Abstract
Advocacy for better drugs and access to treatment has boosted the interest in drug discovery and development for Chagas disease, a chronic infection caused by the genetically heterogeneous parasite, Trypanosoma cruzi. In this work new in vitro assays were used to gain a better understanding of the antitrypanosomal properties of the most advanced antichagasic lead and clinical compounds, the nitroheterocyclics benznidazole, nifurtimox and fexinidazole sulfone, the oxaborole AN4169 and four ergosterol biosynthesis inhibitors – posaconazole, ravuconazole, EPL-BS967 and EPL-BS1246. Two types of assays were developed: one for evaluation of potency and efficacy in dose-response against a panel of T. cruzi stocks representing all current discrete typing units (DTUs) and a time-kill assay. Although less potent, the nitroheterocyclics and the oxaborole showed broad efficacy against all T. cruzi tested and were rapidly trypanocidal, whilst ergosterol biosynthesis inhibitors showed variable activity that was both compound- and strain-specific and were unable to eradicate intracellular infection even after 7 days of continuous compound exposure at most efficacious concentrations. These findings contest previous reports of variable responses to nitroderivatives among different T. cruzi strains and further challenge the introduction of ergosterol biosynthesis inhibitors as new single chemotherapeutic agents for the treatment of Chagas disease.
Similar content being viewed by others
Introduction
Chagas disease or American trypanosomiasis is a neglected chronic tropical infectious disease endemic to Latin America. It is caused by the protozoan parasite Trypanosoma cruzi, found throughout the American continent in a variety of wild mammalian reservoirs and transmitted by the triatomine bug insect vector. Apart from vectorial transmission, humans can be infected by T. cruzi through ingestion of food and drinks contaminated with live parasites, from mother to child during pregnancy and through contaminated blood transfusion or organ transplantation. The WHO estimates that approximately 10 million people are infected with T. cruzi worldwide1, with the highest incidence in Latin America. In recent decades, massive migration of Latin Americans to developed countries has brought a significant number of infected individuals to non-endemic areas such as Europe, North America, Japan and Australia, where T. cruzi transmission can occur through the non-vectorial routes described2,3.
Symptomatic Chagas disease is a leading cause of morbidity and loss of productivity due to infectious disease in Latin America1,4. If not treated during the acute phase, Chagas disease develops into a chronic condition that can be either symptomatic or asymptomatic (also known as the indeterminate form), which is the most frequent clinical presentation. Symptomatic patients develop, usually decades after infection, either the cardiac form, characterized by progressive lesions in cardiac muscle, arrhythmias and heart failure, in up to 30% of patients, or the digestive form, characterized by the enlargement of the esophagus and/or the colon. Some patients may develop a combination of both cardiac and digestive forms5.
Current chemotherapy relies on antiparasitic treatment by either one of the only two registered drugs, benznidazole and nifurtimox. Both are oral nitroheterocyclic compounds that require prolonged treatment (usually 60 days) and are recognized as curative if administrated during the acute phase, whereas their usefulness in the chronic phase is still under investigation6. There is a consensus among the medical and scientific community that antiparasitic treatment is desirable and needed for Chagas disease7 and the ongoing BENEFIT clinical trial1,8,9 aims at understanding whether benznidazole can improve prognosis and clinical outcome in Chagas cardiomyopathic patients. Nonetheless, both benznidazole and nifurtimox cause severe side effects and, consequently, are not well-tolerated and are associated with poor patient compliance with treatment. Additionally, they are contraindicated in some instances, such as during pregnancy2,3,10,11,12,13,14. Therefore, as for other neglected diseases, new drugs with improved efficacy, tolerability and safety are urgently needed.
Recent efforts have advanced several novel chemical entities (NCEs) for chemotherapy of Chagas disease. The triazoles posaconazole and ravuconazole, which target the sterol 14alpha-demethylase enzyme (also known as CYP51), required for ergosterol biosynthesis, are used to treat systemic fungal infections and have been extensively studied against T. cruzi showing potent trypanocidal activity and efficacy both in vitro and in vivo1,4,15,16,17,18,19,20,21,22. These drugs are or have been recently in clinical trials in chronic Chagas disease patients5,23,24,25. Concomitantly, in recent years multiple initiatives have resulted in the launching of several drug discovery programs comprising a range of multidisciplinary activities to enable the discovery and advancement of NCEs for Chagas disease. Among the most advanced and promising compounds is another nitroheterocyclic, fexinidazole, which is currently undergoing clinical trials for human African trypanosomiasis caused by Trypanosoma brucei and is also active against T. cruzi6,26. Parallel efforts culminated in the discovery of the oxaborole class: AN4169 (SCYX-6759) is an oxaborole with curative antichagasic activity in a mouse model and is now an advanced lead for the treatment of Chagas disease7,27. Other recently developed optimized leads are EPL-BS967 (also known as UDD) and EPL-BS1246 (also known as UDO). Both are derivatives of the herbicide fenarimol and have recently been shown to be non-azole inhibitors of T. cruzi CYP5128,29,30. Despite these advances, other NCEs are still much needed due to the possibility of downstream failure of current leads, preclinical and clinical candidates.
Parallel to the ultimate goal of discovery and development of NCEs, there is also a need for a better-defined discovery process and screening sequence in which compounds can be prioritized based on previous supporting data. In this sense, secondary assays that provide data for compound prioritization are a desirable part of drug discovery programs as they may bring relevant biological information about anti-infective agents, such as compound efficacy and pharmacokinetic/pharmacodynamic (PK/PD) relationships, thus providing starting points for the design of in vivo therapy courses. The early identification of some biological characteristics such as lack of efficacy in some key models or unfavorable pharmacodynamics may help to more rapidly identify compounds likely to fail. This strategy would result in economic gains by avoiding expensive clinical trials. Such supporting assays, however, have not yet been established for T. cruzi.
Chagas disease programs also face an additional challenge: T. cruzi is a genetically heterogeneous group of organisms and current its phylogenetic classification comprises six discrete typing units (DTUs)31. Although members of all DTUs are capable of causing Chagas disease, the DTUs I, II, V and VI are more often found in humans, while DTUs III and IV are associated with sylvatic cycles and less often responsible for causing human infections32. Some degree of association between DTUs and different presentations of chronic disease has been previously observed33,34,35,36,37,38, however this association might originate from a geographical overlap between specific T. cruzi DTUs and human populations.
It is well established that T. cruzi strains display differential levels of natural susceptibility (or resistance) to benznidazole and nifurtimox both in vitro and in vivo39,40,41,42,43,44. However, more studies are needed for establishing to what extent the genetic variability of the parasite correlates to response to drugs and ultimately its impact on treatment outcome.
In order to understand whether current and future drugs and drug candidates are active against divergent T. cruzi stocks, we developed an in vitro assay for the evaluation and comparison of compound activity against T. cruzi strains and clones comprising all six DTUs. Three chemical classes with different mechanism of action (MoA) were tested: the oxaborole AN4169; the broad class of nitroherocyclic compounds: benznidazole, nifurtimox and fexinidazole sulfone, the main metabolite of fexinidazole, a new clinical candidate issued from the DNDi pipeline26; and two different scaffolds of CYP51 inhibitors, the triazoles posaconazole and ravuconazole, currently undergoing clinical trials for Chagas disease, together with two fenarimol derivatives issued from the DNDi lead optimization program: EPL-BS967 and EPL-BS124628,30. To aid in the choice of the dosing regimen in vivo, as well as in preliminary PK/PD determination, a time-kill assay was developed to determine the compound concentration and incubation time needed to achieve maximum efficacy in vitro.
Results
Activity assay development with different T. cruzi DTUs
Trypanosoma cruzi is a highly heterogeneous group of organisms and, ideally, new chemotherapy developed for Chagas disease should be active against all circulating genotypes of the parasite. With this aim at least one member belonging to each T. cruzi DTU was adapted to a high content screening assay for assessment of antitrypanosomal activity of the compounds in vitro. The stocks available in our laboratory as epimastigotes, the stage of T. cruzi that multiplies in the gut of triatomine vectors and which are not infective to mammalian cells, were differentiated in vitro to the infective metacyclic trypomastigote form in Grace's medium supplemented with 10% FBS as described in the Materials and Methods section. Metacyclics were used to infect LLC-MK2 cells to generate tissue culture-derived trypomastigotes that were kept cycling in vitro for 2 to 4 weeks prior to development of drug assays to improve the yield of trypomastigotes obtained in the supernatant of infected tissue cultures (data not shown).
The drug assay method consists of infecting the osteosarcoma-derived human cell line U2OS with tissue-derived trypomastigote forms of T. cruzi for 24 h prior to the addition of the compounds to 384-well plates. U2OS cells grow as a monolayer and present a large cytoplasm that allow for improved quantification of T. cruzi amastigotes in high content analysis46. Infected cultures were exposed to compounds for 4 days, except for Dm28c, which were exposed to compounds for 3 days. Plates were processed for high content imaging and normalized compound activity was plotted as dose-response curves as described in the Materials and Methods section.
Compound activity against T. cruzi from all DTUs
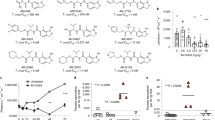
The results are shown in Fig. 1, Supplementary Fig. S1 and in Tables 1 and 2. Figure 1 shows the normalized activity based on infection ratios measurements, while Supplementary Fig. S1 shows curves fitted from raw data of an alternative measurement, the average parasite number per infected cell. The nitroheterocyclic compounds benznidazole, nifurtimox and fexinidazole sulfone, while less potent when compared to other compounds tested, are consistently efficacious against strains and clones from all selected DTUs, reaching more than 90% of maximum activity against all T. cruzi tested. Despite this uniform efficacy, the nitroheterocyclics showed different levels of potency against certain clones and strains and the differences in EC50 values between the most sensitive and most resistant strains were approximately 4-fold for benznidazole (Dm28c vs. Tulahuen) and 8-fold for nifurtimox (Dm28c vs. 92-80 cl2) and fexinidazole sulfone (Y vs. 92-80 cl2). The clone Dm28c and the Y, the two T. cruzi that are the most infective and proliferate faster in vitro among the strains and clones of this panel (data not shown), were less sensitive in terms of potency against the three nitroheterocyclic compounds, with EC50s in the low micromolar range. However these strains could be efficaciously inhibited by all three compounds, with activities reaching more than 98%. Conversely, the other strains and clones (ARMA13 cl1, 92-80 cl2, Tulahuen and CL Brener), which are less infective and grow slower than the Dm28c and Y stocks (data not shown), showed EC50s in the high nanomolar range, suggesting that fast growing strains require higher concentrations of nitroheterocyclic compounds to be efficaciously inhibited. A similar pattern was observed with the oxaborole AN4169, which was active against all the strains and clones in the high nanomolar range, with EC50s varying from 150 nM against the 92-80 cl2 to 440 nM against the Y strain (Table 1 and Fig. 1). Although it showed good efficacy against all the strains and clones, AN4169 was not as efficacious as the nitroheterocyclics.
The nitroheterocyclics benznidazole and nifurtimox are the most efficacious compounds against a phylogenetically broad panel of Trypanosoma cruzi.
Tissue cultures in 384-well plates were infected with one of the following T. cruzi: Dm28c (DTU I, purple), Y (DTU II, red), ARMA13 cl1 (DTU III, orange), ERA cl2 (DTU IV, light green), 92-80 cl2 (DTU V, light blue), CL Brener (DTU VI, dark blue) and Tulahuen (DTU VI, dark green). Compounds were 2-fold diluted and were added to infected cultures 24 h after T. cruzi plating and incubated for 72 (Dm28c) or 96 h (all others) prior to assessment of antiparasitic activity by high content analysis. Dose-response curves of compounds antiparasitic activity normalized to infected and non-infected controls are shown. The X-axis shows log of compound molar (M) concentrations and Y-axis shows the normalized activity, based on the measurement of ratio of infected cells. Data refers to mean values of at least two independent experiments.
A strikingly different pattern of T. cruzi response to drugs was observed with the other classes of compounds, the triazoles posaconazole and ravuconazole and the fenarimol derivatives EPL-BS967 and EPL-BS1246. These compounds all inhibit the ergosterol biosynthesis pathway by targeting the sterol 14 alpha-demethylase, also known as CYP5115,17,29,49. As expected these compounds had EC50s in the range of low nanomolar against some of the T. cruzi strains tested and the triazoles were in general more potent than fenarimols (Fig. 1 and Tables 1 and 2). Noticeably, the Tulahuen strain, representing the DTU VI, was on average the most susceptible to triazoles and fenarimols, with EC50s as low as 0.62 nM for ravuconazole and a maximum efficacy of approximately 90% for posaconazole, for example. The Dm28c (DTU I) and the Y strain (DTU II) were also partially susceptible to both triazoles and fenarimol derivatives, although with greater degrees of variability in potency, with EC50 values as high as approximately 266 nM for EPL-BS1246 against Dm28c and 11.3 nM for posaconazole against Y, compared to EC50s of 18.7 nM for EPL-BS1246 and 1 nM for posaconazole against the Tulahuen strain (Fig. 1 and Table 1). Triazoles and fenarimols derivatives also showed variable levels of efficacy against the Dm28c and Y strains and in most cases were lower than the maximum activity observed against the Tulahuen strain.
The remaining T. cruzi tested, ARMA13 cl1, ERA cl2, 92-80 cl2 and CL Brener, displayed a remarkable and unpredictable pattern of response to both triazoles and fenarimol derivatives: if, for the purpose of this study, resistance is defined as the inability to identify any concentration of compound capable of reducing infection level to a 50% ratio of infected cells – in other words, inability to obtain an EC50 value under the conditions assayed – then all the above mentioned clones were considered to be resistant to at least one of the compounds tested. ARMA13 cl1 (DTU III) was resistant to posaconazole (maximum activity of approximately 33.5%) and both fenarimol derivatives, but not to ravuconazole, which had an EC50 of approximately 0.87 nM against ARMA13 cl1 (similar to that observed for the susceptible strains Y and Tulahuen). However, as shown in Table 2, ravuconazole was less efficacious against ARMA13 cl1 (maximum activity of approximately 60%) compared to the Y and Tulahuen strains (at 79% and 93%, respectively). ERA cl2 (DTU IV) showed partial resistance to all four compounds, with maximum activity ranging from approximately 60% to 75%.
The T. cruzi member of DTU V, 92-80 cl2, showed the highest degree of resistance to posaconazole and ravuconazole among the strains and clones tested: the maximum activity observed was approximately 27% for posaconazole and only 22% for ravuconazole and consequently it was not possible to calculate an EC50 for these compounds against the 92-80 cl2 (Tables 1 and 2 and Fig. 1). However, the 92-80 cl2 was rather sensitive to EPL-BS967 and EPL-BS1246: the difference between triazoles and fenarimols activity was especially marked regarding maximum activity of compounds against 92-80 cl2 and CL Brener (another member of DTU VI), which were more consistent in their response to triazoles and fenarimol derivatives, but were resistant to either one of the two classes.
Importantly, the two CYP51 inhibitors demonstrated more variation in efficacy both across strains and clones and also against a given strain or clone. For instance, posaconazole efficacy is more likely to range from 55 to 100%, approximately, against the Y strain and ravuconazole was as much as 4 times more effective against Tulahuen than against 92-80 cl2 (Table 2). Conversely, fexinidazole sulfone was only 15% more efficacious against ARMA13 cl1 (the most sensitive in this case) than against 92-80 cl2 (least sensitive against fexinidazole sulfone), while AN4169 was only 18% more efficacious against Tulahuen than against Dm28c (Table 2). Indeed, if one considers the ratio of maximum efficacy to minimum efficacy against the different T. cruzi DTUs (Table 2, last column), one gets a clear distinction between the three different classes of compounds tested; the lowest variation being observed among the nitroheterocyclics (fold difference of 1.09 to 1.18), followed by AN4169, representative of the oxaboroles class (fold difference of 1.34) and eventually the CYP51 inhibitors that showed the greatest variation (fold difference between 2.14 and 4.26) but also the lowest efficacy. Altogether, these data suggest that greater efficacy is correlated with lower variability in antichagasic activity.
It can be argued that the poor activity of azoles and fenarimols against some of the stocks was due to pre-existing resistant organisms within the parasite population, as in several cases EPL-BS967, EPL-BS1246, posaconazole and ravuconazole were partially active against some of the stocks – see for instance maximum activity against CL Brener, ranging from approximately 47% to 57% (Fig. 1 and Table 2). Alternatively, it could be argued that amastigotes were not exposed to compounds over a sufficient duration of time to exhaust their entire ergosterol reservoirs. Indeed, triazoles and fenarimols target ergosterol biosynthesis and their activity is dependent on the depletion of the ergosterol stocks15,17.
Time-kill assays
To estimate for how long the amastigotes should be exposed to efficacious compound concentrations in order to reduce cellular infection to undetectable levels, a time-kill assay was developed using a phenotypic readout and in vitro infection conditions similar to the ones described above. The Y strain was chosen for this application as it grows fast and is highly infectious, therefore minimizing day-to-day variations and making the assay more robust. Additionally, the Y strain was one of the strains most sensitive to all compounds tested, providing the optimal combination of growth rate and sensitivity, thus enabling testing of whether prolonged exposure to azoles and fenarimols would be sufficient to reduce cellular infection by T. cruzi to undetectable levels.
Exposure of infected cells to a minimum concentration of 200 μM benznidazole, 50 μM nifurtimox, or 100 μM fexinidazole sulfone for at least 72 h could reduce the infection ratios to levels indistinguishable from those of uninfected controls (Fig. 2 and Table 3). Two-fold lower concentrations were also nearly 100% efficacious against the Y strain following 96-h exposure, in accordance with the maximum activities derived from fitted dose-response curves (Fig. 1 and Table 2). Lower concentrations were also trypanocidal, albeit less efficiently. These results suggest that the trypanocidal activity of these nitroheterocyclic compounds was primarily concentration-dependent, consistent with a shared mechanism of action, albeit efficacy was achieved at different concentrations.
Benznidazole, nifurtimox, fexinidazole sulfone and AN4169 are fast trypanocidal compounds that can eliminate intracellular T. cruzi within 96 h of continuous exposure in vitro.
U2OS cells tissue cultures in 384-well plates were infected with the Y strain for 24 hours prior to addition of compounds in serial dilution by the factor of 2-fold, as indicated the by colors according to the compound gradient (right, top). The following concentrations are shown: 400, 200, 100, 50, 25, 12.5, 6.25, 3.125, 1.56 and 0.78 μM for benznidazole and fexinidazole sulfone; 100, 50, 25, 12.5, 6.25, 3.125, 1.56, 0.78, 0.39 and 0.20 μM for nifurtimox; and 40, 20, 10, 5, 2.5, 1.25, 0.625, 0.31, 0.16 and 0.078 μM for AN4169. Antiparasitic activity was analyzed every 24 h, starting right after compound addition (time point 0 h), up to 96 h. Data points are means and standard deviations of at least two independent experiments.
Similarly to nitroheterocyclics, the oxaborole AN4169 showed a pronounced trypanocidal activity during the first 24 h of exposure with at least 2.5 μM of compound, reducing the infection ratio to approximately 10% of the levels observed in non-treated controls. Half of this concentration (1.25 μM) also produced a strong cidal effect. Subsequent exposure also produced reduction in the infection ratios, however at a slower pace than that observed during the first 24 h (Fig. 2 and Table 3). Curiously, sub-efficacious compound concentrations of 0.625 nM also produced cidal effects on T. cruzi, although slower and steadier than higher concentrations, but reached similar levels of activity to those observed with at least 2-fold higher concentrations at 96 h (Fig. 2). These results suggest that AN4169 has trypanocidal activity that may be both concentration- and time-dependent. However, even following exposure at high concentration for 96 hours, AN4169 did not completely eliminate T. cruzi from the cells and led to an infection ratio of between 4.0–4.5%. Whether a longer exposure would lead to further reduction of the residual infection remains to be determined.
Because it was clear that 96 h of exposure to fenarimols and azoles was not sufficient to achieve 100% in vitro efficacy against the Y strain, the time-kill experiments for these classes were carried out to 144 h of compound exposure. The highest concentrations tested (80 and 40 nM for azoles and 2 and 1 μM for fenarimols) are clearly trypanocidal, as was seen by a gradual reduction in the infection ratio for each 24-h period; however the infection was not completely reduced, with average residual infection levels ranging from 3.7% with ravuconazole and EPL-BS1246 up to 7% with posaconazole even after 144 h exposure to these compounds (Fig. 3 and Table 3). Considering that (i) the Y strain was relatively sensitive to all compounds when compared to other strains and clones tested in the assays here reported and (ii) assuming that exposure of T. cruzi amastigotes to compounds is optimal under the in vitro assay conditions, these results suggest that azoles and fenarimols are not able to efficaciously eliminate T. cruzi infection even after prolonged compound exposure in vitro. Although the fast killing compounds nitroheterocyclics and the oxaborole AN4169 also presented small variability in efficacy and were not capable of reducing infection to undetectable levels in all experiments (Table 3), the slow killing triazoles and fenarimols presented in general higher variability in activity levels and therefore were not as efficacious.
TcCYP51 inhibitors are slow trypanocidal compounds that after prolonged exposure can greatly reduce but not eliminate intracellular infection.
U2OS cells tissue cultures in 384-well plates were infected with the Y strain for 24 hours prior to addition of compounds in serial dilution by the factor of 2-fold and as indicated the by the colors according to compound gradient (right, top). The following concentrations are shown: 80, 40, 20, 10, 5, 2.5, 1.25, 0.625, 0.3125 and 0.156 nM for posaconazole and ravuconazole; and 2000, 1000, 500, 250, 125, 62.5, 31.25, 15.62, 7.81 and 3.91 nM for EPL-BS967 and EPL-BS1246. Antiparasitic activity was analyzed every 24 h, starting right after compound addition (time point 0 h), up to 144 h. Data points are means and standard deviations of three independent experiments.
Discussion
Innovation on processes and assays for successful drug discovery and development for neglected tropical infectious diseases is crucial. A translational platform composed of in vitro and in vivo preclinical assays that can predict with more confidence the efficacy of drug candidates in clinical trials, thus allowing early termination of compounds that do not meet certain criteria, is a real need in Chagas disease drug discovery programs. To achieve this aim, this study reports new assays using a high content screening technology to assist the drug discovery and development process. The first of these assays allowed for sensitive and unbiased detection of compound antiparasitic properties and the systematic comparison of compound activity against divergent T. cruzi clones and strains. The choice of T. cruzi integrating the panel was pragmatic and based on stocks available at the time the study started. While strains presumably reflect better the genetic diversity of a parasite population found in natural T. cruzi infections, competition between clones and clone selection is known to occur in polyclonal populations50,51, suggesting that clones, as more homogenous populations, are less prone to phenotypic changes such as variability in response to drugs. Therefore, a T. cruzi panel composed of both strains and clones is not biased towards one or another type of population and is not detrimental to the objectives of this study.
The assay utilizes a customized high content analysis algorithm that distinguishes intra and extracellular parasites, thus bypassing the need for laborious steps (such as washes) and consequently increasing the speed and throughput of the assay. Normalization of infection ratios (defined as the ratio between the number of infected cells to the number of total cells) against positive (noninfected) and negative (infected, not treated) controls within each plate allowed for quantitative comparison of EC50 values for compounds against the same or across different strains and clones of T. cruzi. We found this type of data analysis advantageous in computer-aided analysis because infection ratios are percentages and thus can be easily normalized to intraplates positive and negative controls48,52 and, in this study, data normalization was necessary to ensure proper comparison of efficacy against the different T. cruzi tested. This point is more easily illustrated by the dose-response curves from raw data of average parasite number per infected cells, as seen in Supplementary Fig. S1: activity measurements based on the average number of T. cruzi per infected cell correlates well with the measurements based on infection ratios (normalized activity, seen in Fig. 1). However, due to intrinsic differences on growth rates among the clones and strains, it is difficult to conclude, from the dataset shown in Supplementary Fig. S1, which compound(s) is(are) more efficacious and which strains and clones are more sensitive or resistant to the compounds.
A few studies have found a lack of correlation between T. cruzi DTUs and susceptibility to benznidazole43,53. To our knowledge this is the first study to compare concomitantly the activity of benznidazole and other clinically relevant compounds against members of all DTUs. Unfortunately, as our study was restricted to only one T. cruzi member of DTUs I – V, or two in the case of DTU VI, it is not possible to conclude whether or not DTUs are linked to susceptibility phenotypes. However, as a different susceptibility profile was observed for CYP51 inhibitors between CL Brener and the Tulahuen strain, both belonging to DTU VI, it is possible to speculate that indeed specific DTUs might not be directly correlated to a specific drug resistance pattern in vitro. Alternatively, the differences observed between these two T. cruzi might be attributed to the fact that one (CL Brener) is a clone and the other is a strain and is therefore possibly more heterogeneous.
Contrary to most published reports39,42,44, we did not find major differences in the susceptibility or resistance of strains and clones to nitroheterocyclics. Although there were differences in EC50 values – up to 8-fold – compounds consistently reached over 90% efficacy in all cases and therefore none of these strains or clones should be considered intrinsically resistant to nitrohetorocyclic compounds. Several reasons for the differences in susceptibility to nitroheterocyclics observed in this study and others reported in the literature can be speculated. First and most common are differences in assay methodology including, but not restricted to, the life cycle stage of the parasite used in the assay. For instance, variation in strain population composition among different labs – one cannot rule out that the differences in susceptibility to compounds observed in lab-adapted and widely available strains such as Y, CL Brener and Tulahuen are due to variations in the genotypes found among different labs. Additionally, some studies are performed with epimastigotes, while in this study tests were performed with amastigotes. To our knowledge, the issue of whether epimastigotes and amastigotes originating from the same T. cruzi stock differ in their susceptibility to drugs has not been thoroughly addressed; differences were reported40 in the activity of several drugs, including nifurtimox and benznidazole, against intracellular amastigotes growing in macrophages in vitro and axenic forms (which included amastigotes and/or epimastigotes) growing in Schneider's medium. Another and related issue is the comparison of in vitro assays and in vivo models: most reports are based on mouse models and it is not possible to affirm that therapeutic failure is due to “natural”, intrinsic T. cruzi resistance as in vivo chemotherapy involves several parameters – that very often are uncontrolled even in experimental models – such as the relationship between exposure and cure, route of administration, time of treatment, treatment regimen and length, mouse lineage and type of immune response, in addition to other Chagas disease related challenges such as definitions of cure criteria and disease stage. All of these factors might interfere with treatment outcome and therefore one should be cautious in attributing therapeutic failure to lack of T. cruzi susceptibility to the drug tested.
Variation in susceptibility to nitroheterocyclics can also be due to emergence of resistance within a population; indeed, it is well known that drug-resistant T. cruzi can be easily generated in the laboratory40,54,55 and a recent study proposed that T. cruzi genome plasticity underlies quick selection of phenotypes resistant to benznidazole56. However, the most important point may be the way resistance is defined: if one considers EC50s – defined as the compound concentration that reduces infection by 50% – as a parameter to measure resistance, our study confirms findings that there are differences in susceptibility to benznidazole, nifurtimox and fexinidazole sulfone (Table 1 and Fig. 1). However, if the efficacy of the compound, here defined as the maximum activity observed, is considered in the evaluation of resistance, then none of the T. cruzi stocks could be considered intrinsically resistant to nitroheterocyclics. We propose that in vitro compound efficacy should be determined in drug assays under controlled conditions and from normalized data and that it should be considered as the primary parameter for determining the level of susceptibility of a T. cruzi strain or clone to a compound, as efficacy is also a parameter that is analyzed in in vivo models assessed by incidence of cure; EC50s should be used as a secondary factor only for potency comparison of equally or similarly efficacious compounds.
The oxaborole AN4169, an advanced lead candidate for Chagas disease and recently shown to be curative in a mouse model27, also had broad spectrum activity against all the strains and clones tested. These data represent an important proof-of-principle for the potential of the oxaborole class as antichagasic drugs. It should however be emphasized that AN4169 was not as efficacious as the nitroheterocyclics in eliminating T. cruzi amastigotes from cells under the conditions tested. The target(s) and antichagasic mechanism of action (MoA) of these oxaboroles are currently unknown and should be determined, as it is desirable to develop drugs with new targets and MoA for Chagas disease treatment. Members of the oxaboroles, a new drug class, have been shown to selectively inhibit Rho-activated kinases in lymphocytes57 and are promising new antibacterials that inhibit leucyl-tRNA synthetase58,59, SCYX-7158 being currently in clinical trials for the treatment of human African trypanosomiasis60.
A different scenario emerged with the ergosterol biosynthesis inhibitors. Although all compounds have the same molecular target, TcCYP5117,29,61,62,63, they displayed variable activity across the panel, suggesting that the clones and strains tested may have CYP51s that differ in sequence and thus in compound affinity to target; alternatively, CYP51 copy number and/or expression might be higher in the resistant stocks. Indeed, Lepesheva and colleagues have shown that posaconazole may induce overexpression of CYP51 and this might constitute a possible resistance mechanism49.
It could be argued that suboptimal activity of triazoles and fenarimol derivatives was due to short compound exposure time as the trypanocidal activity of ergosterol biosynthesis inhibitors is time-dependent and result from the depletion of ergosterol pools in order to be effective17. We attempted to perform time-kill experiments with the triazole-resistant 92-80 cl2; however this clone grows very slowly, hampering the quantitative analysis of initial time points of time-kill experiments (data not shown). However the Y strain, used here in the time-kill experiments (Figs. 2 and 3 and Table 3), can complete its intracellular cycle within 4 days under the experimental conditions of the assay (data not shown). Thus considering that the trypanocidal activity of all CYP51 inhibitors tested against the Y strain could be detected after 48 h of exposure, it is likely that an exposure of 144 h (7 days) was sufficient time to observe compounds maximum activity in vitro. And even if longer exposures are considered – which could not be tested due to the experimental assay limitations – it should be questioned whether it is desirable to have an antichagasic agent that requires a longer time to achieve efficacy when compared to benznidazole, as this may lead to drug resistance, as discussed above. Indeed, our data does not support the choice of CYP51 inhibitors over nitroheterocyclics as antichagasic drugs. It seems rather that there is a correlation between activity against phylogenetically diverse T. cruzi strains and the time a compound needs to exert trypanocidal effects: fast killing compounds – all three nitroheterocyclics and AN4169 – were more efficacious than slow killing CYP51 inhibitors across the T. cruzi panel tested. Posaconazole and ravuconazole were the first NCEs to enter clinical trials for Chagas disease and the publication of results is highly anticipated; however preliminary data points to clinical failure and a performance inferior to benznidazole for both posaconazole and ravuconazole/E1224, probably due to lack of efficacy (I. Molina, personal communication; and DNDi/Eisai E1224 phase 2 trial – site presentation at the ASTMH Symposium November 14, 2013, Washington, DC). The results of our study might partly help to explain these preliminary clinical findings.
Overall, triazoles have been reported to be superior to benznidazole for antiparasitic treatment of T. cruzi-infected mice (as reviewed by64,65), although treatment outcome depends on many parameters such as treatment length and dose, disease stage, mouse and T. cruzi strain, amount of parasites and life stage of the parasite in the inoculum, route of infection, type of cure tests performed and other relevant parameters22,65. Accordingly, posaconazole, ravuconazole, benznidazole and nifurtimox performance as single chemotherapeutic agents in murine models of Chagas disease vary when different in vivo studies are compared and at current stages there is not enough data to support the choice of any of the available mouse models as the most predictive of the performance of drug candidates during clinical trials – or, for the purpose of this study, to correlate the results here reported with those observed in murine models. Ideally, when more clinical data is available and the determinants of antichagasic treatment failure or success are better understood, the knowledge should be applied to the development of “gold standard(s)” for in vivo models (a challenging and time-consuming work, given the current limitations in the field such as the lack of known biomarkers predictive of disease progress and lack of accurate and precise tests to determine parasitological cure of treated patients). Until then, we propose the use of in vitro tests against a panel of strains and time-kill experiments as a first assessment of compound efficacy against different T. cruzi and of intrinsic parasite resistance to compounds in development, as well as determination of compound speed of killing – apparently an important factor to observe.
Time-kill assays are often performed to assist in the design of in vivo therapeutic protocols by determining the pharmacodynamics of compound concentration versus the total time of exposure needed to achieve efficacy and thereby establish in vitro pharmacokinetic/pharmacodynamic correlation with efficacy45,66. Additionally it offers information on the compound mechanism(s) of action, regarding whether the compound activity is concentration- or time-dependent. Our data suggest that benznidazole, as well as nifurtimox and fexinidazole sulfone, are concentration-dependent trypanocidal drugs and therefore more efficacious at higher doses (above 100 μM, or 26 μg/mL, in the case of benznidazole). A recent study has determined the serum concentration of benznidazole in patients with chronic Chagas disease treated with 5 mg/kg/day for 60 days to range from 5 to 6.5 μg/mL on average67. This range is equivalent to 20–25 μM benznidazole, which was a suboptimal concentration (according to time-kill data in this study) against the Y strain for up to 96 h. These results highlight the urgent need for a deeper understanding of how and when benznidazole and other compounds are curative, because only then this knowledge can be applied to the development of in vitro and in vivo assays that can accurately predict clinical performance of new drug candidates.
Altogether these results demonstrate that nitroderivatives are, among the current antichagasic drugs and drug candidates, the compounds with most breadth of activity across multiple divergent T. cruzi genotypes, as well as the fastest T. cruzi killing compounds available, both desirable characteristics if the potential for the development of resistance of T. cruzi to such a drug is considered. The oxaborole AN4169 follows as a candidate with the potential for both broad-spectrum activity and favorable trypanocidal kinetics.
To our knowledge, this is the first study to systematically compare the activity of current drugs and drug candidates against intracellular T. cruzi amastigotes belonging to all current phylogenetic subdivisions and to address exposure duration and compound rates of killing as important characteristics in assessing trypanocidal activity. The portfolio of preclinical assays needs to be further expanded and comparison of in vitro assays, animal models and clinical data must be performed in order to ensure that the research community is moving in the right direction. In this process it is paramount to adopt standards that will enable such comparisons and aid in the development of improved therapy for Chagas disease patients.
Methods
Chemicals
All culture media were purchased from Welgene, Inc. (Daegu, South Korea). Fetal bovine serum (FBS) and penicillin/streptomycin were purchased from Gibco, Inc. (Life Technologies, Carlsbad, CA, USA). DRAQ5™ was purchased from BioStatus, Ltd. (Shepshed, UK). Benznidazole, nifurtimox, EPL-BS967 and EPL-BS1246 were synthesized by Epichem Pty, Ltd. (Murdoch, WA, Australia), posaconazole was extracted from Noxafil and provided by Epichem Pty, Ltd.; fexinidazole sulfone derivative [1-methyl-2-(4-methylsulfonyl phenoxymethyl)-5-nitro imidazole] was prepared by (Axyntis/Centipharm, France) and was provided by the Drugs for Neglected Diseases initiative (DNDi). AN4169 (SCYX-6759) originated from the DNDi Lead optimization program for sleeping sickness45. Ravuconazole (E1224 Raw Material EPS-140) was obtained from Eisai, Ltd. (Tokyo, Japan).
In vitro culture of mammalian cells
The human osteosarcoma cell line U2OS and the Macaca mulatta kidney epithelial cell LLC-MK2 were previously available at our laboratory46. All mammalian cells were cultured in DMEM high-glucose medium supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin and 100 μg/mL streptomycin in a humid atmosphere of 5% CO2 at 37°C. LLC-MK2 tissue cultures were used to support the mammalian cycle of T. cruzi in vitro.
In vitro culture of T. cruzi
Different T. cruzi parasite clones and strains representing each of the six discrete typing units (DTUs) were used in this study (Supplementary Table S1 and Table 1). The DTU classification was based on the current consensus T. cruzi nomenclature31. Clone Dm28c and the Y strain were donated by A. Avila (Instituto Carlos Chagas, Fiocruz, Curitiba, Brazil). CL Brener is a clone obtained from the CL strain and was provided by J. F. Silveira (Universidade Federal de São Paulo, UNIFESP, São Paulo, Brazil). Tulahuen strain was donated by F. Buckner (University of Washington, Seattle, WA, USA) and the clone ARMA13 cl1 was donated by M. Miles (London School of Hygiene and Tropical Medicine, LSHTM, London, UK). The clones 92-80 cl2 and ERA cl2 were also provided by M. Miles, however were originally isolated and prepared by the groups of Michel Tibayrenc (Institut de Recherche pour le Développement, Montpellier, France) and Hernan Carrasco (Universidad Central de Venezuela, Caracas, Venezuela). Axenic cultures of epimastigotes from these different strains were maintained at 28°C in LIT medium containing 10% FBS and 0.2% glucose. Epimastigotes at exponential growth phase were harvested, washed in PBS and resuspended at 3 × 107 trypanosomes/ml in Grace's Insect Medium supplemented with 10% FBS47. Differentiation of epimastigotes to metacyclic trypomastigotes was monitored daily from 5 to 14 days, depending on the strain. When differentiation rate reached a plateau (typically at least 50% of the population would be composed of metacyclic trypomastigotes), parasites were harvested, washed in PBS and transferred to mammalian tissue cultures to infect LLC-MK2 cells in DMEM High Glucose medium supplemented with 10% FBS, 100 U/mL penicillin and 100 μg/mL streptomycin for all strains and clones except for Dm28c, which was cultured in DMEM Low Glucose medium supplemented with 2% FBS, 100 U/mL penicillin and 100 μg/mL streptomycin (for simplicity, both compositions are going to be designated as complete media). Tissue cultures were washed with complete media 48 h after initial infection to remove extracellular metacyclic trypomastigotes and epimastigotes, followed by daily replacement of supernatant by fresh complete media. Typically tissue culture-derived trypomastigotes could be harvested from the supernatants after 5–14 days, depending on the strain or clone. The trypomastigotes were used to infect fresh tissue cultures and maintained in vitro up to 10 infection cycles, at which point cultures were discarded and restarted from a frozen trypomastigote stock.
Compound preparation
Standardized powders were diluted either to 8 mM (AN4169, ravuconazole, posaconazole), 10 mM (EPL-BS967 and EPL-BS1246), or 40 mM (benznidazole, nifurtimox and fexinidazole sulfone) in 100% DMSO and stocked at −80°C. Compound solutions at 100× the final assay plate concentration were two-fold serially diluted in 100% DMSO, transferred to 6% DMSO intermediate plates and finally plated in assay plates of 10 μL/well to yield 1% DMSO, a final concentration that had no apparent effect on parasite growth and viability. Each compound was tested in 14 concentration-points against the panel of T. cruzi strains and clones and 10 points in time-kill assays (see below). Compound stocks were freshly diluted before each experiment and each tested concentration was assayed in three wells per plate. Experiments were performed at least in duplicate.
Antiparasitic activity assays
U2OS cells at 1.75–2.5 × 104 cells/mL of complete media were seeded in 384-well, black, flat-bottomed plates, at 40 μL/well and infected 24 h later with 10 μL of supernatant of T. cruzi trypomastigote cultures at a ratio of 4 (Dm28c and Y), 15 (Tulahuen), 20 (ERA cl2, 92-80 cl2 and CL Brener), or 40 (ARMA 13 cl1) trypomastigotes to 1 U2OS cell seeded 24 h earlier. The trypomastigote to mammalian cell ratios used were determined empirically and were the minimum ratio necessary to achieve optimal infection levels statistically distinguishable from background. After addition of trypomastigotes to tissue culture plates, the final trypomastigote density in wells ranged from 5.6 × 104–8 × 104 trypomastigotes/mL (Y and Dm28c), 2.1–3 × 105 trypomastigotes/mL (Tulahuen), 2.8–4 × 105 trypomastigotes/mL (ERA cl2, 92-80 cl2 and CL Brener) and 5.6–8 × 105 trypomastigotes/mL (ARMA 13 cl1). After 24 h of infection/48 h after first plating cells, 10 μL of compound in DMSO solution were added and plates were incubated at 5% CO2 and 37°C for additional time (72 h for the Dm28c and 96 h for all the other strains or clones). Final DMSO concentration was 1% in all wells. All the plates included multiple wells of infection in the absence of any compound as negative controls and non-infected wells (blank containing only U2OS cells) as positive controls. At the end of compound exposure periods, plates were fixed with 4% paraformaldehyde for 15–30 minutes, followed by an automated wash step with PBS using a Biotek EL402 washer (Biotek, Seoul, South Korea) and DNA staining with DRAQ5™ for microscope imaging. Images were acquired in Operetta® Confocal Microscope (Perkin Elmer, USA) using a 20× objective in the non-confocal mode. Experiments were performed in at least duplicate (i.e., two independent tests performed on different days).
Time-kill assay
U2OS cells were seeded per well in 40 μl complete media at 700 cells/well into 384-well, black, flat-bottomed plates (Greiner Bio-One). At day 2, 2.8 × 103 Y strain trypomastigotes were added per well (ratio of 4 parasites/cell) for a total volume of 50 μL/well, followed by compound plating 24 h later as described above. Final volume in the well was 60 μL. Several replicate plates were prepared in each experiment and at each time point (starting right after the plating of compounds and repeated every 24 h, i.e., cultures were exposed to compound for 0, 24, 48, 72, 96,120, or 144 h); one plate was fixed, stained and imaged as described above. Experiments were performed at least in duplicates.
Data analysis
All the images acquired in the activity assays were analyzed by the Institut Pasteur Korea's proprietary software for high content analysis that had a built-in T. cruzi infection analysis algorithm48, which is able to accurately detect host cell boundary as well as host cell nucleus and T. cruzi nuclear and kinetoplast DNA (both appear as a contiguous structure at 20× magnification). These parameters are quantified to determine, for each well, the total number of cells, the number of infected cells, the ratio of infected cells and the average number of parasites per infected cell. Importantly, only intracellular parasites are scored in the analysis. The values for the ratio of infected cells, or infection ratio, are normalized to the average ratio of infected cells from all negative (1% DMSO) and positive controls (non-infected cells) wells from the same plate to obtain the normalized activity, following the equation WellNA = 100 × [1 − (IRNeg − IRWell)/(IRNeg − IRPos)], where WellNA is the normalized activity for a well, expressed in %; IRNeg is the average infection ratio of negative control wells; and IRPos is the average infection ratio of the positive control wells. As mentioned above, each tested compound concentration was assayed in three wells per plate and the average value was calculated from the normalized data for each concentration-point well to result in the normalized activity for that compound concentration in one particular replicate.
Dose-response curves were fitted and 95% confidence intervals were calculated using the sigmoidal dose-response – variable slope function from the GraphPad Prism version 6.00 for Windows, GraphPad Software, La Jolla California USA, www.graphpad.com. EC50s and maximum activity values (see below) were determined from fitted curves from normalized activity datasets.
Definitions
For the purposes of this study, i) EC50 is defined as the compound concentration capable of reducing the infection in 50% (i.e., inhibiting growth by 50%) as compared to non-treated infected controls and were determined by interpolation from the fitted dose-response curve; ii) potency is defined as the compound ability to reduce infection at a given compound concentration, usually considering the concentration that corresponds to the EC50; and iii) efficacy is defined as the maximum activity (i.e., reduction in infection) observed for a compound regardless of the compound concentration.
References
WHO. Working to overcome the global impact of neglected tropical diseases. Available from http://www.who.int/neglected_diseases/2010report/en/ (2010) (Accessed on 18/02/2014).
Gascon, J., Bern, C. & Pinazo, M.-J. Chagas disease in Spain, the United States and other non-endemic countries. Acta Trop. 115, 22–27 (2010).
Bern, C. & Montgomery, S. P. An estimate of the burden of Chagas disease in the United States. Clin. Infec. Dis. 49, e52–4 (2009).
Lee, B. Y., Bacon, K. M., Bottazzi, M. E. & Hotez, P. J. Global economic burden of Chagas disease: a computational simulation model. Lancet Infect. Dis. 13, 342–348 (2013).
Rassi, A. J., Rassi, A. & Marcondes de Rezende, J. American trypanosomiasis (Chagas disease). Infect. Dis. Clin. North Am. 26, 275–291 (2012).
Coura, J. R. & de Castro, S. L. A critical review on Chagas disease chemotherapy. Mem. Inst. Oswaldo Cruz 97, 3–24 (2002).
Coura, J. R. & Borges-Pereira, J. Chronic phase of Chagas disease: why should it be treated? A comprehensive review. Mem. Inst. Oswaldo Cruz 106, 641–645 (2011).
Marin-Neto, J. A. et al. Rationale and design of a randomized placebo-controlled trial assessing the effects of etiologic treatment in Chagas' cardiomyopathy: the BENznidazole Evaluation For Interrupting Trypanosomiasis (BENEFIT). Am. Heart J. 156, 37–43 (2008).
Marin-Neto, J. A. et al. The BENEFIT trial: testing the hypothesis that trypanocidal therapy is beneficial for patients with chronic Chagas heart disease. Mem. Inst. Oswaldo Cruz 104 Suppl 1, 319–324 (2009).
Castro, J. A., de Mecca, M. M. & Bartel, L. C. Toxic side effects of drugs used to treat Chagas' disease (American trypanosomiasis). Hum. Exp. Toxicol. 25, 471–479 (2006).
Viotti, R. et al. Side effects of benznidazole as treatment in chronic Chagas disease: fears and realities. Expert Rev. Anti Infect. Ther. 7, 157–163 (2009).
Yun, O. et al. Feasibility, drug safety and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala and Bolivia: 10-year experience of Medecins Sans Frontieres. PLoS Negl. Trop. Dis. 3, e488 (2009).
Sosa Estani, S. et al. Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas' disease. Am. J. Trop. Med. Hyg. 59, 526–529 (1998).
Sosa-Estani, S., Colantonio, L. & Segura, E. L. Therapy of Chagas disease: implications for levels of prevention. J. Trop. Med. 2012, (2012).
Urbina, J. A. et al. Antiproliferative effects and mechanism of action of SCH 56592 against Trypanosoma (Schizotrypanum) cruzi: in vitro and in vivo studies. Antimicrob. Agents Chemother. 42, 1771–1777 (1998).
Molina, J. et al. Activities of the triazole derivative SCH 56592 (posaconazole) against drug-resistant strains of the protozoan parasite Trypanosoma (Schizotrypanum) cruzi in immunocompetent and immunosuppressed murine hosts. Antimicrob. Agents Chemother. 44, 150–155 (2000).
Urbina, J. A., Payares, G., Sanoja, C., Lira, R. & Romanha, A. J. In vitro and in vivo activities of ravuconazole on Trypanosoma cruzi, the causative agent of Chagas disease. Int. J. Antimicrob. Agents 21, 27–38 (2003).
Ferraz, M. L., Gazzinelli, R. T., Alves, R. O., Urbina, J. A. & Romanha, A. J. The Anti-Trypanosoma cruzi activity of posaconazole in a murine model of acute Chagas' disease is less dependent on gamma interferon than that of benznidazole. Antimicrob. Agents Chemother. 51, 1359–1364 (2007).
Olivieri, B. P. et al. A comparative study of posaconazole and benznidazole in the prevention of heart damage and promotion of trypanocidal immune response in a murine model of Chagas disease. Int. J. Antimicrob. Agents 36, 79–83 (2010).
Diniz, L. et al. Effects of ravuconazole treatment on parasite load and immune response in dogs experimentally infected with Trypanosoma cruzi. Antimicrob. Agents. Chemother. 54, 2979–2986 (2010).
Veiga-Santos, P. et al. Effects of amiodarone and posaconazole on the growth and ultrastructure of Trypanosoma cruzi. Int. J. Antimicrob. Agents 40, 61–71 (2012).
Buckner, F. S. Experimental chemotherapy and approaches to drug discovery for Trypanosoma cruzi infection. Adv. Parasitol. 75, 89 (2011).
Merck. STOPCHAGAS: a Study of the Use of Oral Posaconazole (POS) in the Treatment of Asymptomatic Chronic Chagas Disease. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2011 May 13 (cited 2013 December 07). Available from <http://clinicaltrials.gov/ct2/show/NCT01377480?term=A+Study+of+the+Use+of+Oral+Posaconazole+%28POS%29+in+the+Treatment+of+Asymptomatic+Chronic+Chagas+Disease&rank=1> Identifier NCT 01377480.
Drugs for Neglected Diseases initiative. Proof of Concept Study of E1224 to Treat Adults Patients with Chagas Disease. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2011 November 24 (cited 2013 December 07). Available from <http://clinicaltrials.gov/ct2/show/NCT01489228?term=chagas+disease&rank=9> Identifier NCT 01489228.
Hospital Universitari Vall d'Hebron Research Institute. Clinical Trial For The Treatment Of Chronic Chagas Disease With Posaconazole And Benznidazole (CHAGASAZOL). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2011 November 24 (cited 2013 December 07). Available from <http://clinicaltrials.gov/ct2/show/study/NCT01162967?term=chagasazol&rank=1> Identifier NCT 01162967.
Bahia, M. T. et al. Fexinidazole: A potential new drug candidate for Chagas disease. PLoS Negl. Trop. Dis. 6, e1870 (2012).
Bustamante, J. M., Craft, J. M., Crowe, B. D., Ketchie, S. A. & Tarleton, R. L. New, combined and reduced dosing treatment protocols cure Trypanosoma cruzi infection in mice. J. Infect. Dis. 10.1093/infdis/jit420 (2013).
Keenan, M. et al. Analogues of fenarimol are potent inhibitors of Trypanosoma cruzi and are efficacious in a murine model of Chagas disease. J. Med. Chem. 55, 4189–4204 (2012).
Hargrove, T. Y. et al. Complexes of Trypanosoma cruzi sterol 14alpha-demethylase (CYP51) with two pyridine-based drug candidates for Chagas disease: structural basis for pathogen-selectivity. J. Biol. Chem. 10.1074/jbc.M113.497990 (2013).
Keenan, M. et al. Design, structure-activity relationship and in vivo efficacy of piperazine analogues of fenarimol as inhibitors of Trypanosoma cruzi. Bioorg. Med. Chem. 21, 1756–1763 (2013).
Zingales, B. et al. A new consensus for Trypanosoma cruzi intraspecific nomenclature: second revision meeting recommends TcI to TcVI. Mem. Inst. Oswaldo Cruz 104, 1051–1054 (2009).
Miles, M. A. et al. The molecular epidemiology and phylogeography of Trypanosoma cruzi and parallel research on Leishmania: looking back and to the future. Parasitology 136, 1509 (2009).
Marcili, A. et al. Comparative phylogeography of Trypanosoma cruzi TCIIc: New hosts, association with terrestrial ecotopes and spatial clustering. Infect. Genet. Evol. 9, 1265–1274 (2009).
Llewellyn, M. S. et al. Genome-scale multilocus microsatellite typing of Trypanosoma cruzi discrete typing unit I reveals phylogeographic structure and specific genotypes linked to human infection. PLoS Pathog. 5, e1000410 (2009).
Lewis, M. D. et al. Genotyping of Trypanosoma cruzi: systematic selection of assays allowing rapid and accurate discrimination of all known lineages. Am. J. Trop. Med. Hyg. 81, 1041–1049 (2009).
Carrasco, H. J. et al. Geographical distribution of Trypanosoma cruzi genotypes in Venezuela. PLoS Negl. Trop. Dis. 6, e1707 EP– (2012).
Burgos, J. M. et al. Molecular identification of Trypanosoma cruzi discrete typing units in end-stage chronic Chagas heart disease and reactivation after heart transplantation. Clin. Infect. Dis. 51, 485–495 (2010).
Cura, C. I. et al. Trypanosoma cruzi discrete typing units in Chagas disease patients from endemic and non-endemic regions of Argentina. Parasitology 139, 516–521 (2012).
Filardi, L. S. & Brener, Z. Susceptibility and natural resistance of Trypanosoma cruzi strains to drugs used clinically in Chagas disease. Trans. R. Soc. Trop. Med. Hyg. 81, 755–759 (1987).
Neal, R. A. & van Bueren, J. Comparative studies of drug susceptibility of five strains of Trypanosoma cruziin vivo and in vitro. Trans. R. Soc. Trop. Med. Hyg. 82, 709–714 (1988).
Veloso, V. M. et al. Variation in susceptibility to benznidazole in isolates derived from Trypanosoma cruzi parental strains. Mem. Inst. Oswaldo Cruz 96, 1005–1011 (2001).
Camandaroba, E. L. P., Reis, E. A. G., Gonçalves, M. S., Reis, M. G. & Andrade, S. G. Trypanosoma cruzi: susceptibility to chemotherapy with benznidazole of clones isolated from the highly resistant Colombian strain. Rev. Soc. Bras. Med. Trop. 36, 201–209 (2003).
Moreno, M. et al. Trypanosoma cruzi benznidazole susceptibility in vitro does not predict the therapeutic outcome of human Chagas disease. Mem. Inst. Oswaldo Cruz 105, 918–924 (2010).
Teston, A. P. et al. In vivo susceptibility to benznidazole of Trypanosoma cruzi strains from the western Brazilian Amazon. Trop. Med. Int. Health 18, 85–95 (2013).
Nare, B. et al. Discovery of novel orally bioavailable oxaborole 6-carboxamides that demonstrate cure in a murine model of late-stage central nervous system African trypanosomiasis. Antimicrob. Agents Chemother. 54, 4379–4388 (2010).
Genovesio, A. et al. Visual genome-wide RNAi screening to identify human host factors required for Trypanosoma cruzi infection. PLoS ONE 6, e19733 (2011).
Heise, N., Raper, J., Buxbaum, L. U., Peranovich, T. M. & de Almeida, M. L. Identification of complete precursors for the glycosylphosphatidylinositol protein anchors of Trypanosoma cruzi. J. Biol. Chem. 271, 16877–16887 (1996).
Moon, S. et al. An image-based algorithm for precise and accurate high throughput assessment of drug activity against the human parasite Trypanosoma cruzi. PLoS ONE 9, e87188 (2014).
Lepesheva, G. I. et al. Structural insights into inhibition of sterol 14alpha-demethylase in the human pathogen Trypanosoma cruzi. J. Biol. Chem. 285, 25582–25590 (2010).
Dvorak, J. A. The natural heterogeneity of Trypanosoma cruzi: biological and medical implications. J. Cell. Biochem. 24, 357–371 (1984).
Engel, J. C., Doyle, P. S. & Dvorak, J. A. Trypanosoma cruzi: biological characterization of clones derived from chronic chagasic patients. II. Quantitative analysis of the intracellular cycle. J Protozool 32, 80–83 (1985).
Siqueira-Neto, J. L. et al. An image-based high-content screening assay for compounds targeting intracellular Leishmania donovani amastigotes in human macrophages. PLoS Negl. Trop. Dis. 6, e1671 (2012).
Villarreal, D., Barnabé, C., Sereno, D. & Tibayrenc, M. Lack of correlation between in vitro susceptibility to benznidazole and phylogenetic diversity of Trypanosoma cruzi, the agent of Chagas disease. Exp. Parasitol. 108, 24–31 (2004).
Buckner, F. S., Wilson, A. J., White, T. C. & Van Voorhis, W. C. Induction of resistance to azole drugs in Trypanosoma cruzi. Antimicrob. Agents. Chemother. 42, 3245–3250 (1998).
Wilkinson, S. R., Taylor, M. C., Horn, D., Kelly, J. M. & Cheeseman, I. A mechanism for cross-resistance to nifurtimox and benznidazole in trypanosomes. Proc. Natl. Acad. Sci. U.S.A. 105, 5022–5027 (2008).
Mejia, A. M. et al. Benznidazole-resistance in Trypanosoma cruzi is a readily acquired trait that can arise independently in a single population. J. Infect. Dis. 206, 220–228 (2012).
Akama, T. et al. Linking phenotype to kinase: Identification of a novel benzoxaborole hinge binding motif for kinase inhibition and the development of high potency rho-kinase inhibitors. J. Pharmacol. Exp. Ther. 10.1124/jpet.113.207662 (2013).
Shapiro, A. B., Gao, N., Hajec, L. & McKinney, D. C. Time-dependent, reversible, oxaborole inhibition of Escherichia coli leucyl-tRNA synthetase measured with a continuous fluorescence assay. Anal. Biochem. 431, 48–53 (2012).
Hu, Q.-H. et al. Discovery of a potent benzoxaborole-based anti-pneumococcal agent targeting leucyl-tRNA synthetase. Sci. Rep. 3, 2475 (2013).
Drugs for Neglected Diseases initiative. Human African Trypanosomiasis: First in Man Clinical Trial of a New Medicinal Product, the SCYX-7158. In: ClinicalTrialsgov [Internet] Bethesda (MD): National Library of Medicine (US). 2012 February 13 (cited 2013 December 07). Available from <http://clinicaltrials.gov/ct2/show/NCT01533961?term=NCT01533961&rank=1> Identifier NCT01533961.
Buckner, F. S. & Urbina, J. A. Recent developments in sterol 14-demethylase inhibitors for Chagas disease. Int. J. Parasitol. Drugs Drug Resist. 2, 236–242 (2012).
Lepesheva, G. I., Villalta, F. & Waterman, M. R. Targeting Trypanosoma cruzi sterol 14α-demethylase (CYP51). Adv. Parasitol. 75, 65–87 (2011).
Chen, C.-K. et al. Structural characterization of CYP51 from Trypanosoma cruzi and Trypanosoma brucei bound to the antifungal drugs posaconazole and fluconazole. PLoS Negl. Trop. Dis. 4, e651 (2010).
Urbina, J. A. Specific chemotherapy of Chagas disease: relevance, current limitations and new approaches. Acta Trop. 115, 55–68 (2010).
Buckner, F. S. Sterol 14-demethylase inhibitors for Trypanosoma cruzi infections. Adv. Exp. Med. Biol. 625, 61–80 (2008).
Jacobs, R. T. et al. SCYX-7158, an orally-active benzoxaborole for the treatment of stage 2 human African trypanosomiasis. PLoS Negl. Trop. Dis. 5, e1151 (2011).
Pinazo, M.-J. et al. Benznidazole-related adverse drug reactions and their relationship in patients with chronic Chagas disease. Antimicrob. Agents Chemother. 57, 390–395 (2013).
Acknowledgements
The authors would like to thank Prof. M. Miles, Prof. F. Buckner, Prof. J. F. da Silveira and Dr. A. Ávila for providing the parasite stocks; M. Park, E. Kim, Dr. J. L. Siqueira-Neto, Dr. M. Lewis and L. M. Alcântara for technical support; Dr. F. Duncanson, Dr. M. Everson and Dr. S. Wells for critically reviewing this manuscript. The authors would like also to thank Eisai, Ltd., for providing ravuconazole, Epichem Pty, Ltd. for providing nifurtimox, AN4169, posaconazole, EPL-BS967 and EPL-BS1246 and Anacor Pharmaceuticals for a collaborative partnership which helped to identify the oxaborole AN4169 and analogues as potent lead candidates for treatment of Chagas Disease. This study was funded by DNDi and the Institut Pasteur Korea (IPK). For the work described in this paper, DNDi received financial support from the Reconstruction Credit Institution-Federal Ministry of Education and Research (KfW-BMBF)/Germany and Médecins Sans Frontières (Doctors without Borders)/International; IPK received a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIP)(No. 2007-00559), Gyeonggi-do and KISTI. The donors had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
C.B.M., M.A.G., S.S., E.C. and L.H.F.J. conceived and designed the experiments; C.B.M., M.A.G., H.K. and C.H.F. performed experiments and data analysis; C.B.M. and L.H.F.J. coordinated and supervised the work; C.B.M., M.A.G., E.C. and L.H.F.J. wrote the main manuscript text; C.B.M., C.H.F. and A.M.A.J. prepared manuscript figures and tables; all authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Table S1 and Figure S1
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. The images in this article are included in the article's Creative Commons license, unless indicated otherwise in the image credit; if the image is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the image. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Moraes, C., Giardini, M., Kim, H. et al. Nitroheterocyclic compounds are more efficacious than CYP51 inhibitors against Trypanosoma cruzi: implications for Chagas disease drug discovery and development. Sci Rep 4, 4703 (2014). https://doi.org/10.1038/srep04703
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04703
This article is cited by
-
Listen to what the animals say: a systematic review and meta-analysis of sterol 14-demethylase inhibitor efficacy for in vivo models of Trypanosoma cruzi infection
Parasitology Research (2024)
-
Anti-Trypanosoma cruzi activity of Coptis rhizome extract and its constituents
Tropical Medicine and Health (2023)
-
Efficacy and safety assessment of different dosage of benznidazol for the treatment of Chagas disease in chronic phase in adults (MULTIBENZ study): study protocol for a multicenter randomized Phase II superiority clinical trial
Trials (2020)
-
Systemic and Target-Site Pharmacokinetics of Antiparasitic Agents
Clinical Pharmacokinetics (2020)
-
Cell death pathways in pathogenic trypanosomatids: lessons of (over)kill
Cell Death & Disease (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.