. A shutter system, controlled byNIS software, adjusted the duration of UV exposure. The polymerization setup isshown schematically in Fig. 1a. The indentation diameterwas controlled by the photomask, with a small amount of variability introducedby the UV exposure duration, as illustrated in Fig. 1b.Indentation depth was controlled by the UV exposure duration; the depthdecreases with longer UV exposure, as illustrated in Fig.1c. Conical indentation was due to defocusing of UV light across thethickness of the PEG/photoinitiator mixture, which leads to polymerization ofthe surrounding mixture.
Fabrication and design of microswimmers.
(a), Schematic of the fabrication setup. PEG solution containingphotosensitive initiator is sandwiched between glass slides. Theswimmers' geometries and the conical shaped indents were createdby exposing the oligomer solution to UV light passing through a maskcontaining the blueprint of the swimmers. (b), Indentation diameter versusUV exposure time. (c), Indentation depth versus UV exposure time. (d),Images showing the decrease in indentation depth for increasing UV exposuretime.
Mechanism of the acoustic microswimmer
The acoustic microswimmer consists of a rectangular polymer body with one or moreconical indentations, as shown in Fig. 2a. When themicroswimmer is submerged in the liquid-filled chamber, an air bubble canspontaneously become trapped in each of its indentations. A piezoelectrictransducer mounted to a glass slide adjacent to the chamber generates theacoustic field, as shown in Fig. 2b (see experimentaldetails in the Methods section). The acoustic cell is designed with absorbingwalls to define a predominately traveling-wave acoustic field and avoidcomplications from the complex nodal structures of standing waves. When thetrapped bubble is exposed to an acoustic field with a wavelength much largerthan bubble diameter, its resulting oscillations induce a steady flow fieldaround itself at a length scale comparable to bubble size. These oscillationsare tracked photographically in Fig. 2c and the resultingflow field is shown in Fig. 2d. When the frequency of thefunction generator driving the transducer approaches a resonance of the trappedbubble, the oscillation amplitude of the liquid-air interface reaches a maximum.We exploit this phenomenon to achieve addressable self-propulsion.
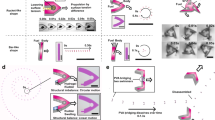
Geometry and experimental design of the acoustic microswimmers.
(a) Fluorescent images of four types of swimmer: linear microswimmers with asingle (false-coloured yellow) or double (red) indent that is symmetricabout the central axis, rotational microswimmers with off-centred (purple)indent and directional microswimmers with (green) indents of differentdiameter. (b), A piezoelectric transducer injects acoustic energy into achamber that is filled with fluid, lined with acoustically-absorbent putty,and enclosed on top and bottom by glass slides. (c), An image sequencerecorded at 360,000 frames per second showing bubble oscillation within theconical indentation, fitted to a sine function. (d), Acoustic oscillation ofthe microswimmer bubbles generates substantial acoustic microstreaming inwater. Both ends of indentations are open.
Bubble size and shape determine the resonance frequency; we control these bychanging the indentation diameter (from 50 to 100 microns), the indentationdepth (from 70 microns to the full length of the swimmer) and the duration ofthe hydrophobic treatment. The symmetry of the bubble position(s) within themicroswimmer determines the type of motion, as shown in Fig.2a. One or two symmetric indentations yield the translational motionshown in Fig. 3a,b and Supplementary Videos1 and 2. Asymmetric or off-centre indentationsproduce the rotational motion of Fig. 3c,d,e and Supplementary Videos 3, 4 and 5. The exposed bubble surface at the interface with the fluidis important for propulsion and is independent of the shape (conical orcylindrical) of the bubble trapped. The motive force and moment (i.e.,torque) created by an acoustically-driven bubble is determined by the intensityof the ambient acoustic field, which is controlled by adjusting the voltageapplied to the piezoelectric transducer. The propulsive force or moment can bemade large enough to achieve very high translational velocities: up to ~ 8mm/sec, which is ~ 50 body lengths per second. Rotational velocities likewisecan be as large as ~ 20 rotations per second in water. The swimmer speed dropsthree orders of magnitude in 50% glycerol/water and speed in viscous hydrogel(discussed later) is much lower but still substantial: 50 µm/s or 3rotations per minute. The acoustic microswimmer rapidly attains its steady-statespeed (see Supplementary Information 1).
High-speed imaging captures the translational and rotation motion of acousticmicroswimmers moving through either a water/microbead mixture orhydrogel.
(a), A single on-centre bubble generates linear motion in water, as does (b),a pair of bubbles of equal size symmetrically placed. An off-centreindentation generates either (c), clockwise or (d), counterclockwise motion.(e), The same rotary motion (or linear motion, not shown) can also beachieved in viscous shear-thinning hydrogel.
In the classic paper “Life at low Reynolds number”, Purcellproposed the scallop theorem: reciprocal motion of a swimmer submerged in fluidat low Reynolds number yields no net motion through the fluid1.In accord with this theorem, natural microswimmers such as sperm or bacteriapropel themselves by means of non-reciprocal motions of flagella, cilia, orother appendages. Artificial microswimmers inspired by biology have likewisetypically sought to achieve non-reciprocal motion. We pursue a differentstrategy here: while our acoustically driven microbubble swimmer does move asa whole at relatively low Reynolds number, the reciprocal oscillationswithin its bubble engine work at moderately high Reynolds number (9< Re < 90 for 0.5 μm < ε< 4 μm) and exploit the nonlinear inertia of fluiddynamics from high-frequency ultrasound. The applied acoustic wave has awavelength on the order of a centimetre, two orders of magnitude larger than themicroswimmer, thus the swimmer is subject to nearly uniform fluid pressure onall sides. This uniform acoustic environment is confirmed by the lack of motionin bubble-free microswimmers that are immersed in an acoustic field of varyingfrequency and amplitude (see Supplementary Information2).
We begin with a high-level discussion of the different potential contributions tothe acoustic propulsion40 in the physical regime of the acousticmicroswimmer, so that we can extract the critical scaling relations that willenable quantitative empirical analysis. The fundamental fluid mechanics fields,density ρ, pressure p and velocity u all haveincident, scattered and streaming components, where the streaming contributionis defined at the zero-frequency component41,42,43. Acousticpropulsion can arise from either acoustic microstreaming44,45,46,47,48,49,50,51,52 or radiation pressure, sinceboth can carry momentum to infinity. The stress tensor Tjj =pδij +ρuiuj can be averaged over theperiod of oscillation to yield  , whichcan then be decomposed into second-order radiative and microstreamingcontributions arising from combinations of incident (i) and scattered(s) fields:
, whichcan then be decomposed into second-order radiative and microstreamingcontributions arising from combinations of incident (i) and scattered(s) fields:  . The propulsiveforce corresponds to the integral of the divergence of this stress tensor overthe region surrounding the microswimmer, out to a surfaceS∞ located at infinity (and remembering thatthe stress tensor associated with the incident plane-wave excitation isdivergence-free). Taking
. The propulsiveforce corresponds to the integral of the divergence of this stress tensor overthe region surrounding the microswimmer, out to a surfaceS∞ located at infinity (and remembering thatthe stress tensor associated with the incident plane-wave excitation isdivergence-free). Taking  to be anoutward normal (i.e., pointing away from the microswimmer), we obtain thepropulsive force
to be anoutward normal (i.e., pointing away from the microswimmer), we obtain thepropulsive force  :
:

The first term here arises from acousticmicrostreaming: this effect has been considered before in the context of pinnedbubbles on substrates, as discussed below. The second term arises from theradiative momentum flux. It has two contributions, one from the scattered fieldalong and the other with contributions from both incident and scatteredfields:

The radiative propulsiveforce will be proportional to the square of the amplitude of the acoustic field(s). Although the (is) and (ss) terms have different contributionsfrom incident and scattered waves, the fact that the scattered wave is a linearresponse to the incident wave implies that the overall radiation-derivedacoustic force at fixed frequency for a given bubble is effectively proportionalto the square of the observed amplitude of the bubble oscillation in both cases.The underlying linear dependence of the (is) term on bubble amplitudecould be revealed by holding the incident wave amplitude fixed and insteadsweeping its frequency across the resonant response peak of the bubble. Sincethe incident wave has larger amplitude, it is reasonable to suppose that thatthe (is) term dominates. Since the (is) term is essentially aninterference term between the incident plane wave and the scattered wave, thiscontribution to the propulsion should vary depending on the orientation of themicroswimmer with respect to the wavevector of the incident plane-wave acousticexcitation. This contribution is also dependent on the acoustic excitationhaving some degree of standing wave character, since the time average of the(is) term would be zero for a pure traveling wave passing over amicroswimmer that is much smaller than the acoustic wavelength.
We now turn our attention to the acoustic microstreaming contribution topropulsion. For purposes of defining an axis of streaming, the bubble must beembedded within an asymmetric acoustic environment. The simplest such asymmetrythat one can consider is a superposition of spherically symmetric oscillationsof the bubble radius of amplitude ε plus transverseoscillations of the bubble centre up and down along a given axis of amplitudeε. Longuet-Higgins showed that a spherical bubble in anunbounded Newtonian fluid engaged in a superposition of radial and transverseoscillations at frequency ω produces a second-order steadyflow that scales as εε and is linear inω53. If the ratio of the radial andtransverse oscillations is fixed, then can be simplified to u∝ ε2ω. Marmottantand Hilgenfeldt extended this result to a bubble oscillating near a wall,finding a toroidal steady flow whose symmetry can be broken by a nearbystructural asymmetry to yield net fluid flow54,55. The quadraticscaling in ε and linear scaling in ω arepreserved in these lower-symmetry situations and similarly should be preservedin our more complex microswimmer geometry, for example, the bubble exposed tofluid on only one side within an indentation. For such a trapped bubble,oscillating with amplitude ε, the flow field around the bubbleis u = εu1 +ε2u2 and in a Newtonianfluid of density ρ, the oscillatory first-orderu1induces a second-order steady flow⟨u2⟩, governed by an Stokes equationwith body force⟨−ρu1·∇u1⟩,which is nonlinear in u1. For completeness, we also note thatin certain cases, the scattered wave can dissipate so rapidly in space thatsubstantial momentum is directly deposited into the ambient fluid to create aradiatively driven streaming flow known as a “quartzwind”. However, in aqueous solution this dissipative mechanism isgenerally significant only at much higher frequencies56 thanthose (kHz) used in this work.
The resonant response of the bubble oscillation amplitude as a function offrequency is mapped out in Fig. 4 with ±1μm accuracy. The resonance is reasonably sharp and for typicalexcitation amplitudes a swimmer that begins at rest will move only when excitedclose to resonance. For air bubbles in water, viscous damping in the form ofacoustic microstreaming dominates over radiation and thermal damping41. Using direct high-speed measurements of the fluid/bubbleinterface, we observed that the amplitude of the bubble oscillation is linearlyproportional to the amplitude V0 of the voltage applied to thesignal generator in water (see Supplementary Information 3).Thus for a given bubble configuration and at a fixed excitation frequency, theacoustic microstreaming and hence swimmer speed derived from bubbleoscillations, should scale as V02 inwater, irrespective of whether the force originates in microstreaming orradiation. Fig. 5a shows that this quadratic relation isreasonably well satisfied by the bubble-powered microswimmer moving in water;the slight deviation from a precise quadratic relation is not surprising,considering that the centre of mass motion of the microswimmer in water is atthe edge of the low Reynolds number regime Re ≤ 1 (see Supplementary Information 4) and the microswimmer carries acomplex acoustic streaming flow pattern around it. The behaviour in 50% glycerolsolution (Fig. 5b) is also reasonably close to theanticipated quadratic behaviour.
Frequency dependence of bubble oscillation amplitude.
The bubble oscillation is largest when the acoustic driving field is resonantwith the fundamental natural frequency of the bubble. The resonance peak fora bubble of diameter 45 µm in water is reasonably narrow.Corresponding to a quality factor Q ~ 25.
Characterization of the acoustic microswimmers.
(a), An acoustic microswimmer immersed in water moves at a speed nearlyproportional to the square of the amplitude of the drive voltage,i.e., the square of the amplitude of the incident acoustic field.This dependence is consistent with the acoustic coupling to motility. (b),An acoustic mciroswimmer immersed in a more viscous solution, 50% glycerol,exhibits similar scaling, with a slightly higher slope. (c), Within theshear-thinning hydrogel, the microswimmer speed varies as the fourth powerof the bubble oscillation amplitude (measured by direct high-speed imaging).This result is consistent with the shear-thinning behaviour and an acousticpropulsion that scales with the square of the oscillation amplitude. Similarresults are obtained for two swimmers with bubble diameters of 30µm (driven at 94.4 kHz, in red) and 67 µm (driven at70.4 kHz, in blue).
Since the scaling relation between speed and applied voltage does not distinguishthe two motive mechanisms, is there any alternative means to establish whetherone or both of these mechanisms is operating here? Fig. 2dshows a strong acoustic microstreaming field in the vicinity of themicroswimmer, with a size comparable to that of the bubble itself andmicrostreaming speed faster than the centre of mass speed of the swimmer. Thisrobust microstreaming pattern strongly suggests that there is net momentum flowand a significant microstreaming-derived acoustic force (see Supplementary Video 8). In addition, there is suggestive but notdefinitive evidence that the radiation force is significant: the speed of themicroswimmer varies significantly (by ~ 25%) as a function of the phase in thecircular orbit of an asymmetric microswimmer37 (see Supplementary Information 5). Since acoustic microstreaming dependsonly on the amplitude of the bubble oscillation and not the wave vector of theincident wave, this variation in speed around the orbit suggests that theradiation force derived from the interference of incident and scatter fields mayalso be significant (with a caveat that acoustic shadowing or residualstanding-wave components of the acoustic field could also be involved). Anability to access a regime in which both of these forces are in fact significantwould provide additional flexibility in tuning microswimmer properties.
Experimental demonstration of the acoustic microswimmer in viscousfluids
Although the microswimmer is small, the powerful bubble engine propels itsufficiently quickly that its Reynolds number is comparable to one. To generatecentre-of-mass motion at lower Reynolds number and simultaneously reduce thespatial extent of the microstreaming pattern surrounding the microswimmer, westudied behaviour of microswimmers in both 50% glycerol solution and viscousshear-thinning hydrogel. The acoustic microstreaming pattern in these moreviscous media is highly localized near the bubble (see Supplementary Information 6). In glycerol solution we obtainRe ≤ 10−2 and in hydrogelRe ~ 10−6 (see SupplementaryInformation 4). These low values ensure that the centre of mass motionoccurs at low Reynolds number, although additional complications arise at thebubble/fluid interface in the case of the hydrogel, since it is shear-thinning.Quantification of drag in a shear-thinning fluid is a complex problem mucheffort has been focused on obtaining approximate expressions in a form similarto the standard Stokes formula, often within a power law model for the variationin viscosity as a function of shear rate τ =K(∂u/∂y)n,where for the hydrogel used K ≈ 9.2 is the flow consistency index andn ≈ 0.49 is the flow behaviour index. If we assume aStokes-like drag expression with a constant correction factor of order unity,then the terminal velocity of the microswimmer in hydrogel should vary as thefourth power of the bubble amplitude. Fig. 5c demonstratesgood agreement to this power law for microswimmers with bubbles of two differentsizes. This analysis, coupled to estimates of typical drag forces, suggests thatthe acoustic microswimmer operating in hydrogel generates forces in themicroNewton range for typical bubble oscillation amplitudes of several microns(see Supplementary Figure S5).
Selective addressability of the acoustic microswimmers
A key advantage of this resonant acoustic mechanism of propulsion is the abilityto selectively address one microswimmer within a group. The quality factor ofthe bubble resonance is reasonably high, on the order of 25 (see Fig. 4), so that even a small difference in bubble diameter willyield a robust separation in frequency response between different swimmers orpossible different bubbles within the same swimmer. To demonstrate this, wefabricated two single-bubble microswimmers with bubbles of different radii andswept the acoustic drive frequency upwards. When the large bubble reachedresonance, the frequency was temporarily held fixed and this swimmer translatedas shown in Fig. 6a and Supplementary Video6. A further ramp of the drive frequency reached the resonance of thesmaller bubble: the original swimmer stopped and newly resonant swimmer beganmoving, as shown in Fig. 6b and Supplementary Video 7. Considering the sharpness of the resonantresponse, it should be possible to selectively address individual microswimmerswithin groups of multiple microswimmers.
Superimposed time-lapse images of selective actuation of an acousticmicroswimmer from within a group.
Two swimmers with bubbles of different size were immersed in an acousticfield of variable frequency. (a), Swimmer A, with the larger bubble, beginsacoustically-driven motion at 74 kHz, with little simultaneous motion ofswimmer B. A video of this behaviour is available as Supplementary Video 6. (b), With further increase in frequency,swimmer A stops; swimmer B begins to move at 91 kHz, with swimmer Aremaining essentially stationary. A video of this behaviour is available asSupplementary Video 7.
Unequal frequency-dependent excitation of different bubbles within a multi-bubbleswimmer should modulate the degree of translational and rotational motion;ultimately, this could enable two-bubble microswimmers that are fully steerablein two dimensions. We have fabricated microswimmers with two bubbles ofdifferent sizes. When the acoustic field excites one bubble more than another,the applied torque leads the swimmer to perform rotational motion as shown inFig. 7a. The orbital radius of the swimmer is largerthan that of a microswimmer with a single off-centre bubble, shown in Fig. 7b, due to some combination of the finite width of theresonances (i.e., the resonances of the two bubbles overlap in frequency)and nonlinear coupling between the bubbles. At this length scale, the effect ofstochastic orientational diffusion is negligible and the swimmer follows anearly perfect circular trajectory with negligible long-run translationaldiffusion57,58. When a different two-bubble swimmer isdriven at a frequency that equally excites both bubbles, it performs lineartranslation, as shown in Fig. 7c.
Superimposed time-lapse images of controlled two-dimensional motion ofdifferent microswimmers with bubbles of different sizes.
(a), When a two-bubble swimmer is driven at the resonance of just one bubble,it rotates in a wide orbit. (b), The orbit of an asymmetric one-bubbleswimmer is much tighter due to its stronger asymmetry. (c), At a frequencyintermediate between the resonances of the two constituent bubbles, atwo-bubble swimmer can move in a straight line.
Tunable Swimmer-swimmer interaction
Tunable swimmer-swimmer interactions are possible: nearby acoustic microswimmersin water can snap into persistent contact whereas similar swimmers in hydrogelcollide and separate. Motion may arise due to overlap of acoustic microstreamingfields or the interaction of re-radiated acoustic waves from nearby swimmers(i.e., the secondary Bjerknes force)59. To demonstratethis phenomenon, we placed two identical rotational swimmers in water in closevicinity. Under acoustic excitation, the swimmers rotate and drift together andeventually come into contact, as shown in Fig. 8a.Thereafter, the swimmers remain in direct contact. The concept has greatpotential in collective behaviour; the interactions of multiple swimmers can becontrolled by the applied acoustic power. In contrast, two rotational swimmersin viscous hydrogel come apart after collide, as shown in Fig.8b. In viscous hydrogel, the acoustic waves from the swimmers wereattenuated more by absorption when compared to that of water3.The lack of sustained contact suggests that significant acoustic microstreamingfields on the order of the swimmer dimensions are important for swimmer-swimmerinteractions.
Interaction of two swimmers in water and hydrogel.
(a), Two swimmers undergoing clockwise motions in water firmly lockthemselves together after snapping into contact, as shown in Supplementary Video 10. (b), Two swimmers in hydrogel, a mediumthat suppresses acoustic streaming, come apart after collision. The swimmertrajectories across the collision are traced in magenta and green. Thisevent is also shown in Supplementary Video 11.
Discussion
These acoustically powered microswimmers achieve significant advances in performance.The acoustic field is inexhaustible and largely unaffected by the ambient chemicalstate, unlike mechanisms of chemically powered motility. Biologically benignlow-power acoustic fields60 can generate sufficient force to propelswimmers through highly viscous fluids, which might be found inside biologicalsystems such as human vasculature. In contrast, electric and magnetic actuationmechanisms often require large, biologically-damaging fields to achieve adequatepropulsion. In addition to this excellent performance on conventional metrics, ourdesign also achieves selective actuation of a single swimmer from among agroup—a first in the field. Selective actuation opens new possibilitiesfor coherent cooperative action within groups of microswimmers. With a third bubble,steering in three dimensions should be possible. Due to the resonant nature of thebubble in acoustic fields, multiple bubbles of dissimilar resonances can becontained in a single microswimmer at different planes. By selectively actuatingeach bubble at resonance, motion in different directions could be obtained. Inaddition, tunable swimmer-swimmer interactions are possible. A collection ofselectively actuated steerable microswimmers, their bubble surfaces stabilized by apolymeric coating, could be deployed in vasculature with actuation provided by anexogenous transducer applied to the skin, the resulting behaviour being tracked byultrasonic or magnetic resonance imaging.
Methods
Materials
The microswimmers were fabricated using a mixture of photo-crosslinkablepolyethylene glycol (PEG) and a photo-initiator. The mixture consisted of 40%(v/v) PEG diacrylate with a molecular weight of 700 (PEG700, fromSigma-Aldrich), 25% (v/v) PEG with a molecular weight of 258 (PEG 258, fromSigma-Aldrich), 15% (v/v) photo-initiator2-Hydroxy-2-methyl-1-phenyl-propan-1-one (Darocur 1173, from Ciba), 15% (v/v) TEbuffer (100 TE, from OmniPur) and 5% (w/v) fluorescein.
Swimmer release
After UV exposure selectively hardened the liquid PEG polymer, the hardenedpolymer bodies of the microswimmers had to be separated from the surroundingliquid polymer. To accomplish this, the swimmers were washed three times inethanol solution containing 0.05% Tween 20 (from Sigma Aldrich) to remove anyliquid PEG residue from the hardened PEG surfaces (including from theindentation).
Trapping of the bubble
A drop of ethanol solution containing the microswimmers was placed onto the glassslide used for observations under the microscope. This glass slide was heatedfor 30 min at 65°C to dehydrate the swimmers. The slide was thenmoved to a vacuum chamber, where the swimmers were treated with 1H, 1H, 2H,2H-perfluorooctyl-trichlorosilane for 20−30 minutes to make theirsurfaces hydrophobic. After this hydrophobicity treatment, drops of liquid(water, 50% glycerol solution, or viscous hydrogel) were added to themicroswimmers, causing air bubbles to be trapped in the swimmer indentations.The size of the trapped air bubble was a function of the indentation diameterand depth and the hydrophobicity treatment duration.
Apparatus for microswimmer characterization
The glass platform holding the microswimmers in ambient liquid (see Section 1.4)was either a rectangular glass slide (6.08 cm × 2.54 cm) or acircular petri dish (9 or 18 cm diameter). The liquid was bounded on theperimeter by acoustically absorbent putty and on the top surface by a glasscover slip. Acoustic waves were introduced to the liquid via the glassslide/petri dish, to which was bonded a piezoelectric transducer driven by afunction generator (Tektronix AFG 3011). The glass slide/petri dish was mountedon the stage of a Nikon TE-2000U optical microscope.
Imaging and tracking
Microswimmer motion was captured using a Photron SA4 fast camera connected to themicroscope. Raw high-speed images were analysed using NIS tracking software todetermine parameters such as translational/rotational velocity.
Additional Information
How to cite this article: Ahmed, D. et al . Selectively manipulable acoustic-powered microswimmers. Sci. Rep. 5, 9744; doi: 10.1038/srep09744 (2015).
References
Purcell, E. M. Life at low Reynolds number. Am. J. Phys. 45, 3 (1977).
Nawroth, J. C. et al. A tissue-engineered jellyfish with biomimetic propulsion. Nat. Biotechnol. 30, 792–7 (2012).
Won, J. M., Lee, J. H., Lee, K. H., Rhee, K. & Chung, S. K. Propulsion of water-floating objects by acoustically oscillating microbubbles. Int. J. Precis. Eng. Manuf. 12, 577–580 (2011).
Patra, D. et al. Intelligent, self-powered, drug delivery systems. Nanoscale 5, 1273–83 (2013).
Gourevich, D. et al. Ultrasound-mediated targeted drug delivery with a novel cyclodextrin-based drug carrier by mechanical and thermal mechanisms. J. Control. release 170, 316–24 (2013).
Nelson, B. J., Kaliakatsos, I. K. & Abbott, J. J. Microrobots for minimally invasive medicine. Annu. Rev. Biomed. Eng. 12, 55–85 (2010).
Mirkovic, T., Zacharia, N. S., Scholes, G. D. & Ozin, G. A. Fuel for thought: chemically powered nanomotors out-swim nature's flagellated bacteria. ACS Nano 4, 1782–9 (2010).
Sacanna, S. et al. Shaping colloids for self-assembly. Nat. Commun. 4, 1688 (2013).
Palacci, J., Sacanna, S., Steinberg, A. P., Pine, D. J. & Chaikin, P. M. Living crystals of light-activated colloidal surfers. Science 339, 936–40 (2013).
Chronis, N. & Lee, L. P. Electrothermally activated SU-8 microgripper for single cell manipulation in solution. J. Microelectromechanical Syst. 14, 857–863 (2005).
Junkin, M., Leung, S. L., Whitman, S., Gregorio, C. C. & Wong, P. K. Cellular self-organization by autocatalytic alignment feedback. J. Cell Sci. 124, 4213–20 (2011).
Liu, M., Zentgraf, T., Liu, Y., Bartal, G. & Zhang, X. Light-driven nanoscale plasmonic motors. Nat. Nanotechnol. 5, 570–3 (2010).
Lee, T.-C. et al. Self-propelling nanomotors in the presence of strong Brownian forces. Nano Lett. 14, 2407–12 (2014).
Schamel, D. et al. Nanopropellers and their actuation in complex viscoelastic media. ACS Nano 8, 8794–801 (2014).
Paxton, W. F. et al. Catalytic nanomotors: autonomous movement of striped nanorods. J. Am. Chem. Soc. 126, 13424–31 (2004).
Wang, Y. et al. Bipolar electrochemical mechanism for the propulsion of catalytic nanomotors in hydrogen peroxide solutions. Langmuir 22, 10451–6 (2006).
Laocharoensuk, R., Burdick, J. & Wang, J. Carbon-nanotube-induced acceleration of catalytic nanomotors. ACS Nano 2, 1069–75 (2008).
Zacharia, N. S., Sadeq, Z. S. & Ozin, G. A. Enhanced speed of bimetallic nanorod motors by surface roughening. Chem. Commun. 5856–5858. doi:10.1039/B911561G. (2009).
Fattah, Z. et al. Straightforward single-step generation of microswimmers by bipolar electrochemistry. Electrochim. Acta 56, 10562–10566 (2011).
Ota, S., Wang, S., Wang, Y., Yin, X. & Zhang, X. Lipid bilayer-integrated optoelectronic tweezers for nanoparticle manipulations. Nano Lett. 13, 2766–70 (2013).
Wu, J. et al. Motion-based DNA detection using catalytic nanomotors. Nat. Commun. 1, 36 (2010).
Calvo-Marzal, P. et al. Electrochemically-triggered motion of catalytic nanomotors. Chem. Commun. 4509–4511. doi:10.1039/B909227G. (2009).
Zhang, L. et al. Characterizing the swimming properties of artificial bacterial flagella. Nano Lett. 9, 3663–7 (2009).
Snezhko, A., Belkin, M., Aranson, I. & Kwok, W.-K. Self-Assembled Magnetic Surface Swimmers. Phys. Rev. Lett. 102, 118103 (2009).
Dreyfus, R. et al. Microscopic artificial swimmers. Nature 437, 862–5 (2005).
Ghosh, A. & Fischer, P. Controlled propulsion of artificial magnetic nanostructured propellers. Nano Lett. 9, 2243–5 (2009).
Sing, C. E., Schmid, L., Schneider, M. F., Franke, T. & Alexander-Katz, A. Controlled surface-induced flows from the motion of self-assembled colloidal walkers. Proc. Natl. Acad. Sci. U. S. A. 107, 535–40 (2010).
Loget, G. & Kuhn, A. Electric field-induced chemical locomotion of conducting objects. Nat. Commun. 2, 535 (2011).
Camacho-Lopez, M., Finkelmann, H., Palffy-Muhoray, P. & Shelley, M. Fast liquid-crystal elastomer swims into the dark. Nat. Mater. 3, 307–10 (2004).
Wang, W., Castro, L. A., Hoyos, M. & Mallouk, T. E. Autonomous motion of metallic microrods propelled by ultrasound. ACS Nano 6, 6122–32 (2012).
Feng, J. & Cho, S. K. Micro propulsion in liquid by oscillating bubbles. Proceedings of the 2013 IEEE 26th International Conference on Micro Electro Mechanical Systems, Taipei, Taiwan, 20–24 January 2013; pp. 63–66 (2013).
Dijkink, R. J., van der Dennen, J. P., Ohl, C. D. & Prosperetti, A. The ‘acoustic scallop’: a bubble-powered actuator. J. Micromech. Microeng. 16, 1653–1659 (2006).
Feng, J. & Cho, S. K. Mini and Micro Propulsion for Medical Swimmers. Micromachines. 5, 97–113 (2014).
Kao, J., Wang, X., Warren, J., Xu, J. & Attinger, D. A bubble-powered micro-rotor: conception, manufacturing, assembly and characterization. J. Micromech. Microeng. 17, 2454–2460 (2007).
Jiang, H.-R., Yoshinaga, N. & Sano, M. Active Motion of a Janus Particle by Self-Thermophoresis in a Defocused Laser Beam. Phys. Rev. Lett. 105, 268302 (2010).
Wilson, D. A., Nolte, R. J. M. & van Hest, J. C. M. Autonomous movement of platinum-loaded stomatocytes. Nat. Chem. 4, 268–74 (2012).
Fomin, V. M. et al. Propulsion Mechanism of Catalytic Microjet Engines. IEEE Trans. Robot. 30, 40–48 (2014).
Ikezoe, Y., Washino, G., Uemura, T., Kitagawa, S. & Matsui, H. Autonomous motors of a metal-organic framework powered by reorganization of self-assembled peptides at interfaces. Nat. Mater. 11, 1081–5 (2012).
Manjare, M., Yang, B. & Zhao, Y.-P. Bubble Driven Quasioscillatory Translational Motion of Catalytic Micromotors. Phys. Rev. Lett. 109, 128305 (2012).
Démoré, C. E. M. et al. Acoustic Tractor Beam. Phys. Rev. Lett. 112, 174302 (2014).
Leighton, T. G. The acoustic bubble. 129 (Academic Press, 1994).
Lighthill, S. J. Acoustic streaming. J. Sound Vib. 61, 391–418 (1978).
Ainslie, M. A. & Leighton, T. G. Review of scattering and extinction cross-sections, damping factors and resonance frequencies of a spherical gas bubble. J. Acoust. Soc. Am. 130, 3184–208 (2011).
Wiklund, M., Green, R. & Ohlin, M. Acoustofluidics 14: Applications of acoustic streaming in microfluidic devices. Lab Chip 12, 2438–51 (2012).
Neild, A., Rogers, P. & Xu, L. Particle sorting using an oscillating microbubble. J. Acoust. Soc. Am. 131, 3302–3302 (2012).
Ding, X. et al. Surface acoustic wave microfluidics. Lab on a Chip, 13, pp. 3626–3649 (2013).
Ahmed, D. et al. Acoustofluidic chemical waveform generator and switch. Anal. Chem. 86, 11803–11810 (2014).
Ozcelik, A. et al. An acoustofluidic micromixer via bubble inception and cavitation from microchannel sidewalls. Anal. Chem. 86, 5083–5088 (2014).
Ahmed, D. et al. Tunable, pulsatile chemical gradient generation via acoustically driven oscillating bubbles. Lab Chip 13, 328–331 (2013).
Ahmed, D., Mao, X., Shi, J., Juluri, B. K. & Huang, T. J. Sub-milliseconds homogenous mixing using single bubble streaming phenomenon. Lab Chip 9, 2738–2741 (2009).
Ahmed, D., Mao, X., Juluri, B. K. & Huang, T. J. Fast microfluidic mixer via acoustic driven sidewall trapped bubbles. Microfluid. and Nanofluid. 7, 727–731 (2009).
Xie, Y. et al. Single-shot characterization of enzymatic reaction constants Km and kcat by an acoustic-driven, bubble-based fast micromixer. Anal. Chem. 84, 7495–7501 (2012).
Longuet-Higgins, M. S. Viscous streaming from an oscillating spherical bubble. Proc. R. Soc. A Math. Phys. Eng. Sci. 454, 725–742 (1998).
Marmottant, P. & Hilgenfeldt, S. Controlled vesicle deformation and lysis by single oscillating bubbles. Nature 423, 153–6 (2003).
Marmottant, P., Raven, J. P., Gardeniers, H., Bomer, J. G. & Hilgenfeldt, S. Microfluidics with ultrasound-driven bubbles. J. Fluid Mech. 568, 109 (2006).
Rife, J. et al. Miniature valveless ultrasonic pumps and mixers. Sensors Actuators A Phys. 86, 135–140 (2000).
Nourhani, A., Byun, Y.-M., Lammert, P. E., Borhan, A. & Crespi, V. H. Nanomotor mechanisms and motive force distributions from nanorotor trajectories. Phys. Rev. E 88, 062317 (2013).
Nourhani, A., Lammert, P. E., Borhan, A. & Crespi, V. H. Chiral diffusion of rotary nanomotors. Phys. Rev. E 87, 050301 (2013).
Yoshida, K., Fujikawa, T. & Watanabe, Y. Experimental investigation on reversal of secondary Bjerknes force between two bubbles in ultrasonic standing wave. J. Acoust. Soc. Am. 130, 135–44 (2011).
Ding, X. et al. Cell separation using tilted-angle standing surface acoustic waves. Proc. Natl. Acad. Sci. U. S. A., 111, 12992–12997 (2014).
Acknowledgements
We thank Thomas E. Mallouk, Adem Ozcelik, Wei Wang, Suzanne Ahmed, Ahsan Ahmed Nawaz,Sumyla Siddique and Michael Ian Lapsley for helpful discussions. We acknowledgesupport from National Institutes of Health (Director's New InnovatorAward, 1DP2OD007209-01), National Science Foundation (1102206) and the Center forNanoscale Science, a National Science Foundation Materials Research Science andEngineering Center supported under DMR-0820404.
Author information
Authors and Affiliations
Contributions
D.A. conceived the initial idea and contributed to the experimental design andperformed the bulk of the experimental work, data analysis and interpretation. M.L.contributed substantially to the experimental work, data analysis, andinterpretation and helped with the theoretical development. A.N. and P.E.L. were theprimary contributors to the theoretical discussion and helped with data analysis.A.N., D.A., Z.S., M.L. and H.S.M. contributed to the writing of the manuscript.V.H.C. and T.J.H. provided overall guidance and contributed to the experimentaldesign and scientific presentation.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Information
Supplementary Information
Supplementary Video 1
Supplementary Information
Supplementary Video 2
Supplementary Information
Supplementary Video 3
Supplementary Information
Supplementary Video 4
Supplementary Information
Supplementary Video 5
Supplementary Information
Supplementary Video 6
Supplementary Information
Supplementary Video 7
Supplementary Information
Supplementary Video 8
Supplementary Information
Supplementary Video 9
Supplementary Information
Supplementary Video 10
Supplementary Information
Supplementary Video 11
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0International License. The images or other third party material in this article areincluded in the article's Creative Commons license, unless indicatedotherwise in the credit line; if the material is not included under the CreativeCommons license, users will need to obtain permission from the license holder inorder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ahmed, D., Lu, M., Nourhani, A. et al. Selectively manipulable acoustic-powered microswimmers. Sci Rep 5, 9744 (2015). https://doi.org/10.1038/srep09744
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep09744
This article is cited by
-
3-D real-time ultrasound tracking of acoustically actuated swimming microdrone
Scientific Reports (2024)
-
Coated microbubbles swim via shell buckling
Communications Engineering (2023)
-
Rolling microswarms along acoustic virtual walls
Nature Communications (2022)
-
Ultrasound-activated ciliary bands for microrobotic systems inspired by starfish
Nature Communications (2021)
-
Machine learning models for the secondary Bjerknes force between two insonated bubbles
Acta Mechanica Sinica (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.