Abstract
This study assessed the clinical accuracy of the correlation between the careHPV test and the HC2 test, as well as the correlation between the careHPV test and the HPV-PCR test. From September 21 to December 31, 2009, 419 cervical specimens from women, 30–49 years, were collected. All women were assessed by Digene HC2 High-Risk HPV DNA Test (HC2), careHPV test and HPV-PCR. The concordance rate between careHPV and HC2 was 93.81% and between careHPV and HPV-PCR 88.12%. The sensitivity and specificity of the careHPV test to detect cancers of equal or greater severity than CIN 2 were 85.71% and 83.15% respectively. Results from careHPV, HC2 and HPV-PCR were highly consistent. The careHPV test has good sensitivity and specificity for the detection of HPV infection and is a promising primary screening method for cervical cancer in low-resource regions.
Similar content being viewed by others
Introduction
Cervical cancer is the second most frequent female malignancy worldwide. High-risk human papillomavirus (HPV) is the main cause of cervical cancer, affecting nearly 500,000 women around the world every year and killing more than 270,000. About 80% of these women live in developing countries that do not have adequate cervical cancer screening or treatment programs1. Most developing countries have insufficient infrastructure for cytology-based screening and lack well-trained cytopathologists. In these countries, only a minority of women receive effective screening and treatment for cervical cancer2. Using cytology as the main method of screening is not feasible in developing countries, thus new screening methods are sought and are being evaluated, for example, visual inspection with acetic acid (VIA) and HPV-DNA testing3.
Persistent infection with HPV is the leading cause of cervical cancer and precancerous cervical lesions4. Infection with HPV is very common in young women after the onset of sexual activity. When infection persists, HPV viral oncoproteins disrupt cell-cycle controls, resulting in cervical intraepithelial neoplasia (CIN)5. Therefore, the high-risk HPV test is an effective cervical cancer screening tool. A large sample study in North America and Europe indicated that the sensitivity of the conventional Pap cytology smear for detecting cervical cancer or cancerous lesions of greater or equal seriousness to CIN 2 is only 53%. This contrasts with the Digene HC2 High-Risk HPV DNA Test (HC2) which has a sensitivity of 96.1%. The specificity of the two methods is 96.3% and 90.7%, respectively6. The Digene HC2 assay is a commercially available test for HPV DNA detection and is approved by the United States Food and Drug Administration (FDA). The HC2 assay can detect 13 types of high-risk HPV associated with cervical cancer: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68. However, the HC2 assay is expensive and requires laboratory equipment, so it is not commonly available in developing countries. An international research project, supported by the Bill & Melinda Gates foundation in China and India from 2003–2007, worked towards developing a new, rapid and economical method of cervical cancer screening. One of the methods developed is a high-risk HPV nucleic acid detection kit (using hybrid capture-chemiluminescence technology) known as the careHPV test, developed by Qiagen (Gaithersburg, MD, USA)7.
The careHPV test is a powerful, rapid and precise HPV-DNA detection method in which antibodies bind to magnetic beads, rapidly capturing specific target HPV nucleic acid sequences, which are then detected using a chemiluminescence signal. This method can detect 14 types of high-risk HPV (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68) and more than 80 specimens can be processed in 2.5 hours. This allows screening and follow-up to be completed in one day.
This study assessed the clinical accuracy of the correlation between the careHPV test and the HC2 test, as well as the correlation between the careHPV test and the HPV-PCR test.
Results
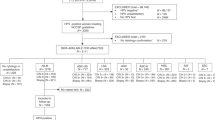
Samples were collected from 419 patients, two of whom failed to return to the clinic for colposcopy and 13 were cases of lesions graded CIN 1+ but were without pathological quality control. There were effectively 404 specimens for analysis: 54 from patients with cervical cancer or precancerous lesions (≥CIN 2); 50 from patients with benign lesions (47 low-grade squamous epithelium intraepithelial lesions graded CIN 1); and 300 from patients with no abnormalities.
The percentage of patients testing positive for HPV with each detection method was: careHPV: 24.01% (97/404); HC2: 25.25% (102/404); and HPV-PCR: 31.93% (129/404) (Tables 1 and 2). Positive detection of CIN 1 lesions by each methodology was: HPV-HC2: 61.70% (29/47); HPV-PCR: 63.83% (30/47); and careHPV: 61.70% (29/47). Positive detection of CIN 2 lesions by each methodology was: HPV-HC2: 90.74% (49/54); HPV-PCR: 90.74% (49/54); and careHPV: 88.89% (48/54).
The results of careHPV test, HC2 test and HPV-PCR test reached agreement (Table 1 and 2). The results of careHPV test had great sensitivity as well as specificity to the lesions graded ≥ CIN 2 (Table 3, 4 and 5) therefore were quite predictive for the lesions graded ≥ CIN 2 (Figure 1). Positive detection of HPV in CIN 1 lesions was low (Figure 2) so the results of careHPV test, HC2 test and HPV-PCR test showed little predictive to CIN 1 lesions.
ROC curve demonstrating sensitivity and specificity of the careHPV, HPV-HC2 and HPV-PCR tests to detect HPV in cervical cancer of equal or greater severity than CIN 2.
The test result variable(s): hc2_code, HPV-PCR_code, careHPVcode has at least one tie between the positive actual state group and the negative actual state group. The smallest cutoff value is the minimum observed test value minus 1 and the largest cutoff value is the maximum observed test value plus 1. All the other cutoff values are the averages of two consecutive ordered observed test values. HPV-HC2 (hc2_code): Z = 0.837; 95% CI: 0.772–0.903; P = 0.0001. HPV-PCR (gl_res_cod): Z = 0.844; 95% CI: 0.767–0.986; P = 0.0001. careHPV (ca_rescodf): Z = 0.844; 95% CI: 0.779–0.909; P = 0.0001. The larger the area under the ROC curve, the higher the diagnostic value of the test.
ROC curve demonstrating sensitivity and specificity of the careHPV, HPV-HC2 and HPV-PCR tests to detect HPV in CIN 1 lesions.
The test result variable(s): hc2_code, gl_res_cod, ca_rescodf has at least one tie between the positive actual state group and the negative actual state group. Statistics may be biased. (a) Under the nonparametric assumption. (b) Null hypothesis: true area = 0.5. HPV-HC2 (hc2_code): Z = 0. 316; 95% CI: 0. 230–0. 403; P = 0.0001. HPV-PCR (gl_res_cod): Z = 0. 365; 95% CI: 0. 278–0. 451; P = 0.0001. careHPV (ca_rescodf): Z = 0. 344; 95% CI: 0. 256–0. 433; P = 0.0001. The area under ROC curve of small, no diagnostic value.
Discussion
This research shows that the careHPV, HC2 and HPV-PCR tests are highly consistent. The HC2 and HPV-PCR tests have been previously licensed by the China Food and Drug Administration(CFDA).
The sensitivity with which the careHPV test detected cancers of grade CIN 2 or higher was 85.71%. This places its power to detect cancer between that of the conventional Pap smear (53%) and the HC2 test (96.1%). The specificity of the careHPV test was lower than the HC2 and the Pap smear. The diagnostic value of the careHPV test was highly significant, as shown by the ROC curve.
There was no statistically significant difference in the positive rate of the careHPV test and HC2 test (P = 0.672). In contrast, the positive detection rate of the careHPV test was significantly lower than the positive detection rate of the HPV-PCR test (P = 0.012).
The HC2 and HPV-PCR tests were both conducted on a single specimen, which may have influenced the results. The detection process requires three days of repeated freeze-thawing of samples and may have impacted upon the results. Nevertheless, the careHPV test has value as a cervical cancer screening method in areas with poor medical resources.
The findings of a randomized trial of cryotherapy based on VIA or HPV-DNA testing in South Africa has prompted the Alliance for Cervical Cancer Prevention to recommend that primary screening in low-resource settings should be based on either VIA or HPV-DNA testing8,9. Amongst young women in China, 3.4% were diagnosed with lesions of CIN grade 2 or worse and 17.1% tested positive for HPV DNA10. The International Agency for Research on Cancer has published several systematic reviews and is now recommending that HPV-DNA testing be used for primary screening, as an alternative to cytology2. A major advantage of HPV-DNA testing is that the negative predictive value can safely extend the interval for rescreening of patients with negative results to 5 years or more11. In our research, the careHPV Test was negative for CIN ≥ 2 and cervical cancer with a negative predictive value of 98.05% (95% CI: 95.98–99.20%).
The careHPV test was designed specifically for application in low-resource public health settings to screen women 30 years of age and older. The test procedure is simpler than the HC2 assay and has a faster time to results, reducing the need for both highly trained personnel and the sophistication of the laboratory equipment required. Professor You-lin Qiao reported the careHPV test (QIAGEN, Gaithersburg, MD, USA),which was able to detect 14 high-risk types of carcinogenic human papillomavirus (HPV) in about 2·5 h, was suitable for screening women in developing regions for cervical intraepithelial neoplasia (CIN). The careHPV test can be done on only a small bench-top workspace and does not require electricity or running water, therefore it is promising as a primary screening method for cervical-cancer prevention in low-resource regions12. In spite of this, the careHPV test still performed adequately under such conditions. In contrast, HC2 testing required experienced professionals and optimal laboratory conditions. The careHPV test is cost-effective and allows women to wait for their results on the day of testing. This enables further management to be provided effectively, which is crucial to the success of programs in low-resource settings where the accessibility of services is restricted13. Goldie and colleagues have shown that in general, a greater cost-effectiveness is achieved when screening and treatment strategies can be completed in one visit and this should be possible with the careHPV test14.
HPV-HC2 testing returns a positive result for over 90% of patients with CIN ≥ 2 and is therefore useful as a screening and prediction tool for these patients. CIN 1 patients with HPV test negative more than 30% of the time using HPV-HC2, limiting its utility as a screening test in such patients.
The duration of infection with high-risk HPV is an important factor causing cervical cancer. High-risk HPV can have vertical transmission; HPV16 infection is often established by vertical transmission15. HPV screening is very important and the careHPV test has clear advantages in certain settings and is worthy of further study. Its simplicity and cost-effectiveness make it worth promoting as a cervical cancer screening method for improved health care in areas with few public health resources.
Methods
Patients
Women with known cervical lesions attending the out-patient clinic of Nanjing Drum Tower Hospital for cervical cancer screening were included in the study if they were between 30 and 59 years old and had been/were sexually active. Women who were confirmed or suspected of being pregnant were excluded (but could participate in the research eight weeks after delivery), as were those with a history of cervical surgery (e.g., hysterectomy). The study was based on the statement of ethical principles in the Declaration of Helsinki.Women participated voluntarily in the study. The purpose and significance of the study, the benefits and possible risks were explained to each patient before they signed informed consent forms. The study received ethical permission from the Hospital Ethics Committee No.2009039.
The sample size was 404 patients, of which it was predicted that there would be 54 patients with cervical cancer or precancerous cervical lesions (≥CIN 2), 50 patients with benign lesions (47 low-grade squamous intraepithelial lesions [CIN 1]) and 300 patients with normal cervixes.
Study course
Demographic, menstrual and reproductive histories were collected from each eligible patient. The general gynecological condition of each patient was recorded before two cervical cell specimens were collected. One specimen was for careHPV detection, the second was for both HPV-PCR and HC2 testing. Patients were numbered in order of enrollment – odd numbered patients provided the careHPV specimen first, while patients with an even number provided their HC2/HPV-PCR specimen first. Cervical cell specimens were then collected for liquid-based cytology (LBC). If there were any adverse events during specimen collection (such as damage and excessive bleeding) then the cervix was visually inspection by VIA. Cotton balls, large enough to cover all of the cervix were soaked in 5% acetic acid and then, gently pressed on the cervix and moved up and down, left and right (including the external aperture of the cervix), so the acetic acid fully saturated the cervical epithelia. After one minute, the response of the cervical epithelium to the acetic acid was observed directly with ordinary light. Any lesion in the region will show white under these conditions. A preliminary diagnosis is made according to the thickness of any white lesions, their boundary and contour, whether they are mosaic in appearance and the regression speed. Results were classified into VIA-negative, VIA-positive and suspected cancer (SFC). If both VIA and LBC were negative, patients were classed into the normal group; if VIA was positive or SFC or LBC showed atypical squamous cells of undetermined significance (ASC-US), then the patients would receive electronic colposcopy. If a lesion was found, then it would be biopsied directly. If the examination by colposcopy was not satisfactory (for example, the integral squamous-columnar epithelium junction could not be seen, or if lesions are in the cervical canals, or if biopsy forceps cannot touch the lesions, or glandular lesions are suspected), endocervical curettage was used. If colposcopy indicated the cervix was normal or the pathology results of the direct biopsy were normal, then these patients were classed as “normal”; if the colposcopy or the biopsy pathology results indicated inflammation, the patient would be assigned to the benign lesion group; if the biopsy pathology results indicated cancerous lesions of equal or greater seriousness than CIN 2, the patient was classed into the precancerous or the cancer group and was treated according to the 2009 ASCCP guideline.
Patients who had already been diagnosed as ≥CIN 2 by colposcopy but were waiting for treatment and were visiting the cervical lesion out-patient clinic were also eligible to participate in this study. These patients were classed in the precancerous lesions or the cancer group and were treated according to the 2009 ASCCP guideline.
Each specimen was coded randomly by the quality control center of the Chinese Academy of Medical Sciences Cancer Institution so that careHPV, HC2 and HPV-PCR testing could be carried out blindly and independently.
careHPV, HC2 and HPV-PCR
careHPV detection
careHPV is a powerful, fast and accurate method for the detection of HPV-DNA. Using an antibody-bound paramagnetic beads technique, the technology can quickly and quantitatively detect 14 types of high risk HPV (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68). The protocol can process more than 80 specimens in 2.5 hours, thus improving the speed and efficiency of screening as well as allowing the results to be known on the same day. The detection instrument can be powered by dry batteries, so can function where there is no electricity available. Furthermore, reagents are pre-prepared in dropper bottles facilitating simple, easy to understand operation. This is a novel, simple, rapid HPV test kit, with cost and time relative to HPV-HC2 equivalent to approximately one-third and one-sixth, respectively.
The careHPV kit was obtained from QIAGEN Biological Engineering (Shenzhen). The experimental operation was performed according to the careHPV instruction manual provided. Briefly, to detect HPV with the careHPV system13, supplied lysate was added to the specimens to dissolve the cells. The exposed double-stranded HPV DNA was denatured to become single-strand DNA by heating the lysate and this single-stranded DNA then hybridizes with the full-length complementary RNA to form HPV DNA/RNA hybrids. Magnetic beads coated with monoclonal antibody for HPV DNA/RNA hybrid mixtures are then added. Alkaline phosphatase is also added to combine with the monoclonal antibody and a chromogenic substrate is acted upon by the alkaline phosphatase. The light intensity generated by the reaction of the chromogenic substrate reflects the amount of HPV DNA contained in the specimen. The ratio of relative light unit (RLU) to the mean RLU of the minimum positive control (RLU/CO) is used for diagnosis. The standard definition for a sample to be positive for HPV DNA is a reading ≥ 0.5 pg/ml in one specimen. RLU/CO = 1.0 is used as a cut-off point.
HC2 detection
Second-generation hybrid capture (HC2, QIAGEN) is a commercial reagent used for HPV DNA detection, which is approved by the U.S. FDA. HC2 can detect 13 types of high-risk HPV associated with cervical cancer: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68. HC2 detection technology had been reviewed by the CFDA. Use of the assay in this study was performed as per manufacturers' protocol. An RLU/CO = 1.0 as the cut-off point, ≥1.0 is a positive result and < 1.0 is a negative result.
HPV-PCR detection
The study used PCR Fluorescence Detection Kit for human papillomavirus (HPV), produced by the Dragon biological technology (Shenzhen) Limited. The kit was approved by SFDA (the number of approval document: STATE MEDICAL PERMITMENT No.S20070001).Using multiple nucleic acid amplification (PCR) method with fluorescence detection and primers and probes designed specifically for target sequences of L1 gene of high-risk HPV, we could detect 13 types of HPV DNA(16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68) from the cervical exfoliative cells in one PCR procedure, but we were not able to distinguish each type.
-
1
The principle of setting the threshold:just above the maximum value of the negative amplification curve.
-
2
Quality control:
-
a
Negative control had no amplification.
-
b
The CT values of critical positive control must be in the range of 30 < CT ≤ 36.
-
c
The CT values of strong positive control must be in the range of 24 < CT ≤ 30.
-
a
-
3
Criteria:
In order to prevent misjudgment, we analysised the amplification curves of the samples that had CT values and defined those with non – “S” curves as negative.
Criteria according to the CT values are as follows:
-
A
CT < 37, reported as high-risk HPV positive.
-
B
No CT values or CT = 40, reported as high-risk HPV negative.
-
C
37 ≤ CT < 40, redo the test; if the re test CT < 40, reported as high-risk HPV positive, otherwise reported as high-risk HPV negative.
-
A
HPV-PCR (Dragon biotechnology) can selectively amplify the HPV DNA sequence in biological specimens. This method can detect the type of HPV DNA and has high sensitivity. Use of the assay in this study was performed as per manufacturers' protocol.
Pathology
The cytology and histopathology of samples was classified using the Bethesda System and the cervical intraepithelial neoplasia system. Samples classed as ASCUS, ASC-H, LSIL, HSIL, SC, AGC, AIS and AC were cytology positive. Those classed as CIN 1, CIN 2, CIN 3/CIS, micro-invasive carcinoma and infiltrating carcinoma were pathology positive.
All pathological diagnoses were verified by a pathologist from the Chinese Academy of Medical Sciences Cancer Institution (CAMSCI) and final diagnoses were obtained after discussion between the pathologists from our hospital and the CAMSCI.
Statistical methods
The final data were analyzed using SPSS version 15.0 statistical software, which calculated the concordance rate and the rate of consistency between results from careHPV and HC2 tests and between careHPV and HPV-PCR. Pathological diagnosis was used as the gold standard, against which calculation of sensitivity, specificity, positive predictive value and negative predictive value for the careHPV test to detected cervical lesions ≥ CIN 2 was compared. From these results, the accuracy and effectiveness of the careHPV test for cervical cancer screening could be evaluated.
References
Boyle, P. & Ferlay, J. Cancer incidence and mortality in Europe, 2004. Ann Oncol. 16, 481–8 (2005).
IARC. World Health Organization. IARC handbooks of cancer prevention: cervix cancer screening. Lyon, France,IARC. 10, 239–41 (2005).
Tsu, V. D. & Pollack, A. E. Preventing cervical cancer in low-resource settings: how far have we come and what does the future hold? Int J Gynaecol Obstet. 89 Suppl 2S55–9 (2005).
De Francesco, M. A. et al. Detection and genotyping of human papillomavirus in cervical samples from Italian patients. J Med Virol. 75, 588–92 (2005).
Cuzick, J. et al. Overview of human papillomavirus-based and other novel options for cervical cancer screening in developed and developing countries. Vaccine. 26 Suppl 10K29–41 (2008).
Cuzick, J. et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer. 119, 1095–101 (2006).
Gravitt, P. E. et al. New technologies in cervical cancer screening. Vaccine. 26 Suppl 10K42–52 (2008).
Denny, L. et al. Screen-and-treat approaches for cervical cancer prevention in low-resource settings: a randomized controlled trial. JAMA. 294, 2173–81 (2005).
Sherris, J. et al. Alliance for cervical cancer prevention: setting the record straight. Am J Public Health. 97, 200–1 (2007).
Hu, S. Y. et al. Prevalence of HPV Infection And Cervical Intraepithelial Neoplasia And Attitudes towards HPV Vaccination among Chinese Women Aged 18–25 in Jiangsu Province. Chin J Cancer Res. 23, 25–32 (2011).
Bulkmans, N. W. et al. Human papillomavirus DNA testing for the detection of cervical intraepithelial neoplasia grade 3 and cancer: 5-year follow-up of a randomised controlled implementation trial. Lancet. 370, 1764–72 (2007).
Qiao, Y. L. et al. A new HPV-DNA test for cervical-cancer screening in developing regions: a cross-sectional study of clinical accuracy in rural China. Lancet Oncol. 9, 929–36 (2008).
Levin, C. E. et al. Cost-effectiveness analysis of cervical cancer prevention based on a rapid human papillomavirus screening test in a high-risk region of China. Int J Cancer. 127, 1404–11 (2010).
Goldie, S. J. et al. Alliance for Cervical Cancer Prevention Cost Working Group. Cost-effectiveness of cervical-cancer screening in five developing countries. N Engl J Med. 353, 2158–68 (2005).
Hong, Y., Li, S. Q., Hu, Y. L. & Wang, Z. Q. Survey of human papillomavirus types and their vertical transmission in pregnant women. BMC Infect Dis. 27(13), 109 (2013).
Acknowledgements
We wish to thank all of our colleagues at the Nanjing Drum Tower Hospital of Obstetrics and Gynecology, especially the HPV testing laboratory. Thanks to Wang Hongyun for writing assistance. Thanks to Edanz group for editing and proofreading work.
Author information
Authors and Affiliations
Contributions
H.Y. participated in project design, performed specific operations, provided materials and analyzed data of table 1–3 and figures. H.Y. wrote the main manuscript text and the final thesis and is in charge of this project in the Nanjing Drum Tower Hospital. F.J. participated in the collection of data and prepared tables 1–3. Z.F. is in charge of the project and prepared figure 1. Q.Y. is the project director and prepared figures 1. H.Y.L. is involved in quality control of Nanjing Drum Tower Hospital test and project guidance. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. The images in this article are included in the article's Creative Commons license, unless indicated otherwise in the image credit; if the image is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the image. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Ying, H., Jing, F., Fanghui, Z. et al. High-risk HPV nucleic acid detection kit–the careHPV test –a new detection method for screening. Sci Rep 4, 4704 (2014). https://doi.org/10.1038/srep04704
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04704
This article is cited by
-
Development and validation of a multiplex qPCR assay for detection and relative quantification of HPV16 and HPV18 E6 and E7 oncogenes
Scientific Reports (2021)
-
Evaluating a community-based cervical cancer screening strategy in Western Kenya: a descriptive study
BMC Women's Health (2018)
-
Cost-effectiveness of cervical cancer screening and preventative cryotherapy at an HIV treatment clinic in Kenya
Cost Effectiveness and Resource Allocation (2017)
-
Cancer screening and prevention in low-resource settings
Nature Reviews Cancer (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.