Abstract
Existing nanoscale chemical delivery systems target diseased cells over long, sustained periods of time, typically through one-time, destructive triggering. Future directions lie in the development of fast and robust techniques capable of reproducing the pulsatile chemical activity of living organisms, thereby allowing us to mimic biofunctionality. Here, we demonstrate that by applying programmed femtosecond laser pulses to robust, nanoscale liposome structures containing dopamine, we achieve sub-second, controlled release of dopamine – a key neurotransmitter of the central nervous system – thereby replicating its release profile in the brain. The fast delivery system provides a powerful new interface with neural circuits and to the larger range of biological functions that operate on this short timescale.
Similar content being viewed by others
Advances in biomaterials and nanotechnology promise the ability to introduce nanoscale devices into living organisms to address, mimic and ultimately control their intrinsic mechanisms1. A first application of this concept has been the development of targeted, site specific drug delivery systems activated by external stimuli2. For example, in nano cancer treatments3, the dosage is delivered slowly and continuously over long periods of time at a specific location in the body4. Equally important to spatial control is gaining temporal, pulsatile control over the drug delivery system5. Numerous vital functions of living biological systems occur in a regulated, repeatable manner with natural rhythms of hours6 to milliseconds7. Mimicking these rhythms – that are essential to life chemistry – demands pulsatile, repeatedly-releasing chemical delivery systems with the appropriate temporal profile. Previous attempts have achieved temporal control of the pulse profile on the order of hours and days4 and typically employ a one-time destructive release mechanism by irreversible breakdown of the containing structure8,9. An important next step of development is towards sub-second control over the temporal drug-delivery profile via a non-destructive release mechanism in order to mimic faster rhythmic biological life cycles10,11.
A particularly important subsecond biological process is the pulsed release of neurotransmitters and neuromodulators in the brain12,13. Chemical synaptic transmission rapidly transmits information between neurons to perform brain functions such as perception and motor control and learning and memory14. Neuromodulators also operate on a subsecond timescales to regulate the activity of large swaths of neural tissue12. Deficiencies in neurochemical signaling in the brain result in neurological disorders, such as Parkinson's disease15. Although replacement therapies have been employed in such disorders, the slow absorption and diffusion of drugs has limited their application to replacement of constant background levels of the neuromodulator16. Better results can be expected by artificially mimicking the neurochemical signal with the appropriate temporal structure. Thus, the ability to reproduce the subsecond release of neurotransmitters and neuromodulators in the brain would be a significant step in controlling brain mechanisms, understanding brain behavior and potentially addressing neurological diseases.
Here, we demonstrate subsecond, pulsatile, on-demand release of dopamine using a nanoscale drug delivery system, thus capable of reproducing neurotransmitter release of the brain. Using a train of femtosecond laser pulses, we stimulate robust, dopamine-filled liposome structures to repeatedly deliver pulsed dopamine concentrations where the delivery time and the concentration are controlled simply by adjusting the intensity and exposure time to the femtosecond laser pulse train.
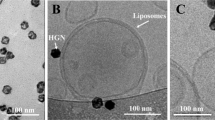
To develop the nanoscale, biocompatible drug delivery system, we prepared liposome structures filled with dopamine, tethered to hollow gold nanoshells (HGN)9 (Figure 1a). The liposomes were prepared by mixing 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC), cholesterol, sphingomyelin, 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] (DSPC-PEG2000) and DSPE-PEG2000-SH at a molar ratio of 100:5:5:4:3.5. On removal of the solvent (chloroform), a phosphate buffered saline (PBS) containing dopamine was added. The mixture is swirled in a water bath at 50°C till all lipid materials are suspended and the liposomes are then extruded through a 200 nm polycarbonate membrane allowing us to regulate their size. The HGN correspond to a size of ~20 nm with absorption in the visible and near-infrared (Figure 1c). HGN suspension was then added periodically to the liposomes with a gold-lipid ratio (mg/mmol) of 100:1. This preparation method resulted in stable liposome structures filled with dopamine and tethered to gold nanoparticles. Based on the fabrication parameters, we estimate that on an average one HGN is attached to each liposome structure. Finally, a carbon fiber of 10 um diameter was dipped in the PBS solution containing the liposome structures for five minutes, thereby allowing a reasonable number of the structures to attach to the fiber. The attachment and consequent immobilization of the liposome structures allowed for their repeatable measurement. We note that the carbon fiber is used for immobilization of liposomes, but is not required for the release mechanism. We confirm this via all-optical experiments, where release of CF dye from the liposome structures into a solution is observed in the absence of a carbon fiber.
Liposome delivery and measurement system.
(A) Dopamine was encapsulated within the liposome's bimolecular lipid membrane. Hollow gold nanoshells (HGN) were tethered to the membrane. Femtosecond laser pulse train induces dopamine release from the liposome structures. (B) The released dopamine was measured using fast scan cyclic voltammetry. Triangular voltage pulses were applied to the carbon fiber electrode at 10 Hz. The current response to the voltage pulse showed an oxidation and reduction peak at the respective potentials of dopamine. (C) The UV/visible/NIR spectra of HGN.
To achieve the release of dopamine from the liposome structures, the carbon fiber with liposomes was submerged in water and illuminated by a train of near-infrared (800 nm) femtosecond pulses. Electron microscope images of the carbon fiber confirmed that the liposomes did not come loose when dipped in tap water and remain largely intact after repeated laser exposures as discussed below. The femtosecond pulse train had a temporal width of 70 fs per pulse, an adjustable intensity of upto 5 W/cm2, in a collimated beam of diameter 0.8 cm and a temporal spacing of 1 ms between pulses (Figure 1a). Thus, for a carbon fiber of diameter 10 μm and length of 250 μm, the laser beam illuminates the fiber with only 200 μJ of total energy in a one-second exposure, corresponding to an energy of 200 nJ/pulse. An electronically operated mechanical shutter controlled the illumination time of the liposome structures, with typical times of a few hundred milliseconds to a second. The potential mechanisms causing the release of chemicals enclosed within gold tethered liposome structures on exposure to light have been discussed previously9,17. Thus, by repeated illumination of the same liposome structures with varied laser exposure times and time between illuminations, we stimulated dopamine delivery with arbitrary temporal profiles.
To measure the release dynamics of dopamine from the light-stimulated liposome structures we used fast scan cyclic voltammetry (FSCV)18. In this technique, triangular voltage waveforms (−0.4 V to 1.3 V and back to −0.4 V at 300 V/s) are applied to a conducting electrode at a 10 Hz frequency. The electrodes are held at −0.4 V between the triangular sweeps (Figure 1b). We measure the dopamine concentration in solution by recording the current flow during oxidation (reduction) of the dopamine molecule at +0.6 V (−0.2 V)18. In order to sensitively measure the release of dopamine from our liposome structures, we use the same carbon fiber to which the liposome structures were attached as the conducting electrode. This improvisation to the FSCV technique thus allows us to measure the dopamine release directly at source with high sensitivity. The increased sensitivity, combined with the immobilization of the liposome structures on the carbon fiber, allows us to measure the repeated, on-demand, pulsatile release dynamics of the liposome structures.
Figure 2a shows the temporal profile of the dopamine concentration released into the solution due to a single one second, 3 W/cm2 illumination with the femtosecond pulse train. We clearly see a rapid and linear rise in the dopamine concentration released while the carbon fiber with the fixed liposome structures is being illuminated. As soon as the laser illumination is shuttered, the dopamine concentration decays exponentially due to diffusion into the solution away from the carbon fiber source. This slow diffusion profile into the solution is expected to vary depending on the solution, dopamine uptake by neighbouring cells and other real-world processes19, which are not the focus of this paper. Here we focus on the sub-second controlled rise in the dopamine concentration (i.e. time profile of dopamine delivery) during laser illumination. The inset in Figure 2a demonstrates pulsatile dopamine delivery achieved via repeated, one-second laser exposures, spaced 40 seconds apart. In each of the dopamine release profiles, we observe the linear and rapid rise in dopamine concentration during the laser exposure, followed by the diffusion of dopamine away from the source.
Pulsatile, repeatable dopamine release.
(A) Rapid increase in dopamine concentration stimulated by a one-second laser exposure followed by a decrease due to diffusion process. (Inset) Pulsatile release by repeated one-second laser exposures over 100 s of seconds. (B) We observe an initial rapid and then slow decrease in dopamine released per exposure after multiple laser exposures. This dynamic can be fitted by bi-exponential curve and is explained by assuming two populations of liposomes with different delivery mechanisms.
By adjusting the laser intensity, the exposure time and the time between exposures, we can program an arbitrary pulsatile dopamine release. Figure 3 demonstrates such an arbitrary pulsatile release profile where the liposome structures were repeatedly illuminated with the femtosecond pulse train using intensities between 2 W/cm2 and 3 W/cm2, exposure times between 500 ms and 1second and the time between exposures ranging from 5 s to 20 s. The insets in Figure 3 show that the released dopamine increases linearly during the period of illumination, with the rate of release determined by the input laser intensity. Hence it is possible to independently control the temporal profile of the released dopamine (via the exposure time to the laser) and the quantity of the released dopamine (via the intensity of the laser pulse). We note that we are not mimicking the native release processes themselves20,21, but instead we have demonstrated an artificial system that is capable of reproducing the temporal profile of the chemical concentration produced by native release processes.
On-demand, repeatable and sub-second drug delivery.
We demonstrate repeated dopamine pulses with arbitrary concentration and temporal profiles controlled via the laser intensity and exposure time respectively. The insets show the linear rise in dopamine concentration during laser exposure, with faster dopamine release rates for higher laser intensities and more prolonged release with longer pulses.
In order to further understand the long term behavior and stability of this dopamine delivery system, we measured the peak dopamine concentration released over repeated one-second, 3 W/cm2 exposures. The exposures were set 40 seconds apart, thereby allowing the dopamine from the previous release to diffuse away. This time between exposures did not alter the results of the experiments. In Figure 2b, we plot the peak dopamine concentration released versus the exposure number. The data can be directly fit to a bi-exponential decay, indicating two different processes contributing to the dopamine delivery mechanism – a fast process that lasts only for the first few exposures and a longer process that lasts for 100 s of exposures.
We mathematically model this behavior by assuming that there are two populations of liposome structures: (i) a ‘fragile’ population with a high probability – αf, of destruction of any one of these liposome structures in a single laser exposure, thereby causing all dopamine within that liposome structure to be released at once9; and (ii) a robust population of liposomes which are not destroyed on laser exposure, but laser exposure increases the permeability of their lipid membranes resulting in the fractional release of dopamine molecules into the solution. Such increases in permeability may be due to thermal17,22 or mechanical9,23 effects from external stimulations as have been proposed previously. With these assumptions, the number of ‘fragile’ liposomes (Nfk) surviving after the kth exposure is given by the exponential decay  , where Nf0 is the initial population of the ‘fragile’ liposomes. The dopamine released in the kth exposure from these liposomes is simply the number destroyed times the dopamine contained in their internal volume -
, where Nf0 is the initial population of the ‘fragile’ liposomes. The dopamine released in the kth exposure from these liposomes is simply the number destroyed times the dopamine contained in their internal volume -  , where C0 and V0 are the concentrations and volumes of each of the as-made liposome structures. For the robust population of liposomes, the number of liposomes doesn't change over time. However, the internal concentration of dopamine continues to deplete due to its partial diffusion into the solution on laser exposure. Assuming that this partial diffusion is merely proportional to the difference in internal and external concentrations (with a proportionality constant of αr) and that the external concentration always goes to zero by the time of the next laser exposure, the dopamine released during the kth exposure is simply
, where C0 and V0 are the concentrations and volumes of each of the as-made liposome structures. For the robust population of liposomes, the number of liposomes doesn't change over time. However, the internal concentration of dopamine continues to deplete due to its partial diffusion into the solution on laser exposure. Assuming that this partial diffusion is merely proportional to the difference in internal and external concentrations (with a proportionality constant of αr) and that the external concentration always goes to zero by the time of the next laser exposure, the dopamine released during the kth exposure is simply  , where Crk is the internal dopamine concentration in the robust liposomes during the kth exposure and Nr0 is the number of robust liposomes. Thus the internal concentration of dopamine follows an exponential decay given by
, where Crk is the internal dopamine concentration in the robust liposomes during the kth exposure and Nr0 is the number of robust liposomes. Thus the internal concentration of dopamine follows an exponential decay given by  where C0 is the as-made initial dopamine concentration. The total dopamine released, given by the sum of the two contributions, thus exhibits a bi-exponential decay with constants αf and αr and magnitudes Nf and Nr respectively.
where C0 is the as-made initial dopamine concentration. The total dopamine released, given by the sum of the two contributions, thus exhibits a bi-exponential decay with constants αf and αr and magnitudes Nf and Nr respectively.
To extract the above parameters from our experiment, we fit the data in Figure 3b with the expected biexponential decay. This gives us values for αf and αr as 0.29 ± 0.08 (standard error; n = 7) and 0.06 ± 0.03 respectively and the ratio of Nr to Nf is 4:1 within experimental error. The large ratio of Nr:Nf indicates that only a small percentage of the liposomes are ‘fragile’. Also, the large value of αf indicates that this ‘fragile’ population contributes to the release only in the few initial exposures before they are essentially all destroyed. On the other hand, the ‘robust’ liposomes constitute a large fraction of the population and the small αr value demonstrates the possibility of repeated release of dopamine over long periods of time. These robust populations also open the door to future dopamine ‘nano-factories’ within the liposomes24, which maintain the internal dopamine concentrations and thus eliminate the slow decay component.
Figure 4 shows an electron microscope image of the liposome structures before and after laser exposure and provides an independent confirmation of this model. As previously discussed, Figure 4c shows numerous liposome structures sticking to the carbon fiber before laser exposure and Figure 4d images the same region of the carbon fiber after repeated laser exposure. Comparison of these two images indicates that only a small population of the liposome structures have been destroyed (about 20% - consistent with the extracted parameters from our mathematical model), while a large fraction remains after the laser irradiation.
The electron microscopy images of liposomes attached to carbon fiber.
(A) Carbon fiber to which liposome structures were fixed for repeated measurement. Rectangle denotes the zoomed in region in (B) before liposome were attached, (C) after liposomes were attached and (D) after laser exposure. One observes a large number of speckles in (C) indicating the attached liposomes and a slightly reduced number in (D) due to losses after laser exposure. A few nominal circles and squares are guides for the eye, with circles as examples of regions where liposomes attach and remain attached after laser exposure (robust population). Squares are examples of regions where liposomes attach but are destroyed after laser exposure (fragile population).
In conclusion, we have demonstrated an on-demand, subsecond, pulsatile, dopamine delivery system using femtosecond lasers as a non-destructive external stimulus. By varying the laser intensity and exposure time, we can arbitrarily and precisely control the concentration and temporal profile of the dopamine delivery. Given the fast timescales on which neural signaling operates, this rapid temporal control provides the ability to mimic the temporal profile of chemicals released during important neurochemical processes. Future directions lie in investigating the applicability of this fast delivery system to a wide range of neurochemicals that can be encapsulated in liposomes25. The technique promises future potential for the delivery of natural and synthetic therapeutic compounds involved in rapid biological signaling; stimulating multiple brain locations simultaneously by combining with recently developed femtosecond techniques to control the size and shape of the stimulated volume26 engineering the response of the delivery system to different laser wavelengths to allow for multi-channel operation; and potentially replacing lost functionality due to neural degeneration via ‘neuro-chemical prosthesis’.
References
Peer, D. et al. Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol 2, 751–760 (2007).
Ganta, S., Devalapally, H., Shahiwala, A. & Amiji, M. A review of stimuli-responsive nanocarriers for drug and gene delivery. J. Control. Release 126, 187–204 (2008).
Arap, W., Pasqualini, R. & Ruoslahti, E. Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science 279, 377–380 (1998).
LaVan, D. A., McGuire, T. & Langer, R. Small-scale systems for in vivo drug delivery. Nat Biotechnol 21, 1184–1191 (2003).
Kikuchi, A. & Okano, T. Pulsatile drug release control using hydrogels. Adv. Drug Deliv. Rev. 54, 53–77 (2002).
Welsh, D. K., Logothetis, D. E., Meister, M. & Reppert, S. M. Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14, 697–706 (1995).
Buzsáki, G. & Draguhn, A. Neuronal oscillations in cortical networks. Science 304, 1926–1929 (2004).
Ellis-Davies, G. C. R. Caged compounds: photorelease technology for control of cellular chemistry and physiology. Nat Meth 4, 619–628 (2007).
Wu, G. et al. Remotely triggered liposome release by near-infrared light absorption via hollow gold nanoshells. J. Am. Chem. Soc. 130, 8175–8177 (2008).
Purvis, J. E. & Lahav, G. Encoding and decoding cellular information through signaling dynamics. Cell 152, 945–956 (2013).
Alberts, B. et al. Molecular Biology of the Cell. Garland Science, New York (2002).
Roitman, M. F., Stuber, G. D., Phillips, P. E. M., Wightman, R. M. & Carelli, R. M. Dopamine operates as a subsecond modulator of food seeking. J Neurosci 24, 1265–1271 (2004).
Wickens, J. R., Budd, C. S., Hyland, B. I. & Arbuthnott, G. W. Striatal contributions to reward and decision making: making sense of regional variations in a reiterated processing matrix. Ann N Y Acad Sci 1104, 192–212 (2007).
Katz, B. Nerve, muscle and synapse. Mcgraw-Hill Book Co, New York (1966).
Parkinson, J. An essay on the shaking palsy. Whittingham and Rowland for Sherwood, Neely and Jones, London (1817).
Arbuthnott, G. W. & Wickens, J. R. Space, time and dopamine. Trends Neurosci 30, 62–69 (2007).
Paasonen, L. et al. Gold nanoparticles enable selective light-induced contents release from liposomes. J. Control. Release 122, 86–93 (2007).
Robinson, D. L., Venton, B. J., Heien, M. L. A. V. & Wightman, R. M. Detecting subsecond dopamine release with fast-scan cyclic voltammetry in vivo. Clin Chem 49, 1763–1773 (2003).
Cragg, S. J. & Rice, M. E. DAncing past the DAT at a DA synapse. Trends Neurosci 27, 270–277 (2004).
Amatore, C., Oleinick, A. I. & Svir, I. Reconstruction of aperture functions during full fusion in vesicular exocytosis of neurotransmitters. Chemphyschem 11, 159–174 (2010).
Mellander, L. J. et al. Two modes of exocytosis in an artificial cell. Sci Rep 4, 3847 (2014).
Djanashvili, K. et al. Development of a liposomal delivery system for temperature-triggered release of a tumor targeting agent, Ln(III)-DOTA-phenylboronate. Bioorg Med Chem 19, 1123–1130 (2011).
Oerlemans, C., Deckers, R., Storm, G., Hennink, W. E. & Nijsen, J. F. W. Evidence for a new mechanism behind HIFU-triggered release from liposomes. J. Control. Release 168, 327–333 (2013).
Schroeder, A. et al. Remotely activated protein-producing nanoparticles. Nano Lett. 12, 2685–2689 (2012).
Sharma, A. & Sharma, U. S. Liposomes in drug delivery: progress and limitations. Int. J. Pharm. 154, 123–140 (1997).
Papagiakoumou, E. et al. Scanless two-photon excitation of channelrhodopsin-2. Nat Meth 7, 848–854 (2010).
Author information
Authors and Affiliations
Contributions
T.N., J.W. and K.D. wrote the main manuscript text and T.N. prepared figures 1–4. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Nakano, T., Chin, C., Myint, D. et al. Mimicking subsecond neurotransmitter dynamics with femtosecond laser stimulated nanosystems. Sci Rep 4, 5398 (2014). https://doi.org/10.1038/srep05398
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep05398
This article is cited by
-
Dynamic control of neurochemical release with ultrasonically-sensitive nanoshell-tethered liposomes
Communications Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.