Abstract
Preeclampsia (PE) is an excessive systemic inflammation response with dysfunction of endothelial. Our study was to investigate the association between genetic variations in IL-1 and the susceptibility to PE in Chinese Han population. 402 PE patients and 554 normal pregnant women of third trimester were enrolled. The polymorphisms of rs315952 in IL1RN and rs17561 in IL1A were genotyped by TaqMan allelic discrimination real-time PCR. Obviously statistic difference of the genotypic frequencies were found in both of IL1RN rs315952 and IL1A rs17561 between cases and controls (for rs315952, P = 0.001; for rs17561, P = 0.021.). For rs315952, the C allele was associated with development of PE (P = 0.003, OR = 1.319, 95%CI 1.099–1.583). Patients with CC or CT genotype were less likely to develop severe PE than patients carrying TT genotype(P< 0.001, OR = 0.24, 95%CI 0.15–0.40). For rs17561, the C allele was the risk factor for predisposition to PE (P = 0.012, OR = 1.496, 95%CI 1.089–2.055). Our results suggest IL1RN and IL1A may involve in the development of PE in Chinese Han population.
Similar content being viewed by others
Introduction
As a pregnancy-specific disorder, preeclampsia (PE) is diagnosed by the onset of hypertension and proteinuria after 20 gestational weeks. Because PE is often accompanied with multi-organ disorders and there is no effective treatment other than terminating pregnancy, it becomes a leading cause of perinatal morbidity and mortality1. Furthermore, hypertension is the most common risk factor for the initial myocardial infarction, which may affect the hospital mortality2. Therefore, the onset of PE must get our attention. Although unremitting efforts have been taken for many years, the etiology and pathogenesis of PE remain unclear. Several pathological factors are proposed to explain the development of PE, such as immune maladaptation, exaggerated inflammation, abnormal trophoblast invasion and genetic involvement. The susceptibility to PE is affected by both genetic and environment interaction. To date, a variety of candidate genes have been tested to have associations with PE, including ERAP2, COMT, TNF-α and so on3,4,5,6,7. Moreover, some candidate genes, such as CYP11B2 and ACE, were demonstrated to be linked with hypertension8,9.
PE is an excessive systemic inflammation response with dysfunction of endothelial10. The serum levels of several cytokines, such as IL-1, TNF-α, IL-6, IL-8, are increased in PE patients11,12,13. IL-1 is a critical mediator of the inflammatory response and important factor that stimulates the structural and functional alterations in endothelial cells10. It consists of three molecules namely IL-1α,IL-1β and IL-1 receptor antagonist (IL-1Ra), which is encoded by IL-1A, IL-1B and IL-1RN, respectively. IL-1α and IL-1β are pro-inflammatory cytokines, which bind to the IL-1 receptor (IL-R) to activate signal transduction and exert biological effects while IL1Ra is the anti-inflammatory cytokine by competing for binding sites on the IL-1R11. The polymorphisms of promoter and coding regions in cytokine genes may affect the production and function of cytokines14. The rs315952, located in exon 7 of IL-1RN, has been suggested to be associated with several autoimmune disorders, such as ankylosing spondylitis, systemic lupus erythematosus, knee osteoarthritis15,16,17. The plasma levels of IL1RA were found to be higher with the allele C of rs315952, especially for the homozygous carriers of the C allele in acute respiratory distress syndrome18. The IL1A rs17561 located in the exon 5, is a nonsynonymous variant (Ala114Ser)19, is reported to have associations with inflammatory disorders and reflect increased IL-1α production in carriers of this allele T and the allele T of rs180058720. The SNP of rs17561 is associated with the risk of endometriosis, ovarian cancer, ankylosing spondylitis and so on21,22,23. However, the mechanism of the functional role of these polymorphisms has not been identified.
Considering the exaggerated inflammation response and the changes in serum levels of inflammatory cytokines of PE patients, we hypothesized polymorphisms of IL-1 may influent the susceptibility and clinical symptoms of PE. The aim of this study is to investigate the association between polymorphisms of IL1RN rs315952 and IL1A rs17561 and susceptibility to PE in Chinese Han population.
Results
Demographic and clinical characteristics
The clinical characteristics of people enrolled and p-value for comparison between cases and controls were summarized in table 1. The mean age of cases and controls was 30.74 ± 5.70 and 30.67 ± 4.48 years old, respectively. As compared with controls, PE patients had earlier gestational weeks at delivery (36.31 ± 2.93weeks vs. 39.23 ± 1.35 weeks, P < 0.001), lower birth weight of offspring (2684 ± 909 g vs. 3382 ± 394 g, P < 0.001) and higher blood pressure (P < 0.001). In addition, there was higher serum levels of the count of white blood cell and neutrophil, triglycerides, ALT, AST, urea nitrogen, creatinine in PE patients (P < 0.05). There is no statically significant difference in weight increased during pregnancy, number of abortion, age of menarche, serum level of total cholesterol between the two groups (P > 0.05).
Genetic analysis
The population of controls was in Hardy–Weinberg equilibrium for both SNPs (for rs315952, χ2 = 0.024, P = 0.877; for rs17561, χ2 = 0.0005, P = 0.983). Table 2 showed the genotypic and allelic frequencies of rs315952 and rs17561 in cases and controls. For rs315952, there was significant difference of genotypic and allelic frequencies between two groups (χ2 = 13.741, P = 0.001 by genotype; χ2 = 8.869, P = 0.003 by allele). When subdividing these samples into CC + CT/TT groups or TT + CT/CC groups, we found a significant difference between cases and controls in TT + CT and CC groups (P < 0.001, OR = 1.775, 95%CI 1.308–2.411). The C allele of rs315952 was associated with development of PE (OR = 1.319, 95%CI 1.099–1.583). Similarly, for rs17561, the distribution of genotypes differed significantly between cases and groups (χ2 = 7.725, P = 0.021) and there is a statistical difference in AA + AC and CC groups (P = 0.007, OR = 1.605, 95%CI 1.137–2.266). The C allele was the risk allele for predisposition to PE (P = 0.012, OR = 1.496, 95%CI 1.089–2.055).
Analysis of genotype-phenotype relationship
The results were present in table 3, 4. For rs17561, patients who had genotype CC showed higher urea nitrogen than those who had genotype AA and AC (4.81 ± 2.09 mmol/L vs. 4.10 ± 1.36 mmol/L, P = 0.002). Similarly, the level of serum creatinine was higher in patients carrying CC genotype than patients carrying AA and AC genotype (69.64 ± 19.89 umol/L vs. 63.45 ± 20.51 umol/L, P = 0.041). We did not found associations between rs315952 and demographic characteristics and results of serum biochemistry. However, the distribution of the severe preeclampsia in this three genotypes of rs315952 was significantly different (P < 0.001). Patients with CC or CT genotype were less likely to develop severe PE than patients carrying TT genotype (P < 0.001, OR = 0.24, 95%CI 0.15–0.40).
Discussion
The basic pathological change of PE is the spasm of systemic small vessel24, thus resulting a reduction of blood flow in various organs. Other than the most common symptoms hypertension and proteinuria, PE is often companied with additional disturbance of multi-organs, such as activation of the clotting system, impaired liver and renal function, pulmonary edema in cases of severe PE particularly. Due to unclear pathogenesis, there are no effectively preventive methods and the treatments of PE is limited to deal with clinical symptoms and terminate pregnancy1.In our study, PE patients had earlier gestational weeks at delivery and lower birth weight of offspring, which demonstrated the fact that PE patients usually have to choose induced preterm delivery to relieve the clinical symptoms. What's more, the PE patients enrolled in our study had higher serum level of ALT, AST, urea nitrogen and creatinine compared with controls. Moreover, the count of white blood cells and neutrophil were higher in cases than controls, which revealed a status of exaggerated inflammation in PE.
The normal pregnancy is suggested to be a condition of controlled mild maternal systematic inflammation. And exaggerated inflammation is proposed to play an important role in the development of PE10,11,12,13. There is Th1/Th2 imbalance in PE patients and Th1 immunity is predominant in the immune and inflammatory response25. Among the Th1-type pro-inflammatory cytokines, IL-1 initiates and perpetuates inflammatory response. The levels of IL-1 synthesized both from decidual lymphocytes and peripheral blood mononuclear cells are higher in PE patients10,11. IL-1 can stimulate expression and activity of matrix metalloproteinase (MMP) 9 and MMP2, thus regulate trophoblast differentiation along the invasive pathway, which may affect the process of placentation26. Moreover, IL-1 can alter the structure and function of endothelial cells. In vitro, IL-1 produced by placenta altered the proliferation of umbilical vein endothelial cell and induced the secretion of soluble ICAM and IL-627, whose serum levels were increased in PE patients. Therefore, IL-1 is a potential mediator of endothelial dysfunction and may involve in the development of PE.
Previously, genetic researches, including the polymorphic and functional studies, have been carried out in the investigations of mechanisms of PE or hypertension28,29,30. Santulli28 et al. found that CaMK4 deletion induced hypertension through the influence on the synthase activity of endothelial nitric oxide and further confirmed the association between rs10491334 variant and a reduction in the expression levels of CaMKIV in hypertensive patients. Among G-protein-coupled receptor kinase (GRK) family, the GRK5 Leu41 allele was reported to decrease the risk for adverse cardiovascular outcomes in treated hypertensive patients29. What's more, GRK2 abundancy was related with hypertension through impairment of β-adrenergic mediated vasodilation, which was also present in PE30. Furthermore, the associations between polymorphisms of IL-1 and the risk of PE have also been investigated. However, because of mutations, genetic recombination, human mobility and natural selection, the frequencies of genotypes and alleles are different in the population from different race or region, therefore the results of these studies are controversial6,31,32,33,34. Goddard et al. analyzed 775 SNPs in 190 candidate genes, founding that rs3783550 in IL1A had a statistically significant association with PE in Chile (P = 0.0014)31. Faisel et al. studied on 133 PE patients and 112 controls from Finland, suggesting an association between the variable copy number of 86-bp tandem repeats (VNTR) of the IL1RN polymorphism and PE32. However, Valencia et al. did not find the associations between polymorphism of IL1RN VNTR and PE in Mexican-Mestizo and Maya-Mestizo women33. And polymorphism of rs16944 and rs1143634 in IL-1β showed no associations with the risk of PE in Taiwanese34.
In our study, we selected IL-1RN rs315952 and IL1A rs17561 to investigate the associations with PE. Both rs315952 and rs17561 polymorphisms had a significant association with PE in Chinese Han population. For rs315952, the C allele was the risk allele for the development of PE. Patients with CC or CT genotype were more inclined to develop severe PE than patients with TT genotype. For rs17561, the frequency of the C allele was higher in PE patients. The levels of serum urea nitrogen and creatinine in patients carrying CC genotype were higher than those carrying AA or AC genotype. Our results suggested that IL1RN and IL1A may involve in the development of PE, which is consistent with the studies of Goddard and Faisel31,32. To our knowledge, it is the first time to examine whether the SNPs of rs315952 and rs17561 in IL-1 are associated with the risk of PE.
There are several limitations in this study that should to be noted. Firstly, the sample size was relatively small and all participants were ethnic Han Chinese. Because the results are affected by ethnic or region, our study could not represent other human races. Secondly, other variants of IL-1A and IL-1RN were not genotyped for the associations with PE, thus genetic linkage analysis could not be conducted. Further studies on the associations of genetic variants of IL-1 with PE will be necessary. Thirdly, the serum level of IL-1 were not measured in our population, thus the relationships between the SNPs and IL-1 levels were not observed. In spite of some limitations, our study suggested that IL1RN and IL1A may involve in the development of PE in Chinese Han population. Our results need to be validated in a larger sample and in other races with functional analyses to clarify the potential mechanisms underlying the links between SNPs of IL-1 and susceptibility to PE.
Methods
Subjects
402 PE patients and 554 normal pregnant women of third trimester admitted to the Affiliated Hospital of Qingdao University, Linyi People's Hospital and Heze Municipal Hospital were enrolled in our study. Age was matched in the two groups. The mean age of PE patients was 30.74 ± 5.70 years old and the controls were 30.67 ± 4.48 years old. The controls requested pregnant women without multiple pregnancy or any pathological states, such as premature rupture of membrane, placenta previa, poly- or oligo-hydramnios, threatened abortion, diabetes mellitus, hypertension, autoimmune disease and so on. If the fetus was congenital malformations or macrosomia, the maternal sample was removed from the control group. The demographic and clinical characteristics of participators, such as pregnancy and family history, clinical symptoms, the results of blood routine test, blood clotting state, liver and renal function, complication of fetus, were collected to build a clinical database. The study was approved by Ethics Committee of hospital and all the participants signed informed consent.
PE was defined as onset of hypertension during the third trimester (≥140/90 mmHg on two occasions) and detectable urinary protein (≥1+ by dipstick or ≥300 mg/24 h). Patients with one or more of the following criteria were diagnosed severe PE: high blood pressure (≥160/110 mmHg), proteinuria (≥3+ by dipstick or ≥300 mg/24 h on two random urine samples), oliguria (<500 mL/24 h), cerebral or visual disturbances, pulmonary edema or cyanosis, impaired liver functions, epigastric or right upper-quadrant pain and fetal growth restriction35.
Extraction of DNA and genotyping of IL-1
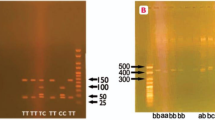
Genomic DNA was extracted from 300 ml peripheral blood by using Qiagen DNA extraction kit. The polymorphisms of rs315952 in IL1RN and rs17561 in IL1A were genotyped by TaqMan allelic discrimination real-time PCR. The Taqman probes and primers were designed by Applied Biosystems of Life Technologies. For rs315952, the sequence of forward and reverse primer is 5′-GCTTCGCCTTCATCCGCTCAGACAG-3′ and 5′-GGCCCCACCACCAGTTTTGAGTCTG-3′ respectively. For rs17561, the forward primer is 5′-ACATTGCTCAGGAAGCTAAAAGGTG-3′ and reverse primer is 5′-TGACCTAGGCTTGATGATTTCTAAA-3′. The polymerase chain reaction (PCR) was conducted in 25 ul reaction mixture, containing 20 × SNP Genotyping Assay 1.25 ul, 2 × PCR Master Mix 12.5 ul, DNA and DNase-free water 11.25 ul. The amplifications were carried out by C1000TM thermal cycler system with the following conditions: 95°C for 3 min, followed by 45cycles at 95°C for 15 sec and 60°C for 1 min. For each cycle, the fluorescent signal from the VIC- or FAM-labeled probes was determined. The discrimination of genotypes was conducted with Bio-Rad CFX manager 3.0 software.
All experiments were carried out in accordance with relevant guidelines and regulations.
Statistical analysis
All analyses were performed by statistical software package SPSS19.0. Hardy-Weinberg equilibrium was examined in control group with goodness-of-fit χ2 test. Student's t test was used to test the comparison of demographic and clinical characteristics between cases and controls. An analysis of variance (ANOVA) was used to conduct genotype-phenotype analysis. The level of statistical significance was defined as p-value<0.05. The allelic and genotypic distributions of cases and controls were compared by Pearson's χ2 test (if expected values were below 5, Fisher's exact test was used) and P-values <0.025 were considered significant when Bonferroni's correction was made.
References
Steegers, E. A., von Dadelszen, P., Duvekot, J. J. & Pijnenborg, R. Pre-eclampsia. Lancet 376, 631–644 (2010).
Canto, J. G. et al. Number of coronary heart disease risk factors and mortality in patients with first myocardial infarction. JAMA 306, 2120–2127 (2011).
Lachmeijer, A. M. et al. Searching for preeclampsia genes: the current position. Eur. J. Obstet. Gynecol. Reprod. Biol. 105, 94–113 (2002).
Liang, S. et al. Association between Val158Met functional polymorphism in the COMT gene and risk of preeclampsia in a Chinese population. Arch. Med. Res. 43, 154–158 (2012).
Hill, L. D. et al. Fetal ERAP2 variation is associated with preeclampsia in African Americans in a case-control study. BMC Med. Genet. 12, 64 (2011).
Mohajertehran, F., Tavakkol Afshari, J., Rezaieyazdi, Z. & Ghomian, N. Association of single nucleotide polymorphisms in the human tumor necrosis factor-alpha and interleukin 1-beta genes in patients with pre-eclampsia. Iran. J. Allergy Asthma Immunol. 11, 224–229 (2012).
Vural, P. et al. Tumor necrosis factor alpha, interleukin-6 and interleukin-10 polymorphisms in preeclampsia. J. Obstet. Gynaecol. Res. 36, 64–71 (2010).
Takeuchi, F. et al. Reevaluation of the association of seven candidate genes with blood pressure and hypertension: a replication study and meta-analysis with a larger sample size. Hypertens Res 35, 825–831 (2012).
Chen, Z., Xu, F., Wei, Y., Liu, F. & Qi, H. Angiotensin converting enzyme insertion/deletion polymorphism and risk of pregnancy hypertensive disorders: a meta-analysis. J Renin Angiotensin Aldosterone Syst 13, 184–195 (2012).
Rusterholz, C., Hahn, S. & Holzgreve, W. Role of placentally produced inflammatory and regulatory cytokines in pregnancy and the etiology of preeclampsia. Semin. Immunopathol. 29, 151–162 (2007).
Amash, A., Holcberg, G., Sapir, O. & Huleihel, M. Placental secretion of interleukin-1 and interleukin-1 receptor antagonist in preeclampsia: effect of magnesium sulfate. J. Interferon Cytokine Res. 32, 432–441 (2012).
Szarka, A., Rigo, J., Jr, Lazar, L., Beko, G. & Molvarec, A. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol. 11, 59 (2010).
Rinehart, B. K. et al. Expression of the placental cytokines tumor necrosis factor alpha, interleukin 1beta and interleukin 10 is increased in preeclampsia. Am. J. Obstet. Gynecol. 181, 915–920 (1999).
Bidwell, J. et al. Cytokine gene polymorphism in human disease: on-line databases. Genes Immun. 1, 3–19 (1999).
Jin, G. X. et al. Association between IL-1RN gene polymorphisms and susceptibility to ankylosing spondylitis: a large Human Genome Epidemiology review and meta-analysis. Genetics and molecular research: GMR 12, 1720–1730 (2013).
Tahmasebi, Z. et al. Interleukin-1 gene cluster and IL-1 receptor polymorphisms in Iranian patients with systemic lupus erythematosus. Rheumatol. Int. 33, 2591–2596 (2013).
Wu, X. et al. IL-1 receptor antagonist gene as a predictive biomarker of progression of knee osteoarthritis in a population cohort. Osteoarthritis Cartilage 21, 930–938 (2013).
Meyer, N. J. et al. IL1RN coding variant is associated with lower risk of acute respiratory distress syndrome and increased plasma IL-1 receptor antagonist. Am J Respir Crit Care Med 187, 950–959 (2013).
Liu, Y. et al. Genetic variants in IL1A and IL1B contribute to the susceptibility to 2009 pandemic H1N1 influenza A virus. BMC Immunol. 14, 37 (2013).
Sata, F. et al. Proinflammatory cytokine polymorphisms and the risk of preterm birth and low birthweight in a Japanese population. Mol Hum Reprod 15, 121–130 (2009).
Hata, Y. et al. A nonsynonymous variant of IL1A is associated with endometriosis in Japanese population. J. Hum. Genet. 58, 517–520 (2013).
White, K. L. et al. Ovarian cancer risk associated with inherited inflammation-related variants. Cancer Res. 72, 1064–1069 (2012).
Mfuna Endam, L., Cormier, C., Bosse, Y., Filali-Mouhim, A. & Desrosiers, M. Association of IL1A, IL1B and TNF gene polymorphisms with chronic rhinosinusitis with and without nasal polyposis: A replication study. Arch. Otolaryngol. Head Neck Surg. 136, 187–192 (2010).
Kobayashi, T., Tokunaga, N., Isoda, H., Kanayama, N. & Terao, T. Vasospasms are characteristic in cases with eclampsia/preeclampsia and HELLP syndrome: proposal of an angiospastic syndrome of pregnancy. Semin. Thromb. Hemost. 27, 131–135 (2001).
Darmochwal-Kolarz, D., Rolinski, J., Leszczynska-Goarzelak, B. & Oleszczuk, J. The expressions of intracellular cytokines in the lymphocytes of preeclamptic patients. Am. J. Reprod. Immunol. 48, 381–386 (2002).
Meisser, A., Chardonnens, D., Campana, A. & Bischof, P. Effects of tumour necrosis factor-alpha, interleukin-1 alpha, macrophage colony stimulating factor and transforming growth factor beta on trophoblastic matrix metalloproteinases. Mol. Hum. Reprod. 5, 252–260 (1999).
Rusterholz, C., Gupta, A. K., Huppertz, B., Holzgreve, W. & Hahn, S. Soluble factors released by placental villous tissue: Interleukin-1 is a potential mediator of endothelial dysfunction. Am. J. Obstet. Gynecol. 192, 618–624 (2005).
Santulli, G. et al. CaMK4 Gene Deletion Induces Hypertension. J Am Heart Assoc 1, e001081 (2012).
Lobmeyer, M. T. et al. Polymorphisms in genes coding for GRK2 and GRK5 and response differences in antihypertensive-treated patients. Pharmacogenet Genomics 21, 42–49 (2011).
Santulli, G., Trimarco, B. & Iaccarino, G. G-protein-coupled receptor kinase 2 and hypertension: molecular insights and pathophysiological mechanisms. High Blood Press Cardiovasc Prev 20, 5–12 (2013).
Goddard, K. A. et al. Candidate-gene association study of mothers with pre-eclampsia and their infants, analyzing 775 SNPs in 190 genes. Hum. Hered. 63, 1–16 (2007).
Faisel, F. et al. Polymorphism in the interleukin 1 receptor antagonist gene in women with preeclampsia. J. Reprod. Immunol. 60, 61–70 (2003).
Valencia Villalvazo, E. Y. et al. Analysis of polymorphisms in interleukin-10, interleukin-6 and interleukin-1 receptor antagonist in Mexican-Mestizo women with pre-eclampsia. Genetic testing and molecular biomarkers 16, 1263–1269 (2012).
Kang, L., Chen, C. H., Yu, C. H., Chang, C. H. & Chang, F. M. Interleukin-1beta gene is not associated with preeclampsia in Taiwanese. Taiwan. J. Obstet. Gynecol. 51, 240–244 (2012).
Bulletins--Obstetrics, A. C. o. P. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstet. Gynecol. 99, 159–167 (2002).
Acknowledgements
We are grateful to all participants who have made the completion of this study. This work was supported by the National Natural Science Foundation of China (81371499 and 30971586).
Author information
Authors and Affiliations
Contributions
J.L. carried out the initial analyses, reviewed and revised the manuscript. M.L. carried out the initial analyses and approved the final manuscript as submitted. J.L.* and M.L.* contribute equally to this work. J.Z. and P.T. designed the data collection instruments. J.W. coordinated and supervised data collection. X.W. approved the final manuscript as submitted. Y.Y. reviewed the manuscript. S.L. conceptualized and designed the study, drafted the initial manuscript and write manuscript. X.L. conceptualized and designed the study.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Li, J., Liu, M., Zong, J. et al. Genetic variations in IL1A and IL1RN are associated with the risk of preeclampsia in Chinese Han population. Sci Rep 4, 5250 (2014). https://doi.org/10.1038/srep05250
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep05250
This article is cited by
-
Exploration of CYP21A2 and CYP17A1 polymorphisms and preeclampsia risk among Chinese Han population: a large-scale case-control study based on 5021 subjects
Human Genomics (2020)
-
Interleukin-23 receptor (IL-23R) gene polymorphisms and haplotypes associated with the risk of preeclampsia: evidence from cross-sectional and in silico studies
Journal of Assisted Reproduction and Genetics (2019)
-
Polymorphisms of the IL27 gene in a Chinese Han population complicated with pre-eclampsia
Scientific Reports (2016)
-
Contribution of TIMP3 polymorphisms to the development of preeclampsia in Han Chinese women
Journal of Assisted Reproduction and Genetics (2015)
-
Investigation of polymorphisms in pre-eclampsia related genes VEGF and IL1A
Archives of Gynecology and Obstetrics (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.