Abstract
Modern acidification by the uptake of anthropogenic CO2 can profoundly affect the physiology of marine organisms and the structure of ocean ecosystems. Centennial-scale global and regional influences of anthropogenic CO2 remain largely unknown due to limited instrumental pH records. Here we present coral boron isotope-inferred pH records for two periods from the South China Sea: AD 1048–1079 and AD 1838–2001. There are no significant pH differences between the first period at the Medieval Warm Period and AD 1830–1870. However, we find anomalous and unprecedented acidification during the 20th century, pacing the observed increase in atmospheric CO2. Moreover, pH value also varies in phase with inter-decadal changes in Asian Winter Monsoon intensity. As the level of atmospheric CO2 keeps rising, the coupling global warming via weakening the winter monsoon intensity could exacerbate acidification of the South China Sea and threaten this expansive shallow water marine ecosystem.
Similar content being viewed by others
Introduction
The present-day level of atmospheric CO2 is the highest known to occur for the past 2 million years1. One half of the total modern anthropogenic CO2 emissions have been absorbed by the ocean with an estimated rate of up to 1 million metric tons per hour2,3, resulting in a reduction of seawater pH (pHsw) and so-called ocean acidification. Sea surface pHsw dropped by ~0.1 unit relative to preindustrial time, based on modeling estimates4 and is projected to decrease by another 0.3–0.4 units by AD 2100 under the IS92a “business-as-usual” scenario5. Ocean acidification could lead to significant shifts in the structure and dynamics of ocean ecosystems. One of the direct impacts of acidification is that it may lead to a decrease in the saturation state of surface ocean waters with respect to CaCO3. This is a threat to marine organisms that rely on the process of calcification to construct their skeletons6,7,8. These studies are based on modern observations and model predictions4 with a pHsw decrease rate of ~0.002 year−1 (ref. 5). However, instrumental pHsw records are sparse and span no more than three decades9,10. Natural archives offer an alternative approach to reconstruct pre-instrumental pHsw records much further back in time.
Biogenic carbonate δ11B data have been suggested as a promising proxy to reconstruct ambient pHsw since the 1990s (refs. 11,12). Recent studies13,14,15 showed that coral δ11B could faithfully record pH values of extracellular calcifying fluids (pHcf) that depend directly on ambient (external) seawater pHsw. Only two published δ11B-derived pHsw records provide decadal to centennial timescales records16,17. The two records from Flinders16 and Arlington Reefs17 of the Great Barrier Reef (GBR) (Fig. 1) express a signature of pHsw reduction over the last 50 years. However, the inconsistency of pHsw variations during AD 1800–2000 for the two records (Supplementary Fig. 4) cannot provide convincing evidence for a decrease in pHsw that can be unambiguously attributed to atmospheric CO2 rising.
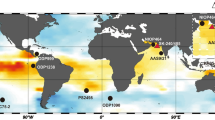
Maps with study sites.
Modern and fossil Porites cores were drilled from (a) Xiaodonghai Reef (star), located off (b) southern Hainan in (c) the northern South China Sea (SCS). Circle, triangle and square symbols denote the locations of Arlington Reef17 and Flinders Reef16 in the Great Barrier Reef (GBR) and Station of ALOHA9 in Hawaii, respectively. The maps created using NCAR Command Language (http://www.ncl.ucar.edu/) and MapPlot the Earth.4 dataset (http://www.ncl.ucar.edu/Document/HLUs/Classes/MapPlotData4_1_earth_4.shtml). Representative X-radiographs of slices of (d) modern coral sample Song-5 and (e) fossil coral sample Dong-5 are also shown. Red lines denote the subsampling transect along the maximum growth axis.
In this study, we present a new pHsw record inferred from skeletal δ11B data of modern and fossil corals from Xiaodonghai Reef (18°12.46′N, 109°29.93′E), located in the South China Sea (SCS) (Fig. 1). The modern coral record spans from AD 1838–2001, before the beginning of the Industrialization Period and covers the current warm period (CWP, since 1950s). The δ11B–inferred record of a precisely 230Th-dated18 fossil coral records values from AD 1048–1079, within the Medieval Warm Period (MWP)19. Understanding pHsw variations in the MWP of the last millennium without anthropogenic CO2 perturbation will be helpful in understanding modern acidification in the CWP.
Results
Measured δ11B values of modern and fossil corals are given in Supplementary Figure 3 and Supplementary Tables 2–4. The pHsw values (total scale) were calculated from measured δ11B data (see Methods and ref. 15). The coral-reconstructed pHsw record from AD 1838–2001 varies from 7.82 to 8.21 (Fig. 2b). There is a ~30-yr period with maximal pHsw values of 8.20–8.21 during AD 1830–1870. A minimum pHsw value of 7.82 occurred from AD 1990–1993. Striking features of this record includes a decreasing trend of −0.0015 ± 0.0002 pH year−1 from AD 1838–2001 (R2 = 0.63, p < 0.0001) and inter-decadal variations with amplitudes of 0.1–0.2 units (Fig. 2b). The reconstructed bi-weekly pHsw data in AD 2000 displays strong seasonality, from high values of ~8.1 in winter to low values of ~7.6 in summer (Fig. 3b). This seasonal pHsw change is consistent with local instrumental data (Supplementary Fig. 7) and follows intra-annual seawater pCO2 variations of SCS reef water, from a high value of ~500 μatm in summer to a low value of ~380 μatm in winter20,21. A 32-yr fossil coral-derived pHsw record is characterized by a high average value of 8.17 ± 0.08 (2σ, n = 8) (Fig. 2c).
Coral-inferred pHsw in the SCS and atmospheric CO2 (ref. 32).
Comparison of the pHsw (red circles and lines) based on boron isotope and atmospheric CO2 (gray dots and yellow line) from (a) AD 1000–2000 and two enlarged sections during (b) AD 1030–1100 and (c) AD 1830–2000. CO2 data after 1958 (included) are annual averages of direct observations from Mauna Loa and the South Pole32. CO2 data before 1958 are from ice cores33. Vertical red bars denote the 2σ uncertainty of pHsw. Dark blue triangle in (c) indicates position of 230Th-dated layer. The chronology of fossil sample was set by year intervals relative to the dating layer.
Biweekly-resolution coral data and environmental parameters in AD 2000 and time series of the reconstructed pHsw based on boron isotope and Siberian High index24 from AD 1838–2001.
(a) Coral δ18O (pink) and sea surface temperature (SST) (black line) and (b) coral-inferred pHsw (red), wind speed (cyan line) and Siberian High index24 (blue line) in AD 2000. Monthly SST data (17–19°N, 115–117°E) with a 1° resolution were from the Optimum Interpolation Sea Surface Temperature, Version 2, National Oceanic and Atmospheric Administration (NOAA): (http://www.esrl.noaa.gov/psd/data/gridded/data.noaa.oisst.v2.html). Wind data (17–19°N, 115–117°E) with a 0.25° resolution were from the Blended Sea Winds provided by the NOAA National Climate Data Center: (http://www.ncdc.noaa.gov/oa/rsad/air-sea/seawinds.html). Intra-annual data were calendared by aligning coral δ18O maxima with SST minima and applying a linear interpolation between anchor points. (c) Comparison of coral-inferred pHsw data and 4-yr averaged Siberian High index24 (blue line) and (d) detrended pHsw data and Siberian High index. Vertical red bars denote the 2σ uncertainty of pHsw.
Discussion
The longest in-situ pHsw record at Station ALOHA in Hawaii shows a significant decreasing trend of ~0.018 pH year−1 in surface water of the central North Pacific Ocean over the past two decades (AD 1988–2011) (Supplementary Fig. 4)9. Our coral-inferred pHsw reconstruction from the SCS provides the first evidence of a decreasing trend, traced back to the Industrialization Period (Fig. 2b). Atmospheric CO2 increased slightly between AD 1840 and 1950 at a rate of 0.270 ± 0.010 ppm year−1 (R2 = 0.97, p < 0.0001), after which the rate increases dramatically to 1.135 ± 0.062 ppm year−1 (R2 = 0.97, p < 0.0001) (Fig. 2). Our pHsw sequence follows the temporal pattern of CO2. The coral-inferred pHsw values of 8.1–8.2 in AD 1830–1870 are not significantly different from those in the MWP (Fig. 2). The values decreased at a rate of −0.0011 ± 0.0003 pH year−1 (R2 = 0.35, p = 0.0009) in a 110-yr interval after AD 1840. This rate of decline almost tripled to −0.0029 ± 0.0013 pH year−1 (R2 = 0.33, p = 0.04) after AD 1950. This pHsw follows changes observed in the CO2 time series (Pearson correlation coefficient: R2 = 0.63, n = 41, p < 0.0001). The long-term decreasing trend of pHsw also corresponds to a shift to more negative δ13C values (−0.0037 ± 0.0010 year−1, R2 = 0.26, p = 0.0007, AD 1838–2001) (Supplementary Fig. 5b), where a significant positive correlation (R2 = 0.21, n = 41, p = 0.003) exists between pHsw and δ13C. This suggests synchronous changes between seawater acidification and decreasing dissolved inorganic carbon (DIC) in this region. The δ13C change could be attributed to the “Suess effect”, which is due to uptake of atmospheric CO2 by the oceans that has been progressively depleted in 13C by combustion of fossil fuels, as supported by previous studies16,17. These signatures all reflect the remarkable absorption of anthropogenic CO2 by the ocean and induced acidification in response to the rapid rise in atmospheric CO2 since the Industrialization Period.
While atmospheric CO2 governs the long-term SCS pHsw trend, the inter-decadal cycles of 0.1–0.2 pH must be linked to other mechanism, in the absence of significant inter-decadal variability in atmospheric CO2. The physical-biogeochemical conditions of the SCS are strongly influenced by the Asian monsoon22,23. During summer, this region is affected by the southwest Asian summer monsoon (ASM), which brings warm and wet tropical air masses to the study area, resulting in increased precipitation (Supplementary Fig. 1b). However there is no correlation between the coral-inferred pHsw and the precipitation record (Supplementary Fig. 6). The Asian winter monsoon (AWM) prevails during November to March, bringing cold and dry continental air associated with the Siberian High. From AD 1994 to 2011, the monthly mean wind speed varied from 0.4 to 11 m s−1, with prominent peaks occurring during the winter monsoon season (Supplementary Fig. 1c). We found a significant correlation (R2 = 0.20, n = 41, p = 0.003) between the pHsw and Siberian High pressure, an index of AWM intensity24. Low and high pHsw values respectively co-vary with weak and strong Siberian High phases on an inter-decadal timescale (Fig. 3c). It is a robust feature that the significant correlation (R2 = 0.18, n = 41, p = 0.006) still exists for the two detrended sequences (Fig. 3d). This concurrence suggests that the AWM may contribute to changes in regional pHsw in the SCS.
The linkage between pHsw and AWM is attributable to the flushing efficiency of waters over the reefs as observed in the GBR16 and monsoon-driven productivity. During weak AWM periods with reduced wind forcing22 in the SCS, the build-up of CO2 due to calcification and respiration could lower pHsw due to poor flushing of reef waters by open seawater. In contrast, periods of relatively strong AWM with strengthened surface currents22 could result in higher pHsw values. At the same time, surface primary productivity of the SCS is also controlled by AWM intensity23. A weak (strong) AWM would cause the nutricline to deepen (shallow) and result in lower (higher) surface productivity23 and decreased (increased) pHsw. This mechanism is strongly supported by the co-variation of biweekly-resolved coral-inferred pHsw and AWM wind speed data from AD 2000 (Fig. 3b).
The most acidic condition in AD 1990 is coincident with a sharp drop in the Siberian High index (Fig. 3c), an unprecedented weak event during the past four centuries24. Global warming preferentially warms the high latitudes of Eurasia and this decreases the land-sea pressure gradient and weakens the AWM25. The expected rise in atmospheric CO2 will likely reduce the AWM intensity in the coming decades25 and exacerbate the impact of acidification on the SCS and threaten the most diverse collection of shallow water marine organisms on Earth26. Under the stress of current accelerated ocean acidification, an in-depth understanding of the ecological and socioeconomic repercussions in the SCS and other monsoon regions is imperative.
Methods
Coral cores
A 3-m long core of Poritessp. coral, Song-5, was drilled from Xiaodonghai Reef (18°12.46′N, 109°29.93′E), at a water depth of 8.5 m offshore south Hainan in April 2002 (Fig. 1). Slabs, 7 mm in thickness, were sectioned, washed with ultrapure water and then dried for X-ray images. Annual banding counts were used to establish the chronology. Subsamples were milled in continuous 4-yr intervals down the full length of the core along the maximum growth axis for the period AD 1838 to 2001. A set of biweekly resolution subsamples was milled from the AD 2000 segment. One more 1-m long core fossil Porites sp. coral, Dong-5, was also drilled. Subsamples were milled at 4-year intervals along the maximum growth axis on a 32-yr section. X-ray diffraction analysis shows coral samples are 100% aragonite and scanning electron microscopy image indicates an absence of secondary aragonite. After ground into a homogeneous powder using a pre-cleaned agate, all the subsamples (1–3 mg) were treated with 10% NaOCl and ultrapure water (18.2 MΩ) in an ultrasonic bath.
Isotopic analysis and dating
Micro-sublimation techniques were used for boron purification27. Aliquots (~50 μL) containing 50–100 ng of boron were then diluted to 20 ppb boron for the isotopic measurements. δ11B compositions were determined by a multi-collector inductively coupled plasma mass spectrometer (MC-ICP-MS), Thermo Neptune, at the Earth Dynamic System Research Center, National Cheng Kung University. Instrumental mass bias was calibrated using standard-sample bracketing techniques27,28 with NIST SRM 951 boric acid. At each analytical sequence, two JCp-1 and JCt-1 carbonate standards (Poritessp. and giant clam Geological Survey of Japan) and one in-house standard Alfa-B (B ICP-MS solution, Alfa Aesar) were used to monitor the micro-sublimation procedure and plasma condition in order to maintain analytical accuracy. The JCp-1 and JCt-1 standard yielded a mean value of 24.20 ± 0.24‰ (2σ, n = 9) and 16.21 ± 0.05‰ (2σ, n = 3) respectively. The external precision of δ11B measurements are better than ±0.28‰ (2σ)27. The procedure blank is <5 pg of boron. Instrument sensitivity for 20 ppb B was 0.4–0.5 V for 11B signal intensity and the effect of total blanks (<0.01 V, including procedure blank and instrument background) can be negligible. Boron isotopic ratios are expressed are expressed using delta (δ) notation relative to the NIST SRM 951. The δ13C and δ18O measurements were carried out using an isotope ratio mass spectrometer MAT-252 equipped with a micro carbonate automatic sample input Kiel II device in the Institute of Earth Environment, Chinese Academy of Sciences. Results are expressed using delta (δ) notation relative to the Vienna Pee-Dee Belemnite (V-PDB) standard. The external error of the laboratory standard is ±0.1‰ and ±0.2‰ (1σ) for δ13C and δ18O, respectively.
The fossil coral 5 Dong-was dated by 230Th techniques18 (Supplementary Table 1) by a MC-ICP-MS, Thermo Neptune, at the High-Precision Mass Spectrometry and Environment Change Laboratory (HISPEC), National Taiwan University. The first annual banding of the 32-yr section was sampled for dating.
pH calculation
The pHcf was converted by boron isotope systematics based on a following equation29:

where δ11Bsw and δ11Bcoral represents δ11B in seawater (δ11Bsw = 39.5‰)28 and in coral, respectively and α(B3–B4) = 1.0272 (ref. 30). The dissociation constant of boric acid pKB is 8.597 at 25°C and a salinity of 35 (ref. 31). The pHsw value was calculated using the empirical equation: [pHsw = (pHcf − 4.72)/0.466), R2 = 0.99] (refs. 14,15). The external uncertainty of ±0.28‰ (2σ) (ref. 27) for δ11B data corresponds to a precision of better than ±0.02 units for pHcf and ±0.04 for pHsw. The biweekly pHsw was corrected by temperature effect31 on pKB.
References
Hönisch, B., Hemming, N. G., Archer, D., Siddall, M. & McManus, J. F. Atmospheric carbon dioxide concentration across the Mid-Pleistocene Transition. Science 324, 1551–1554 (2009).
Sabine, C. L. et al. The oceanic sink for anthropogenic CO2 . Science 305, 367–371 (2004).
Brewer, P. G. A changing ocean seen with clarity. Proc. Natl. Acad. Sci. USA. 106, 12213–12214 (2009).
Caldeira, K. & Wickett, M. E. Anthropogenic carbon and ocean pH. Nature 425, 365 (2003).
Orr, J. C. et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437, 681–686 (2005).
Hoegh-Guldberg, O. et al. Coral reefs under rapid climate change and ocean acidification. Science 318, 1737–1742 (2007).
Pandolfi, J. M., Connolly, S. R., Marshall, D. & Cohen, A. L. Projecting coral reef futures under global warming and ocean acidification. Science 333, 418–422 (2011).
Inoue, S., Kayanne, H., Yamamoto, S. & Kurihara, H. Spatial community shift from hard to soft corals in acidified water. Nature Clim. Change 3, 683–687 (2013).
Dore, J. E., Lukas, R., Sadler, D. W., Church, M. J. & Karl, D. M. Physical and biogeochemical modulation of ocean acidification in the central North Pacific. Proc. Natl. Acad. Sci. USA. 106, 12235–12240 (2009).
Byrne, R. H., Mecking, S., Feely, R. A. & Liu, X. Direct observations of basin-wide acidification of the North Pacific Ocean. Geophys. Res. Lett. 37, L02601; 10.1029/2009GL040999 (2010).
Vengosh, A., Kolodny, Y., Starinsky, A., Chivas, A. R. & Mcculloch, M. T. Coprecipitation and isotopic fractionation of boron in modern biogenic carbonates. Geochim. Cosmochim. Acta 55, 2901–2910 (1991).
Hemming, N. G. & Hanson, G. N. Boron isotopic composition and concentration in modern marine carbonates. Geochim. Cosmochim. Acta 56, 537–543 (1992).
Al-Horani, F. A., Al-Moghrabi, S. M. & de Beer, D. The mechanism of calcification and its relation to photosynthesis and respiration in the scleractinian coral Galaxea fascicularis. Mar. Biol. 142, 419–426 (2003).
Trotter, J. et al. Quantifying the pH ‘vital effect’ in the temperate zooxanthellate coral Cladocora caespitosa: Validation of the boron seawater pH proxy. Earth Planet. Sci. Lett. 303, 163–173 (2011).
McCulloch, M. T., Falter, J., Trotter, J. & Montagna, P. Coral resilience to ocean acidification and global warming through pH up-regulation. Nature Clim. Change 2, 623–627 (2012).
Pelejero, C. et al. Preindustrial to modern interdecadal variability in coral reef pH. Science 309, 2204–2207 (2005).
Wei, G., McCulloch, M. T., Mortimer, G., Deng, W. & Xie, L. Evidence for ocean acidification in the Great Barrier Reef of Australia. Geochim. Cosmochim. Acta 73, 2332–2346 (2009).
Shen, C.-C. et al. High-precision and high-resolution carbonate 230Th dating by MC-ICP-MS with SEM protocols. Geochim. Cosmochim. Acta 99, 71–86 (2012).
Moinuddin, A. et al. Continental-scale temperature variability during the past two millennia. Nature Geosci. 6, 339–346 (2013).
Dai, M. et al. Diurnal variations of surface seawater pCO2 in contrasting coastal environments. Limnol. Oceanogr. 54, 735–745 (2009).
Yan, H. et al. Coral reef ecosystems in the South China Sea as a source of atmospheric CO2 in summer. Chinese Sci. Bull. 56, 676–684 (2011).
Xu, D. & Malanotte-Rizzolib, P. The seasonal variation of the upper layers of the South China Sea (SCS) Circulation and the Indonesian through flow (ITF): An ocean model study. Dynam. Atoms. Oceans 63, 103–130 (2013).
Liu, K.-K. et al. Inter-annual variation of chlorophyll in the northern South China Sea observed at the SEATS Station and its asymmetric responses to climate oscillation. Biogeosciences Discuss. 10, 6899–6938 (2013).
D'Arrigo, R., Wilson, R., Panagiotopoulos, F. & Wu, B. On the long-term interannual variability of the East Asian winter monsoon. Geophy. Res. Lett. 32, L21706; 10.1029/2005GL023235 (2005).
Hori, M. E. & Ueda, H. Impact of global warming on the East Asian winter monsoon as revealed by nine coupled atmosphere-ocean GCMs. Geophys. Res. Lett. 33, L03713; 10.1029/2005GL024961 (2006).
Morton, B. & Blackmore, G. South China Sea. Mar. Pollut. Bull. 12, 1236–1263 (2001).
Wang, B.-S. et al. Direct separation of boron from Na- and Ca-rich matrices by sublimation for stable isotope measurement by MC-ICP-MS. Talanta 82, 1378–1384 (2010).
Foster, G. L. Seawater pH, pCO2 and [CO32−] variations in the Caribbean Sea over the last 130 kyr: A boron isotope and B/Ca study of planktic foraminifera. Earth Planet. Sci. Lett. 271, 254–266 (2008).
Zeebe, R. E., Sanyal, A., Ortiz, J. D. & Wolf-Gladrow, D. A. A theoretical study of the kinetics of the boric acid-borate equilibrium in seawater. Mar. Chem. 73, 113–124 (2001).
Klochko, K., Kaufman, A. J., Yao, W., Byrne, R. H. & Tossell, J. A. Experimental measurement of boron isotope fractionation in seawater. Earth Planet. Sci. Lett. 248, 276–285 (2006).
Dickson, A. G. Thermodynamics of the dissociation of boric acid in synthetic seawater from 273.15 to 318.15 K. Deep Sea Res. Part A 37, 755–766 (1990).
Keeling, C. D. et al. Atmospheric CO2 and 13CO2 exchange with the terrestrial biosphere and oceans from 1978 to 2000: observations and carbon cycle implications. in A History of Atmospheric CO2 and its effects on Plants, Animals and Ecosystems. (Eds Ehleringer, J. R. et al.) 83–113 (Springer Verlag, New York, 2005).
Meure, M. C. et al. Law Dome CO2, CH4 and N2O ice core records extended to 2000 years BP. Geophys. Res. Lett. 33, L14810; 10.1029/2006GL026152 (2006).
Acknowledgements
This study was jointly supported by National Key Basic Research Program of China (2013CB956102 to Y.L.), Strategic Priority Research Program of the CAS (XDA05080300 to K.F.Y.), National Science Foundation of China (41003002 to Y.L.), NSC and MOE Top University Program (120-2116-M006-006 to C.F.Y.), MOST and NTU grants (101-2116-M-002-009, 102-2116-M-002-016 and 101R7625 to C.C.S.).
Author information
Authors and Affiliations
Contributions
Y.L., Z.P. and C.-F.Y. designed and initiated the research. Y.L., Y.-B.L. and C.-F.Y. analyzed B isotope. S.S. and L.X. drilled the cores. C.-C.W. and C.-C.S. conducted 230Th dating. W.L. analyzed O and C isotopes. R.Z., C.-F.Y., K.Y., G.W. and G.B. contributed significantly to the discussion of results and manuscript. Y.L. and C.-C.S. wrote the paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
SUPPLEMENTARY INFORMATION
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. The images in this article are included in the article's Creative Commons license, unless indicated otherwise in the image credit; if the image is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the image. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Liu, Y., Peng, Z., Zhou, R. et al. Acceleration of modern acidification in the South China Sea driven by anthropogenic CO2. Sci Rep 4, 5148 (2014). https://doi.org/10.1038/srep05148
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep05148
This article is cited by
-
Recent ocean acidification trends from boron isotope (δ11B) records of coral: Role of oceanographic processes and anthropogenic CO2 forcing
Journal of Earth System Science (2022)
-
Surface ocean pH variations since 1689 CE and recent ocean acidification in the tropical South Pacific
Nature Communications (2018)
-
What is at stake? Status and threats to South China Sea marine fisheries
Ambio (2017)
-
Transcriptome and biomineralization responses of the pearl oyster Pinctada fucata to elevated CO2 and temperature
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.