Abstract
The last case of infection with wild-type poliovirus indigenous to China was reported in 1994. In 2011, a poliomyelitis outbreak caused by imported wide-type poliovirus occurred in Xinjiang Uighur Autonomous Region. Here, we report the results of enterovirus (EV) isolation from Xinjiang students that returned to school in Shandong after summer vacation during this outbreak. Stool specimens from 376 students were collected and 10 EV strains were isolated including 4 polioviruses (All Sabin strains), 1 coxsackievirus (CV) A13, 3 CVA17 and 2 EV-C99. VP1 sequence analysis revealed these CVA13, CVA17 and EV-C99 strains had 71.3–81.8%, 76.5–84.6% and 74.2–82.9% nucleotide similarity with strains from other countries within a serotype, respectively. EV-C99 strains had 82.7–92.8% VP1 similarity with two previously reported Xinjiang strains. Complete genome analysis on EV-C99 strains revealed intra-serotypic genetic recombination events. These findings reflect great genetic divergence between Chinese strains and strains from other countries of the three types and provide valuable information on monitoring EV transmission over long distance.
Similar content being viewed by others
Introduction
Enteroviruses (EV) are small, non-enveloped, positive-sense RNA viruses belonging to genus Enterovirus of the Picornaviridae family and are associated with a spectrum of human diseases such as fever, hand, foot and mouth disease (HFMD), paralysis, aseptic meningitis, encephalitis, myocarditis and neonatal enteroviral sepsis1,2,3.
EVs are amongst the most common viruses infecting humans worldwide. Although most EV infections are asymptomatic, estimates are that as many as 5 to 10 million symptomatic EV infections occur each year in the United States1. EV infection can cause serious illnesses in children <15 years of age and it has become a global public health problem.
Serotypes of human EVs have traditionally been classified into echoviruses (E), coxsackieviruses (CV) group A and B and polioviruses (PV) by neutralization test. In 1999, molecular typing method was introduced which suggests strains with <70% VP1 nt similarity are classified as different types and the strains with >75% VP1 nt similarity are classified as members of same type and led to the discovery of a large number of new EV types4,5,6,7. Currently, human EVs comprise more than 100 types which are classified into 4 species, EV-A to EV-D3,8. EV-B has been demonstrated the predominant species in most parts of the world (including Shandong Province of China), whilst a study conducted in Cameroon uncovered a high prevalence of EV-C2,9,10,11,12,13.
PV infection is known to be associated with acute paralytic poliomyelitis14. No case due to indigenous WPV has been identified in China since 1994. However, a poliomyelitis outbreak caused by imported WPV occurred in Xinjiang Uighur Autonomous Region of China in 2011. Altogether 21 cases were confirmed infection with WPV and they were identified between July 3 and October 915. In September, many Xinjiang students returned to school in Shandong after summer vacation. In support of global polio eradication, we conducted an investigation on EV infection in these students and analyzed the genetic characterization of three scarcely isolated EV-C types.
Results
Overview
The locations of Xinjiang and Shandong Provinces are illustrated in Fig. 1. There is a long geographic distance (~2600 km) between the two provinces. Altogether 3113 Xinjiang students are enrolled in schools in 11 out of 17 cities in Shandong province in 2011. Ages ranged from 7 to 22 years; most (74.8%) were 15–19 years of age. Altogether 397 (13.5%) students had received all 4 doses of OPV immunization whilst 224 (7.6%) did not; OPV immunization status of the rest 2311 (78.8%) students was not available.
Locations of Shandong and Xinjiang Province.
Maps were created using Mapinfo software; data are from the National Fundamental Geographic Information System (NFGIS) website (http://ngcc.sbsm.gov.cn/).
Virus isolation and typing
Stool specimens of 376 students from southern Xinjiang were collected and enteroviruses were isolated from 10 students. Four isolates were identified as PV, including 1 PV1, 2 PV3 and 1 mixture of PV1 + PV3. Sequencing on entire VP1 coding region revealed that they were all Sabin strains with number of substitutions ranging from 1 to 2. No WPV was identified.
Molecular typing on the rest 6 non-polio enteroviruses (NPEV) revealed 1 CVA13, 3 CVA17 and 2 EV-C99. All were recovered in HEp-2 cell line except 1 CVA17 in RD cell. They were isolated from 4 males and 2 females and ages ranged from 15 to 20 years.
VP1 sequence analysis of NPEV
The complete VP1 sequences of the NPEV isolates from Xinjiang Students were aligned with global sequences obtained from GenBank.
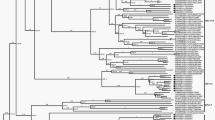
CVA13 prototype strain Flores was isolated in Mexico in 1952, whilst most (73.3%, 33/45) of available VP1 sequences of CVA13 came from African countries including Cameroon, Madagascar and Central African Republic. Others included Bangladesh, USA, Australia and Argentina with relatively low frequency. To the best of our knowledge, this is the first identification of CVA13 in China. Homologous comparison on VP1 region showed that strain QD62/SD/CHN/11 from a Xinjiang student had 71.3–81.8% nucleotide (nt) and 84.4–97.7% amino acid similarity with strains from other countries, reflecting that it had distant genetic relationship with other strains. Phylogenetic analysis revealed that although this strain was segregated into a group mainly comprised of African strains, it did not have close relationship with any CVA13 strain (Fig. 2).
Phylogenetic tree based on complete VP1 sequences of CVA13, CVA17 and EV-C99.
Circles indicate prototype strains, triangles indicate strains obtained in this study and squares indicate strains from Shandong AFP cases. Rranches in green, light blue, deep blue and red indicate strains from Africa, Australia, Asia and America, respectively.
Altogether 10 CVA17 VP1 sequences can be obtained from GenBank. They were isolated from Cameroon (n = 3), Madagascar (n = 3), Bangladesh (n = 2), South Africa (n = 1, prototype strain) and Cambodia (n = 1). Besides, 3 CVA17 strains were isolated during the acute flaccid paralysis (AFP) surveillance conducted in Shandong Province from 1988 to 2013 (2 in 1990 and 1 in 2013) and they were included in the sequence analysis. Strains from Xinjiang students had 93.8–100% VP1 nucleotide similarity with each other, 81.0–95.8% with Shandong AFP isolates and 76.5–84.6% with strains from other countries. Phylogenetic analysis revealed that strains from Xinjiang students had closest genetic relationship with an AFP strain 13061 in 2013 (Fig. 2) and the average p-distance between them is 0.067.
Altogether 9 EV-C99 VP1 sequences from GenBank were available and used for sequence analysis. EV-C99 prototype strain BAN00-10461 was isolated from an AFP case in Bangladesh in 2000. Other strains were from USA, Madagascar, Bangladesh and Oman. Two strains previously isolated from healthy children in Xinjiang Province in 201116 were also included. Strains from Xinjiang students had 92.1% VP1 nucleotide similarity with each other, 82.7–92.8% with previously reported Xinjiang strains and 74.2–82.9% with strains from other countries. Phylogenetic analysis revealed that strains from Xinjiang students were clustered together with one previously reported Xinjiang strain KF129412/KSSC-ALXHH01F/XJ/CHN/2011.
Analysis of the EV-C99 genome
Complete genomes of EV-C99 strains YT23 and YT31 consisted of 7454 and 7456 nucleotides respectively and encoding polypeptides of 2211 amino acids. The difference in genome length resulted from a 2-nt insertion at nucleotide position 101 in 5′ UTR of strain YT31. Strain YT23 and YT31 had 78.5–93.2% complete genomic similarity with other nine EV-C99 strains and 92.9% with each other. In the polypeptide region, strain YT23 and YT31 had 77.8–93.0% nucleotide and 91.4–97.4% amino acid similarity with other nine EV-C99 strains and 92.6% nucleotide and 97.4% amino acid similarity with each other.
Recombination analysis
Similarity plot and bootscanning analysis was performed on EV-C99 strains from Xinjiang students with other nine global EV-C99 strains (Fig. 3). The two strains in this study had high similarity with another previous reported Xinjiang strain (KF129412) throughout the complete genome. Nevertheless, still some intratypic recombination events were revealed. The highest similarity was observed in different regions with different strains. In the P1 coding region, strain YT23 had the highest similarity with strain KSSC-ALXHH01F. In nt positions 4,800–4,200 (partial P2 coding region), it had the highest similarity with strain YT31. In nt positions 5,500–6,000 (the partial P3 coding region), it had the highest similarity with strain HT-XEBGH09F/XJ/CHN/2011. Also, a similarity plot analysis with all EV-C genomic sequences available in GenBank (n = 97) was performed while no more intimate genomic sequence was observed.
Similarity plot (A) and bootscanning analysis (B) of complete genome of strain YT23/SD/CHN/2011with other EV-C99 strains.
Each point represents the similarity between the query sequence and a given heterologous sequence, with a 400 nt window moving in 20 nt steps. Positions containing gaps were excluded from the analysis.
Discussion
The last indigenous WPV in China was isolated in 1994. However, importation of WPV into China has appeared several times, including importation from Myanmar in 1995 and 1996, from India in 1999 and most recently from Pakistan to Xinjiang in 201115,17. In September 2011, when school opened after summer vocation, many Xinjiang students returned to school in Shandong. Since the students from southern Xinjiang had the possibility of being infected by WPV, in order to monitor and prevent the potential transmission by these students, we conduct the virological investigation on this population. The zero isolation of WPV provides valuable information for the control of the poliomyelitis outbreak.
EV-B is the predominant species from EV surveillance in many parts of the world2,10,11,12,13. Similarly, during the AFP surveillance conducted in Shandong Province from 1988 to 2013, EV-B was the most frequently isolated species and EV-C only accounted for 5.1% of isolation. According to the results of EV surveillance in USA from 1970 to 2005, CVA13 and CVA17 only accounted for 0.06% and 0.016% of total reports2. However, a study in Cameroon uncovered a high prevalence of EV-C9, suggesting various composition of circulating EV types in different parts of the world. In this study, all NPEVs from Xinjiang students were EV-C types including CVA13, CVA17 and EV-C99. So, it is most likely that EV-C frequency in Xinjiang is higher than eastern China provinces (such as Shandong). CVA13 and EV-C99 had not been previously identified in Shandong yet. Their isolation from Xinjiang students reveals the existence of long-distance spread and provides the possibility of local circulation in Shandong. Continuous surveillance on EV in the future can provide further information on understanding the circulation of these serotypes.
Since the introduction of molecular typing method, many newer enterovirus serotypes have been identified from AFP cases in Shandong and other provinces of China7,13,16,18,19,20. Besides two EV-C99 strains isolated from students from southern Xinjiang in 2011, two other EV-C99 strains had also been previously reported to be isolated from healthy children in southern Xinjiang during the outbreak in 201116. High genetic divergence (82.7–92.8% VP1 similarity) among these Xinjiang strains was observed, suggesting EV-C99 was not a new virus in southern Xinjiang and has circulated for a long period. Similarity plot and bootscanning analysis on all EV-C99 isolates provided evidence of recombination in the complete genomes, but the percent support values in bootscanning analysis were not high (<95%) and no strains had high than 99% similarity in any of the genomic regions. So, none of these strains can be simply designated as recombination partners. Technically, since the recombination frequency of EV is very high and available genomic sequences from GenBank is very limited and point mutation in EV genome occurs frequently (high substitution rate), it is difficult to identify the exact recombination partner currently. So, via the recombination analysis in this study, we can only get the evidence of occurrence of recombination in the genome of these EV-C99 strains, but cannot conclude where, when, with whom recombination happened. More genomic sequences of currently circulating strains from different spatial and temporal circumstances are needed to understand the evolution and recombination of EV types.
VP1 sequence analysis revealed CVA13, CVA17 and EV-C99 strains from Xinjiang students had great genetic divergence with strains from other countries. Most CVA13 strains available in GenBank came from African countries, CVA17 strains came from African and Asian countries and most EV-C99 strains came from Asia. The absence of European stains is most likely due to the low prevalence of these three serotypes there. All the strains of the three serotypes available in GenBank were isolated from stool specimens from healthy people or AFP cases, so to date little information is available for their pathogenic properties. More surveillance data are needed to understand the evolutionary genetics and epidemiology of these viruses.
Methods
Ethics statement
The ethical approval was given by Ethics Review Committee of the Shandong Center for Disease Control and Prevention and the study was conducted in compliance with the principles of the Declaration of Helsinki. Written informed consents for the use of their clinical samples were obtained from the students or their legal guardians.
Virus isolation
Stool samples from the Xinjiang students were collected and processed according to standard procedures recommended by the WHO21. The ethical approval was given by Ethics Review Committee of the Shandong Center for Disease Control and Prevention and the study was conducted in compliance with the principles of the Declaration of Helsinki. Written informed consents for the use of their samples were obtained from the students or legal guardians. RD, HEp-2 and L20B cell lines were used for EV isolation21. A total of 200 μl of chloroform-treated stool solution was added to each of the cell culture tubes and the inoculated cells were examined daily. After 7 d, the tubes were re-passaged and another 7-d examination was performed. Infected cell cultures were harvested after complete cytopathic effect was obtained. To avoid cross contamination, cell tubes of normal cells served as negative controls. Isolates from RD or HEp-2 cell lines were re-passaged to L20B cell line and were designated as NPEV if no cytopathic effect was observed.
VP1 amplification, sequencing and molecular typing
Total RNA was extracted from 140 μl of the infected cell culture using QIAamp viral RNA mini kit (Qiagen, Valencia, CA, USA) and reverse transcription-PCR (RT-PCR) was performed by using Access RT-PCR System (Promega, USA) according to the manufactures' protocol. The full-length VP1 sequences were determined by RT-PCR and sequencing using the following primer pairs (in the 5′ to 3′ direction): UF1 (GAT GAY TWY ACI GMI GGI GG) and UR1 (CCR TCI TAR AAR TGI SWR TAI GC) for 5′ end amplification and 0408 (ATG TAY RTI CCI MCI GGI GC) and DR1 (GCY WTR TTY TGR TGI CCR AA) for 3′ end amplification. Combination of 5′ and 3′ end sequences yielded entire VP1 coding sequences. PCR positive control was offered in Access RT-PCR System and performed according to the manufacture's protocol. To avoid cross contamination, reagent control and negative control were also included in the RT-PCR reaction. PCR positive products were purified and sequenced with the BigDye Terminator v3.0 Cycle Sequencing kit (Applied Biosystems, Foster City, CA) and sequences were analyzed by ABI 3130 genetic analyzer (Applied Biosystems, Hitachi, Japan). Molecular typing based on VP1 sequences was performed using online Enterovirus Genotyping Tool version 0.122.
Homologous comparison and phylogenetic analysis
Nucleotide sequence alignments were carried out by BioEdit 7.0.5.3 software23. Phylogenetic trees were constructed by Mega 4.0 using neighbor-joining method after estimation of genetic distance using the Kimura two-parameter method24. A bootstrapping test was performed with 1,000 duplicates.
Complete genome sequencing and recombination analysis
EV-C99 is a newly identified EV serotype of species Enterovirus C. Its prototype strain (BAN00-10461) was isolated in Bangladesh in 200025. Current information on EV-C99 or other new EV serotypes in circulation is very limited and many properties of these new types remain unknown. So, whole genome sequencing was performed on two EV-C99 strains in this study. The RT-PCR amplification on the rest of the genome of EV-C99 strains was performed using five pairs of primers designed in this study (Table 1). Positive products were purified and sequenced bi-directionally as described above. The similarity plot and bootscanning analysis on EV-C99 strains was analyzed by using SimPlot 3.5.1 program26, with a 400 nt window moving in 20 nt steps and Jukes-Cantor correction. The available EV-C99 strains (with GenBank accession numbers) used for sequence comparisons were: HEV-99_68229 (JF260926), USA-GA84-10636 (EF555644), USA-Ok85-10627 (EF015012), BAN04-10697 (EF015010), BAN01-10582 (EF015009), BAN00-10461 (EF015008), HT-XEBGH09F/XJ/CHN/2011 (KF129411), KSSC-ALXHH01F/XJ/CHN/2011 (KF129412) and OMA99-10696 (EF015011).
References
Enteroviruses: polioviruses, coxsackieviruses, echoviruses and newer enteroviruses. Fields Virology, 5th ed. (Lippincott Williams & Wilkins, Philadelphia, 2007).
Khetsuriani, N., Lamonte-Fowlkes, A., Oberst, S. & Pallansch, M. A. Centers for Disease Control and Prevention. Enterovirus surveillance--United States, 1970–2005. MMWR Surveill. Summ. 55, 1–20 (2006).
Picornaviridae. Virus Taxonomy: Classification and Nomenclature of Viruses: Ninth Report of the International Committee on Taxonomy of Viruses. (Elsevier, San Diego, 2011).
Oberste, M. S. et al. Molecular identification of 13 new enterovirus types, EV79-88, EV97 and EV100-101, members of the species Human Eneterovirus B. Virus Res. 128, 34–42 (2007).
Oberste, M. S. et al. Molecular identification and characterization of two proposed new enterovirus serotypes, EV74 and EV75. J. Gen. Virol. 85, 3205–3212 (2004).
Oberste, M. S. et al. Enteroviruses 76, 89, 90 and 91 represent a novel group within the species Human enterovirus A. J. Gen. Virol. 86, 445–451 (2005).
Tang, J. et al. Complete genome characterization of a novel enterovirus type EV-B106 isolated in China, 2012. Sci. Rep. 4, 4255 (2014).
Oberste, M. S., Maher, K., Kilptrick, D. R. & Pallansch, M. A. Molecular evolution of the human enteroviruses: correlation of serotype with VP1 sequence and application to picornavirus classification. J. Virol. 73, 1941–1948 (1999).
Sadeuh-Mba, S. A. et al. High frequency and diversity of species C enteroviruses in Cameroon and neighboring countries. J. Clin. Microbiol. 51, 759–770 (2013).
Trallero, G. et al. Enteroviruses in Spain over the decade 1998–2007: Virological and epidemiological studies. J. Clin. Virol. 47, 170–176 (2010).
Rao, C. D., Yergolkar, P. & Shankarappa, K. S. Antigenic diversity of enteroviruses associated with nonpolio acute flaccid paralysis, India, 2007–2009. Emerg. Infect. Dis. 18, 1833–1840 (2012).
Blomqvist, S., Paananen, A., Savolainen-Kopra, C., Hovi, T. & Roivainen, M. Eight years of experience with molecular identification of human enteroviruses. J. Clin. Microbiol. 46, 2410–2413 (2008).
Bingjun, T. et al. Molecular typing and epidemiology of non-polio enteroviruses isolated from Yunnan province, the People' s Republic of China. J. Med. Virol. 80, 670–679 (2008).
Roberts, L. Polio eradication. Looking for a little luck. Science 323, 702–705 (2009).
Luo, H. et al. Identification and Control of a Poliomyelitis Outbreak in Xinjiang, China. N. Engl. J. Med. 369, 1981–1990 (2013).
Sun, Q. et al. Complete genome sequence analysis of two human enterovirus C99 strains isolated in Xinjiang Uighur Autonomous Region, China, in 2011. Arch. Virol. 159, 359–364 (2014).
From the Centers for Disease Control and Prevention: importation of wild poliovirus into Qinghai Province — China, 1999. JAMA 283, 1414–1415 (2000).
Tao, Z. et al. Isolation and genomic characterization of three enterovirus 90 strains in Shandong, China. Arch. Virol. 158, 479–483 (2013).
Tao, Z. et al. Complete genome sequence of enterovirus 87 isolated from a child with acute flaccid paralysis in China in 2000. Virus Genes 47, 156–159 (2013).
Tao, Z. et al. Complete genome sequence of an enterovirus 80 strain isolated in China. J. Virol. 86, 13129–13130 (2012).
Isolation and identification of polioviruses. WHO Polio Laboratory Manual, 4th edn. (World Health Organization, Geneva, 2004).
Kroneman, A. et al. An automated genotyping tool for enteroviruses and noroviruses. J. Clin. Virol. 51, 121–125 (2011).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids Symp. Ser. 41, 95–98 (1999).
Tamura, K., Dudley, J., Nei, M. & Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 24, 1596–1599 (2007).
Brown, B. A. et al. Resolving ambiguities in genetic typing of human enterovirus species C clinical isolates and identification of enterovirus 96, 99 and 102. J. Gen. Virol. 90, 1713–1723 (2009).
Lole, K. S. et al. Full-length human immunodeficiency virus type 1 genomes from subtype C-infected seroconverters in India, with evidence of intersubtype recombination. J. Virol. 73, 152–160 (1999).
Acknowledgements
This study was supported by a grant from the Science and Technology Department of Shandong Province (2012YD18044), a grant from the National Natural Science Foundation of China (81302481) and the Key Technologies Reaseach & Development Program of National Ministry of Science and Technology (2013ZX10004-202).
Author information
Authors and Affiliations
Contributions
Z.T., Q.Y., A.X. and M.W. conceived the study and drafted the paper, X.L., S.W., Y.L., F.J. and L.S. gathered and analyzed the data and P.X. and N.C. helped to interpret results and contributed to the writing. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Accession codes: Nucleotide sequences used in this study were deposited in GenBank under accession numbers KJ857500–KJ857508.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Tao, Z., Yuan, Q., Lin, X. et al. Molecular Characterization of Enteroviruses Including a New Type EV-C99 Isolated from Xinjiang Students in Shandong, China in 2011. Sci Rep 4, 6564 (2014). https://doi.org/10.1038/srep06564
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep06564
This article is cited by
-
Complete Genome Analysis of an Enterovirus EV-B83 Isolated in China
Scientific Reports (2016)
-
Disposable optics for microscopy diagnostics
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.