Abstract
BRCA2 gene plays an important role in homologous recombination. Polymorphic variants in this gene has been suggested to confer cancer susceptibility. Numerous studies have investigated association between BRCA2 N372H polymorphism and risk of several cancers, especially breast cancer. However, the results were inconsistent. We performed a comprehensive meta-analysis to provide a more precise assessment of the association between N372H and cancer risk, following the latest meta-analysis guidelines (PRISMA). Forty six studies involving 36299 cases and 48483 controls were included in our meta-analysis. The crude ORs and the 95% CIs were used to evaluate the strength of the association. The results indicated that the BRCA2 N372H variant was significantly associated with an increased risk of overall cancer (dominant model: OR = 1.07, 95% CI = 1.01–1.13; recessive model: OR = 1.12, 95% CI = 1.02–1.23). Moreover, stratified analyses by the cancer type and source of control observed significantly increased risk associated with BRCA2 N372H in subgroups with ovarian cancer, non-Hodgkin lymphoma and population-based controls, but not breast cancer or hospital-based controls. We also found such association among Africans. Overall, the meta-analysis suggested that BRCA2 N372H may be a cancer susceptibility polymorphism. Well-designed and large-scale studies are needed to substantiate the association between BRCA2 N372H polymorphism and cancer risk.
Similar content being viewed by others
Introduction
Cancer remains one of the leading causes of death worldwide, with approximately 8.2 million cancer-related deaths in 2012 (GLOBOCAN 2012). It has long been recognized that the imbalance between DNA damage and repair plays pivotal role in carcinogenesis. As an example, environmental agents such as mutagenic chemicals and certain types of radiation may induce various DNA alterations, which, if not repaired properly, would cause genetic instability, gene mutations and chromosomal alterations1. Therefore, the host DNA repair systems are important in protecting the genomic integrity against cancer-causing agents.
There are a variety of DNA repair pathways in humans, with each responsible for repairing a certain type of DNA damage2. Relatively simple single stranded DNA damages are usually restored via three common mechanisms, base excision repair, nucleotide excision repair and DNA mismatch repair1,2. In contrast, the repair of double-strand DNA breaks (DSBs), the most severe damage, requires more complex mechanisms including homologous recombination (HR) and non-homologous end-joining (NHEJ) repair pathways, that involve BRCA1, BRCA2, RAD51, etc3,4. BRCA2 gene is a well-known cancer susceptibility gene. Its protein product, comprised of 3418 amino acid residuals, has multiple cellular functions in repairing of DSBs by HR5,6,7. One of mechanisms, by which BRCA2 participate in DSB repair is through regulating the intracellular shuttling and function of RAD51, another critical protein in HR8. Several lines of evidence demonstrated that BRCA2 protein is essential for DSB repair through HR, but not NHEJ. HR repair of DSBs, not NHEJ, are suppressed in BRCA2-null cells and consequently abnormality in chromosome structure (i.e., chromosome breaks) would mount up during cell cycle progression9. It has been know that mutation in BRCA2 is related to not only increased breast cancer risk, but also increased risk of the ovary, prostate, pancreas and male breast, partially due to impaired capacity of repairing DNA DSBs as mentioned above. Accumulating studies have indicated that the polymorphic variants in BRCA2 gene may also confer genetic susceptibility to cancer because of the alteration in DNA repair capacity.
The N372H is the only common non-synonymous polymorphism in the BRCA2 gene, resulting in the single amino acid substitution of histidine (His, H) for asparagine (Asn, N)10. And the consequential amino acid substitution falls into residues 290–453 of BRCA2, a region mediating interaction between BRCA2 and the histone acetyltransferase P/CAF and transcriptional activation of target genes11. Therefore, BRCA2 N372H polymorphism may influence transcriptional activation function of BRCA2 protein.
Over the past decade, many studies were conducted to evaluate the association between BRCA2 N372H polymorphism and the risk of cancer, mainly breast cancer. Several studies reported that homozygous carriers of the H allele of BRCA2 N372H have an increased risk of breast cancer12,13,14, when compared to controls. However, one meta-analyses conducted in 2006 including 12 studies showed no significant association between the polymorphism and breast cancer risk15. Qiu et al.16 performed an updated pooled analysis of 22 studies in 2010 and confirmed the result. However, Qiu et al. also observed a significant association that was restricted to the population-based studies. These conflicting findings may be ascribed to the relatively small sample size in previous studies. Moreover, no meta-analyses have ever been carried out to investigate the relationship between N372H polymorphism and risks of other types of cancer, such as ovarian cancer. To provide a more precise assessment of the association between N372H and cancer risk, we performed a comprehensive meta-analysis by including the most recent and relevant articles.
Results
Study characteristics
As shown in Figure 1, a total of 1278 relevant articles were retrieved from PubMed, EMBASE and CNKI using search terms described in the methods section. After title and abstract screening, 1222 publications, which did not investigate the association between cancer risk and the polymorphism of interest, were excluded; and then, the remaining 56 publications were carefully reviewed according to the criteria described in the ‘materials and methods’ section. Twenty six publications were further removed, among which 10 publications were overlapped with others, 10 were not case-control studies, two used cancer patients as controls, two were prognostic and survival analysis, one was conducted in male patients and last one departed from HWE. After the removal of all studies that didn't meet our criteria, 46 studies from 30 publications including 36299 cases and 48483 controls were finally included in our meta-analysis (Table 1). Among them, there were 30 breast cancer studies12,13,14,15,17,18,19,20,21,22,23,24,25,26,27,28, four ovarian cancer studies29,30,31, six non-Hodgkin lymphoma (NHL) studies32,33,34,35,36,37, two prostate cancer studies extracted from a publication that contained two different populations (Caucasians and African-Americans)38 and three other cancer studies39,40,41 (bladder cancer, esophageal squamous cell carcinoma, melanoma). Additionally, one study42 combined all the tobacco related cancers together without detailed information for each cancer type. Therefore, this study was only included in our overall analysis, while not in the subgroup analysis by cancer type. Furthermore, 23 studies were considered as low quality (quality score < 9) and 23 were considered as high quality (quality score ≥ 9). All of the cases were histological confirmed and most controls were matched by sex, age and ethnicity.
Quantitative synthesis
As indicated in Table 2, pooled analysis yielded a statistically significant association between BRCA2 N372H variant and overall cancer risk. The overall ORs and 95% CIs among all cancer types were as follows: [homozygous model: OR (95% CI) = 1.13 (1.03–1.23) (Figure 2), dominant model: OR (95% CI) = 1.07 (1.01–1.13); and recessive model: OR (95% CI) = 1.12 (1.02–1.23)]. In the stratified analysis by cancer type, significantly increased cancer risk was found with the minor allele among ovarian cancer [heterozygous: OR (95% CI) = 1.14 (1.02–1.27); dominant: OR (95% CI) = 1.13 (1.02–1.26)] and NHL [recessive: OR (95% CI) = 1.23 (1.01–1.43)] and other cancers [homozygous: OR (95% CI) = 1.37 (1.08–1.73); recessive: OR (95% CI) = 1.35 (1.07, 1.70)]. We further conducted the stratified analysis by ethnicity. As a result, a statistically significant association was found among Africans [dominant: OR (95% CI) = 1.36 (1.10–1.69)] and Mixed group [recessive: OR (95% CI) = 1.33 (1.01, 1.75)]. The stratified analysis by source of controls, observed a statistically significant association in the PB subgroup under all the four models [homozygous model: OR (95% CI) = 1.18 (1.06–1.32); heterozygous model: OR (95% CI) = 1.04 (1.01–1.08); dominant model: OR (95% CI) = 1.11 (1.03–1.19); recessive model: OR (95% CI) = 1.14 (1.01, 1.29)] (Figure 3), but not in the HB subgroup. Finally, when studies were stratified by quality score, a statistically significant association was observed in the high score subgroup under three models [homozygous model: OR (95% CI) = 1.17 (1.03–1.32); dominant model: OR (95% CI) = 1.06 (1.02–1.10); recessive model: OR (95% CI) = 1.09 (1.01, 1.17)], while in low score subgroup, statistically significant association was only observed under recessive model [OR (95% CI) = 1.16 (1.06, 1.26)] (Table 2).
Q test and I2 statistic were used to assess the heterogeneity among the studies. There was no heterogeneity under the heterozygous model (P = 0.642, I2 = 0.0%) in the overall analysis. In contrast, significant heterogeneity was observed under the homozygous (P < 0.001, I2 = 49.1%), dominant (P < 0.001, I2 = 61.6%) and recessive model (P < 0.001, I2 = 53.8%). Then, we use meta-regression to determine the source of heterogeneity by cancer type, ethnicity and source of controls. As is shown in Table 3, we found that ethnicity contributed to the heterogeneity in the meta-analysis (homozygous model: P = 0.017; recessive model: P = 0.001), but not cancer type (homozygous model: P = 0.459; heterozygous model: P = 0.437; dominant model: P = 0.250; recessive model: P = 0.577), source of controls (homozygous model: P = 0.962; heterozygous model: P = 0.797; dominant model: P = 0.853; recessive model: P = 0.971) and quality score (homozygous model: P = 0.340; heterozygous model: P = 0.936; dominant model: P = 0.931; recessive model: P = 0.782).
Sensitivity analysis
We performed leave-one-out sensitivity analysis by excluding a study at a time and recalculating ORs and 95% CIs. There was no substantial change found in the pooled ORs and 95% CIs during the overall analysis, suggesting the stability of our analysis.
Publication bias
Begg's funnel plot was used to check the existence of publication bias. There was no evidence of publication bias for the association between polymorphism of BRCA2 N372H and overall cancer risk under the heterozygous, dominant, or recessive model by using Egger's weighted regression test (P = 0.866, P = 0.376, P = 0.341) (Figure 4), whereas a strong degree of publication bias was found under the homozygous model (P = 0.032).
Discussion
In this comprehensive meta-analysis for the association between BRCA2 N372H polymorphism and cancer risk, 46 studies with a total of 36299 cases and 48483 controls were included. This great sample size provided adequate statistical power to detect potential association with cancer risk. Pooled analysis suggested a major role of the polymorphism in shaping over cancer risk. Specifically, BRCA2 N372H polymorphism was shown to confer 13%, 7% and 13% increases in cancer risk under the homozygous, dominant and recessive models, respectively. Moreover, while stratified analyses were performed by cancer type, significant associations were observed for ovarian cancer, non-Hodgkin lymphoma and other cancers (a combination of prostate cancer, bladder cancer and esophageal cancer), but not for breast cancer. Moreover, stratified analyses by ethnic group and the source of control identified the association among Africans and mixed groups, as well as subgroup with PB controls. To the best of our knowledge, this is the first meta-analysis to evaluate the association between BRCA2 N372H polymorphism and overall cancer risk.
The BRCA2 gene is located at chromsome13q13.1 and composed of 27 exons. The BRCA2 protein directly interacts with the RAD51 recombinase to regulate homologous recombination5. The N372H polymorphism in the exon 10 is the only identified common non-synonymous polymorphism in BRCA2 gene, which may confer genetic cancer predisposition. It has been shown that BRCA2 modulates transcriptional activation of other genes by bonding to histone acetyltransferase P/CAF11. BRCA2 N372H, the non-conservative amino acid substitution-causing polymorphism may influence transcriptional activation function of BRCA2 protein. And a review of epidemiological literatures suggests that this polymorphism may be involved in carcinogenesis43. Two meta-analyses have been carried out to attempt to evaluate the association between BRCA2 N372H and risk of breast cancer. A meta-analysis conducted in 2006 including 15627 cases and 15968 controls revealed no significant association under all the models15. With inclusion of more studies, a meta-analysis by Qiu et al. in 2010 also observed no significant association with breast cancer in the overall analysis. Nevertheless, subjects with HH homozygous genotype were reported to have a mild increased risk of breast cancer (OR = 1.11, 95% CI = 1.01–1.21) in PB subgroup16. Our results substantiated the previously findings that there was no evidence of association between BRCA2 N372H and breast cancer risk. However, this polymorphism was shown to significantly increase the risk of ovarian cancer and NHL in stratification analyses by cancer type. Four29,30,31 and six studies32,33,34,36,37,44 on ovarian cancer and non-Hodgkin lymphoma have been conducted before, respectively, but the results were conflicting. The inconsistency may, in part, result from lack of power in individual studies. Moreover, a significant increased cancer risk was also found in PB subgroup, but not in HB or nested case-control subgroup. The null association in HB subgroup is probably due to that controls recruited from hospitals failed to represent the general population from which the cases originated, which may lead to attenuation of risk value.
Moreover, although subgroup analysis by ethnicity found association among Africans, the results should be considered with caution. Since there were only two studies included in the African group, the result may be unstable and false positive, although it was confirmed by meta-regression analysis. Stratification analyses also revealed the between-study heterogeneity observed in the overall analysis might be attributed to the differences among ethnic groups. Many factors may contribute to the strong heterogeneity among overall analysis. The ethnicity-dependent association may be attributed to the discrepancies in genotype distributions among controls of different ethnic groups. Cancer is complicated disease as a result of gene-environment interaction. Therefore different genetic backgrounds among different ethnicities help to explain the ethnicity-dependent results. For example, different populations usually have different linkage disequilibrium types. It is possible that BRCA2 N372H polymorphism may be in close linkage with another nearby causal variant in one ethnic population but not in another. Moreover, clinical features (e.g., years from illness onset, disease severity) or lifestyle habits (e.g., age, sex, diet) may also explain the heterogeneity of ethnicity. More studies are needed to explore the heterogeneity in the future.
Finally, several limitations in this meta-analysis should be taken into consideration. First, individual studies included in some subgroup analysis, like ovarian cancer and among Africans (<5 studies), may be insufficient. And this could diminish statistical power to detect the potential association. Second, because of lacking the original data from individual studies, our analysis was based on the risk estimates (ORs) without adjustment for other confounding factors. As a result, our findings might suffer from potential confounding bias. Therefore, if possible, future studies should take into account other potential confounding factors to improve the precision of risk estimates for this polymorphism and cancer risk. Third, publication bias also may be one of the concerns because studies with positive results were more prone to be published than those with negative results. The funnel plot and Egger's test suggested that potential publication bias may exist under the homozygous model. Trim-and-fill methods were employed to infer the existence of unpublished hidden studies and yield unbiased pooled estimates. The OR did not significantly change after the publication bias was adjusted (1.13 vs. 1.08). Overall, due to these limitations, the results of this meta-analysis should be interpreted with caution.
In conclusion, this systematical meta-analysis regarding the association between BRCA2 N372H polymorphism and cancer risk revealed that this polymorphism was significantly associated with an increased risk of overall cancer and the association was also observed for ovarian cancer. In addition, the significant association was observed in population-based studies while not in hospital-based studies. Finally, well-designed, large-scale studies will be needed to investigate these findings.
Methods
The meta-analysis was conducted according to the latest meta-analysis guidelines (PRISMA), including literature search, data collection, inclusion, etc.
Identification of the eligible studies
A comprehensive literature retrieval was performed using the search terms as “BRCA2 or N372H”, “polymorphism or variation or variant” and “cancer or carcinoma or tumor” through the PubMed and EMBASE up to May 2014 for all relevant studies with no language restriction, according to the latest meta-analysis guidelines (PRISMA)45. We also searched the references of the potential relevant publications and bibliographies of the original articles manually in order to find more eligible studies.
Inclusion criteria
The studies included in our meta-analysis should meet the following criteria: (i) only case-control study or cohort studies were taken into account; (ii) assessed the association between BRCA2 and risk of cancer; (iii) provided sufficient detail of the genotype frequency or data for estimating odd ratios (ORs) and corresponding 95% confidence intervals (CIs); In addition, studies were excluded if significant departures from Hardy–Weinberg equilibrium (HWE) (PHWE < 0.05) in controls were observed, unless there were further evidence from other polymorphism of BRCA2 gene satisfy HWE. When studies had overlapping subjects with others, only the latest or the most complete study was included.
Data collation
The detailed information was extracted from all the eligible publications independently by two authors as the criteria described above. Conflicts were fully discussed by the two authors until consensuses were reached. The following information from the individual study was collected: name of the first author, year of publication, country of origin, ethnicity, numbers of cases and controls for the BRCA2 N372H genotypes or the OR and 95% CI under different genetic models, source of controls, genotype method and the P-value of HWE in controls. The subgroup analysis was performed by cancer type (cancer subgroups contained less than three individual studies were combined and defined as “others cancer type” group), ethnicity (categorized as Asians, Caucasians, Africans or Mixed which contained more than one ethnic group), the source of controls (HB: hospital-based controls and PB: population-based controls) and quality score (low quality: quality score < 9; high quality: quality score ≥ 9)46. We did not define the minimum number of patients for inclusion in our analysis. And publications were divided into different categories or studies when they included different ethnics, cancer types or from different countries.
Statistical method
The crude ORs and the corresponding 95% CIs were used to evaluate the association between polymorphism of BRCA2 and cancer risk. For BRCA2 N372H, the pooled ORs were performed for the homozygous model (CC vs. AA), heterozygous model (CA vs. AA), dominant model (CA + CC vs. AA) and recessive model (CC vs. AA + CA), respectively. Z test was used to determine whether an association was statistically significant. Cochran Q-test and I2 statistic were used to assess and quantify the between-study heterogeneity (A statistically significant heterogeneity was considered when P < 0.10 for Q-test and I2 represents the proportion of variation in a meta-analysis that is explained by heterogeneity across studies rather than by sampling error.). Fixed-effect model was used if there was no heterogeneity observed when P > 0.1 for Q-test; otherwise, a random-effect model was used. Leave-one-out sensitivity analysis was performed by sequentially excluding one single study at a time. Chi-square test was conducted to test if the study is departed from HWE. Additionally, the symmetry of the funnel plot was assessed by Egger's liner regression test to detect the potential publication bias, when at least 4 available dataset were included in subgroup analysis. A meta-regression analysis was performed to investigate the major sources of the heterogeneity across the studies in our meta-analysis.
All statistical tests were performed by STATA version 11.0 (STATA Corporation, College Station, TX), all the P values were two-side test and P < 0.05 was considered statistically significant.
References
Wood, R. D., Mitchell, M., Sgouros, J. & Lindahl, T. Human DNA repair genes. Science 291, 1284–9 (2001).
Christmann, M., Tomicic, M. T., Roos, W. P. & Kaina, B. Mechanisms of human DNA repair: an update. Toxicology 193, 3–34 (2003).
Jackson, S. P. Sensing and repairing DNA double-strand breaks. Carcinogenesis 23, 687–96 (2002).
Pierce, A. J. et al. Double-strand breaks and tumorigenesis. Trends Cell Biol 11, S52–9 (2001).
Lord, C. J. & Ashworth, A. RAD51, BRCA2 and DNA repair: a partial resolution. Nat Struct Mol Biol 14, 461–2 (2007).
Davies, O. R. & Pellegrini, L. Interaction with the BRCA2 C terminus protects RAD51-DNA filaments from disassembly by BRC repeats. Nat Struct Mol Biol 14, 475–83 (2007).
Esashi, F., Galkin, V. E., Yu, X., Egelman, E. H. & West, S. C. Stabilization of RAD51 nucleoprotein filaments by the C-terminal region of BRCA2. Nat Struct Mol Biol 14, 468–74 (2007).
Pellegrini, L. et al. Insights into DNA recombination from the structure of a RAD51-BRCA2 complex. Nature 420, 287–93 (2002).
Venkitaraman, A. R. Cancer susceptibility and the functions of BRCA1 and BRCA2. Cell 108, 171–82 (2002).
Pharoah, P. D. et al. Polygenic susceptibility to breast cancer and implications for prevention. Nat Genet 31, 33–6 (2002).
Fuks, F., Milner, J. & Kouzarides, T. BRCA2 associates with acetyltransferase activity when bound to P/CAF. Oncogene 17, 2531–4 (1998).
Healey, C. S. et al. A common variant in BRCA2 is associated with both breast cancer risk and prenatal viability. Nat Genet 26, 362–4 (2000).
Millikan, R. C., Player, J. S., Decotret, A. R., Tse, C. K. & Keku, T. Polymorphisms in DNA repair genes, medical exposure to ionizing radiation and breast cancer risk. Cancer Epidemiol Biomarkers Prev 14, 2326–34 (2005).
Spurdle, A. B. et al. The BRCA2 372 HH genotype is associated with risk of breast cancer in Australian women under age 60 years. Cancer Epidemiol Biomarkers Prev 11, 413–6 (2002).
Breast Cancer Association Consortium. Commonly studied single-nucleotide polymorphisms and breast cancer: results from the Breast Cancer Association Consortium. J Natl Cancer Inst 98, 1382–96 (2006).
Qiu, L. X. et al. BRCA2 N372H polymorphism and breast cancer susceptibility: a meta-analysis involving 44,903 subjects. Breast Cancer Res Treat 123, 487–90 (2010).
Cox, D. G., Hankinson, S. E. & Hunter, D. J. No association between BRCA2 N372H and breast cancer risk. Cancer Epidemiol Biomarkers Prev 14, 1353–4 (2005).
Dombernowsky, S. L. et al. Missense polymorphisms in BRCA1 and BRCA2 and risk of breast and ovarian cancer. Cancer Epidemiol Biomarkers Prev 18, 2339–42 (2009).
Freedman, M. L. et al. Common variation in BRCA2 and breast cancer risk: a haplotype-based analysis in the Multiethnic Cohort. Hum Mol Genet 13, 2431–41 (2004).
Garcia-Closas, M. et al. Polymorphisms in DNA double-strand break repair genes and risk of breast cancer: two population-based studies in USA and Poland and meta-analyses. Hum Genet 119, 376–88 (2006).
Hu, R. et al. [Association of polymorphisms of N372H in BRCA2 gene and 135G/C in RAD51 gene and breast cancers]. Sichuan Da Xue Xue Bao Yi Xue Ban 39, 973–5 (2008).
Ishitobi, M. et al. Association of BRCA2 polymorphism at codon 784 (Met/Val) with breast cancer risk and prognosis. Clin Cancer Res 9, 1376–80 (2003).
Johnson, N. et al. Counting potentially functional variants in BRCA1, BRCA2 and ATM predicts breast cancer susceptibility. Hum Mol Genet 16, 1051–7 (2007).
Li, X. et al. Association study between signal nucleotide plymorphism of BRCA2 and susceptibility to sporadic breast cancer in Han Chinese women from Jiangxi province. Chin J Cancer Prev Treat 18, 489–493 (2011).
Menzel, H. J. et al. Association of NQO1 polymorphism with spontaneous breast cancer in two independent populations. Br J Cancer 90, 1989–94 (2004).
S, D. E. S. et al. Novel sequence variants and common recurrent polymorphisms of BRCA2 in Sri Lankan breast cancer patients and a family with BRCA1 mutations. Exp Ther Med 2, 1163–1170 (2011).
Seymour, I. J. C. S. & Zampiga, V. Disease family history and modification of breast cancer risk in common BRCA2 variants. Oncol Rep 19, 783–786 (2008).
Sun, X., Li, Z. & Ma, Y. The relationship between BRCA2 N372H polymorphism and risk factor exposure in sporadic breast cancer. Conference papers of tumor etiology research and comprehensive diagnosis (2009).
Auranen, A. et al. BRCA2 Arg372Hispolymorphism and epithelial ovarian cancer risk. Int J Cancer 103, 427–30 (2003).
Beesley, J. et al. Association between single-nucleotide polymorphisms in hormone metabolism and DNA repair genes and epithelial ovarian cancer: results from two Australian studies and an additional validation set. Cancer Epidemiol Biomarkers Prev 16, 2557–65 (2007).
Wenham, R. M. et al. Polymorphisms in BRCA1 and BRCA2 and risk of epithelial ovarian cancer. Clin Cancer Res 9, 4396–403 (2003).
Hill, D. A. et al. Risk of non-Hodgkin lymphoma (NHL) in relation to germline variation in DNA repair and related genes. Blood 108, 3161–7 (2006).
Salagovic, J., Klimcakova, L., Ilencikova, D. & Kafkova, A. Association of follicular lymphoma risk with BRCA2 N372H polymorphism in Slovak population. Med Oncol 29, 1173–8 (2012).
Scott, K. et al. RAG1 and BRCA2 polymorphisms in non-Hodgkin lymphoma. Blood 109, 5522–3 (2007).
Shen, M. et al. Polymorphisms in DNA repair genes and risk of non-Hodgkin's lymphoma in New South Wales, Australia. Haematologica 92, 1180–5 (2007).
Shen, M. et al. Polymorphisms in DNA repair genes and risk of non-Hodgkin lymphoma among women in Connecticut. Hum Genet 119, 659–68 (2006).
Rudd, M. F., Sellick, G. S., Webb, E. L., Catovsky, D. & Houlston, R. S. Variants in the ATM-BRCA2-CHEK2 axis predispose to chronic lymphocytic leukemia. Blood 108, 638–44 (2006).
Agalliu, I. et al. Genetic variation in DNA repair genes and prostate cancer risk: results from a population-based study. Cancer Causes Control 21, 289–300 (2010).
Hu, N. et al. Common genetic variants of TP53 and BRCA2 in esophageal cancer patients and healthy individuals from low and high risk areas of northern China. Cancer Detect Prev 27, 132–8 (2003).
Wu, X. et al. Bladder cancer predisposition: a multigenic approach to DNA-repair and cell-cycle-control genes. Am J Hum Genet 78, 464–79 (2006).
Debniak, T. et al. Common variants of DNA repair genes and malignant melanoma. Eur J Cancer 44, 110–4 (2008).
Kotnis, A. et al. Multiple pathway-based genetic variations associated with tobacco related multiple primary neoplasms. PLoS One 7, e30013 (2012).
Goode, E. L., Ulrich, C. M. & Potter, J. D. Polymorphisms in DNA repair genes and associations with cancer risk. Cancer Epidemiol Biomarkers Prev 11, 1513–30 (2002).
Shen, M. et al. Polymorphisms in DNA repair genes and risk of non-Hodgkin's lymphoma in New South Wales, Australia. Haematologica 92, 1180–1185 (2007).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339, b2700 (2009).
He, J. et al. Association of MTHFR C677T and A1298C polymorphisms with non-Hodgkin lymphoma susceptibility: Evidence from a meta-analysis. Sci Rep 4, 6159 (2014).
Acknowledgements
This study was supported by grants from the National Science Fund for Distinguished Young Scholars (Grant No. 81325018), the key project for International Cooperation and Exchange of the National Natural Science Foundation of China (Grant No. 81220108022) and Special Financial Grant from the China Postdoctoral Science Foundation (Grant No. 2014T70836).
Author information
Authors and Affiliations
Contributions
All authors contributed significantly to this work. W.X., Y.H., J.Z. and J.M. performed the research study and collected the data; W.X., Y.H. and J.Z. analyzed the data; W.J. and J.H. designed the research study; W.X., Y.H., J.Z., J.H. and W.J. wrote the paper and W.X. and Y.H. prepared Figures 1–4, Tables 1–3 and Supplemental Table 1. All authors reviewed the manuscript. In addition, all authors approved the final draft.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Dataset 1
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Xue, WQ., He, YQ., Zhu, JH. et al. Association of BRCA2 N372H polymorphism with cancer susceptibility: A comprehensive review and meta-analysis. Sci Rep 4, 6791 (2014). https://doi.org/10.1038/srep06791
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep06791
This article is cited by
-
Association of IL-6 174G/C (rs1800795) and 572C/G (rs1800796) polymorphisms with risk of osteoporosis: a meta-analysis
BMC Musculoskeletal Disorders (2020)
-
Spectrum of genetic variants of BRCA1 and BRCA2 in a German single center study
Archives of Gynecology and Obstetrics (2017)
-
Association between XRCC3 Thr241Met polymorphism and nasopharyngeal carcinoma risk: evidence from a large-scale case-control study and a meta-analysis
Tumor Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



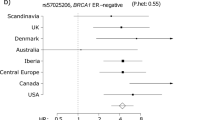


 , pooled ORs and its 95% CIs.
, pooled ORs and its 95% CIs.
 , pooled ORs and its 95% CIs.
, pooled ORs and its 95% CIs.