Abstract
Evaluation of volcanic and hydrothermal fluxes to the surface environments is important to elucidate the geochemical cycle of sulphur and the evolution of ocean chemistry. This paper presents S/3He ratios of vesicles in mid-ocean ridge (MOR) basalt glass together with the ratios of high-temperature hydrothermal fluids to calculate the sulphur flux of 100 Gmol/y at MOR. The S/3He ratios of high-temperature volcanic gases show sulphur flux of 720 Gmol/y at arc volcanoes (ARC) with a contribution from the mantle of 2.9%, which is calculated as 21 Gmol/y. The C/S flux ratio of 12 from the mantle at MOR and ARC is comparable to the C/S ratio in the surface inventory, which suggests that these elements in the surface environments originated from the upper mantle.
Similar content being viewed by others
Introduction
Volcanic and hydrothermal activity discharge sulphur and carbon from the Earth's mantle to the atmosphere and hydrosphere. Sulphur and carbon also dissolve in seawater and are incorporated into sediments before being recycled back into the mantle1 through subduction. The global flux of sulphur gas from sub-aerial arc volcanoes is well constrained1,2 based on measurements of SO2 gas from volcanoes using correlation spectrometry (COSPEC3), UV spectroscopy and satellite remote sensing2. However the main mantle flux of volatile elements is derived from MOR-type volcanism on divergent plate boundaries of the Earth. Therefore, submarine flux must be studied together with sub-aerial flux to evaluate a mass balance of sulphur and carbon on the Earth's surface. However, the sulphur flux from submarine volcanism, which includes the amount released from the magma and that resulting from dissolution of solidified igneous rocks, is not well understood due to difficulties inherent to ocean bottom measurements.
A sulphur flux of 7.8 × 1010 mol/y was estimated from seawater–basalt sulphur exchange during hydrothermal alteration4. This value is markedly lower than the estimate of 1.64 × 1012 mol/y based on oceanic crust production and its sulphur content5. 3He is a useful geochemical tracer because of its primordial origin and inert behaviour and its mantle flux has been used for calculations of other volatile fluxes. Mantle carbon flux of 2 × 1012 mol/y was derived from the MOR 3He flux and CO2/3He ratio in MOR basalt glass6. However, except for a very rough estimate7, no report in the literature has described an estimation of mantle sulphur flux at MOR as calibrated against the mantle 3He flux because no S/3He ratios in MOR basalt glasses have yet been reported.
This report describes the sulphur flux at MOR based on crushing of basalt glass, data of high-temperature submarine vent chemistry and recent estimates of the mantle 3He flux8,9. Additionally, we present the amount and the origin of sulphur in arc magmas based on calculations using δ34S values and S/3He ratios. The results enable us to compare the total natural flux with anthropogenic emissions of sulphur. We also verify the global mass balance of carbon to discuss the evolution of the atmosphere.
Results
Vesicles
We analyzed MORB glass samples collected at six sites on the East Pacific Rise, Mid-Atlantic Ridge and Central Indian Ridge (Fig. 1). The 3He/4He ratios and 3He contents of MOR basalt glass vesicles were 7.9 Ra–9.4 Ra (where Ra is the atmospheric ratio of 1.382 × 10−6)10 and from 1.9 × 10−15 to 5.1 × 10−15 mol/g (Table 1), respectively. The values agree well with data presented in an earlier report11. The samples show similar total sulphur contents in vesicles with an average of 1.25 × 10−7 mol/g. The average value of S/3He ratios in vesicles was (4.2 ± 1.2) × 107 (1σ). Evaluating δ34S values of vesicle sulphur was difficult because the amount was less than the blank contribution from the filtering system used to precipitate BaSO4.
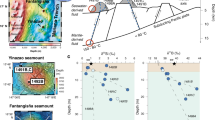
Map showing sampling sites of MOR basalt (RY380-R03b, CL DR01, 418R002, CH31 DR12, KH10-6 DR15-glass and KH10-6 DR16-glass) analyzed in this study (stars), major hydrothermal vents (circles) and major subaerial volcanoes (triangles), together with helium isotopic signatures: R denotes the 3He/4He ratio in each site; Ra is the 3He/4He ratio in air of 1.382 × 10−6.
All data are from Tables and Supplementary Tables. Lines on the ocean area show oceanic ridges. This figure was prepared using the Ocean Data View software52.
Glass matrix
The 3He contents of MOR basalt glass matrix are listed in Supplementary Table 1. The sulphur contents of MOR basalt glass matrix measured using a secondary ion mass spectrometer (NanoSIMS; Cameca SAS, Gennevilliers, France) listed in Supplementary Table 1 are well within the variation of sulphur contents in MOR glass obtained using a conventional method12. The average value of S/3He ratios in the glass matrix was (1.0 ± 0.2) × 1010 (1σ). Observed δ34S values are consistent with those of MOR basalt and mantle values12,13, suggesting a typical mantle sulphur signature.
Discussion
The S/3He ratio and 3He flux at MOR are needed for sulphur flux calculations. The observed S/3He ratios of vesicles in MOR basalt glass are 2.0 × 107–9.9 × 107. These values are lower than those in the glass matrix (See Table 1 and Supplementary Table 1). This observation suggests higher solubility of sulphur than of helium in basaltic melt, which is also supported by recent laboratory experiments14. We consider the average vesicle S/3He ratio of 4.2 × 107 as the minimum for MOR for our flux calculations. The other independent means to estimate the S/3He ratio of MOR is using the chemistry of high-temperature submarine vents. The average S/3He ratio is (3.4 ± 0.7) × 108 (1σ) among 10 high-temperature (>200°C) hydrothermal sites worldwide (Supplementary Table 2). The δ34S value of H2S in hot vent fluids is variable13, but the original value before the incursion of seawater is similar to the MOR basalt values4,15. A small part of H2S might be generated from the reduction of seawater SO4 from the recharge zone4, although it is difficult to deconvolve the contribution quantitatively. Therefore the vent S/3He ratio of 3.4 × 108 is expected to be the maximum estimate at MOR. We take an average of these two independent estimates (1.9 ± 1.5) × 108 as the upper mantle S/3He ratio in the current study. The S/3He ratio in MOR glass matrix is higher than this upper mantle value (Supplementary Table 1), which implies that helium has degassed from the melt before it was quenched to a glass and therefore these ratios should not be used for the sulphur flux estimate.
Based on the saturation anomaly of 3He in deep seawater of the eastern Pacific, a value of 1070 ± 270 mol/y was calculated for the 3He flux from MOR16. A more recent estimate of the MOR 3He flux is 530 ± 100 mol/y derived from an ocean circulation model which also considers radiocarbons and chlorofluorocarbons8. This MOR 3He flux, when combined with the average S/3He ratio obtained in this study, provides the MOR sulphur flux of (1.0 ± 0.8) × 1011 mol/y. This mantle flux is consistent with the estimate based on seawater–basalt sulphur exchange during hydrothermal alteration4, but it is about an order of magnitude smaller than the value calculated from the production rate of the oceanic crust and sulphur contents therein5. This difference suggests that most sulphur remains in the magma and solidifies as sulphides in the MOR crust and does not contribute to the MOR flux that discharges into the ocean. When we consider the mass balance of sulphur in global ocean water, the mantle flux of 1.0 × 1011 mol S/y is a second-order flux 'compared to the riverine input of 8.9 × 1011 mol S/y to the ocean and the output of 5.5 × 1011 mol S/y as sedimentary pyrite and evaporitic sulphate17. However, the sulphur flux from the upper mantle into the ocean represents a deep Earth contribution and is therefore distinct from riverine sulphur input that is continent-derived.
Sulphur and helium isotopic compositions are useful for investigating the origin of sulphur at arc volcanoes. The thermodynamic equilibrium between SO2 and H2S together with their δ34S values might provide constraints on the evolution of volcanic gases, such as an isochemical cooling path, under the assumption that the initial δ34SΣS value is 0‰13, where δ34SΣS denotes the total sulphur isotopic ratio of SO2 and H2S. In addition, the δ34SΣS values might provide information related to the origin of sulphur in ARC volcanic gases, even though they might be affected by a gas-melt separation and related fractionation processes13,18. Available data of 3He and total sulphur contents and δ34SΣS values for high-temperature volcanic gases (>200°C) in subduction zones were compiled from the literature (Table 2). Their 3He/4He ratios are consistent with the range of subduction-type He1,19. The δ34SΣS values are generally positive, except for one outlier from Galeras. This similarity suggests that the sulphur signature of an ARC magma source is due to incorporation of subducted sulphate partly derived from a seawater component18,20,21 with high δ34S values. The average value of S/3He ratios among these high-temperature ARC gases is (6.5 ± 1.1) × 109 (1σ), which is significantly higher than that of the upper mantle, suggesting enrichment of sulphur in the ARC mantle source by subduction processes.
Fig. 2 presents the relation between S/3He ratios and δ34SΣS values of volcanic gases in subduction zones. The figure particularly shows end-member data for the upper mantle, sedimentary pyrite with reduced sulphur derived from slab and subducted sulphate. The δ34S values of sedimentary pyrite vary considerably due to the result of bacterial reduction of seawater sulphate and have a mean value of −20.9‰ in the Western Pacific22. Results of a recent study23 of the oceanic basement in northern Italy suggest that low-temperature serpentinization produces a negative δ34SΣS value with (−8.9 ± 8.0)‰. Then the δ34S value of sedimentary pyrite is defined as (−14.9 ± 6.0)‰. No source of primordial helium exists in the pyrite and the slab may have lost the original mantle helium as well24. It is therefore possible to adopt S/3He larger than 1 × 1013 for the sedimentary pyrite. Seawater sulphate has a δ34S value of +21.0‰25. Metasomatic fluids released from sediment, of which the sulphur is mostly in the form of sulphate, have a δ34S value of +14‰ when their sulphur compositions resemble the bulk sediment composition21. Using these values, the δ34S value of subducted sulphate is here defined as (+17.5 ± 3.5)‰. A defined S/3He larger than 1 × 1013 for sedimentary sulphate is consistent with the seawater SO4/3He of 1.0 × 1014.
Correlation diagram between δ34S and S/3He ratios of high-temperature volcanic gases in circum-Pacific regions.
Model end-members of the upper mantle, sedimentary pyrite and subducted sulphate are included. The curve shows mixing among the end-members. #Data are of the following volcanoes: (#1) Avacha, (#2) Mutonovsky, (#3) Kudryavy, (#4) Usu, (#5) Kuju, (#6) Satsuma-Iwojima, (#7) Lewotolo, (#8) White Island, (#9) Ngauruhoe, (#10) Momotombo, (#11) Galeras, (#12) Colima. All data are from Table 2.
The distribution of volcanic gas data in the S/3He-δ34S diagram (Fig. 2) suggests that ARC samples are explained by three-component mixing. When sulphur in a sample is a mixture of the upper mantle, subducted sedimentary pyrite and subducted sulphate having respective masses M, P and S, the following equations can be derived:


Therein, the following relation holds:

In those equations, subscripts V, M, P and S respectively denote the volcanic gas, the upper mantle, subducted sedimentary pyrite and subducted sulphate. Taking values of δ34SM = 0‰, δ34SP = −14.9‰, δ34SS = +17.5‰, (S/3He)M = 1.9 × 108, (S/3He)P = 1.0 × 1013 and (S/3He)S = 1.0 × 1013, one can calculate the percentage of the three components M, P and S quantitatively in ARC samples (Table 2). The uncertainty of estimated contributions is assessed in the Supplementary Discussion. The contribution of mantle sulphur is 1.5%–19% (2.9% average) in ARC samples and the main contribution derives from subducted sulphate and sedimentary pyrite (See Supplementary Discussion for calculations). Volcanic gas from Satsuma-Iwojima shows the highest subducted sulphate contribution with the highest δ34SΣS value. To explain the heavy δ34SΣS values of ARC volcanic gases, incorporation of a seawater component in the magma source has been inferred since the 1970s20. Here we first provide a quantification of the relative amount of the seawater sulphate contribution to ARC gases, which allows us to evaluate the recycling capacity of ARC volcanoes within the global sulphur cycle.
A conventional ARC 3He flux was estimated from the MOR flux, given the assumption that the magma production rate of ARC is about 20% of that of MOR9. This percentage is consistent with the estimate of global magma emplacement and volcanic output averaged over the last 180 m.y.26. Recently, the MOR 3He flux was calculated to be 530 mol/y8 which would result in an ARC 3He flux of 110 ± 20 mol/y. This flux is consistent with the value obtained by summation of 3He flux at arc volcanoes worldwide1. The average S/3He ratio of (6.5 ± 1.1) × 109 (1σ) is obtained from high-temperature volcanic gases. Therefore, the ARC sulphur flux is estimated to be (7.2 ± 1.8) × 1011 mol/y based on the 3He flux of 110 mol/y. This value is considerably larger than MOR sulphur flux calculated in this study. However the upper mantle contribution to ARC volcanic gases is only (2.9 ± 0.5)% of total sulphur, on average. The sulphur flux from the wedge mantle at ARC then becomes (2.1 ± 0.6) × 1010 mol/y, which is less than the mantle sulphur flux discharging into the ocean at MOR. The major contribution of the ARC sulphur flux is derived from subducted sedimentary pyrite and subducted sulphate partly derived from the seawater component.
A summary of the global sulphur flux is depicted in Fig. 3a. Present hot spot magmatism likely does not contribute substantially to the global flux of sulphur (See Supplementary Discussion). The total volcanic flux of sulphur is estimated as 8.2 × 1011 mol/y and represents about one-third of the anthropogenic emissions due to coal burning and sulphide ore smelting27. This natural flux, if it has remained constant over 4.55 billion years of geological time, engenders an accumulation of 3.7 × 1021 mol. This value is greater than the surface inventory of 5.3 × 1020 mol1. If we take the MOR flux together only with the mantle wedge flux of 2.1 × 1010 mol/y, then the accumulation becomes 5.6 × 1020 mol in total, which is equivalent to the surface inventory. When steady-state recycling of sulphur is applied, the total subducting flux becomes 8.2 × 1011 mol/y.
As new 3He flux data at MOR have been reported8, we revise the carbon geodynamics along with sulphur. The CO2/3He ratio at MOR was calculated to be (2.2 ± 0.7) × 109 using CO2/3He data for MOR basalt glass and hydrothermal fluids28. This ratio, combined with the new MOR 3He flux, engenders the global MOR CO2 flux of (1.2 ± 0.4) × 1012 mol/y, which is consistent with the most recent estimate based on vesicularities of MORB worldwide29.
For ARC volcanism, we selected 24 volcanic gas and steam well data with temperatures higher than 200°C (Supplementary Table 3). Their carbon source is well explained by the mixing of three components: The upper mantle (M), organic sediment (S) and limestone with a slab component (L) (Fig. 4; Ref. 30). These end-member components are described in Supplementary Table 3. Using those values, we calculate the respective percentages of the three components in the ARC samples (Supplementary Table 3). The contribution of the upper mantle carbon is 3.2%–36% (average 11%), whereas a major part is attributable to subducted carbonate and organic carbon. Because the average CO2/3He ratio of these data is (2.0 ± 0.3) × 1010, the carbon flux from ARC is (2.2 ± 0.5) × 1012 mol/y using the ARC 3He flux of 110 ± 20 mol/y, which is also consistent with the recent estimate using volcanic gas observations worldwide31.
Correlation diagram between δ13C and CO2/3He ratios of high-temperature volcanic gases in circum-Pacific regions.
Model end-members of the upper mantle, sediment and limestone are included. The curve shows mixing among the end-members. #Data are those of the following volcanoes: (#1) Klyuchevskoy, (#2) Koryak, (#3) Avacha, (#4) Mutnovsky, (#5) Kudryavy, (#6) Usu, (#7) Kuju, (#8) Unzen, (#9) Satsuma-Iwojima, (#10) Merapi, (#11) Lewotolo, (#12) Ngawha, (#13) White Island, (#14) Ngauruhoe, (#15) Cerro Negro, (#16) Momotombo, (#17) Pacaya, (#18) Galeras, (#19) Cumbal, (#20) Colima, (#21) La Primavera. All data are from Supplementary Table 3.
A summary of global carbon flux is depicted in Fig. 3b. The total volcanic flux of carbon is 3.4 × 1012 mol/y, which is two orders of magnitude smaller than anthropogenic emission by fossil fuel combustion and cement production32. The MOR flux combined with the wedge mantle flux is 1.4 × 1012 mol/y. This value, if accumulated for 4.55 billion years, results in 6.6 × 1021 mol of carbon, which closely approximates the surface inventory of 7.0 × 1021 mol1. If steady-state recycling of carbon is applied, then the total subduction flux becomes 3.4 × 1012 mol/y. This estimate is consistent with the influx of carbon1.
In conclusion, the best estimates of MOR sulphur and carbon flux are 1.0 × 1011 mol/y and 1.2 × 1012 mol/y, respectively at present, which are less than their volcanic fluxes at ARC. Sulphur and carbon fluxes from only the mantle wedge to the surface environment at ARC are calculated as 2.1 × 1010 mol/y and 2.4 × 1011 mol/y, respectively. These data provide a C/S flux ratio of 12 which is similar to the C/S ratio in the surface inventory of 13 (Ref. 1). Our results suggest that the main source of sulphur and carbon is the upper mantle. To balance the mass between the crust and the mantle, the sulphur subducted into the mantle and not immediately recycled to the surface is expected to be equivalent to 1.2 × 1011 mol/y, which is about 17% of the recycling sulphur of 7.0 × 1011 mol/y. We calculated sulphur and carbon fluxes from the mantle based on the plausible S/3He and C/3He ratios and the recently reported 3He flux at MOR, which constrained geochemical cycles of sulphur and carbon and evolutionary histories of the atmosphere and hydrosphere.
Methods
Glass vesicle
It is difficult to measure the abundance of sulphur species such as H2S and SO2 in vesicles of MOR basalt glass together with 3He because the gases are highly reactive. They easily adhere to the inner surface of a vacuum crushing vessel. We have developed a gas-extraction method, ‘Frozen Crushing Method’, by which sulphur gases are fixed immediately in semi-frozen alkaline solution during mechanical fracturing of glass7. The abundance of helium and 3He/4He ratios were measured using a noble gas mass spectrometer (VG5400; Waters Corp.) at the Atmosphere and Ocean Research Institute (AORI). Subsequently, the vacuum was broken and the S-bearing solution was filtered. All sulphur compounds were converted into sulphate ion by oxidation with hydrogen peroxide. The concentration was measured using an ion chromatography system (ICS-2100; Thermo Fisher Scientific Inc.) at AORI. Blank contributions of sulphur and helium were considerably smaller than the actual amounts in samples. Experimental details are presented in an earlier report7. Sulphate ion in the alkaline solution was converted into BaSO4 precipitations by adding BaCl2 solution and δ34S values obtained with an elemental analyzer (vario PYRO cube; Elementar Analysensysteme, GmbH) coupled to an isotope-ratio mass spectrometer (Delta XP; Thermo Fisher Scientific Inc.) via an interface (ConFlo IV; Thermo Fisher Scientific Inc.) at the University of Ottawa.
Glass matrix
Sulphur contents in glass matrix were measured (NanoSIMS; Cameca SAS, Gennevilliers, France) at AORI, whereas δ34S values were obtained using an elemental analyzer isotope-ratio mass spectrometer system33 (Isoprime-EA; Isoprime Ltd.) at the University of Tsukuba.
References
Hilton, D. R., Fischer, T. P. & Marty, B. Noble gases and volatile recycling at subduction zones. In: Noble Gases in Geochemistry and Cosmochemistry. Reviews in Mineralogy and Geochemistry 47 (eds Porcelli D., Ballentine C. J., & Wieler R.) 319–370 (Mineralogical Society of America, 2002).
Bluth, G. J. S., Schnetzler, C. C., Krueger, A. J. & Walter, L. S. The contribution of explosive volcanism to global atmospheric sulphur dioxide concentrations. Nature 366, 327–329 (1993).
Stoiber, R. E. & Jepsen, A. Sulfur dioxide contributions to the atmosphere by volcanoes. Science 182, 577–578 (1973).
Alt, J. C. Sulfur isotopic profile through the oceanic crust: Sulfur mobility and seawater-crustal sulfur exchange during hydrothermal alteration. Geology 23, 585–588 (1995).
Hansen, K. W. & Wallmann, K. Cretaceous and Cenozoic evolution of seawater composition, atmospheric O2 and CO2: A model perspective. Am. J. Sci. 303, 94–148 (2003).
Marty, B. & Jambon, A. C/3He in volatile fluxes from the solid Earth: implications for carbon geodynamics. Earth Planet. Sci. Lett. 83, 16–26 (1987).
Kagoshima, T. et al. Estimation of sulfur, fluorine, chlorine and bromine fluxes at Mid Ocean Ridges using a new experimental crushing and extraction method. Geochem. J. 46, e21–e26 (2012).
Bianchi, D. et al. Low helium flux from the mantle inferred from simulations of oceanic helium isotope data. Earth Planet. Sci. Lett. 297, 379–386 (2010).
Torgersen, T. Terrestrial helium degassing fluxes and the atmospheric helium budget: Implications with respect to the degassing processes of continental crust. Chem. Geol. 79, 1–14 (1989).
Sano, Y., Marty, B. & Burnard, P. Noble gases in the atmosphere. In: The Noble Gases as Geochemical Tracers. Advances in Isotope Geochemistry. (ed Burnard P.) 17–31 (Springer–Verlag, 2013).
Graham, D. W. Noble gas isotope geochemistry of mid-ocean ridge and ocean island basalts: characterization of mantle source reservoirs. In: Noble Gases in Geochemistry and Cosmochemistry. Reviews in Mineralogy and Geochemistry 47 (eds Porcelli D., Ballentine C. J., & Wieler R.) 247–317 (Mineralogical Society of America, 2002).
Sakai, H., Des Marais, D. J., Ueda, A. & Moore, J. G. Concentrations and isotope ratios of carbon, nitrogen and sulfur in ocean-floor basalts. Geochim. Cosmochim. Acta 48, 2433–2441 (1984).
Marini, L., Moretti, R. & Accornero, M. Sulfur isotopes in magmatic-hydrothermal systems, melts and magmas. In: Sulfur in magmas and melts: Its importance for natural and technical processes. Reviews in Mineralogy and Geochemistry 73 (eds Behrens H., & Webster J. D.) 423–492 (Mineralogical Society of America, 2011).
Webster, J. D. & Botcharnikov, R. E. Distribution of sulfur between melt and fluid in S–O–H–C–Cl-bearing magmatic systems at shallow crustal pressures and temperatures. In: Sulfur in magmas and melts: Its importance for natural and technical processes. Reviews in Mineralogy and Geochemistry 73 (eds Behrens H., & Webster J. D.) 247–283 (Mineralogical Society of America, 2011).
Gamo, T. Wide variation of chemical characteristics of submarine hydrothermal fluids due to secondary modification processes after high temperature water-rock interaction: a review. In: Biogeochemical processes and ocean flux in the Western Pacific (eds Sakai H., & Nozaki Y.) 425–451 (Terrapub, 1995).
Craig, H., Clarke, W. B. & Beg, M. A. Excess 3He in deep water on the East Pacific Rise. Earth Planet. Sci. Lett. 26, 125–132 (1975).
Holser, W. T., Schidlowski, M., Mackenzie, F. T. & Maynard, J. B. Geochemical cycles of carbon and sulfur. In: Chemical cycles in the evolution of the earth (eds Gregor C. B., Garrels R. M., Mackenzie F. T., & Maynard J. B.) 105–173 (John Wiley & Sons, 1988).
de Moor, J. M. et al. Sulfur degassing at Erta Ale (Ethiopia) and Masaya (Nicaragua) volcanoes: Implications for degassing processes and oxygen fugacities of basaltic systems. Geochem. Geophys. Geosyst. 14, 4076–4108 (2013).
Sano, Y. & Fischer, T. P. The analysis and interpretation of noble gases in modern hydrothermal systems. In: The Noble Gases as Geochemical Tracers. Advances in Isotope Geochemistry (ed Burnard P.) 249–317 (Springer–Verlag, 2013).
Sakai, H. & Matsubaya, O. Stable isotopic studies of Japanese geothermal systems. Geothermics 5, 97–124 (1977).
Alt, J. C., Shanks III, W. C. & Jackson, M. C. Cycling of sulfur in subduction zones: The geochemistry of sulfur in the Mariana Island Arc and back-arc trough. Earth Planet. Sci. Lett. 119, 477–494 (1993).
Alt, J. C. & Burdett, J. W. Sulfur in Pacific deep-sea sediments (Leg 129) and implications for cycling of sediment in subduction zones. Proc. ODP, Sci. Results 129, 283–294 (1992).
Alt, J. C. et al. Uptake of carbon and sulfur during seafloor serpentinization and the effects of subduction metamorphism in Ligurian peridotites. Chem. Geol. 322–323, 268–277 (2012).
Hiyagon, H. Retention of solar helium and neon in IDPs in deep sea sediment. Science 263, 1257–1259 (1994).
Rees, C. E., Jenkins, W. J. & Monster, J. The sulfur isotopic composition of ocean water sulphate. Geochim. Cosmochim. Acta 42, 377–381 (1978).
Crisp, J. A. Rates of magma emplacement and volcanic output. J. Volcanol. Geotherm. Res. 20, 177–211 (1984).
Moller, D. Estimation of the global man-made sulphur emission. Atoms. Environ. 18, 19–27 (1984).
Marty, B. & Tolstikhin, I. N. CO2 fluxes from mid-ocean ridges, arcs and plumes. Chem. Geol. 145, 233–248 (1998).
Chavrit, D., Humler, E. & Grasset, O. Mapping modern CO2 fluxes and mantle carbon content all along the mid-ocean ridge system. Earth Planet. Sci. Lett. 387, 229–239 (2014).
Sano, Y. & Marty, B. Origin of carbon in fumarolic gas from island arcs. Chem. Geol. 119, 265–274 (1995).
Fischer, T. P. Fluxes of volatiles (H2O, CO2, N2, Cl, F) from arc volcanoes. Geochem. J. 42, 21–38 (2008).
Ciais, P. et al. Carbon and other biogeochemical cycles. In: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker T. F., et al.) 465–570 (Cambridge University Press, 2014).
Maruoka, T., Koeberl, C., Hancox, P. J. & Reimold, W. U. Sulfur geochemistry across a terrestrial Permian–Triassic boundary section in the Karoo Basin, South Africa. Earth Planet. Sci. Lett. 206, 101–117 (2003).
Taran, Y. A. Geochemistry of volcanic and hydrothermal fluids and volatile budget of the Kamchatka-Kuril subduction zone. Geochim. Cosmochim. Acta 73, 1067–1094 (2009).
Taran, Y. A., Connor, C. B., Shapar, V. N., Ovsyannikov, A. A. & Bilichenko, A. A. Fumarolic activity of Avachinsky and Koryaksky volcanoes, Kamchatka, from 1993 to 1994. Bull. Volcanol. 58, 441–448 (1997).
Taran, Y. A., Pilipenko, V. P., Rozhkov, A. M. & Vakin, E. A. A geochemical model for fumaroles of the Mutnovsky volcano, Kamchatka, USSR. J. Volcanol. Geotherm. Res. 49, 269–283 (1992).
Taran, Y. A., Hedenquist, J. W., Korzhinsky, M. A., Tkachenko, S. I. & Shmulovich, K. I. Geochemistry of magmatic gases from Kudryavy volcano, Iturup, Kuril Islands. Geochim. Cosmochim. Acta 59, 1749–1761 (1995).
Marty, B., Jambon, A. & Sano, Y. Helium isotopes and CO2 in volcanic gases of Japan. Chem. Geol. 76, 25–40 (1989).
Kasasaku, K., Minari, T., Mukai, H. & Murano, K. Stable sulfur isotope ratios of the gases from Mt. Sakurajima and Satsuma-Iwojima volcanoes –Assessment of volcanic sulfur on rainfall sulfate in Kagoshima Prefecture. Nippon Kagaku Kaishi 7, 479–486 (1999).
Mizutani, Y., Hayashi, S. & Sugiura, T. Chemical and isotopic compositions of fumarolic gases from Kuju-Iwoyama, Kyushu, Japan. Geochem. J. 20, 273–285 (1986).
Nagao, K., Takaoka, N. & Matsubayashi, O. Rare gas isotopic compositions in natural gases of Japan. Earth Planet. Sci. Lett. 53, 175–188 (1981).
Saito, G., Shinohara, H. & Kazahaya, K. Successive sampling of fumarolic gases at Satsuma-Iwojima and Kuju volcanoes, southwest Japan: Evaluation of short-term variations and precision of the gas sampling and analytical techniques. Geochem. J. 36, 1–20 (2002).
Sano, Y. & Williams, S. N. Fluxes of mantle and subducted carbon along convergent plate boundaries. Geophys. Res. Lett. 23, 2749–2752 (1996).
Poorter, R. P. E., Varekamp, J. C., Poreda, R. J., Van Bergen, M. J. & Kreulen, R. Chemical and isotopic compositions of volcanic gases from the east Sunda and Banda arcs, Indonesia. Geochim. Cosmochim. Acta 55, 3795–3807 (1991).
Varekamp, J. C., Kreulen, R., Poorter, R. P. E. & Van Bergen, M. J. Carbon sources in arc volcanism, with implications for the carbon cycle. Terra Nova 4, 363–373 (1992).
Marty, B. & Giggenbach, W. F. Major and rare gases at White Island volcano, New Zealand: Origin and flux of volatiles. Geophys. Res. Lett. 17, 247–250 (1990).
Giggenbach, W. The chemical and isotopic composition of gas discharges from New Zealand andesitic volcanoes. Bull. Volcanol. 45, 253–255 (1982).
Giggenbach, W. F., Sano, Y. & Wakita, H. Isotopic composition of helium and CO2 and CH4 contents in gases produced along the New Zealand part of a convergent plate boundary. Geochim. Cosmochim. Acta 57, 3427–3455 (1993).
Giggenbach, W. F. Are Tokaanu chloride waters the outflow from Ketetahi or Hipaua? Proceedings of the 18th New Zealand Geothermal Workshop. University of Aukland. 18, 175–182 (1996).
Menyailov, I. A., Nikitina, L. P., Shapar, V. N. & Pilipenko, V. P. Temperature increase and chemical change of fumarolic gases at Momotombo volcano, Nicaragua, in 1982–1985: Are these indicators of a possible eruption? J. Geophys. Res. 91, 12, 199–12, 214 (1986).
Taran, Y. A. et al. Chemistry and mineralogy of high-temperature gas discharges from Colima volcano, Mexico. Implications for magmatic gas-atmosphere interaction. J. Volcanol. Geotherm. Res. 108, 245–264 (2001).
Schlitzer, R. Ocean Data View, http://odv.awi.de (2013). Date of access: 2014 January 23.
Acknowledgements
We would like to thank Dr. Paul Middlestead of the University of Ottawa for cooperating in the analyses of sulphur isotopes in MOR basalt vesicles. CL DR01 and CH31 DR12 were supplied by Dr. Bernard Marty of Université de Lorraine. KH10-6 DR15-glass and KH10-6 DR16-glass were supplied by Dr. Hiroshi Sato of Senshu University. RY380-R03b and 418R002 were supplied from the sample archives of JAMSTEC, retrieved from following cruises under international projects or co-operations: MODE'98 and RidgeFlux. This work was partly supported by a Grant-in-Aid for JSPS Fellows (25-6922). T.K. is a JSPS Research Fellow.
Author information
Authors and Affiliations
Contributions
T.K. was responsible for the research and the analyses of MOR basalt vesicles with Frozen Crushing Method. Y.S. supervised the research and made important contributions in producing the manuscript: data compilation of hydrothermal vent and volcanic gas chemistry, figure and table preparation and discussion of volatile cycles. N.T. is a manager of VG-5400 and NanoSIMS laboratories and supported data interpretation. T.M. analyzed samples using Isoprime-EA and interpreted data. T.P.F. made important comments on compiled data of arc volcanic gases and volatile flux. K.H. made important comments on sulphur chemistry and discussion of volatile cycles. T.K. and Y.S. wrote the manuscript based on other authors' comments.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary materials
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kagoshima, T., Sano, Y., Takahata, N. et al. Sulphur geodynamic cycle. Sci Rep 5, 8330 (2015). https://doi.org/10.1038/srep08330
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08330
This article is cited by
-
Rates of seafloor and continental weathering govern Phanerozoic marine phosphate levels
Nature Geoscience (2023)
-
Uncovering and quantifying the subduction zone sulfur cycle from the slab perspective
Nature Communications (2020)
-
Subducting carbon
Nature (2019)
-
The emissions of CO2 and other volatiles from the world’s subaerial volcanoes
Scientific Reports (2019)
-
Oxidising agents in sub-arc mantle melts link slab devolatilisation and arc magmas
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.