Abstract
As one of the key enzymes in the oxidative pentose phosphate pathway, glucose-6-phosphate dehydrogenase (G6PDH) plays a role in response to abiotic stresses and pathogenesis. Here, a full-length cDNA was obtained, designed as ScG6PDH from sugarcane. The ScG6PDH gene is 1,646 bp long with a 1,524-bp long ORF encoding 507 amino acid residues. Analysis of a phylogenetic tree indicated that this gene is a member of the cytosolic G6PDH gene family, which is consistent with results from a subcellular localization experiment. Based on a real-time quantitative RT-PCR performed under salt, drought, heavy metal (CdCl2) and low temperature (4°C) treatments, the transcription levels of the ScG6PDH gene were higher compared with transcription levels where these treatments were not imposed, suggesting a positive response of this gene to these environmental stresses. Furthermore, G6PDH activity was stimulated under 4°C, CdCl2, NaCl and PEG treatments, but the increments varied with treatment and sampling time, implying positive response to abiotic stresses, similar to the transcript of the G6PDH gene. Ion conductivity measurements and a histochemical assay provided indirect evidence of the involvement of the ScG6PDH gene in defense reactions to the above-mentioned abiotic stresses.
Similar content being viewed by others
Introduction
When plants grow in the light, photosynthesis in green tissues can generate nicotinamide adenine dinucleotide phosphate (NADPH), which is used in processes that include carbon fixation, fatty acid synthesis and nitrogen assimilation. When plants grow in the dark, however, NADPH in photosynthetic or non-photosynthetic tissues is supplied by the oxidative pentose phosphate pathway (OPPP)1,2. Glucose-6-phosphate dehydrogenase (G6PDH, EC1.1.1.49) is an important regulating enzyme of the OPPP that catalyzes the conversion of glucose-6-phosphate to 6-phosphogluconolactone, provides NADPH for biosynthesis and supplies pentose for nucleic acid synthesis3,4. G6PDH exists widely in plants, animals and prokaryotes. It is generally accepted that G6PDH can be detected in the cytoplasm or plasmids of plants5. Based on this distribution, G6PDH is divided into cytosolic G6PDH and plastidic G6PDH. Two sites, a substrate-binding site (IDHYLG) and NADP-binding site (NEFVIRLQP), are highly conserved in the cytosolic G6PDH gene sequence6. In addition, two different types of plastidic G6PDH, P1 and P2, which evolved from a common ancestral gene, were discovered by researchers based on the alignment of mature proteins and signal peptides. The difference in signal peptides between P1 and P2 exists in the cleavage site, which is designated as (I/V/L)X(S/T/K)↓(S) for P1 and (I/V)X(S/T/A)↓(S/T/P/Q) for P27. Furthermore, Wendt et al. observed that the P1 and P2 isoforms showed different gene expression patterns in Solanum tuberosum L.8.
G6PDH is not only a rate-limiting enzyme, but also plays a role in response to biotic and abiotic stresses. The response of plant G6PDH has been examined under different types of environmental stresses. For example, Scharte et al.9 reported an increase in G6PDH activity in the resistant Nicotiana tabacum Samsun NN cultivar, but not in the susceptible Xanthi cultivar after infection with Phytophthora nicotianae. In addition, the role of the G6PDH isoenzyme in tobacco drought tolerance and flowering was verified using the RNAi method. These results demonstrate that G6PDH may play a crucial role during early-stage drought stress, plant development and pathogen defense. The tolerance of G6PDH to low temperature has been reported by several researchers10,11. A phenotypic difference between wild-type and T1 generation transgenic tobacco plants containing the PsG6PDH gene from Populus suaveolens was observed in a cold treatment experiment12. The results indicated that wild-type plants suffered earlier and recovered later from cold injury compared with transgenic lines subjected to a temperature change from 25°C to 4°C without cold acclimation. These results were consistent with the trends in peroxidase (POD) activity, malondialdehyde (MDA) content and superoxide dismutase (SOD) activity. Meanwhile, gene expression related to cold stress in transgenic tobacco plants over-expressing PsG6PDH was induced, indicating the role of PsG6PDH in the development of cold tolerance in tobacco. In Triticum aestivum L. subjected to 0.15 M NaCl stress, the transcript level of G6PDH was quantified using northern hybridization, which increased over time and reached a maximum at 12 h13. Zhang et al. discovered the key role of G6PDH in Oryza sativa suspension cells under salt stress, which was the result of the coordination of G6PDH and NAPDH oxidase to maintain cell redox balance14. The activity of the G6PDH enzyme responsive to heavy metal stresses in Phaseolus vulgaris L. and wheat has been reported to be modulated by zinc or aluminum15,16.
Sugarcane (Saccharum officinarum L.) is an important sugar and economic crop. In the past decade, sugarcane has accounted for more than 90% of sugar production in China and 80% of the world sugar production in 2012. The negative impact of abiotic stresses, such as cold, salt and drought, on plant metabolism, physiology and productivity results in yield loss. Among these factors, drought and cold are the two major environmental stresses which can affect or limit sugarcane growth and thus result in a serious decrease in sugarcane production, especially in China. Breeding resistant sugarcane cultivars is one effective strategy to overcome these problems. Considering of modern sugarcane variety is a complex of S. officinarum and S. spontaneum with a highly complicated genetic background of polyploidy and aneuploid and with about 120 or more chromosomes, it is not easy to obtain an ideal cultivar by means of traditional crossing and selection due to an adverse linkage of important industrial and agricultural traits. With the development of modern genetic engineering, it is practical to obtain highly resistant cultivars by introducing resistance genes to improve gene expression in modern cultivar derived from traditional crossing. While, unfortunately, sugarcane genome sequencing is just beginning and only very limited genome information is obtained. Therefore, to better understand the molecular mechanism of the stress response and the ability to avoid stress or increase tolerance to adverse growing conditions, it is beneficial to identify stress responsive genes in sugarcane. In recent years, some genes related to environmental stress have been cloned from sugarcane and investigated. For example, Guo et al. reported a ScDir gene cloned from sugarcane, which was involved in the response to drought, salt and oxidant stresses17. Su et al. reported two stress-related genes, i.e., ScGluA1 and ScGluD118 and a novel stress-induced Scdr1 gene19, which was isolated from sugarcane and displayed tolerance to drought, salt and oxidative stresses in transgenic tobacco plants. Transgenic tobacco plants containing the gene showed higher tolerance to drought, salinity and oxidative stress. To date, although the G6PDH gene has been isolated from several other plant species and investigated4,10,20, it remains unreported in sugarcane.
In this study, we investigated the role of the G6PDH gene in sugarcane (Accession number: KJ620023) subjected to CdCl2, polyethylene glycol (PEG), NaCl and cold treatments. Expression analysis determined using a real-time quantitative RT-PCR (RT-qPCR) and increased enzyme activities showed that ScG6PDH responded positively to environmental stresses. In addition, the role of this enzyme in the hypersensitive response (HR) in tobacco and its subcellular location was determined.
Results
Cloning and sequence analysis of ScG6PDH
A full length cDNA containing 1,646 bp, termed ScG6PDH (Accession number: KJ620023), was obtained from Yacheng05-179 and had an open reading frame (ORF) of 1,524 bp encoding 507 amino acid residues. In addition, ScG6PDH contained a 5′-UTR of 56 bp and a 3′-UTR of 66 bp (see Fig. 1A). Two conserved sites, i.e., a substrate-binding (IDHYLG) and NADP-binding site (NEFVIRLQP), were also found as shown in Fig. 1A. The predicted primary structural information is presented in Supplementary Table S1.
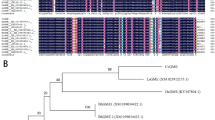
The cDNA sequence and coding amino acid sequence of ScG6PDH (A) and phylogenetic tree of the deduced amino acid sequences of G6PDHs from Saccharum officinarum and other plant species (B).
The substrate-binding site and NADP-binding site are underlined in (A). The plant species in (B) include Oryza sativa, Nicotiana tabacum, Solanum tuberosum, Petroselinum crispum, Triticum aestivum, Medicago sativa and Spinacia oleracea.
The amino acid sequence of ScG6PDH shares 77.50%, 91.34%, 77.34% and 83.63% homologies with the cytosolic G6PDH from tobacco (CAA04994), rice (CAC09489), potato (CAA52442) and wheat (BAA97663), respectively (see Fig. S1). The prediction results did not reveal an N-terminal signal peptide or transmembrane in ScG6PDH (see Supplementary Fig. S2 and Fig. S3). A phylogenetic tree shows that ScG6PDH belongs to the cytosolic G6PDH family (see Fig. 1B).
Subcellular localization
The ScG6PDH gene was fused with the pCAMBIA 2300 vector between the 35S promoter and GFP to generate the subcellular localization vector pCAMBIA 2300-ScG6PDH-GFP (see Fig. S4 and Fig. S5). When Agrobacterium-mediated transformation of Nicotiana benthamiana leaves had been initiated, green fluorescence was evident and appeared in the cytoplasm and cell nucleus under an laser scanning confocal microscope, while green fluorescence in the pCAMBIA 2300-ScG6PDH-GFP group was only observed in the cytoplasm (see Fig. 2 and Fig. S6).
Tissue specific expression analysis of ScG6PDH
The transcript level of ScG6PDH gene in different tissues in sugarcane variety Yacheng 05-179 was investigated with the GAPDH being used as an internal control. As showed in Fig. 3, the ScG6PDH transcript was observed in all the six types of tissues, including leaf, bud, root, stem epidermal, stem pith and leaf sheath. The ScG6PDH transcript level in the stem pith and bud was obviously higher than the other four tissue types, which was 2.9 times and 2.7 times higher respectively compared with sheath. The transcript level of the ScG6PDH in sheath was also significantly lower than leaf (1.4 times), bud (2.1 times) and root (1.6 times).
Tissue-specific expression analysis of the ScG6PDH in different tissues in sugarcane variety Yacheng05-179.
All data are normalized to the GAPDH transcript level and a data point is the mean ± SE (n = 3) Different lowercase letters indicate a significant difference, as determined by the least-significant difference test (p-value < 0.05).
Expression of ScG6PDH gene in E. coli and its growth under abiotic stresses
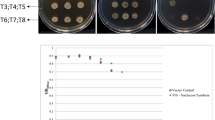
The results of recombinant protein, the blank and the mock induced for 2 h using isopropyl β-D-1-thiogalactopyranoside (IPTG) were shown in Fig. 4A. Target protein of ScG6PDH expressed successfully in E. coli Rosetta cells was obviously observed with the band approximate 60 kDa molecular mass (Lane 6 in Fig. 4A). Due to the presence of 6-HIS tag in the pET28a vector17, the molecular mass of the specific protein showed in Fig. 4A was higher than that of the estimated 57 kDa.
The expression of ScG6PDH gene in E. coli and its growth under abiotic stresses.
Protein expression of pET28a-ScG6PDH in E. coli Rosetta cells induced by IPTG was shown in (A). Lane 1: blank without induction; 2: blank with induction for 2 h; 3: mock without induction; 4: mock with induction for 2 h; 5: pET28a-ScG6PDH without induction; 6: pET28a-ScG6PDH with induction for 2 h; 7: protein marker. The black arrow showed the ScG6PDH protein. Liquid culture assay of Rosetta/pET28a-ScG6PDH in LB media with 250 mM NaCl, 15% PEG and 750 μM CdCl2 was shown in (B), (C) and (D) respectively (n = 3).
The growth curves of Rosetta strain cells in liquid culture under different stresses were shown in Fig. 4. Interestingly, when NaCl, CdCl2 or PEG was added in the media respectively, Rosetta cells expressed ScG6PDH protein showed decreased growth in cell quantity than those without ScG6PDH. It seemed the protein ScG6PDH expressed in Rosetta cells didn't increase the growth ability in these abiotic stresses.
Expression profiles of the ScG6PDH gene under environmental stresses in sugarcane
To investigate the role of the ScG6PDH gene in response to environmental stresses in sugarcane plantlets of Yacheng05-179, the expression profiles were detected using RT-qPCR under CdCl2 (500 μM), NaCl (250 mM), PEG 8000 (25%) and 4°C treatments. As shown in Fig. 5, the up-regulation expression trends of ScG6PDH were similar after exposure to different environmental stresses. However, the ScG6PDH transcript was strongly induced by exogenous CdCl2 and low temperature stresses and was 43 times higher after 12 h (CdCl2 stress) and 129 times higher after 24 h (4°C stress) compared with the control. Although the increase in ScG6PDH transcript levels induced by NaCl and PEG was significant, the increment was considerably lower than those induced by CdCl2 and 4°C stresses.
RT-qPCR analysis of the ScG6PDH gene expression profiles in Yacheng05-179 plantlets under various abiotic stresses.
Data were normalized to the GAPDH transcript level. (A–D): The transcript level of ScG6PDH subjected to 500 μM CdCl2, low temperature (4°C), 250 mM NaCl and 25% PEG 8000, respectively. The data points represent the means ± SE (n = 3). Different lowercase letters indicate significant differences determined using the least significant difference test (p-value < 0.05).
Changes in enzyme activity of G6PDH under various environmental stresses
Based on the different degrees of increased transcript levels of ScG6PDH gene under the four stress treatments, the activity of G6PDH was measured to investigate changes in protein abundance. As shown in Fig. 6, G6PDH activity peaked after 12 h (175.9% of the control level) and remained higher than that of the control though with a little of decrease after treatment with 500 μM CdCl2, while its activity remained enhanced during the time course of 4°C stress and reached a peak of 269.2% of the control on 24 h, showing the same trends with the transcript levels. The salt treatment on seedlings resulted in only a little change before 24 h and a sharp enhancement was observed at 24 h to 48 h (193.2% of the control level at 48 h), also showing its positive role in response to salt stress. Meanwhile, a similar change tendency was observed under the treatment 25% PEG 8000 and the maximum at 48 h was 163.6% of that of the control.
Transient expression of ScG6PDH induces a defense response in tobacco
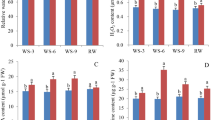
To investigate the role of ScG6PDH in the hyper-sensitive response and immunity in plants, an overexpressed pCAMBIA 1301-ScG6PDH vector was constructed and an Agrobacterium-mediated transient expression method was performed to identify the effect of ScG6PDH expression on the induction of the defense response in tobacco leaves. The expression of H2O2 can be used as an early signal molecule of the plant-pathogen interaction that reflects the extent of plant hypersensitive cell death. Here, H2O2 was observed as a brown leaf color using the 3,3'-diaminobenzidine solution (DAB) staining method. As shown in Fig. 7, a typical hypersensitive response with enhanced ion conductivity (see Fig. 7A) and deeper DAB staining color (see Fig. 7B) in leaves expressing ScG6PDH was observed in 48 h after infection. This was significantly higher than that of the control without ScG6PDH. Both the ion conductivity and histochemical assays demonstrated that the ScG6PDH gene may play a role in hypersensitive cell death response and defense reaction.
The transient expression of ScG6PDH in tobacco.
(A): Ion conductivity to assess the cell death response in leaf discs infiltrated by Agrobacterium tumefaciens strain EHA105 for 48 h (n = 5). (B): DAB staining of ScG6PDH in Nicotiana benthamiana leaves infiltrated by Agrobacterium tumefaciens strain EHA105 for 48 h to assess H2O2 production (n = 5). Images were captured using a Canon camera.
Discussion
As a key enzyme of the pentose phosphate pathway, G6PDH can catalyze the synthesis of 6-phosphogluconolactone and the production of NAPDH. Because of the connection between protein location and function, knowledge of the G6PDH location is useful to determine its role in plants. In previous reports, G6PDH was classified into two types based on location, i.e., plastidic and cytosolic G6PDH, both encoded by the nuclear gene and with similar molecular mass (approximately 56 kDa)5. One report provided further evidence for the functional role of one cytosolic and two plastidic (P1 and P2) isoforms, suggesting each isoform appeared to have a different regulatory mechanism and played distinct roles in growth and stress tolerance1. In this study, we isolated the ScG6PDH gene (Accession number: KJ620023) from sugarcane. This gene contains two conserved sites, i.e., a substrate-binding (IDHYLG) and a NADP-binding site (NEFV IRLQP) (see Fig. 1A), sharing high homology (77.34%–91.34%) in amino acid sequences with cytosolic G6PDH from other plant species (see Fig. S1). In addition, according to the phylogenetic tree (see Fig. 1B), ScG6PDH was classified as part of the cytosolic G6PDH family and bearing the closest relationship to cytosolic OsG6PDH (Accession number: CAC09489) (http://www.ncbi.nlm.nih.gov/protein/CAC09489). This agrees with our prediction of no signal peptide or transmembrane, indicating that the ScG6PDH presented here may belong to the cytosolic isoform. Based on the subcellular location results of the recombinant protein that exists in the cytoplasm, we suggest that ScG6PDH belongs to the cytosolic G6PDH family.
In recent years, studies on G6PDH have focused on its response to abiotic stresses. Transgenic tobacco lines carrying PsG6PDH from poplar exhibited an increasing cold tolerance compared with wild plants and a previous hypothesis that increased cytosolic G6PDH activity might involve the induction of freezing resistance in poplar cuttings was also confirmed12. The overexpression of Cvcg6pdh from Chlorella vulgaris can lead to improved freezing tolerance in Saccharomyces cerevisiae, indicating that G6PDH would be an indirect factor in the development of freezing tolerance10. In the present study, the ScG6PDH transcript level was significantly increased when sugarcane was subjected to 4°C stress. Consistent with these previous reports, G6PDH activity increased at a protein level, indicating that ScG6PDH may be involved in the response to cold stress in sugarcane.
In comparison, reports on the role of the G6PDH gene in response to metal stress are limited. Based on a previous study, when the metal (zinc and cadmium) content of bean leaves exceeded a toxic threshold value, an increase in the activity of the G6PDH enzyme was measured16. Moreover, a rapid increase in G6PDH activity in aluminum-resistant wheat cultivars was observed during the first 10 h of treatment with 100 μM aluminum, while no change was detected in sensitive cultivars during 24 h exposure, suggesting that G6PDH may be associated with the mechanism of aluminum tolerance through regulation of the OPPP15. In addition, changes in G6PDH transcript levels were investigated, showing a 43-fold increase after 12 h exposure to CdCl2 treatment compared with the control; these high levels were maintained until 24 h. In our study, the G6PDH activity also showed an increase and reached a maximum level after 12 h. Further, the higher transcript levels (in comparison to the control) were maintained for the duration of the investigation period, implying possible involvement of G6PDH in response to metal stress in sugarcane, similar to previous research.
Several studies have shown that G6PDH plays a key role in resistance to salt stress. For example, G6PDH played a role in maintaining cell redox balance in rice suspension cells under salt stress, indicating the coordination of G6PDH and NADPH oxidase14. In Phragmites communis Trin subjected to salt stress, G6PDH played a crucial role in maintaining the glutathione (GSH) pool21. In addition, in wheat treated with high salt levels, there was a significant increase in either the G6PDH transcript or its enzymatic activity13. However, in salt-stressed Arabidopsis, the G6PDH transcript was down-regulated based on a global analysis of expression profiles22. Interestingly, in all rice plants treated with high salinity, cold temperatures, PEG or abscisic acid (ABA) under experimental conditions, the transcript of the G6PDH gene remained constant, unlike the G6PDH genes in wheat (up-regulated) and Arabidopsis (down-regulated). Therefore, the role of G6PDH in salt stress tolerance is unclear based on the distinct expression patterns in different plant species. In the current study, we performed RT-qPCR to investigate the ScG6PDH expression pattern in sugarcane subjected to salt stress. At 12 h after treatment, there were indications of up-regulation of the transcript of the ScG6PDH gene (see Fig. 5), which is a similar response to the G6PDH gene in wheat. In addition, a distinct increase in the ScG6PDH enzymatic activity measured at 24 to 48 h and which corresponded to the transcript of the ScG6PDH gene under salt treatment, implied a positive response to salt stress in sugarcane.
Drought is a common abiotic stress limiting plant growth and crop production. To date, many reports have shown the positive role of the G6PDH gene to drought stress. G6PDH could provide GSH and ascorbate by utilizing NADPH to participate in the regulation of root development after drought stress. This was recently confirmed in Glycine max roots subjected to drought stress23. Further, the role of G6PDH in the cytosol as a key factor in determining plant development, drought stress and early pathogen defense has been demonstrated9. In sugarcane, we observed an increase in both the ScG6PDH transcript and its enzymatic activity after drought stress (stimulated by PEG) (see Fig. 5, Fig. 6), which was consistent with the G6PDH increase in soybean roots23, indicating the likely involvement of the ScG6PDH gene in drought stress in sugarcane.
It has been shown that reactive oxygen species (ROS) accumulate under unfavourable conditions that include cold temperatures, salt and drought21,24,25. Lower levels of ROS could be related to signal transduction pathways, while high levels of ROS accumulation may affect cell redox states and result in oxidative damage26. Plant defense reactions under oxidative stress rely on the balance between ROS production and scavenging. There are two efficient antioxidative defense systems in plants to scavenge excessive ROS. One system is composed of enzymes (e.g., SOD, catalase (CAT), ascorbate peroxidase (APX) and POD) involved in hydrogen peroxide and superoxide radical scavenging; the other system is comprised of non-enzymatic antioxidants, including a variety of secondary metabolites such as ascorbate and GSH. These systems maintain appropriate oxidative and reductive states in plants exposed to various types of stress25,27,28,29. NADPH, which is an important reductant molecule, is required in the ascorbate-glutathion cycle involved in oxidative stress30. This molecule plays a vital role in maintaining the GSH content in cells. Therefore, NADPH is considered as the most important molecule to determine the potential antioxidant capacity of the cell. Furthermore, the OPPP, with G6PDH as the key enzyme, is the major source of NADPH production. When plants suffer oxidative stress, more NADPH is required to maintain a normal redox state. This may result in an increase of G6PDH, which was confirmed by our results. Therefore, we can conclude that G6PDH plays a vital role in various oxidative stresses by supplying sustainable levels of NADPH to maintain oxidative-reductive balance.
Our conclusion of the role of G6PDH in abiotic stresses was also supported by the ion conductivity and DAB staining results. Hypersensitive response cell death can restrict pathogen growth as well as stimulate the defense reaction of nearby tissues and systemic acquired resistance31,32 of the entire plant. Here, DAB staining showed a deep brown color in the presence of H2O2 in tobacco leaves after 48 h infiltration and increased electrolyte leakage, suggesting the ScG6PDH gene was associated with HR cell death, which is consistent with a previous report33. The production of H2O2 through oxidative burst has been reported to be closely connected with HR cell death34. We deduce that the transient over-expression of the ScG6PDH gene may be related to HR, providing indirect evidence that ScG6PDH is involved in the plant defense reaction.
Wendt et al. ever demonstrated that in contrast with the plastidic P1 isoform, which mRNA amount was accumulated in the leaves, stolons harvested from tissue growing above ground and roots harvested from tissue growing hydroponically in northern blots in potato5, the P2 was also expressed ubiquitously more or less by northern blot analysis in potato, which was similar to the expression pattern of the cytosolic G6PDH gene8. In Arabidopsis, several G6PDH isoforms was studied based on genome-wide analysis. Among them, two cytosolic G6PDH isoforms of AtG6PDH5 and AtG6PDH6 showed different expression pattern in tissues, with the mRNA from AtG6PDH5 being detected at high levels in leaves and AtG6PDH6, as the major cytosolic isoform of G6PDH, showing similar expression levels in all tissues throughout the plants1. In our study, the tissue specific transcript analysis of ScG6PDH showed to be more or less ubiquitously in six distinct sugarcane tissues, including leaf, bud, root, stem pith, stem epidermal and sheath, with the highest expression (see Fig. 3), being consistent with the previous reports. The distribution pattern of ScG6PDH may be related to its vital function in sugarcane closely. We speculate that it showed higher expression level in the stem pith and epidermal because of its vital role as the key enzyme in glucose metabolism, as the stem is the key tissue to accumulate sucrose. Besides, it also may be related to the sucrose transport in sugarcane, as the NADPH is one important hydrogen dornor. Meanwhile, the high level of ScG6PDH gene expression in buds may be resulted from its involvement in the redox reaction under abiotic stresses above-mentioned, as the redox reaction is stronger in meristem. In the same way, its expression level in sheath is lower than others because of its main protection role with lower redox reaction in it.
As showing in Fig. 4, ScG6PDH was induced by IPTG strongly in E. coli Rosetta strain, making it convenient and possible to do the further protein study. In spite of the consistent induction results were observed in sugarcane and in other plants17,18, however, according to our experiments, the tolerance to the stresses of CdCl2, NaCl, PEG and low temperature (4°C) in recombinant procaryotic Rosetta cells declined, unlike previous results18, which maybe due to the difference between the prokaryotic and eukaryotic expression system. Dai et al. also found the similar phenomenon that the chicken interferon-gamma gene expression level and biologic activity of its product in E. coli were different from the expression pattern in COS-1 cells35. Struhl also illuminated the different logic of gene regulation between eukaryotes and prokaryotes36. Therefore, it is reasonable to deduce the disparity may be due to the great differences in the physiological and biological metabolism and in genomic background between eukaryotes and prokaryotes, such as lack of post-translation modification in prokaryotes. In addition, it has been proven that excess NADPH represses the growth of E. Coli cells but it could be recovered by introducing a NADPH-consuming pathway to some extent37. So, we also deduced that our result may result from the inhibitory effect of excess NADPH which couldn't be consumed by E. coli cells in time. Of course, this is only a preliminary speculation which needs further research.
In summary, we isolated the ScG6PDH gene (Accession number: KJ620023) from sugarcane. Based on the results of bioinformatic prediction, the analysis of the phylogenetic tree and the subcellular location experiment, the ScG6PDH gene is a member of the cytosolic G6PDH gene family. On the basis of up-regulated ScG6PDH transcript levels and an increase in enzymatic activities under CdCl2, low temperature (4°C), NaCl and PEG treatments, we propose that the ScG6PDH gene may play a positive role in sugarcane in response to environmental stresses such as heavy metals, cold temperatures, salt and drought. In addition, the DAB staining and ion conductivity experiments suggest the expression of the ScG6PDH gene is related to HR and therefore provides indirect evidence for the involvement of this gene in defense reactions.
Methods
Materials
The sugarcane variety Yacheng05-179 was provided by the key laboratory of Sugarcane Biology and Genetic Breeding, Ministry of Agriculture (Fuzhou, China). The Escherichia coli rosette, prokaryotic expression vector pET28a, the subcellular localization vector pCAMBIA 2300 and the plant expression vector pCAMBIA 1301 were obtained from Abmart, Inc. (Tokyo, Japan). The restriction enzymes SalI, SacI, XhoI, NheI, T4 DNA ligase, Ex-Taq enzyme, PrimeScript RT-PCR Kit, TaKaRa LA PCRTM in vitro Cloning Kit, DNA and protein molecular marker were purchased from TaKaRa (Tokyo, Japan). RQ1 RNase-Free DNase was purchased from Promega Corporation (Beijing, China), the SYBR Green PCR Master Mix Kit was provided by Roche (Shanghai, China) and the NADPNa2 and D-glucose 6-phosphate disodium salt were purchased from Sigma (San Francisco, CA, USA).
Cloning and sequence analysis of the ScG6PDH cDNA
Using the Zea maysG6PDH gene (Accession number: LOC100284317) as the querying probe, several highly homologous sugarcane expressed sequence tags (ESTs) were obtained from the sugarcane EST database. These selected ESTs were clustered and spliced. Then, the new sugarcane contig was obtained and used as a new probe to search for more sugarcane ESTs. Using this method, a putative novel gene sequence was obtained. In order to isolate this gene from sugarcane, the primers ScG6PDHF: 5′-TCTCGCTCCGCCAGAAATCTCG-3′ and ScG6PDHR: 5′-AGGGCACTCATCC TAGATAGTAGAT-3′ were designed according to the assembled sequence. The 25-μL PCR system contained 2.5 μL 10× PCR buffer, 2.0 μL deoxynucleotide triphosphates (dNTPs, 2.5 mM), 1.0 μL each of forward and reverse primers (10 μM), 2.0 μL cDNA of the sugarcane variety Yacheng05-179 (100 ng·μL−1), 0.125 μL Ex-Taq enzyme (5.0 U·μL−1) and 17.375 μL ddH2O. The PCR program included pre-denaturation for 4 min at 94°C, denaturation for 30 s at 94°C, annealing for 45 s at 58°C, extension for 90 s at 72°C, for 35 cycles and final extension for 10 min at 72°C.
The open reading frame (ORF) of the full-length cDNA sequence of the ScG6PDH gene was predicted using the ORF Finder online tool (http://www.ncbi.nlm.nih.gov/projects/gorf/). The signal 4.1 server program (http://www.cbs.dtu.dk/services/SignalP/) and TMHMM server 2.0 software (http://www.cbs.dtu.dk/services/TMHMM-2.0/) were used to analyze signal peptide and transmembrane segments. The homology alignment among the ScG6PDH and other G6PDHs was performed using DANMAN software. A phylogenetic tree of the deduced amino acid sequences of G6PDHs was constructed according to the neighbor-joining method using 4.1 MEGA software.
Subcellular localization
The ScG6PDH gene was sub-cloned with SalI and SacI sites into the pCAMBIA 2300 vector and then transformed into the Agrobacterium tumefaciens strain EHA105. First, the EHA105 cells were inoculated into LB medium containing kanamycin (50 μg·mL−1) and rifampicin (34 μg·mL−1), shaken overnight at 200 rpm and 28°C and then 1.0 mL LB medium was inoculated into a new LB medium containing kanamycin (50 μg·mL−1) and rifampicin (34 μg·mL−1). After culturing at 28°C with shaking (200 rpm) for approximately 8 h, the OD600 of the culture was measured and diluted to OD600 = 0.8 using Murashige and Skoog (MS) liquid medium (containing 200 μM acetosyringone). A syringe was used to infiltrate the diluted bacterial suspension into tobacco leaves. Injected plants were cultured under 12 h light/12 h dark photoperiod18. After two days, the leaves were collected and the subcellular localization was observed using laser scanning confocal microscopy (Leica, Wetzlar, Germany).
The protolast isolation and transient expression of ScG6PDH-GFP fusion gene were also carried out. Choose well-expended leaves from 4-week-old tabacco plants before flowing. Cut 1 mm leaf strips from the middle part of a leaf using a fresh sharp razor blade without tissue crushing at the cutting site. The leaf strips were transfered quickly and gently into the prepared solutiob containing 20 mM Fatty Acid Methyl Ester Sulfonate (MES) pH 5.7, 1.5% cellulase R10, 0.4% macerozyme R10, 0.4 M mannitol and 20 mM KCl, 0.1% bovine serum albumin (BSA) and 10 mM CaCl2. The digestion was carried out at room tempreature with gently shaking for 4 h. Then check for the release of protaplasts in the solution under microscope. Then dilute the enzyme solution with an equal volume of W5 solution (2 mM MES pH 5.7, 154 mM NaCl, 125 mM CaCl2 and 5 mM KCl) before filtration to remove undigested leaf tissues with a clean 70 μM nylon mesh. The flowpthrough was centrifuge at 130 rpm for 2 min to pellet the protoplasts in a 50 mL round-bottomed tube. Remove as much supernatant as possible and re-suspend the protoplast pellet by gentle swirling. Remove the W5 solution as much as possible without touching the protoplast pellet after resting the protoplas by keeping on ice for 30 min. Then the MMG solution (4 mM MES pH 5.7, 0.4 M mannitol, 100 mM CaCl2) was added to re-suspend protoplasts at room temperature. Add 10 μg DNA, 100 μL protoplasts and 110 μL PEG solution (30% PEG 4000, 0.2 M mannitol, 100 mM CaCl2) to a 2-mL microfuge tube and mix gently by tapping the tube. The transfection mixture was incubated at room temperature for 10 min and diluted with 400 μL W5 solution gently to stop the transfection process. Centrifuge at 130 rpm for 2 min and remove supernatant. 1 mL WI solution (4 mM MES pH 5.7, 0.5 M mannitol and 20 mM KCl) was add to resuspend protoplasts in each well of a 6 - well tissue culture plate. At last, the protoplasts were observed with laser scanning confocal microscope (Leica, Wetzlar, Germany) after incubation at room temperature for 18 h.
SDS-PAGE analysis of prokaryotic expression product
In order to study the prokaryotic expression of the target gene, the pET28a-ScG6PDH recombinant plasmid was constructed. Firstly, the ScG6PDH gene ORF with the XhoI and NheI sites was amplified and then inserted into the plasmid pET28a (+). The recombinant vector pET28a-ScG6PDH was verified by the PCR and sequencing. After that, the positive clone was transformed into the E. coli Rosetta strain to generate the putative recombinants.
The empty pET28a (+) and pET28a-ScG6PDH were separately transformed into the E. coli Rosetta strains, respectively. The single clone from the Luria-Bertani (LB) medium plate was transferred into the liquid LB medium with kanamycin (50 μg·mL−1) and chloramphenicol (170 μg·mL−1). After shaking overnight at 200 rpm at 37°C, 300 μL cells were inoculated into new LB medium containing kanamycin (50 μg·mL−1) and chloramphenicol (170 μg·mL−1) and shaken at 200 rpm at 37°C until the OD600 reached 0.4–0.6. 300 μL LB medium was collected as control and the isopropyl β-D-1-thiogalactopyranoside (IPTG) was added into the rest LB medium which was up to a final concentration of 1.0 mM. After 2 h of inducing, 300 μL LB medium was collected. In this process, both of the LB medium with empty vector of pET28a (+) (Rosetta) and the blank E. coli Rosetta strains were also induced with IPTG by the same method as the pET28a-ScG6PDH (Rosetta). The collected cells were mixed with 30 μL 5× protein loading buffer and then boiled for 5 min at 100°C. After centrifugation, 8 μL supernatant was taken to conduct electrophoresis in 12% SDS-PAGE loading. The gel was colored with coomassie brilliant blue dye solution.
Liquid culture assay
The effects of NaCl, PEG, CdCl2 on the growth of E. coli cells with recombinant plasmid and vector alone were investigated. As described in the study17, after the cultured E. coli Rosetta cells were diluted to OD600 = 0.6, about 300 μL of cells were inoculated into 200 mL of LB liquid medium (50 μg·mL−1 kanamycin and 170 μg·mL−1 chloramphenicol) containing 250 mM NaCl, 15% PEG (water potential of −0.4 Mpa) and 750 μM CdCl2, respectively. Then the LB liquid medium was cultured at 37°C with shaken at 200 rpm for growth measurement by harvesting every 2 h, measured with ultraviolet spectrophotometer (Purkinje General, Beijing, China).
Expression profiles of the ScG6PDH gene under various environmental stresses and in different tissues
Healthy and constantly growing sugarcane plantlets, derived from tissue culture of Yacheng05-179, were grown under a 16 h light/8 h dark photoperiod at 28°C for one week and then treated with different exogenous stresses (i.e., 500 μM CdCl2, 250 mM NaCl, 25% PEG 8000 (water potential of −0.8 MPa) and 4°C temperature). The sampling times for these treatments were 0 h, 12 h and 24 h and all samples were stored in a −80°C refrigerator until RNA extraction.
RNA extraction was conducted using the TRIzol reagent (Invitrogen, Shanghai, China) procedure and reverse transcription was carried out following the instructions of the Prime-Script™ RT Reagent Kit (TaKaRa, Japan). The GAPDH (glyceraldehyde-3-phosphate dehydrogenase) (5′-CACGGCCACTGGAAGCA-3′ and 5′-TCCTCAGGGTTCCTGATGCC-3′) gene was used as an internal control during the real-time quantitative PCR (RT-qPCR)38. The primers used in the RT-qPCR were designed using the primer 5.0 software according to the ScG6PDH sequence. The forward primer was ScG6PDHQF: 5′-AAGCCTGGGTTGGAAATGG-3′ and the reverse primer was ScG6PDHQR: 5′-CTCTGCGGACGAAGTGCTG-3′. The RT-qPCR was performed using the ABI PRISM7500 real-time PCR system (Applied Biosystems, Shanghai, China) and the procedure was conducted for 2 min at 50°C, 10 min at 95°C, 15 s at 94°C and 60 s at 60°C for 40 cycles. The 2−ΔΔCT method was used to analyze the results and three biologic replicates and three technical replicates were used for each sample. Significant differences in gene expression levels were assessed using Student's t-test (p-value < 0.05).
Determination of G6PDH activity
Briefly, 0.5 g of tissue-cultured seedlings were ground and suspended in 1.5 mL of extract buffer containing 50 mM Hepes-Tris (pH 7.8), 3 mM MgCl2, 1 mM ethylene diamine tetraacetic acid (EDTA), 1 mM phenylmethylsulfonyl fluoride and 1 mM dithiothreitol. The homogenates were centrifuged (12,000 rpm) at 4°C for 20 min. Then, a 100-μL aliquot of extract was added to the assay buffer containing 50 mM Hepes-Tris (pH 7.8), 3.3 mM MgCl2, 0.5 mM D-glucose 6-phosphate disodium salt and 0.5 mM NADPNa2. The reduction of NADP to NADPH was measured as the change rate of the absorbance at 340 nm for the initial 5 min. One unit (U) of enzyme activity was defined as the amount of enzyme that increased a 0.01 of absorbance at 340 nm per minute under the assay condition. Significant differences in gene expression levels were assessed using Student's t-test (p-value < 0.05).
Ion conductivity measurement
Cell death was quantified by measuring ion leakage as described previously39,40. Ion conductivity was determined by washing six discs/leaves (10 mm diameter) in 20 mL of double distilled water followed by incubation with gentle shaking for 1 h at room temperature. Conductivity was measured using a conductivity meter (Mettler Toledo, Shanghai, China). Five biologic replications were carried out.
Histochemical assay
DAB was used to stain H2O2-producing leaves41. The leaves were incubated in DAB solution (1.0 mg·mL−1, pH = 5.8) overnight (dark conditions). Then, the leaves were placed into 95% alcohol and treated with boiling water for 5 min until the green color faded. The leaves were rinsed in 95% alcohol, photographed and examined. Five biologic replications were carried out.
References
Wakao, S. & Benning, C. Genome-wide analysis of glucose-6-phosphate dehydrogenases in Arabidopsis. Plant J. 41, 243–256; 10.1111/j.1365-313X.2004.02293.x (2005).
Wakao, S., Andre, C. & Benning, C. Functional analyses of cytosolic glucose-6-phosphate dehydrogenases and their contribution to seed oil accumulation in Arabidopsis. Plant Physiol. 146, 277–288; 10.1104/pp.107.108423 (2008).
Copeland, L. & Turner, J. F. The regulation of glycolysis and the pentose-phosphate pathway. The biochemistry of plants 107–125 (Academic Press, New York, 1987).
Graeve, K., von Schaewen, A. & Scheibe, R. Purification, characterization and cDNA sequence of glucose-6- phosphate dehydrogenase from potato (Solanum tuberosum L.). Plant J. 5, 353–361; 10.1111/j.1365-313X.1994.00353.x (1994).
Von Schaewen, A., Langenkamper, G., Graeve, K., Wenderoth, I. & Scheibe, R. Molecular characterization of the plastidic glucose-6-phosphate dehydrogenase from potato in comparison to its cytosolic counterpart. Plant Physiol. 109, 1327–1335; 10.1104/pp.109.4.1327 (1995).
Lin, Y. Z. et al. Cloning and sequence analysis of a glucose-6-phosphate dehydrogenase gene PsG6PDH from freezing-tolerant Populus suaveolens. For. Stud. China 7, 1–6; 10.1007/s11632-005-0048-2 (2005).
Wendt, U. K. et al. Evidence for functional convergence of redox regulation in G6PDH isoforms of cyanobacteria and higher plants. Plant Mol. Biol. 40, 487–494; 10.1023/A,1006257230779 (1999).
Wendt, U. K., Wenderoth, I., Tegeler, A. & von Schaewen, A. Molecular characterization of a novel glucose-6-phosphate dehydrogenase from potato (Solanum tuberosum L.). Plant J. 23, 723–733; 10.1046/j.1365-313x.2000.00840.x (2000).
Scharte, J., Schön, H., Tjaden, Z., Weis, E. & von Schaewen, A. Isoenzyme replacement of glucose-6-phosphate dehydrogenase in the cytosol improves stress tolerance in plants. PNAS 106, 8061–8066; 10.1073/pnas.0812902106 (2009).
Honjoh, K. et al. Molecular cloning and characterization of a cDNA for low-temperature inducible cytosolic glucose 6-phosphate dehydrogenase gene from Chlorella vulgaris and expression of the gene in Saccharomyces cerevisiae. Plant Sci. 172, 649–658; 10.1016/j.plantsci.2006.12.004 (2007).
Lin, S. Z. et al. Characterization and role of glucose-6-phosphate dehydrogenase of Populus suaveolens in induction of freezing resistance. For. Stud. China 6, 1–7; 10.1007/s11632-004-0022-4 (2004).
Lin, Y. Z., Lin, S. Z., Guo, H., Zhang, Z. Y. & Chen, X. Y. Functional analysis of PsG6PDH, a cytosolic glucose-6-phosphate dehydrogenase gene from Populus suaveolens and its contribution to cold tolerance improvement in tobacco plants. Biotechnol. Lett. 35, 1509–1518; 10.1007/s10529-013-1226-2 (2013).
Nemoto, Y. & Sasakuma, T. Specific expression of glucose-6-phosphate dehydrogenase (G6PDH) gene by salt stress in wheat (Triticum aestivum L.). Plant Sci. 158, 53–60; 10.1016/S0168-9452(00)00305-8 (2000).
Zhang, L., Liu, J., Wang, X. M. & Bi, Y. R. Glucose-6-phosphate dehydrogenase acts as a regulator of cell redox balance in rice suspension cells under salt stress. Plant Growth Regul. 69, 139–148; 10.1007/s10725-012-9757-4 (2013).
Ślaski, J. J., Zhang, G. C., Basu, U., Stephens, J. L. & Taylor, G. J. Aluminum resistance in wheat (Triticum aestivum) is associated with rapid, Al-induced changes in activities of glucose-6-phosphate dehydrogenase and 6-phosphogluconate dehydrogenase in root apices. Physiol. Plantarum 98, 477–484; 10.1111/j.1399-3054.1996.tb05702.x (1996).
Van Assche, F., Cardinaels, C. & Clijsters, H. Induction of enzyme capacity in plants as a result of heavy metal toxicity: Dose-response relations in Phaseolus vulgaris L., treated with zinc and cadmium. Environ. Pollut. 52, 103–115; 10.1016/0269-7491(88)90084-X (1988).
Guo, J. L. et al. A novel dirigent protein gene with highly stem-specific expression from sugarcane, response to drought, salt and oxidative stresses. Plant cell Rep. 31, 1801–1812; 10.1007/s00299-012-1293-1 (2012).
Su, Y. C. et al. Molecular cloning and characterization of two pathogenesis-related β-1, 3-glucanase genes ScGluA1 and ScGluD1 from sugarcane infected by Sporisorium scitamineum. Plant Cell Rep. 32, 1503–1519; 10.1007/s00299-013-1463-9 (2013).
Begcy, K. et al. A novel stress-induced sugarcane gene confers tolerance to drought, salt and oxidative stress in transgenic tobacco plants. PLoS One 7, 1–14; 10.1371/journal.pone.0044697 (2012).
Lin, S. Z. et al. Role of glucose-6-phosphate dehydrogenase in freezing-induced freezing resistance of Populus suaveolens. J. Plant Physiol. Mol. Biol. 31, 34–40; (2005).
Wang, X. M. et al. Glucose-6-phosphate dehydrogenase plays a central role in modulating reduced glutathione levels in reed callus under salt stress. Planta 227, 611–623; 10.1007/s00425-007-0643-7 (2008).
Kreps, J. A. et al. Transcriptome changes for Arabidopsis in response to salt, osmotic and cold stress. Plant Physiol. 130, 2129–2141; 10.1104/pp.008532 (2002).
Liu, J., Wang, X. M., Hu, Y. F., Hu, W. & Bi, Y. Glucose-6-phosphate dehydrogenase plays a pivotal role in tolerance to drought stress in soybean roots. Plant Cell. Rep. 32, 415–429; 10.1007/s00299-012-1374-1 (2013).
Gong, H. L. et al. Involvement of G6PDH in regulation of heat stress tolerance in the calli from Przewalskia tangutica and tobacco. Biol. Plantarum 56, 422–430; 10.1007/s10535-012-0072-8 (2012).
Hasanuzzaman, M. & Fujita, M. Selenium pretreatment upregu-lates the antioxidant defense and methylglyoxal detoxification system and confers enhanced tolerance to drought stress in rapeseed seedlings. Biol. Trace Elem. Res. 143, 1758–1776; 10.1007/s12011-011-8998-9 (2011).
Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 7, 405–410; 10.1016/S1360-1385(02)02312-9 (2002).
Gill, S. S. & Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 48, 909–930; 10.1016/j.plaphy.2010.08.016 (2010).
Li, Y., Liu, Y. & Zhang, J. Advances in the research on the AsA-GSH cycle in horticultural crops. Front. Agric. China 4, 84–90; 10.1007/s11703-009-0089-8 (2010).
Noctor, G. & Foyer, C. H. Ascorbate and glutathione: keeping active oxygen under control. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 249–279; 10.1146/annurev.arplant.49.1.249 (1998).
Leterrier, M., del Rio, L. A. & Corpas, F. J. Cytosolic NADP-isocitrate dehydrogenase of pea plants: genomic clone characterization and functional analysis under abiotic stress conditions. Free Radic. Res. 41, 191–199; 10.1080/10715760601034055 (2007).
Hammond, K. E. & Jones, J. D. G. Plant disease resistance genes. Plant Mol. Biol. 48, 575–607; 10.1146/annurev.arplant.48.1.575 (1997).
Greenberg, J. T. & Yao, N. The role and regulation of programmed cell death in plant pathogen interactions. Cell Microbiol. 6, 201–211; 10.1111/j.1462-5822.2004.00361.x (2004).
Asai, S. et al. A plastidic glucose-6-phosphate dehydrogenase is responsible for hypersensitive response cell death and reactive oxygen species production. J. Gen. Plant Pathol. 77, 152–162; 10.1007/s10327-011-0304-3 (2011).
Levine, A., Tenhaken, R., Dixon, R. & Lamb, C. H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79, 583–593; 10.1016/0092-8674(94)90544-4 (1994).
Dai, J. H., Qin, A. J., Xu, J. J., Liu, Y. L. & Jin, W. J. Cloning and expression of chicken interferon-gamma gene. Chinese J. Prev. Vet. Med. 25, 249–253 (2003).
Struhl, K. Fundamentally different logic of gene regulation in eukaryotes and prokaryotes. Cell 98, 1–4; 10.1016/S0092-8674(00)80599-1 (1999).
Kabir, M. M. & Shimizu, K. Fermentation characteristics and protein expression patterns in a recombinant Escherichia coli mutant lacking phosphoglucose isomerase for poly(3-hydroxybutyrate) production. Appl. Microbiol. Biot. 62, 244–255; 10.1007/s00253-003-1257-z (2003).
Que, Y. X. et al. Selection of control genes in real-time qPCR analysis of gene expression in sugarcane. China J. Trop. Crop. 30, 274–278 (2009).
Hwang, I. S. & Hwang, B. K. The pepper mannose-binding lectin gene CaMBL1 is required to regulate cell death and defense responses to microbial pathogens. Plant Physiol. 155, 447–463; 10.1104/pp.110.164848 (2011).
Lee, D. H., Choi, H. W. & Hwang, B. K. The pepper E3 ubiquitin ligase RING1 gene, CaRING1, is required for cell death and the salicylic acid-dependent defense response. Plant Physiol. 156, 2011–2025; 10.1104/pp.111.177568 (2011).
Hwang, I. S. & Hwang, B. K. Requirement of the cytosolic interaction between pathogenesis-related protein10 and leucine-rich repeat protein1 for cell death and defense signaling in pepper. Plant Cell 24, 1675–1690; 10.1105/tpc.112.095869 (2012).
Acknowledgements
This work was funded by the earmarked fund for the Modern Agriculture Technology of China (CARS-20) and the 948 Program on the Introduction of International Advanced Agricultural Science and Technique of Department of Agriculture (2014-S18).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Y.Y. and L.X. Performed the experiments: Y.Y., Z.F., Y.S., X.Z. and G.L. Analyzed the data: Y.Y., Z.F. and Y.S. Wrote the paper: Y.Y. and L.X. Revised the final version of the paper: L.X., J.G. and Y.Q. Approved the final version of the paper: L.X.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Yang, Y., Fu, Z., Su, Y. et al. A cytosolic glucose-6-phosphate dehydrogenase gene, ScG6PDH, plays a positive role in response to various abiotic stresses in sugarcane. Sci Rep 4, 7090 (2014). https://doi.org/10.1038/srep07090
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07090
This article is cited by
-
Biotechnological Intervention for Sugarcane Improvement Under Salinity
Sugar Tech (2023)
-
Ethylene insensitive mutation alleviates cadmium-induced photosynthesis impairment in Arabidopsis plants
Acta Physiologiae Plantarum (2022)
-
Nitric oxide and hydrogen peroxide increase glucose-6-phosphate dehydrogenase activities and expression upon drought stress in soybean roots
Plant Cell Reports (2020)
-
Comparative physiology and transcriptome analysis allows for identification of lncRNAs imparting tolerance to drought stress in autotetraploid cassava
BMC Genomics (2019)
-
Mechanism(s) of action of heavy metals to investigate the regulation of plastidic glucose-6-phosphate dehydrogenase
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.