Abstract
Asexual propagules are the dominant means of propagation in most submerged macrophytes. To improve the understanding of how water nutrient concentrations and population density influence the turion production of Potamogeton crispus L., the turions were planted in mesocosms with three water nutrient conditions (ambient lake water, high P and high N) and two plant density levels (4 and 15 turions m−2). After a 9-month experiment, the +P in the water column significantly increased the total turion number per plant under both of the plant density treatments. However, the +N in the water column did not affect the turion number per plant under low plant density. The +P in the water and high plant density significantly reduced the turion individual biomass. An examination of 3210 turion individuals from all treatments revealed that the increased water nutrient concentrations and plant density impacted the turion size by producing different stem diameters of individual turions. Most of the scale leaf morphological traits of the turions were significantly increased under higher water nutrients, but these traits were similar between the different plant density treatments. These results demonstrate that the water P concentration interacts with plant density, affecting both the production and traits of turions.
Similar content being viewed by others
Introduction
Most submerged macrophytes produce asexual propagules, which are the major propagules used for the dispersal and propagation of submerged macrophytes1,2,3. For submerged macrophytes, asexual propagules generally include the rhizome, creep stem, tuber, turion and stem fragment4,5. Compared to seeds, turions are relatively large and they are able to photosynthesize and/or take up nutrients; thus, these asexual propagules are considered to have a greater potential than seeds for successful population expansion in submerged macrophytes2,3,6. Therefore, a comprehensive understanding of asexual propagule production, which is closely related to the environment, is important in submerged macrophyte population ecology.
Most rooted submerged macrophytes are able to take up nutrients (e.g., nitrogen and phosphorus) through roots, but they also can utilize nutrients from water column via shoots7,8,9,10. However, few studies have investigated the effect of water nutrient concentrations on asexual reproduction in submerged macrophytes. For instance, Smith et al.11 found that the water potassium concentration did not significantly affect the auto-fragment number and biomass of Myriophyllum spicatum L. However, Wang et al.12 concluded that Potamogeton crispus L. significantly decreased its turion number and the dry matter production of turions per plant under a high water P concentration (25 mg L−1). Although many previous studies have indicated that high nutrient conditions may reduce the asexual propagation of submerged macrophytes, most of these studies in aquatic habitats have focused on single factors (e.g., nutrient availability), whereas evidence of multiple factors is lacking.
Plant density is another important factor that affects the growth and propagation of submerged macrophytes, as it can influence the absorption or uptake of nutrients and/or light5,13. Previous studies have demonstrated that differences in resource availability among patches (i.e., environmental heterogeneity) can be created even at small scales5,14. Competition among neighboring plants can also magnify the individual size difference on a small scale, as it can alter the resource distribution within a patch and the patchiness of resource availability15. For instance, Guan et al.16 found that plant density significantly affected individual vegetative production in Alternanthera philoxeroides. Many studies have focused primarily on the growth and morphological parameters of submerged macrophytes (e.g., biomass and stem number), but few experimental studies have addressed the role of plant density on the propagules of submerged macrophytes13,17.
Several previous studies have also demonstrated that plant density has a feedback regulation effect on environmental resources, suggesting interactions between nutrient availability and plant density18,19. Indeed, in environments with increased plant density, nutrient absorption is generally reduced due to inactive root activity and an insufficient energy supply19. However, if a high nutrient supply is also available, plants might gain constructive material for root growth and stem elongation to increase nutrient acquisition5,19. In addition, high levels of nutrients can promote shoot production5,20 and might increase the total amount of stored carbohydrates (i.e., soluble sugar and starch) in submerged macrophytes21, which could help the plants to resist the effects of high plant density. Nevertheless, our understanding of the mechanisms by which the growth and propagation of submerged macrophytes are affected by the interaction between nutrient availability and plant density remains limited.
Potamogeton crispus L. is a perennial submerged macrophyte that is a dominant species in freshwater regions throughout eastern Asia22,23. In the subtropical or warmer temperate regions of the Northern Hemisphere (e.g., eastern Asia), the life cycle of P. crispus is different from that of most submerged macrophytes. P. crispus sprouts in autumn, grows throughout winter and increases its biomass rapidly in the following spring24,25. P. crispus flowers and produces bur-like turions almost continuously from late May until the aboveground portions decompose in the summer25,26. The seed production of P. crispus is high, but the seed germination is very low (approximately 0.001%)24. Therefore, the primary means of propagation for P. crispus is via turions24,27,28. The newly formed turions are always green and exhibit a high sprouting rate (approximately 100%); however, the turions turn brown and exhibit a relatively low sprouting rate after a period of summer dormancy20,29.
The turions of P. crispus are formed by specialized shoot apices that are composed of modified leaves (also called scale leaves) and an enlarged stem (Fig. 1). The turions are normally formed when temperature is higher than 20°C and the photoperiod is longer than 12 h26. Wang et al.12 and Xie and Yu20 reported that the low water/sediment nutrient concentrations would stimulate the turion formation processes (increase turion number or turion biomass fraction). A typical turion also contains 3 to 7 dormant buds, which are directly related to the (potential) sprouting performance of P. crispus plantlets27,30. Turions of P. crispus store large amounts of total nonstructural carbohydrates (30–70% dry weight) and mineral nutrients (TN and TP)20,28,31. As turions cannot take up nutrients from the surrounding environment until new roots/shoots are formed, the main function of turions' storage could thus be supporting the growth of new roots/shoots32,33,34,35. Previous studies have mainly focused on how environmental factors influence turion germination27, formation20 and dispersal29. Our previous study also revealed that high sediment nutrients concentration would reduce the turion size (biomass) of P. crispus20. However, few studies have investigated the combined effect of the water nutrient level and plant density on the turion production of P. crispus. In this study, we tested the effects of different water nutrient conditions and plant densities on turions and the morphological differences among turions. We hypothesize that increasing water nutrient and plant density may reduce the P. crispus turion sizes through morphological characteristics changes.
Methods
Materials
This experiment was conducted at the National Observation and Research Field Station for Lake Ecosystem in Liangzi Lake, China (30°05′–30°18′N, 114°21′–114°39′E). From 15–17 October 2011, mature, healthy turions of similar sizes (not grazed or broken, 5 or 6 green scale leaves) were collected from the sediment of Liangzi Lake (3 collection sites, 100 turions per site). These turions have been on the sediment for at least 2 months. The water depths at the collection sites were 0.5 to 1.2 m, the sediment compositions were 2.35–2.88 mg g−1 TN, 0.10–0.15 mg g−1 TP, 9.33–12.18% organic matter, 1.20–1.66 g cm−3 bulk density and 40–72% water content and the sites were covered with monospecific stands of P. crispus. The 300 turions were kept in a greenhouse in containers that were filled with fresh lake water to a depth of 20 cm (water temperature 21.4 ± 3.8°C, mean ± SE). These turions were kept for approximately 3 weeks until they pre-sprouted and 135 turions (turion fresh weight 1.58 ± 0.02 g, mean ± SE), each with one healthy plantlet (plantlet length: 7 to 10 cm), were selected for the subsequent experiment.
Experimental design
This experiment was conducted outdoors from 22 November 2011 to 10 August 2012. At the beginning of the experiment, the selected sprouting turions were cultivated in 25-cm-deep muddy substratum (2.75 mg g−1 TN, 0.10 mg g−1 TP, 10.53% organic matter, 1.33 g cm−3 bulk density and 57% water content) that was extracted from Liangzi Lake with three water nutrient and two plant density level treatments. Each treatment was replicated three times (n = 3), with 45 turions per variant within one repetition. All of the turions were cultivated in fiberglass tanks (depth 100 cm (gap); 785 L, 18 tanks in total). Three different water nutrient treatments were tested: the ambient nutrient treatment used the fresh lake water from Liangzi Lake (TN 0.83 ± 0.14 mg L−1 and TP 0.04 ± 0.01 mg L−1 during the experimental period); the +N treatment, 2.62 g NH4NO3 added to each tank (with a water TN concentration of approximately 2.0 mg L−1); and the +P treatment, 0.57 g KH2PO4 added to each tank (with a water TP concentration of approximately 0.2 mg L−1). The nutrient values for the water nutrient treatments were based on a eutrophic lake that is approximately 30 km away from Liangzi Lake (Yanglan Lake, 30°00′–30°06′N, 114°32′–115°05′E) (TN 2.53 ± 0.17 mg L−1 and TP 0.13 ± 0.03 mg L−1)36. The nutrient concentrations (TN and TP) of each tank were measured at 10-d intervals using an IL-500 N nitrogen analyzer and an IL-500P phosphorus analyzer (Hach Company, Loveland, Colorado, USA). To maintain constant concentrations of nutrients, NH4NO3 and KH2PO4 were added at 10-d intervals after the nutrient concentrations were measured. Within each water nutrient treatment, two different turion densities were tested: low density, with 3 turions per tank (equivalent to 4 plants m−2) and high density, with 12 turions per tank (equivalent to 15 plants m−2). These densities approximate those occurring in natural populations of P. crispus in Liangzi Lake (turion density: 3 to 22 m−2).
During the experimental period, under water light intensity and water temperature were recorded daily at noon (see Fig. 2). The water turbidities (ranged from 0.66 to 1.99 NUT) and water pH values (ranged from 7.6 to 9.6) in tanks were recorded at 10-d intervals. All parameters were measured at a depth of 20 cm.
Turion harvest
The newly formed turions were harvested when they were mature and when the aboveground portions of P. crispus decomposed (on 10 August 2012). Upon collection, the turions were first classified according to the number of scale leaves and these turions were then cleaned with light jet water and divided into scale leaves and stems. The following turion characteristics were measured: number of turions per tank, number of turions per type, length and diameter of turion stems and length and width of turion scale leaves. If the turion number was too large to measure, 80 turions per type were randomly collected for each treatment replicate. All of the turions were dried at 70°C for 72 h and then weighted to obtain the dry-weight biomass.
Statistical analysis
All of the experimental data were transformed using the log(x) or log(x + 1) function to satisfy the conditions of the homogeneity of variance or normal distribution of the residuals. The transformed data were then analyzed. The turion number and biomass per plant and average turion biomass were analyzed using a two-way ANOVA with water nutrients and plant density as the fixed factors. After harvest, all of the turions had a scale leaf number from 3 to 11. However, turions with a scale leaf number from 8 to 11 were rare (5 turions in total). Therefore, all of the turions in our experiment were classified based on a scale leaf number from 3 to 7. The variances in the morphological traits (i.e., turion stem length, turion stem diameter and length and width of turion scale leaf) of the turions with different scale leaf numbers among the treatments were analyzed using a generalized linear model (GLM) ANOVA. Duncan's comparison test was used to test the significance of all the above differences (P < 0.05). The data were analyzed using SPSS 18.0 software (SPSS, Chicago, IL, USA).
Results
During the experimental period, all of the plantlets survived. The water nutrient conditions significantly affected the total number per plant of the P. crispus turions (F2,17 = 37.6, P < 0.001) and the interactions of water nutrient and plant density were significant (F2,17 = 11.6, P = 0.002). However, the plant density did not significantly affect the total number per plant or total biomass of turions (F1,17 = 2.2, P = 0.160). Under both of the plant density treatments, the +P treatment significantly increased the total turion number. However, the +N increased the total turion number per plant only in the high-plant-density treatment (Fig. 3 A). Under both of the plant density conditions, the average turion biomass was similar among the nutrient treatments, whereas the average turion biomass was significantly increased in both the control (ambient lake water) and +N treatments under low-plant-density conditions (Fig. 3 B).
Differences in the turion number per plant (A) and average turion biomass (B) between the water nutrient and plant density treatments. The data are presented as means ± SE (n = 3). The bars with different letters indicate significant differences among the treatments (P < 0.05, two-way ANOVA with Duncan's correction). The data were transformed using the log(x) function.
The turion size varied among the treatments. Under low-plant-density conditions, P. crispus grown in lake water and in the +N environment produced larger propagules (turions with 6 and 7 scale leaves) than those produced in the +P environment. In addition, higher percentages of large turions were produced by the plants that grew in a low-plant-density environment (Fig. 4 and Table 1).
Percentages of turion number classified with different scale leaf number under different water nutrient conditions and at different plant densities (high plant density: open symbols; low plant density: filled symbols).
The different symbols indicate the different water nutrient treatments ( Lake water,
Lake water,  +P and
+P and  +N). All of the data were transformed using the log(x + 1) function.
+N). All of the data were transformed using the log(x + 1) function.
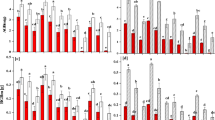
Neither the water nutrient conditions nor the plant density significantly affected the stem length of the turions across most of the turion types that were classified with different scale leaf numbers (Appendix fig. 1 A–E, Table 2). However, in the low-plant-density treatment, the lake water and +N treatments significantly increased the turion stem diameter across all types of turions (except for turions with 4 scale leaves) (Appendix fig. 1 F–J and Table 2). In addition, in most types of turions, the increase in the N and P concentrations in the water significantly increased the length and width of the turion scale leaves (except for the length of the scale leaves on turions with 3 scale leaves). However, the plant density did not significantly influence the length and/or width of the turion scale leaves in most of the turion types (Appendix fig. 2 A–J and Table 2).
Discussion
Previous studies have demonstrated that slightly increasing the P concentration in the water column (0.25 mg L−1) only slightly increased the turion production in P. crispus, whereas the turion number and biomass were considerably reduced when the water P concentration was increased to 25 mg L−112. However, in our experiment, the increase in the water P concentration (0.20 mg L−1) significantly increased the total number of formed turions per plant in both plant density treatments. These conflicting conclusions may have resulted from different responses of plants to different experimental protocols (e.g., the long-term outdoor experiment in our study). Appenroth and Nickel37 indicated that the timing of turion formation increased (i.e., took more time to form turion) with the higher water P concentration. In addition, turion production was not solely influenced by the N and P concentrations in the water column but was also dependent on an adequate daily temperature range and photoperiod38,39. Therefore, a long-term perspective should be considered when studying the nutrient impacts on the asexual propagation of submerged macrophytes in future studies. In the field, however, an increase in nutrient concentrations in the water column may cause a heavily phytoplankton- and epiphyton-dominated state, thereby negatively impacting the growth or propagation of submerged macrophytes36,40. However, in our experiment, the water was relative clear (water turbidities ranged from 0.66 to 1.99 NUT). Neither increase in the N and P concentrations in the water column influenced the water clarity. The increase in the nitrogen concentration of the water did not significantly increase the turion number per plant at low plant density, which may contribute to the significant acute biochemical changes in submerged macrophytes due to relative high ammonium levels (approximately 0.7 mg L−1), possibly leading to substantial biochemical damage41,42 and a decrease in asexual propagule production.
Sediments are the main nutrient source of rooted macrophytes. One of our previous studies indicated that under nutrient-poor sediment (0.74 mg g−1 TN and 0.03 mg g−1 TP) P. crispus allocated more biomass to turion fraction and produced more large-sized turions (>100 mg)20, indicating that under low-nutrient-availability conditions, the plants decreased their investment in the leaf and stem biomass, resulting in a decreased supply of resources to growing propagules, but not to new propagules and allowing the plants to produce larger turions20,43. Similarly, results from present experiment were partly consistent with those conclusions that P. crispus produced more small sized turions in low-nutrient conditions (in both water column and sediment). Path analysis from our previous study20 also revealed that leaf and stem biomass of P. crispus had direct negative effects on total turion biomass, which consequently affected turion size and number. Unfortunately, we failed to harvest the P. crispus aboveground biomass in this experiment due to the aboveground portions decomposition at high summer temperature. Solid evidence cannot be provided in this experiment that how nutrients affect the total plant size and consequently affected the turion size. Therefore, in our future studies, the plant sizes, which can be estimated by some modern technologies (e.g., photo image technology), should be thoroughly considered and the different effects of sediment nutrients and water column nutrients on plant reproduction should also be considered.
Most previous studies reported that high plant density can cause growth inhibition in submerged macrophytes, consequently decreasing the asexual propagule size (biomass)5,18. This result was consistent with our finding that P. crispus decreased its individual turion biomass with the increase in plant density. Wang et al.44 showed that plants from the high-density treatment usually produced less shoot biomass than did plants from the low-density treatment due to light and/or nutrient competition. Indeed, it is common for perennial clonal plants to not produce asexual propagules in habitats with high plant density because of low asexual propagule survival45,46. However, the turion number and turion biomass were similar between the different plant density treatments, whereas the interactions between the water nutrient and the plant density treatments were significant. Therefore, the effect of plant density on turion formation is a result of the indirect negative effect that interacted with water nutrient treatments. These results are consistent with previous studies in reproductive allocation5,15. Moreover, the water pH values in high-density treatments (ranged from 7.8 to 9.6) were higher than that in low-density treatments (ranged from 7.6 to 8.3) in our experiment. The water pH value changes may also explain the turion size differences between different density treatments. Because the high water pH value condition would reduce the photosynthetic rate in submerged macrophytes47 and, thus, may also reduce the turion sizes.
The scale leaf number of individual turions varied among the treatments. Under low-plant-density conditions, the P. crispus plants grown in an environment with a low water P concentration (i.e., ambient lake water and high water N treatments) produced larger propagules, whereas the plants grown in an environment with a high water P concentration produced smaller propagules, which was consistent with previous studies of many submerged macrophytes in sediment nutrients5,20,43. Previous studies pointed out that changes in the water nutrient concentrations (i.e., N and P) also trigger changes in the internal C–N metabolism in P. crispus12,48, which has a direct effect on chlorophyll synthesis and photosynthesis, thereby affecting turion production due to variations in carbohydrate synthesis12. However, the direct evidence on such process is limited.
The present study also demonstrated that P. crispus produces larger turions when under less competition. The survival of a propagule is key to a plant's ability to reproduce optimally in various environments45, as the ability to produce larger turions leads to a higher probability of survival29,49. Therefore, it is reasonable to believe that under high-plant-density environments, propagule development is constrained due to high intra-specific competition, leading to the production of smaller propagules5. Under high-plant-density conditions, plants tend to produce more shoot biomass (i.e., leaves and stems) when nutrient levels increase in response to enhanced light competition5,13,50, which directly reduces turion production and indirectly reduces the turion size and number20. However, our study did not directly support this and further deeper studies are needed.
Previous studies have demonstrated that P. crispus produced more and smaller turions under nutrient-rich than under nutrient-poor conditions12,20. Recent studies have also indicated that turions with larger sizes produced longer plantlet heights and sprout biomass than did those with smaller sizes51,52. The turion biomass was used as an index to indicate the turion size in these studies. However, the scale leaf number of turions had a more direct correlation with the turion sprouting abilities27, which were significantly correlated with turion morphology. Therefore, the scale leaf number should be used to classify the turions of P. crispus.
Recent studies have demonstrated that the non-structural carbohydrate or soluble protein reserves in the asexual propagules of aquatic macrophytes could be readily mobilized and used for sprouting or regrowth52,53,54,55. Unfortunately, we did not measure the reserve concentration in the turions of P. crispus. Future studies should pay more focus on how these reserves in the turion stems are mobilized and used for the sprouts of P. crispus. In the present study, increases in the water P concentration and plant density treatment significantly reduced the turion stem diameter in most turions with different scale leaf numbers. There is evidence that most turions in the field give rise to only one sprout27. Evidence in other aquatic plants has demonstrated that large stems increase the survival rate and growth of stem fragments55,56. Therefore, the potential establishment abilities of turions under high P and plant density conditions may decrease due to the small turion stem size produced under such environments57, which may be an adaptive strategy of P. crispus to cope with severe disturbances (e.g., water nutrient and plant density increase) in the field.
Conclusions
In the present study, the +P treatment significantly increased the total number of P. crispus turions per plant under both high and low plant densities. However, the average individual turion biomass was reduced due to the increased P concentration in the water column. An assessment of 3210 turion individuals with different scale leaf numbers revealed that the stem diameters were lower under increased P and high-plant-density treatments. In addition, most of the scale leaf morphological traits were affected by the water nutrient treatments but not the plant density treatments across all types of turions.
Some studies have documented that abiotic stresses can influence the regeneration abilities of asexual propagules by affecting the sizes of the storage organs55,58. Our results support these findings showing that increased P concentration and plant density reduce the turion stem diameters in P. crispus; our results also highlight that the potential sprouting abilities of P. crispus turions may differ in different environments. This strategy may contribute greatly to the sprouting of turions and facilitate the expansion of P. crispus into frequently disturbed, locally submerged communities.
References
Santamaría, L. Why are most aquatic plants widely distributed? Dispersal, clonal growth and small-scale heterogeneity in a stressful environment. Acta Oecol. 23, 137–154 (2002).
Riis, T. & Sand-Jensen, K. Dispersal of plant fragments in small streams. Freshwater Biol. 51, 274–286 (2006).
Boedeltje, G., Ozinga, W. A. & Prinzing, A. The trade-off between vegetative and generative reproduction among angiosperms influences regional hydrochorous propagule pressure. Global. Ecol. Biogeogr. 17, 50–58 (2008).
Barrat-Segretain, M. H. Strategies of reproduction, dispersion and competition in river plants: a review. Plant Ecol. 123, 13–37 (1996).
Xie, D. & Yu, D. Size-related auto-fragment production and carbohydrate storage in auto-fragment of Myriophyllum spicatum L. in response to sediment nutrient and plant density. Hydrobiologia 658, 221–231 (2011).
Boedeltje, G., Bakker, J. P., Bekker, R. M., Van Groenendael, J. M. & Soesbergen, M. Plant dispersal in a lowland stream in relation to occurrence and three specific life-history traits of the species in the species pool. J. Ecol. 91, 855–866 (2003).
Carignan, R. An empirical model to estimate the relative importance of roots in phosphorus uptake by aquatic macrophytes. Can. J. Fish Aquat. Sci. 39, 243–247 (1982).
Denny, P. Sites of nutrient absorption in aquatic macrophytes. J. Ecol. 60, 819–829 (1972).
Takayanagi, S., Takagi, Y., Shimizu, A. & Hasegawa, H. The shoot is important for high-affinity nitrate uptake in Egeria densa, a submerged vascular plant. J. Plant Res. 125, 669–678 (2012).
Madsen, T. V. & Cedergreen, N. Sources of nutrients to rooted submerged macrophytes growing in a nutrient-rich stream. Freshwater Biol. 47, 283–291 (2002).
Smith, D. H., Madsen, J. D., Dickson, K. L. & Beitinger, T. L. Nutrient effects on autofragmentation of Myriophyllum spicatum. Aquat. Bot. 74, 1–17 (2002).
Wang, L. et al. Growth and turion formation of Potamogeton crispus in response to different phosphorus concentrations in water. Aquat. Ecol. 47, 87–97 (2013).
Xie, D., Yu, D., You, W. H. & Xia, C. X. The propagule supply, litter layers and canopy shade in the littoral community influence the establishment and growth of Myriophyllum aquaticum. Biol. Invasions 15, 113–123 (2013).
Xiao, K., Yu, D. & Wang, J. Habitat selection in spatially heterogeneous environments: a test of foraging behaviour in the clonal submerged macrophyte Vallisneria spiralis. Freshwater Biol. 51, 1552–1559 (2006).
Sugiyama, S. & Bazzaz, F. A. Size dependence of reproductive allocation: the influence of resource availability, competition and genetic identity. Funct. Ecol. 12, 280–288 (1998).
Guan, B., An, S., Schooler, S. & Cai, Y. Influence of substrate phosphorus concentration and plant density on growth and phosphorus accumulation of Alternanthera philoxeroides. J. Freshwater Ecol. 25, 219–225 (2010).
Rodríguez-Gironés, M. A., Sandsten, H. & Santamaría, L. Asymmetric competition and the evolution of propagule size. J. Ecol. 91, 554–562 (2003).
Jiang, J. et al. Sediment type, population density and their combined effect greatly charge the short-time growth of two common submerged macrophytes. Ecol. Eng. 34, 79–90 (2008).
Xie, Y., An, S., Wu, B. & Wang, W. Density-dependent root morphology and root distribution in the submerged plant Vallisneria natans. Environ. Exp. Bot. 57, 195–200 (2006).
Xie, D. & Yu, D. Turion production and nutrient reserves in Potamogeton crispus are influenced by sediment nutrient level. Aquat. Biol. 14, 21–28 (2011).
Xie, D., Yu, D., You, W.-H. & Wang, L.-G. Morphological and physiological responses to sediment nutrients in the submerged macrophyte Myriophyllum spicatum. Wetlands 33, 1095–1102 (2013).
Xie, Y., Yu, D. & Geng, X. Effects of elevated CO2 concentration on phenotypic, physiological and biochemical characteristics of submersed plant Potamogeton crispus leaf. Acta Phytoecol. Sin. 27, 218–222 (2003).
Kunii, H. Life cycle and growth of Potamogeton crispus L. in a shallow pond, Ojaga-ike. J. Plant Res. 95, 109–124 (1982).
Rogers, K. H. & Breen, C. M. Growth and reproduction of Potamogeton crispus in a South African lake. J. Ecol. 68, 561–571 (1980).
Sastroutomo, S. S. Turions formation, dormancy and germination of curly pondweed, Potamogeton crispus L. Aquat. Bot. 10, 161–173 (1981).
Wu, J., Cheng, S., Liang, W., He, F. & Wu, Z. Effects of sediment anoxia and light on turion germination and early growth of Potamogeton crispus. Hydrobiologia 628, 111–119 (2009).
Jian, Y., Li, B., Wang, J. & Chen, J. Control of turion germination in Potamogeton crispus. Aquat. Bot. 75, 59–69 (2003).
Woolf, T. E. & Madsen, J. D. Seasonal biomass and carbohydrate allocation patterns in Southern Minnesota curlyleaf pondweed populations. J. Aquat. Plant Manage. 41, 113–118 (2003).
Xie, D., Yu, D., Xia, C. & You, W. Stay dormant or escape sprouting? Turion buoyancy and sprouting abilities of the submerged macrophyte Potamogeton crispus L. Hydrobiologia 726, 43–51 (2013).
Bolduan, B. R., van Eeckhout, G. C., Quade, H. W. & Gannon, J. E. Potamogeton crispus-the other invader. Lake Reserv. Manag. 10, 113–125 (1994).
Kunii, H. Continuous growth and clump maintenance of Potamogeton crispus L. in Narutoh river, Japan. Aquat. Bot. 33, 13–26 (1989).
Adamec, L. Dark respiration and photosynthesis of dormant and sprouting turions of aquatic plants. Fund. Appl. Limnol. 179, 151–158 (2011).
Adamec, L. Tissue mineral nutrient content in turions of aquatic plants: does it represent a storage function? Fund. Appl. Limnol. 176, 145–151 (2010).
Yu, J. et al. Effects of salinity and water depth on germination of Phragmites australis in coastal wetland of the Yellow River Delta. Clean – Soil Air Water 40, 1154–1158 (2012).
Yi, N. et al. Eichhornia crassipes cleans wetlands by enhancing the nitrogen removal and modulating denitrifying bacteria community. Clean – Soil Air Water 41, 1–10 (2013).
Xie, D., Yu, D., You, W. & Wang, L. Algae mediate submerged macrophyte response to nutrient and dissolved inorganic carbon loading: A mesocosm study on different species. Chemosphere 93, 1301–1308 (2013).
Appenroth, K. J. & Nickel, G. Turion formation in Spirodela polyrhiza: The environmental signals that induce the developmental process in nature. Physiol. Plant. 138, 312–320 (2010).
Netherland, M. D. Turion Ecology of Hydrilla. J. Aquat. Plant Manage. 35, 1–10 (1997).
Thullen, J. S. Production of axillary turions by the dioecious Hydrilla verticillata. J. Aquat. Plant Manage. 28, 11–15 (1990).
Sousa, W. T. Z. Hydrilla verticillata (Hydrocharitaceae), a recent invader threatening Brazil's freshwater environments: a review of the extent of the problem. Hydrobiologia 669, 1–20 (2011).
Cao, T., Ni, L. & Xie, P. Acute biochemical responses of a submersed macrophyte, Potamogeton crispus L., to high ammonium in an aquarium experiment. J. Freshwater Ecol. 19, 279–284 (2004).
Cao, T., Ni, L., Xie, P., Xu, J. & Zhang, M. Effects of moderate ammonium enrichment on three submersed macrophytes under contrasting light availability. Freshwater Biol. 56, 1620–1629 (2011).
Hangelbroek, H. H., Santamaría, L. & de Boer, T. Local adaptation of the pondweed Potamogeton pectinatus to contrasting substrate types mediated by changes in propagule provisioning. J. Ecol. 91, 1081–1092 (2003).
Wang, J. W., Yu, D., Xiong, W. & Han, Y. Q. Above-and belowground competition between two submersed macrophytes. Hydrobiologia 607, 113–122 (2008).
Takada, T. & Nakajima, H. The optimal allocation for seed reproduction and vegetative reproduction in perennial plants: an application to the density-dependent transition matrix model. J. Theor. Biol. 182, 179–191 (1996).
Jongejans, E., de Kroon, H. & Berendse, F. The interplay between shifts in biomass allocation and costs of reproduction in four grassland perennials under simulated successional change. Oecologia 147, 369–378 (2006).
Mvungia, E. F., Lyimo, T. J. & Björk, M. When Zostera marina is intermixed with Ulva, its photosynthesis is reduced by increased pH and lower light, but not by changes in light quality. Aquat. Bot. 102, 44–49 (2012).
Zhang, M. et al. Ammonium, microcystins and hypoxia of blooms in eutrophic water cause oxidative stress and C–N imbalance in submersed and floating-leaved aquatic plants in Lake Taihu, China. Chemosphere 82, 329–339 (2011).
Barrat-Segretain, M. H., Henry, C. P. & Bornette, G. Regeneration and colonization of aquatic plants fragments in relation to the disturbance frequency of their habitat. Arch. Hydrobiol. 145, 111–127 (1999).
Xie, D., Yu, D., Yu, L. F. & Liu, C. H. Asexual propagations of introduced exotic macrophytes Elodea nuttallii, Myriophyllum aquaticum and M. propinquum are improved by nutrient-rich sediments in China. Hydrobiologia 655, 37–47 (2010).
Xie, D., Zhou, H., Ji, H. & An, S. A. The growth and establishment of floating turions of Potamogeton crispus are not favored by summer climate. CLEAN - Soil, Air, Water doi/10.1002/clen.201300836 (2014).
Xie, D., Zhou, H., Ji, H., Chen, Y. & An, S. Effects of buoyancy and season on turion dispersal of submerged macrophyte Potamogeton crispus L. CLEAN - Soil, Air, Water doi/10.1002/clen.201300833 (2014).
Deng, Z. M. et al. Effects of size and vertical distribution of buds on sprouting and plant growth of the clonal emergent macrophyte Miscanthus sacchariflorus (Poaceae). Aquat. Bot. 104, 121–126 (2013).
Dong, B. C. et al. How internode length, position and presence of leaves affect survival and growth of Alternanthera philoxeroides after fragmentation? Evol. Ecol. 24, 1447–1461 (2010).
Puijalon, S., Piola, F. & Bornette, G. Abiotic stresses increase plant regeneration ability. Evol. Ecol. 22, 493–506 (2008).
Dong, B. C., Zhang, M. X., Alpert, P., Lei, G. C. & Yu, F. H. Effects of orientation on survival and growth of small fragments of the invasive, clonal plant Alternanthera philoxeroides. PLoS ONE 5, e13631 (2010).
Shen, J., Xu, W. & Shi, F. Effects of weight and store temperature on turion germination and seedling growth of Potamogeton crispus L. B. Bot. Res. 28, 477–481 (2008).
Puijalon, S., Lena, J.-P. & Bornette, G. Interactive effects of nutrient and mechanical stresses on plant morphology. Ann. Bot. 100, 1297–1305 (2007).
Acknowledgements
Ligong Wang provided laboratory/field assistance. Dr. Zhong Wang, Shufeng Fan, Chengxing Xia, Zhigang Wu and Jinning Zhu provided useful comments in discussing the manuscript. This research was supported by the National Natural Science Foundation of China (31370382).
Author information
Authors and Affiliations
Contributions
Q.C. and D.X. wrote the main manuscript text. Q.C., D.X. and D.Y. designed and executed the technical assays. Q.C. and W.H.Y. contributed to planning the experiments, the interpretation of the data and writing and editing the manuscript. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Appendix Figures
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Qian, C., You, W., Xie, D. et al. Turion morphological responses to water nutrient concentrations and plant density in the submerged macrophyte Potamogeton crispus. Sci Rep 4, 7079 (2014). https://doi.org/10.1038/srep07079
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07079
This article is cited by
-
Asexual reproduction for overwintering of the submersed macrophyte Vallisneria spinulosa at different light intensities
Aquatic Sciences (2022)
-
How does turions production of Potamogeton crispus L. respond to parental population biomass and living environment
Environmental Science and Pollution Research (2022)
-
Nitrogen and phosphorus removal in simulated wastewater by two aquatic plants
Environmental Science and Pollution Research (2021)
-
The contribution of Potamogeton crispus to the phosphorus budget of an urban shallow lake: Lake Monger, Western Australia
Limnology (2016)
-
Differences in the regeneration traits of Potamogeton crispus turions from macrophyte- and phytoplankton-dominated lakes
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.