Abstract
Prevention of quality of life (QOL) deterioration is associated with the inhibition of geriatric diseases and the regulation of brain function. However, no substance is known that prevents the aging of both body and brain. It is known that polyamine concentrations in somatic tissues (including the brain) decrease with increasing age and polyamine-rich foods enhance longevity in yeast, worms, flies and mice and protect flies from age-induced memory impairment. A main source of exogenous polyamines is the intestinal lumen, where they are produced by intestinal bacteria. We found that arginine intake increased the concentration of putrescine in the colon and increased levels of spermidine and spermine in the blood. Mice orally administered with arginine in combination with the probiotic bifidobacteria LKM512 long-term showed suppressed inflammation, improved longevity and protection from age-induced memory impairment. This study shows that intake of arginine and LKM512 may prevent aging-dependent declines in QOL via the upregulation of polyamines.
Similar content being viewed by others
Introduction
Polyamines (PAs) such as putrescine (PUT), spermidine (SPD) and spermine (SPM) are present in the cells of all mammalian species. PAs have various bioactivities, such as the synthesis and stabilization of DNA, RNA and protein and stimulation of cell proliferation or differentiation1,2. PAs also have strong anti-inflammatory functions3,4; they are, therefore, useful for inhibition of chronic inflammation, which is one of the main symptoms of geriatric diseases. There is a close association between PAs and maintenance of intestinal mucosal barrier functions, which are required for the secretion of mucous or secretory IgA from intestinal epithelial cells5, recovery of damaged mucosal layers6 and upregulation of E-cadherin7 and occluding proteins8 that are bound by tight junctions to intestinal epithelial cells. Moreover, PAs suppress mutagenic events by repressing the occurrence of frameshift mutations9 and DNA alkylation10. Recent studies demonstrated that PAs can enhance longevity in bacteria, yeast, worms, flies and mice and that this is due to promotion of autophagy11. In the case of mammals, Soda et al. extended the lifespan of mice by administering PA-rich food12. Consistent with this, we previously found that increasing intestinal PA concentrations by supplementing chow with Bifidobacterium animalis subsp. lactis LKM512 inhibited senescence and enhanced longevity in mice13. PAs also regulate brain function, which is one of the most important criteria upon which quality of life (QOL) is evaluated. N-methyl-D-aspartate (NMDA) receptors, which play a key role in activation-dependent synaptic plasticity and formation of memory in rodents, are regulated by PAs14. Chronic treatment with difluoromethylornithine (DFMO), a potent and irreversible inhibitor of ornithine decarboxylase (ODC), depletes PAs and impairs spatial learning and memory15. In contrast, striatal injection of SPM improved recognition memory16. Recently, it has been reported that PA-rich diets protect fruit flies from age-induced memory impairment17.
In addition to their cellular biosynthesis, PAs are derived from exogenous foodstuffs in the diet and from intestinal bacteria. Intracellular production of PAs and their concentration in tissue and organs decreases with aging18,19. Therefore, intake of exogenous PAs may be beneficial in the treatment of some geriatric diseases. The major route for delivery of PAs is oral consumption, since many food ingredients contain these molecules. A long-term PA-rich diet or PA-rich food increases PA concentration in the blood of mice or humans, respectively20. Most dietary PAs are absorbed in the small intestine21; on the other hand, most of the PUT and SPD in the colon are provided by intestinal bacteria22. Rather than dietary PAs, intestinal bacterial PAs are required to maintain and repair the large intestine, which is more susceptible to senescence than the small intestine23. We reported that fecal PA levels of elderly people are lower than those of younger individuals24 and that ingestion of LKM512 as a probiotic resulted in altered composition of intestinal bacteria25, an increased amount of PAs in the intestine and an inhibition of systemic inflammation26. However, the increase in PA concentration upon ingestion of LKM512 varied between individuals and ranged from 10–1000 μM; moreover, PA levels did not increase at all in certain individuals. Together, these results indicate that ingestion of LKM512 per se is not sufficient for elevating intestinal PA concentration.
About 1,000 species of bacteria can colonize the human intestine and approximately 160 of these are present in any healthy individual at a given time27. Depending on PA concentration in its growth environment, Escherichia coli can transport PAs into the cell through PA-specific transporters, or synthesize them when levels of cellular uptake are not sufficient28,29,30. In addition, during logarithmic growth, E. coli can also excrete31 PAs. This indicates that controlling PA concentration in the large intestine is synonymous with controlling the PA metabolism of intestinal microorganisms. Cellular absorption of PAs is affected by the concentration of amino acids or growth factors32,33,34, indicating that environmental components can modulate PA absorption and release in intestinal bacteria. Although little is known regarding the identity of intestinal bacterial metabolites, we have recently characterized metabolites with low molecular weight produced by intestinal bacteria using dynamic metabolomics analysis with CE-TOFMS22.
Here, we use CE-TOFMS metabolomics to identify the molecular components that correlate with PA concentration in the intestine. Furthermore, we demonstrate that oral administration of PA-upregulating substances, which we found via an in vitro screening approach, elevated intestinal PA concentration, inhibited systemic inflammation, prolonged longevity and promoted spatial learning and memory in mice.
Results
Identification of PA-upregulating substances using metabolomic analysis
Fecal samples from 12 volunteers who were served identical meals were subjected to metabolomic analysis. Hijiki seaweed, an indigestible ingredient, was served for breakfast on day 4 to measure the food retention time in the digestive tract. The seaweed was not detected in 2 out of 12 samples, which were then excluded from further analysis since results derived from them may have been affected by food ingredients that were not existent in other samples. CE-TOFMS metabolomics detected 221 metabolites, including 125 cationic and 96 anionic substances. The metabolites that correlated with levels of PUT, which is the most abundant PA in the samples, were determined (Table 1). Twenty-eight metabolites, in particular, N-acetylputrescine, γ-aminobutyric acid (GABA), 5-aminovaleric acid, arginine (Arg), N5-ethylglutamine, lysine (Lys), 3-methyl-2-oxovaleric acid, 5-hydroxylysine and proline (Pro), were significantly correlated with PUT concentration (p < 0.001).
Screening of candidate metabolites that regulate PUT using bacterial and fecal culture methods
We then performed a bacterial culture screen with the metabolites that correlated with PUT concentration (p < 0.01). We specifically tested GABA, Arg, Lys, Pro, fumaric acid, Tyr and Asp, since they are associated with safety in humans and low cost. The results show that addition of GABA, Arg and fumaric acid increased PA concentration more than 2-fold (Fig. 1A). In particular, Arg increased PUT concentration more than 7-fold in E. coli culture. Fecal culture tests were also performed with these candidate substances. Culturing with Arg increased PUT concentration significantly between 6 h and 12 h (Fig. 1B). Other candidates elicited no effect in fecal culture. In contrast, the effect of Arg was present in all fecal samples (Supplementary Fig. S1A) in spite of large individual differences in intestinal bacterial composition (Supplementary Fig. S2). The SPD and SPM concentrations tended to increase in response to the other metabolites, including Arg, at 24 h and 12 h, respectively (Supplementary Fig. S1B). However, this depended on the individual examined and did not reach statistical significance.
Screening of putrescine-upregulating substances by culture methods.
(a) PA production of a single culture of E. coli MG1655, Blautia producta JCM 1471T, or Bacteroides thetaiotaomicron JCM 5827T with candidate substances (GABA, Arg, Lys, Pro, fumaric acid, Asp and Ser). Candidates that increased PA concentration more than 2-fold were used in experiments that required fecal culture. (b) PUT concentration stimulated by candidate substances with human fecal culture. Data are expressed as relative ratio that calculate PUT concentration from 0 h to 1 h. Results of each fecal specimen are shown in Supplementary Fig. S1A. Data are represented as mean ± SD **p < 0.01, ***p < 0.001. (c) Dose-dependent effect of Arg on PUT production with human fecal culture.
Effects of oral administration of arginine on fecal polyamine concentration in vivo
Oral administration of Arg to mice increased PUT concentration in a dose-dependent manner (Fig. 2A). In mice, fecal PUT concentrations increased significantly (p < 0.05) when Arg intake was more than 0.6 mg/g of body weight. In rats, PUT concentration tended to increase following oral administration of Arg at 0.9 mg/g of body weight (p = 0.078) and SPD concentration was increased by oral administration of Arg at 0.3 mg/g of body weight (p < 0.05). In contrast, antibiotic treatment before Arg ingestion completely eliminated the effect of Arg on PUT production (Fig. 2B, black bar). When ornithine, the main precursor of PAs in mammalian cells, was administrated orally instead of Arg, there was no change in concentration of any PAs (Fig. 2B, blue bar).
Effects of Arg administration on putrescine concentration in rodents.
(a) Fecal PA concentration after oral administration of Arg in ICR mice (left) and SD rats (right) (n = 8) *p < 0.05, **p < 0.01, ***p < 0.001 (vs. control = 0 mg). (b) Influences of oral administration of antibiotics on PA production due to Arg and effects of ornithine substitution on PA production in ICR mice (n = 8). Data are represented as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001 (vs. Arg 0.6 mg/g body). nt: not tested. (c) Polyamine concentration in the colonic lumen and tissue after oral Arg administration in SD rats (n = 4). (d) Changes of blood PA concentration after oral Arg administration in rats with a jugular catheter (n = 5). *p < 0.05 (vs. pre-administration). (e) Fecal PA concentration after injection of Arg through a proximal colon catheter (n = 5). *p < 0.05, **p < 0.01 (vs. control = 0 mg).
Detection of stable isotope-labeled putrescine in the colon contents
We observed that stable isotope-labeled PUT was detected from colonic content following injection of 13C6,15N4-labeled Arg via a colon catheter. In extracts of colonic content, both stable isotope-labeled PUT (m/z 128, 171 and 361) and unlabeled PUT (m/z 126, 166 and 355) were detected and the proportion of stable isotope- labeled PUT was found to be 44% (Fig. 3).
Effects of oral administration of arginine on colonic tissue and blood polyamine concentration in vivo
Although oral administration of Arg to rats increased colonic luminal PUT significantly (p < 0.05), the PUT concentration in colonic tissue did not increase significantly. On the other hand, the SPD concentration in colonic tissue tended to increase (Fig. 2C). Using a jugular catheter, changes in blood PA concentrations were measured in rats (Fig 2D). Blood SPD concentration increased significantly (p < 0.05) at 4 h and 8 h after oral Arg administration (mean ± SEM: 30.74 ± 1.32 μM at pre-consumption, 36.72 ± 1.83 μM at 4 h, 38.25 ± 2.08 μM at 8 h after administration), but PUT concentration was very low and did not change. Following injection of Arg through a proximal colon catheter, fecal PUT and SPM concentrations increased significantly (p < 0.05); this was observed when Arg intake was more than 4.4 mg/g of body weight. Fecal SPD concentration also increased significantly (p < 0.05) when Arg intake was 6.6 mg/g of body weight (Fig. 2E).
Effects of 6-months' oral administration of Arg, LKM512 and Arg & LKM512 mix on ICR mice
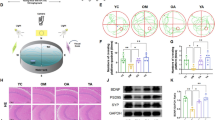
We then subjected mice between 12 and 18 months of age to a 6-month treatment of either Arg or the probiotics LKM512, or a combination of the two. Serum inflammatory cytokine concentrations are shown in Fig. 4A. Levels of IL-6 (p < 0.05) in the Arg group, IL-6 (p < 0.05) and MIP-2 (p < 0.05) in the LKM512 group and MIP-2 (p < 0.05) and TNF-α (p = 0.07) in the double treatment group were reduced compared with those in the controls. Concentrations of IL-1β, MIP-2 and TNF-α in the double treatment group were lowest. Urinary 8-OHdG, which is indicative of oxidative DNA damage, tended to be lower in the double treatment group than in controls (Supplementary Fig. S3A). Senescence marker protein-30 (SMP-30), a protein expressed in multiple organs and tissues, including the liver and kidney, likely plays a role in in protecting organs from oxidative stress during aging and its tissue levels decrease with aging35,36, SMP-30 levels in liver tissue in the LKM512 and double treatment groups was higher than that in the control group (Fig. 4B). This finding is consistent with a report from Soda et al.37 who found that SMP-30 levels were increased in the liver and the kidney in mice following oral intake of PA-rich food.
Effects of 6-month oral administration of Arg, LKM512, or Arg & LKM512 mix in aging ICR mice.
The test sample was administered 3 times a week, starting when the mice were 12 months old. Data were obtained from 7 mice in the Arg and LKM512 groups, 6 mice in the Arg & LKM512 mix group (a mouse died in the study period) and 4 mice in the control group (3 mice died in the study period). (a) Serum inflammatory cytokine concentrations *p < 0.05, **p < 0.01. (b) SMP-30 content in liver tissue (right: western blot, left: immunohistochemical staining). (c) A dendrogram of fecal microbiota of mice after a 6-month treatment based on the T-RFLP pattern. (d) Bacterial PUT synthesis pathway from an Arg precursor. Pathway 1: decarboxylation of ornithine generated from Arg hydrolysis. Pathway 2: direct conversion of agmatine generated from Arg decarboxylation, catalyzed by agmatine ureohydrolase. Pathway 3: indirect conversion of agmatine generated from Arg decarboxylation via N-carbamoylputrescine, catalyzed by agmatine deiminase/iminohydrolase and N-carbamoylputrescine amidohydrolase (e) Comparison of the proportion of PUT synthesis-related genes detected from both the Arg & LKM512 mix group and the control group by fecal metagenome analysis.
The dendrogram of fecal microbiota of mice after 6-month treatment (i.e., at 18 months of age) based on the T-RFLP pattern is shown in Fig. 4C. Following the administration of LKM512, Arg, the double treatment, or PBS (control), the fecal microbiota of mice could be divided into 4 clusters, with some exceptions. Fecal PUT concentration in all treatment groups tended to be higher than that in the control group, although this did not reach statistical significance due to individual variation (Supplementary Fig. S3B). Metagenome analysis was performed with fecal samples from the double treatment mice and control animals following 6-month oral administration of Arg & LKM512 mix. About 75,000,000 reads in each sample passed a quality control, although 5% of the total reads were discarded. Approximately 682,000 contigs were assigned with a 423-bp length on average after assembly with Meta-velvet. All sequence data were submitted to NCBI through DDBJ (Bio project # PRJDB1459-PRJDB1464). Significant differences between the groups were revealed in these categories: transcription, nuclear structure, cell wall/membrane/envelope biogenesis and posttranslational modification/protein turnover/chaperones (Supplementary Fig. S4). We focused on genes in the bacterial pathway for PA synthesis from Arg (Fig. 4D). The comparison of detection rate of PUT synthesis-related enzymes showed that arginine decarboxylase (SpeA) was more frequently detected (p < 0.05) in the double treatment group. N-carbamoylputrescine amidase (p = 0.102) and agmatinase (SpeB) (p = 0.193) were also more frequently in the double treatment group than in the control group (Fig. 4E).
Effects of 6-month oral administration of Arg & LKM512 mix on the metabolome of the prefrontal cortex in ICR mice
A total of 260 metabolites (CE-TOFMS,190 metabolites; LC-TOFMS, 70 metabolites) were identified from the prefrontal cortex metabolome in both the Arg & LKM512 mix and control groups (Supplementary Table S1). Metabolites that were present in significantly higher concentrations in the Arg & LKM512 mix group included 2-deoxyglucose 6-phosphate, pelargonic acid, N5-ethylglutamine, acylcarnitine (20:1), acylcarnitine (17:0), decanoic acid, heptanoic acid and octanoic acid. In contrast, metabolites that were present in significantly lower concentrations in the Arg & LKM512 mix group were coenzyme A, 1-methyladenosine, 2-oxoglutaric acid, threonine, citrulline and carnosine (Table 2). No significant differences were observed in prefrontal cortex PA concentrations between the Arg & LKM512 mix-treated group and controls.
Effects of long-term oral administration of Arg & LKM512 mix on lifespan in ICR mice
We observed that female ICR mice treated with the Arg & LKM512 mix lived longer than controls (p < 0.05) until we reached the point of 12 months' treatment (26-month-old). At this time, we had to terminate the study for ethical considerations, since many mice lost the ability to swim during the Morris water maze task and also frequently developed tumors. Male ICR mice treated with the Arg & LKM512 mix lived significantly longer than controls (p < 0.01) (Fig. 5A). On the other hand, in both female and male mice, there was no significant weight difference between the groups (Fig. 5B). Although mice in both treatment groups were given a standard pellet chow diet ad libitum, the quantity of chow intake was not significantly different between the groups except at month 4 (18-month-old). It is important to note that the longevity observed in the Arg & LKM512 mix-treated mice was not related to caloric restriction (CR), which has been shown to increase longevity in a variety of species.
Long-term oral administration of Arg & LKM512 mix delays aging in ICR mice.
The Arg & LKM512 mix was administered 3 times a week, starting when the mice were aged 14 months (female Arg & LKM512 mix group: n = 62, female control group: n = 66, male Arg & LKM512 mix group: n = 10, male control group: n = 10). *p < 0.05, **p < 0.01. (a) Kaplan-Meier survival curves. (b) Differences in body weight and chow intake between the Arg & LKM512 mix group and control group. (c) Change in escape latency. (d) Time spent in four quadrants in probe test. *p < 0.05 (Student's t-test), †p < 0.05 (paired t-test).
Morris water maze
Animals were trained to find a hidden platform for 8 days in each trail (pretreatment and month 6).
On the third day of the trial at month 6 (20-month-old animals), the latency of the Arg & LKM512 mix-treated mice with respect to finding the hidden platform was significantly lower than that of the control (p < 0.05) (Fig. 5C). For the probe test before administration (12-month-old), there were no differences between groups in terms of the percentage of time at the former platform quadrant. However, for the probe test after 6 months (20-month-old animals) of administration, the Arg & LKM512 mix-treated mice spent significantly more time at the former platform quadrant than the control (p < 0.05). While the time spent at the former platform quadrant after 6 months of treatment in mice treated with the Arg & LKM512 mix increased significantly (p < 0.05) compared with those in pretreatment, there was no corresponding change in the control group (Fig. 5D).
Discussion
To our knowledge, this is the first report describing the induction of the production of specific target bioactive substances by intestinal microbiota. Our results clearly show that there is an immense variety of individual intestinal microbes, hampering our aim of specifically producing polyamines in the intestine in order to counteract aging. Despite this, however, we have clearly demonstrated that, collectively, these intestinal microbes confer the expected health benefits in terms of their anti-aging effects.
Recently, many studies have focused on the microbiome of the large intestine in order to clarify the composition of intestinal microbiota and the metabolic pathways that are operant in the intestinal tract38,39,40. However, studies focused on the actual identification of metabolites in the intestinal tract are lacking. To the best of our knowledge, there are few studies that have been designed in order to produce specific metabolites using intestinal bacteria, with the exception of the production of short chain fatty acids such as butyrate41,42. Sato et al.43 reported that synbiotic ingestion of the lactate-utilizing butyrate-producer Anaerostipes caccae strain L2 and indigestible oligosaccharides alters microbial fermentation and promotes the formation of butyrate in the gut. However, there are several problems in those methods, such as safety for oral administration and resistance of this bacterium to stomach acids. We suggest that our current results provide a clinically more viable strategy, as oral Arg supplementation is safe and can stimulate elevated production of PAs by the normal intestinal microbiota.
There are three pathways for bacterial PUT biosynthesis, depending on its precursors: Arg or ornithine44. PA concentration in the feces did not increase when ornithine was orally administered to mice (Fig. 2B), suggesting that the majority of bacteria derive PUT from the Arg (rather than the ornithine) precursor and that orally administered ornithine was absorbed by the host. When Arg was administered to mice after treatment with antibiotics, PUT was not synthesized in the intestinal tract, indicating that it was produced by intestinal bacteria and did not originate from host cells. In fact, using stable isotope labeling of Arg, we demonstrated that colonic luminal PUT was produced from Arg metabolism by intestinal bacteria (Fig. 3).
The number of genes that were upregulated in the pathway controlling Arg conversion to PUT increased (Fig. 4e), indicating that long term administration of Arg conferred the intestinal microbiota with an increased ability to produce PUT from Arg. Thus, we successfully created an intestinal microbiota producing a target bioactive substance in the intestinal lumen. Extrapolation of the effective Arg dose used in rats (4.4 mg/kg body weight) to humans reveals that this approach is suitable for clinical use. Importantly, the SPD concentration in colon tissue (Fig. 2C) and in blood (Fig. 2D) was temporarily increased by the oral administration of Arg, despite the fact that the intracellular PA concentration is strictly regulated1. Most of the PUT absorbed by the host cell from the intestinal lumen is degraded in the intestinal epithelial cells and excess PUT is transferred to the circulatory system and converted to SPD and SPM45. Therefore, the increase of SPD in the colon tissue and blood is likely due to the metabolism of PUT. It is also possible that increased SPD in the lumen of the colon (Fig. 2C) is absorbed directly into colonic tissue and blood.
Our central hypothesis is that increased PA concentration in the intestinal tract represses senescence and promotes longevity by maintaining or restoring intestinal barrier function, or by exerting anti-inflammatory activity, anti-mutagenic activities, anti-oxidative properties and autophagy13,46. In the present study, the most robust anti-inflammatory and anti-oxidative activities were obtained in the Arg & LKM512 mix group, in which PA concentration was highest, supporting our hypothesis. Cobun et al. reported that Arg administration suppressed inflammation in a mouse model of DSS colitis but did not increase PA concentration in the colon tissue47. However, it is very likely that most of the Arg administered in that study was mainly absorbed in the small intestine because Arg was administered in drinking water. The authors suggested that Arg transferred to blood was converted to PAs and that this exerted beneficial anti-inflammatory effects, but the level of PA synthesized was below the limit of detection in the colon tissue.
We investigated a large-scale and long-term treatment using a Arg & LKM512 mix, whose fecal PA production, anti-inflammatory and anti-oxidative activity were highest, starting when the mice were aged 14 months. As a result, we observed a significant increase in longevity, even though we began our treatment 4 months later than that of our previous study13. Although in this experiment, we did not obtain blood PA data because we were concerned that the inflammatory reaction induced by blood collection would confound our results, blood PA concentrations appear to increase because their levels are enhanced concomitantly with the rise in intestinal PAs elicited by oral Arg administration in rats in other experiment in this study (Fig. 2D). In this study, we selected ICR mice because this strain has closed colony-dependent genetic polymorphism. Further studies are required to investigate the effects of PAs or Arg & LKM512 mix on older mice and inbred mouse strain. Assimilating our current results with those from previous studies, we advocate that PAs, in particular those produced by intestinal commensal bacteria, are important environmental factors that can directly influence longevity independently of CR.
Liu et al.48 demonstrated age-related region-specific changes in PAs in memory-associated structures. For example, in the prefrontal cortex, there was an age-related decrease in PUT and a significant increase in SPD and SPM levels. Decreased levels of PUT and SPD in the hippocampus and prefrontal cortex following DFMO treatment impaired spatial learning and memory, implicating endogenous PAs as functionally important for learning and memory15. However, it is unclear that similar effects are elicited by the Arg & LKM512 mix, because no significant differences were observed in prefrontal cortex PA concentrations between the Arg & LKM512 mix-treated group and controls (Supplementary Table S1). In this study, we succeeded in detecting 260 metabolites from the prefrontal cortex, which we believe is the largest number identified to date. Fourteen of the 260 metabolites (Table 2) were differently regulated in the Arg & LKM512 mix group and the control group, indicating that these metabolites are related to the promotion of spatial learning and memory. Differences in the levels of these metabolites likely depend on the precise composition of intestinal microbiota (Fig. 3B), since the cerebral metabolome is influenced by intestinal microbiota49. Acylcarnitines were higher in the Arg & LKM512 mix group when compared with the control group. The principle role of acylcarnitines is to transport long-chain fatty acids into mitochondria for β-oxidation, especially in the muscle and heart50. However, this supplementation may also have beneficial effects for the treatment of various neurological diseases, even though fat is not the major fuel for brain oxidative metabolism. Brain acylcarnitines have a role in lipid synthesis, altering and stabilizing membrane composition, modulating genes and proteins, improving mitochondrial function, increasing antioxidant activity and enhancing cholinergic neurotransmission51. Acylcarnitines also seem to play an important role in learning and memory in the Arg & LKM512 mix-treated group. However, there have been no reports on the relationship between PAs and acylcarnitines. Furthermore, the neuronal effects of other metabolites that were differentially detected between treatment groups are unclear. Recently, Gupta et al.17 reported that PA-feeding protected fruit flies from age-induced memory impairment in an autophagy-dependent manner. In our previous study, we confirmed that colonic autophagy was promoted by PA upregulation of LKM51213. However, the relationship between autophagy and promotion of learning and memory was not determined in this study and further studies are required to determine any associations.
Since the discovery that PA was involved in the progression of cancer52, a large number of studies have investigated this relationship in more detail. This has led to the tentative conclusion that PAs are causative agents in cancer, a conclusion that was bolstered by the finding that progression of familial adenomatous polyposis and colorectal adenoma are prevented by concomitant administration of anti-inflammatory agents and DFMO53,54. However, most of the historical studies tested the effects of PAs on existing tumors or on the growth of already established tumors55,56. Additionally, we have found no evidence that increased intake of PAs promotes oncogenic transformation in normal cells and healthy animals. In spite of this reassuring evidence, we fully understand concerns that PA may act as a tumor promoter. Therefore, the technology developed in the present study to increase the intestinal luminal PA concentration should be applied carefully to patients who are at risk of, or who already have, cancer. Similarly, although Arg is also reported to promote colorectal cancer53, we have not observed Arg-induced cancer in healthy animals. Indeed, recent reports mention that PAs actually function as cancer chemopreventive agents. For example, Scuoppo et al.57 reported that adenosylmethionine decarboxylase 1, which is involved in SPD biosynthesis, is a tumor suppressor using a human lymphoma model. Soda et al.58 demonstrated that increased polyamine intake was associated with a decreased incidence of colon tumors induced by 1,2-demethylhydrazine administration in BALB/c mice. Furthermore, there are many reports that PAs are indispensable for homeostatic processes in a diverse group of organisms ranging from cells and bacteria to plants and mammals59,60. We note that PAs are essential metabolic building blocks of life, since PAs stimulate mRNA translation by altering mRNA structure in both prokaryotic and eukaryotic cells61 and SPD is required for maturation of eIF5A62, an indispensable factor in protein synthesis. We hope that the results presented here encourage more researchers to investigate the diverse and important bioactivities of PAs.
Conclusion
Oral administration of Arg, which was selected from the fecal metabolome as a colonic luminal PA-upregulating substance, increased the concentration of PUT in the colon and of SPD and SPM in the blood. ICR mice diets were supplemented with Arg, LKM512, Arg & LKM512 mix, or PBS from12 months of age. Six months later, fecal PUT concentration was increased by both Arg and LKM512, although a mixture of the two elicited the greatest increase. These supplements inhibited the production of inflammatory cytokines and again the effect of the Arg & LKM512 mix was the strongest. Long-term administration of the Arg & LKM512 mix suppressed inflammation, improved longevity and protected from age-induced memory impairment. This study demonstrates that intake of Arg with the probiotics LKM512 may prevent aging-induced QOL deterioration via upregulation of polyamines. However, the relationship between the protection of memory impairment and PAs remains unclear and there is no evidence of intestinal luminal polyamine transport to the brain via the blood.
Methods
Human volunteers and controlled diet
Twelve healthy men (age, 25–37 years; average, 31.3 years) volunteered for the experiments. Three identical meals a day were served on a fixed schedule. The meals, which are Japanese standard meals, were supervised by a nutritionist (Supplementary Fig. S5). Details about the controlled diet used are shown in Supplementary Methods. At the tenth meal, the intestinal retention time of food was measured by including Hijiki seaweed (an indigestible ingredient easily recognized in feces) in the meal. Informed consent was obtained from all participants and the study conformed to the Helsinki Declaration. This human study protocol was approved by the Institutional Review Board of Kyodo Milk (Permit Number: 2009h-01).
Fecal samples for metabolomics analysis
Fecal samples were collected at day 5 in the morning. Immediately after excretion, feces were collected in a tube and kept on ice. Fecal solution for metabolomics was prepared within 3 h after collection by resuspending 0.5–1.0 g feces in 4 volumes of Dulbecco's phosphate-buffered saline (pH 7.2; D-PBS, GIBCO) with vigorous mixing (3 times for 1 min, each time followed by 5 min on ice).
CE-TOFMS metabolomics of fecal samples
Metabolomics measurements and data processing were performed as described in our previous report22 using an Agilent Capillary Electrophoresis System. This measurement was carried out through a facility service at Human Metabolome Technology Inc., Tsuruoka, Japan.
Searching of PA-upregulating metabolites using a single culture of bacteria
A wild-type Escherichia coli MG1655 and the intestinal predominant bacterial strains Blautia producta JCM 1471T and Bacteroides thetaiotaomicron JCM 5827T were used for the screening of candidate metabolites: γ-aminobutyric acid, arginine, lysine, proline, fumaric acid, tyrosine and aspartic acid. Details about the cultivation used are shown in Supplementary Methods. After incubation, the supernatant was collected and PA concentration was measured.
Identification of PA-upregulating metabolites using human fecal bacterial culture
Fecal samples were collected from 5 individuals under no dietary constraints. Fecal medium was prepared by resuspending the fresh fecal samples in D-PBS at a 40% (w/v) concentration and intestinal bacteria were cultured in the media at 37°C under anaerobic conditions. Candidate metabolites that were selected during previous single culture tests were added to the fecal medium at a final concentration of 0.2% (w/v). Details are provided in Supplementary Methods.
Preparation of fecal extract
Feces were diluted 10-fold with D-PBS and extracted three times by intense mixing for 1 min and followed by a standing period of 5 min on the icebox. At 1 min after extraction, the upper aqueous (precipitation-free) portion at the bottom was collected and centrifuged (12,000 × g for 10 min at 4°C) and 200 μL of supernatant was centrifugally filtered (0.45-μm filter, Millipore). The filtrate was stored at −80°C until use.
Determination of fecal PA concentration
The PA concentration in the supernatants of single culture medium, fecal extracts and blood was measured as described in Reguera et al.63, with some modifications, using Acquity ultra performance LC (UPLC) system with FLR Detector (Waters, Milford, MA). Details about this measurement are provided in Supplementary Methods.
Mice and rats
All mice and rats were obtained from Charles River Laboratories Japan Inc. (Kanagawa, Japan). All animal experiments were approved by the Kyodo Milk Animal Use Committee (Permit Number: 2010-01) and were in accordance with the Guide for the Care and Use of Laboratory Animals, published by the National Academies Press. Details are provided in Supplementary Methods.
Oral administration of arginine to mice and rats
The assay timeline is presented in Supplemental Fig. S6A. Seven-week-old male ICR mice (n = 8) were obtained and housed for 2 weeks in the animal facility. After 3 h of starvation, 0, 0.2, 0.4, 0.6 and 1.0 mg/g of body weight Arg in D-PBS (pH 7.0) was given orally to the mice with a sonde. Ornithine (0.6 mg/g of body weight), which is one of precursors of PUT, was also given orally to the mice instead of Arg. Kanamycin was also given to mice to evaluate the contribution of intestinal bacteria. Kanamycin (75 mg/d per mouse, Wako, Tokyo, Japan) was provided to mice twice, at 12 and 3 h before administering Arg (Supplemental Fig. S6B). Seven-week-old male SD rats (n = 8) were obtained and housed for 2 weeks in the animal facility. After 3 h of starvation, 0, 0.3 and 0.9 mg/g of body weight Arg in D-PBS was given orally to the rats with a sonde. Mice and rats were housed individually and after each meal, fecal samples of mice and rats were collected at 5–6 h and 7–8 h, respectively. All dose and treatment experiments of oral Arg administration were performed using the same animals.
Determination of PA concentration in colonic tissue and luminal content in rats
After 3 h of starvation, 0.9 mg/g of body weight Arg in D-PBS (pH 7.0) was given orally to the rats with a sonde (n = 4). After 8 h, rats were sacrificed and the colonic tissues and content were obtained for the measurement of PA concentrations. Control rats were administered D-PBS not containing Arg (n = 4). Colon tissues were lysed using RIPA Buffer (Thermo, IL, USA) with Protease Inhibitor Cocktail Kit (Thermo) following the manufacturer's protocols. In addition to the procedure for fecal extracts, colonic luminal content extracts and analysis of PA concentration in colonic tissue lysates was performed by high performance liquid chromatography.
Determination of blood PA concentration using jugular catheter rats
After 3 h of starvation, 0.9 mg/g of body weight Arg in D-PBS (pH 7.0) was given orally to the rats with a sonde. Before and after 2, 4, 6, 8, 10, 12 and 14 hours of Arg administration, 100 μL blood was collected through a jugular catheter. Pretreatment measurement of blood PA concentration was performed as described in Soda et al37.
Arg injection through colon catheter rats
Five male SD rats (seven-week-old) with proximal colon catheters were provided from Charles River Laboratories Japan, Inc. and kept for 1 week in the animal facility. Arg (0.66, 3.3, 4.4, 6.6, 33, 66 mg/kg body weight) in D-PBS (pH 7.0) was injected into rats through the colon catheter. After injection, feces discharged were collected every hour from 3 to 6 h. All Arg dosing experiments were performed using the same rats.
Detection of stable isotope-labeled putrescine in colonic contents
Eight-week-old SD rats fitted with proximal colon catheters were provided from Charles River Laboratories Japan, Inc. and kept for 1 week in the animal facility. Stable isotope-labeled Arg (13C6,15N4 Arg·1HCl, 6.6 mg/kg body weight) in saline (pH 7.0) was injected into rats through a colon catheter. Six hours post-injection, rats were euthanized and colonic contents were obtained. The contents were diluted 5-fold with D-PBS and extracted per the fecal extracts and PUT was analyzed by GC-MS (GCMS-QP2010, Shimadzu Co., Kyoto, Japan). PUT was derived using ethylchloroformate (ECF) and trifluoroacetic acid anhydride (TFAA) as described in Chen et al.64.
Six-months' oral administration of Arg, LKM512 and Arg and LKM512 mixtures in mice
Female ICR retired mice (8-month-old, previously used solely as breeders) were obtained from Charles River Laboratories Japan Inc. Mice were gavaged with a D-PBS solution containing Arg (n = 7), B. animalis subsp. lactis LKM512 (n = 7), Arg and LKM512 mixture (Arg & LKM512 mix) (n = 7), or nothing (control treatment; n = 7). The solution was administered 3 times a week, starting when the mice were aged 12 months. Details are provided in Supplementary Methods and Supplemental Fig. S6C.
Determination of serum cytokine content
Serum IL-1β, IL-6, TNF-α, IL-12 and IFN-g were measured using the commercially available sandwich ELISA kits and the Murine Mini ELISA Development Kit for each cytokine (PEPRO TECH, NJ, USA).
Determination of urinary 8-OHdG (8-hydroxydeoxyguanosine) concentration
Urinary 8-OHdG was measured with 8-OHdG Check ELISA Kits (JaICA, Fukuroi, Japan) using 10-fold diluted urine. 8-OHdG was converted into the quantity of creatinine required for measurement with a QuantiChrom Creatinine Assay Kit (BioAssay Systems).
Immunohistochemical examination and western blot of senescence marker protein-30 (SMP-30)
Livers used for histological analysis were fixed with 15% neutral formalin, embedded in paraffin, sectioned at 3 mm thickness. Sections were stained with SMP-30 Regucalcin Gluconolactonase (GNL) WesternBlot·ImmunoStain Kit (SHIMA LABORATORIES, Tokyo, Japan). Details are provided in Supplementary Methods.
T-RFLP analysis of fecal microbiota
Isolation of fecal bacterial DNA and T-RFLP analysis were isolated as described in our previous report22. PCR products were digested with HaeIII and AluI.
Metagenome analysis
Metagenome analysis was performed with three fecal samples of mice chosen randomly from long-term experiments-1 at the point of 6-month administrations. Fecal DNA was extracted using the QIAamp DNA Stool Mini Kit (Qiagen). Sequence analysis was commissioned to Takara Bio Dragon Genomics (Takara Bio, Mie Japan) and performed on a HiSeq 2000 System (Illumina) by using pair-end sequencing. Total sequences of about 70 Mbp were obtained from each sample and genome assembly was carried out with Meta-Velvet software Ver 0.7.62. (http://metavelvet.dna.bio.keio.ac.jp/). The gene prediction analysis was performed using MetaGeneAnnotator65 with all contigs. Homology search and pathway assignments were based on the COG and KEGG databases, respectively.
CE-TOFMS and LC-TOFMS metabolomics of prefrontal cortex
Brains were obtained from 4 randomly selected mice in each group after 6 months of treatment. The brain was resected on ice and the prefrontal cortex was sliced between 2.5 and 3.5 mm anterior to bregma within 5 min of sacrifice. CE-TOFMS analysis was performed as described previously66. LC-TOFMS measurement and metabolic profiling analysis was performed using an Agilent LC System (Agilent 1200 series RRLC system SL) equipped with an Agilent 6230 Time of Flight mass spectrometer (Agilent Technologies, Santa Clara, CA, USA). These measurements and metabolic profiling analyses were carried out through a facility service at Human Metabolome Technology Inc. Details are shown in Supplementary Methods.
Long-term oral injection of Arg & LKM512 mix in aging ICR mice
One hundred and forty female and 40 male ICR retired mice (8 months old, previously used solely as breeders) were obtained from Charles River Laboratories Japan Inc. Mice were randomly divided into 2 groups as cage units. Mice were gavaged with a D-PBS solution containing an Arg (0.2 mg/g body weight) and LKM512 (108 cfu/mice) mixture or nothing (control treatment). The solution was administered 3 times a week, starting when the mice were aged 14 months. Details are shown in Supplementary Methods and Supplemental Fig. S6D.
Morris water maze task
This task was performed using the methods described by Morris67 with some modifications using the water maze pool was a black circular tank measuring 120 cm in diameter and 30 cm in height. Mice were trained with 3 trials per day for 8 days. The probe test was performed 3 times on the 9th day and latency value was expressed as the average. Details are shown in Supplementary Methods.
Statistical analyses
The relationships between concentration of PA and metabolites were calculated by correlation analysis. PA concentrations and the markers for each experiment were compared using Mann-Whitney U tests. Changes in fecal PA concentrations due to the administration of Arg were compared with baseline values using a paired t-test. In long-term treatment 1, differences in relative quantity were evaluated for individual metabolites detected from prefrontal cortex by Welch's t-test. Clustering analysis in fecal microbiota was processed by Excel multivariate analysis (Esumi Co. Ltd, Tokyo, Japan). Survival curves were drawn using the Kaplan–Meier method and the survival rate was compared with the Log-rank test. StatMate IV (ATMS Co. Ltd., Tokyo, Japan) was used to conduct all statistical analyses.
References
Pegg, A. E. & McCann, P. P. Polyamine metabolism and function. Am. J. Physiol. 243, C212–221 (1982).
Medina, M. A., Urdiales, J. L., Rodriguez-Caso, C., Ramirez, F. J. & Sanchez-Jimenez, F. Biogenic amines and polyamines: similar biochemistry for different physiological missions and biomedical applications. Crit. Rev. Biochem. Mol. Biol. 38, 23–59 (2003).
Zhang, M. et al. Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: A counterregulatory mechanism that restrains the immune response. J. Exp. Med. 185, 1759–1768 (1997).
Li, L., Rao, J. N., Bass, B. L. & Wang, J. Y. NF-kappaB activation and susceptibility to apoptosis after polyamine depletion in intestinal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 280, G992–G1004 (2001).
Buts, J.-P., de Ketser, N., Kolanowski, J., Spkal, E. & van Hoof, F. Maturation of villus and crypt cell functions in rat small intestine. Role of dietary polyamines. Dig. Dis. Sci. 38, 1091–1098 (1993).
Lux, G. D., Marton, L. J. & Baylin, S. B. Ornithine decarboxylase is important in intestinal mucosal maturation and recovery from injury in rats. Science 210, 195–198 (1980).
Guo, X. et al. Regulation of adherens junctions and epithelial paracellular permeability: a novel function for polyamines. Am. J. Physiol. Cell Physiol. 285, C1174–1187 (2003).
Guo, X. et al. Polyamines are necessary for synthesis and stability of occludin protein in intestinal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 288, G1159–1169 (2005).
Clarke, C. H. & Shankel, D. M. Antimutagens against spontaneous and induced reversion of a lacZ frameshift mutation in E. coli K-12 strain ND-160. Mutat. Res. 202, 19–23 (1988).
Della Croce, C., Galli, A., Minnucci, S., Rosellini, D. & Bronzetti, G. in XVII EEMS Meeting. Abstract P-8-14.
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Soda, K. et al. Long-term oral polyamine intake increases blood polyamine concentrations. J. Nutr. Sci. Vitaminol. (Tokyo). 55, 361–366 (2009).
Matsumoto, M., Kurihara, S., Kibe, R., Ashida, H. & Benno, Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS One 6, e23652 (2011).
Williams, K., Zappia, A. M., Pritchett, D. B., Shen, Y. M. & Molinoff, P. B. Sensitivity of the N-methyl-D-aspartate receptor to polyamines is controlled by NR2 subunits. Mol. Pharmacol. 45, 803–809 (1994).
Gupta, N., Zhang, H. & Liu, P. Chronic difluoromethylornithine treatment impairs spatial learning and memory in rats. Pharmacol. Biochem. Behav. 100, 464–473 (2012).
Velloso, N. A. et al. Spermine improves recognition memory deficit in a rodent model of Huntington's disease. Neurobiol. Learn. Mem. 92, 574–580 (2009).
Gupta, V. K. et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat. Neurosci. 16, 1453–1460 (2013).
Das, R. & Kanungo, M. S. Activity and modulation of ornithine decarboxylase and concentrations of polyamines in various tissues of rats as a function of age. Exp. Gerontol. 17, 95–103 (1982).
Nishimura, K., Shiina, R., Kashiwagi, K. & Igarashi, K. Decrease in polyamines with aging and their ingestion from food and drink. J. Biochem. 139, 81–90 (2006).
Soda, K. et al. Long-term oral polyamine intake increases blood polyamine concentrations. J. Nutr. Sci. Vitaminol. 55, 361–366 (2009).
Uda, K., Tsujikawa, T., Fujiyama, Y. & Bamba, T. Rapid absorption of luminal polyamines in a rat small intestine ex vivo model. J. Gastroenterol. Hepatol. 18, 554–559 (2003).
Matsumoto, M. et al. Impact of intestinal microbiota on intestinal luminal metabolome. Sci Rep 2, 233 (2012).
Steegenga, W. T. et al. Structural, functional and molecular analysis of the effects of aging in the small intestine and colon of C57BL/6 J mice. BMC Med Genomics 5, 38 (2012).
Matsumoto, M. & Benno, Y. The relationship between microbiota and polyamine concentration in the human intestine: a pilot study. Microbiol. Immunol. 51, 25–35 (2007).
Matsumoto, M., Sakamoto, M. & Benno, Y. Dynamics of fecal microbiota in hospitalized elderly fed probiotic LKM512 yogurt. Microbiol. Immunol. 53, 421–432 (2009).
Matsumoto, M., Ohishi, H. & Benno, Y. Impact of LKM512 yogurt on improvement of intestinal environment of the elderly. FEMS Immunol. Med. Microbiol. 31, 181–186 (2001).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Kurihara, S., Kato, K., Asada, K., Kumagai, H. & Suzuki, H. A putrescine-inducible pathway comprising PuuE-YneI in which gamma-aminobutyrate is degraded into succinate in Escherichia coli K-12. J. Bacteriol. 192, 4582–4591 (2010).
Kurihara, S. et al. A novel putrescine utilization pathway involves gamma-glutamylated intermediates of Escherichia coli K-12. J. Biol. Chem. 280, 4602–4608 (2005).
Kurihara, S. et al. gamma-Glutamylputrescine synthetase in the putrescine utilization pathway of Escherichia coli K-12. J. Biol. Chem. 283, 19981–19990 (2008).
Kashiwagi, K., Miyamoto, S., Suzuki, F., Kobayashi, H. & Igarashi, K. Excretion of putrescine by the putrescine-ornithine antiporter encoded by the potE gene of Escherichia coli. Proc. Natl. Acad. Sci. U. S. A. 89, 4529–4533 (1992).
Buts, J. P. et al. Response of rat immature enterocytes to insulin: regulation by receptor binding and endoluminal polyamine uptake. Gastroenterology 106, 49–59 (1994).
Bauske, R., Milovic, V., Turchanowa, L. & Stein, J. EGF-stimulated polyamine accumulation in the colon carcinoma cell line, Caco-2. Digestion 61, 230–236 (2000).
Chabanon, H., Persson, L., Wallace, H. M., Ferrara, M. & Brachet, P. Increased translation efficiency and antizyme-dependent stabilization of ornithine decarboxylase in amino acid-supplemented human colon adenocarcinoma cells, Caco-2. Biochem. J. 348 Pt 2, 401–408 (2000).
Fujita, T., Uchida, K. & Maruyama, N. Purification of senescence marker protein-30 (SMP30) and its androgen-independent decrease with age in the rat liver. Biochim. Biophys. Acta 1116, 122–128 (1992).
Sato, Y. et al. Hydrogen-rich pure water prevents superoxide formation in brain slices of vitamin C-depleted SMP30/GNL knockout mice. Biochem. Biophys. Res. Commun. 375, 346–350 (2008).
Soda, K., Dobashi, Y., Kano, Y., Tsujinaka, S. & Konishi, F. Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 44, 727–732 (2009).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012).
Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Hague, A., Singh, B. & Paraskeva, C. Butyrate acts as a survival factor for colonic epithelial cells: further fuel for the in vivo versus in vitro debate. Gastroenterology 112, 1036–1040 (1997).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547.
Sato, T. et al. Isolation of lactate-utilizing butyrate-producing bacteria from human feces and in vivo administration of Anaerostipes caccae strain L2 and galacto-oligosaccharides in a rat model. FEMS Microbiol Ecol 66, 528–536 (2008).
Burrell, M., Hanfrey, C. C., Murray, E. J., Stanley-Wall, N. R. & Michael, A. J. Evolution and multiplicity of arginine decarboxylases in polyamine biosynthesis and essential role in Bacillus subtilis biofilm formation. J. Biol. Chem. 285, 39224–39238 (2010).
Milovic, V. Polyamines in the gut lumen: bioavailability and biodistribution. Eur. J. Gastroenterol. Hepatol. 13, 1021–1025 (2001).
Matsumoto, M. & Kurihara, S. Probiotics-induced increase of large intestinal luminal polyamine concentration may promote longevity. Med. Hypotheses 77, 469–472 (2011).
Coburn, L. A. et al. L-arginine supplementation improves responses to injury and inflammation in dextran sulfate sodium colitis. PLoS One 7, e33546 (2012).
Liu, P., Gupta, N., Jing, Y. & Zhang, H. Age-related changes in polyamines in memory-associated brain structures in rats. Neuroscience 155, 789–796 (2008).
Matsumoto, M. et al. Cerebral low-molecular metabolites influences by intestinal microbiota: a pilot study. Front in Syst Neurosci 7, (in press) (2013).
Bremer, J. Carnitine--metabolism and functions. Physiol. Rev. 63, 1420–1480 (1983).
Jones, L. L., McDonald, D. A. & Borum, P. R. Acylcarnitines: role in brain. Prog. Lipid Res. 49, 61–75 (2010).
Russell, D. H. & Levy, C. C. Polyamine accumulation and biosynthesis in a mouse L1210 leukemia. Cancer Res. 31, 248–251 (1971).
Gerner, E. W. Impact of dietary amino acids and polyamines on intestinal carcinogenesis and chemoprevention in mouse models. Biochem. Soc. Trans. 35, 322–325 (2007).
Thompson, P. A. et al. Levels of rectal mucosal polyamines and prostaglandin E2 predict ability of DFMO and sulindac to prevent colorectal adenoma. Gastroenterology 139, 797–805, 805 e791 (2010).
Sarhan, S., Knodgen, B. & Seiler, N. Polyamine deprivation, malnutrition and tumor growth. Anticancer Res. 12, 457–466 (1992).
Cipolla, B. G., Havouism, R. & Moulinoux, J. P. Polyamine contents in current foods: a basis for polyamine reduced diet and a study of its long term observance and tolerance in prostate carcinoma patients. Amino Acids 33, 203–212 (2007).
Scuoppo, C. et al. A tumour suppressor network relying on the polyamine-hypusine axis. Nature 487, 244–248 (2012).
Soda, K., Kano, Y., Chiba, F., Koizumi, K. & Miyaki, Y. Increased polyamine intake inhibits age-associated alteration in global DNA methylation and 1,2-dimethylhydrazine-induced tumorigenesis. PLoS One 8, e64357 (2013).
Takahashi, T. & Kakehi, J. Polyamines: ubiquitous polycations with unique roles in growth and stress responses. Ann. Bot. 105, 1–6 (2010).
Medina, M. A., Urdiales, J. L., Rodriguez-Caso, C., Ramirez, J. & Sanchez-Jimenez, F. Biogenic amines and polyamines: similar biochemistry for different physiological missions and biomedical applications. Crit. Rev. Biochem. Mol. Biol. 38, 23–59 (2003).
Igarashi, K. & Kashiwagi, K. Characterization of genes for polyamine modulon. Methods Mol. Biol. 720, 51–65 (2011).
Park, M. H., Nishimura, K., Zanelli, C. F. & Valentini, S. R. Functional significance of eIF5A and its hypusine modification in eukaryotes. Amino Acids 38, 491–500 (2010).
Reguera, R. M., Balana-Fouce, R., Showalter, M., Hickerson, S. & Beverley, S. M. Leishmania major lacking arginase (ARG) are auxotrophic for polyamines but retain infectivity to susceptible BALB/c mice. Mol. Biochem. Parasitol. 165, 48–56 (2009).
Chen, G. G., Turecki, G. & Mamer, O. A. A quantitative GC-MS method for three major polyamines in postmortem brain cortex. J. Mass Spectrom. 44, 1203–1210 (2009).
Noguchi, H., Taniguchi, T. & Itoh, T. MetaGeneAnnotator: detecting species-specific patterns of ribosomal binding site for precise gene prediction in anonymous prokaryotic and phage genomes. DNA Res. 15, 387–396 (2008).
Matsumoto, M. et al. Cerebral low-molecular metabolites influenced by intestinal microbiota: a pilot study. Front Syst Neurosci 7, 9 (2013).
Morris, R. G., Garrud, P., Rawlins, J. N. & O'Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 297, 681–683 (1982).
Acknowledgements
This study was supported by the Program for Promotion of Basic and Applied Researches for Innovations in Bio-oriented Industry by the Bio-oriented Technology Research Advancement Institution (BRAIN), Japan. We thank and Mr. Takuho Kuroda, Mr. Ryo Fujii, Mr. Hiroyuki Harashima, Mr. Takeshi Saeki, Dr. Kuruto Hara, Mr. Taito Yamada, Mr. Hiroaki Ozaki, Mr. Satoshi Kanakubo and Mr. Takahiro Seto (Kyodo Milk Industry Co. Ltd.) for the providing of fecal samples and Ms. Manami Kondo for the management of control diet test (Kyodo Milk Industry Co. Ltd.).
Author information
Authors and Affiliations
Contributions
M.M. designed this study. R.K., S.K., T.O. and M.M. wrote the paper. R.K., S.K., Y.S., H.S. and Y.B. performed and analyzed the bacterial culture data. T.O. performed CE-TOFMS and LC-TOFMS metabolomics and T.O., E.S. and M.M. analyzed metabolomics data. A.N. performed and analyzed the test of stable isotope labeling of Arg. K.M., E.S., Y.K., A.Y., M.K. and M.M. performed the animal experiments and analyzed the Morris water maze data.
Ethics declarations
Competing interests
This work was supported by the BRAIN, JAPAN. This work was also funded by Kyodo Milk Industry Co. Ltd and Human Metabolome Technologies, Inc. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. E. Sawaki, K. Muramatsu, A. Nakamura, A. Yamashita, Y. Kitada and M. Matsumoto are employees of Kyodo Milk Industry Co. Ltd. and had a role in study design, data analysis, preparation of the manuscript and decision to publish the manuscript. T. Ooga is employee of Human Metabolome Technologies, Inc. and had a role in data analysis and decision to publish the manuscript. All of the other authors declare that they have no conflict of interest.
Electronic supplementary material
Supplementary Information
Supplementary Information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. The images in this article are included in the article's Creative Commons license, unless indicated otherwise in the image credit; if the image is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the image. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kibe, R., Kurihara, S., Sakai, Y. et al. Upregulation of colonic luminal polyamines produced by intestinal microbiota delays senescence in mice. Sci Rep 4, 4548 (2014). https://doi.org/10.1038/srep04548
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04548
This article is cited by
-
Spermidine from arginine metabolism activates Nrf2 and inhibits kidney fibrosis
Communications Biology (2023)
-
Annotation-free discovery of functional groups in microbial communities
Nature Ecology & Evolution (2023)
-
Gut microbiota bridges dietary nutrients and host immunity
Science China Life Sciences (2023)
-
Caloric restriction-mimetics for the reduction of heart failure risk in aging heart: with consideration of gender-related differences
Military Medical Research (2022)
-
Microbial-derived metabolites as a risk factor of age-related cognitive decline and dementia
Molecular Neurodegeneration (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.