Abstract
Knowledge of genes essential for the life-extending effect of dietary restriction (DR) in mammals is incomplete. In this study, we found that neuropeptide Y (Npy), which mediates physiological adaptations to energy deficits, is an essential link between DR and longevity in mice. The lifespan-prolonging effect of lifelong 30% DR was attenuated in Npy-null mice, as was the effect on the occurrence of spontaneous tumors and oxidative stress responses in comparison to wild-type mice. In contrast, the physiological processes activated during adaptation to DR, including inhibition of anabolic signaling molecules (insulin and insulin-like growth factor-1), modulation of adipokine and corticosterone levels and preferential fatty acid oxidation, were unaffected by the absence of Npy. These results suggest a key role for Npy in mediating the effects of DR. We also provide evidence that most of the physiological adaptations to DR could be achieved in mice without Npy.
Similar content being viewed by others
Introduction
Moderate restriction of dietary calories while providing essential nutrients, known as dietary restriction (DR), inhibits ageing-dependent disorders and increases the lifespan in many organisms, including yeast, invertebrates and mammals1,2. DR also extends the disease-free lifespan of non-human primates3,4, although it may not increase overall survival4. These findings suggest that the DR paradigm may extend the healthy lifespans of humans by preventing age-related disorders. Accordingly, it is essential to identify the genes encoding the key proteins that mediate the effects of DR. Epistasis analyses using genetic mutations have led to the identification of several genes critical for the effects of DR in invertebrates, including sirtuins, DAF-16, SKN-1 and the target of rapamycin (TOR)1. However, similar studies in mammals have been limited.
An evolutionary view predicts that the effects of DR are due to the processes used by animals to adapt to harsh environmental conditions, especially famine5,6. In mammals, neurons in the hypothalamic arcuate nuclei (ARC) sense the states of energy intake and storage through the levels of circulating hormones7. A negative energy balance and a subsequent decrease in adipose tissue reduce the circulating levels of leptin, insulin and insulin-like growth factor (IGF)-1 but increase the levels of ghrelin and adiponectin. These hormonal changes subsequently activate neuropeptide Y (Npy) neurons in the ARC. Many of these neurons co-express agouti-related protein (Agrp), which attenuates the activity of proopiomelanocortin (Pomc) neurons in the ARC. Changes in the activities of these first-order neurons inhibit the second-order hypothalamic neurons, including those secreting growth hormone-releasing hormone (Ghrh), gonadotropin-releasing hormone (Gnrh) and thyrotropin-releasing hormone (Trh), but activate corticotropin-releasing hormone (Crh) neurons7,8. These hypothalamic changes ultimately suppress the somatotropic, reproductive and thyroid axes and activate the adrenal glucocorticoid axis7,8. In fact, many of these neuroendocrinological changes occur in rodents subjected to DR8. The neuroendocrine equilibrium maintained during DR is believed to favor longevity over growth and reproduction. By contrast, genetic inhibition of the Ghrh–GH axis and the subsequent reduction in plasma IGF-1 levels extends the lifespan of rodents without restricted food intake. This suggests that inhibition of anabolic signaling pathways mediates the effects of DR.
In this study, we focused on the role of Npy in mediating the effects of DR because of its unique properties that differ from those of other hypothalamic neuropeptides. Npy is required to inhibit Ghrh neurons and GH secretion into the circulation in fasted rats9,10, whereas Agrp did not inhibit GH secretion11. Thus, DR may exert some of its effects via Npy-induced inhibition of the Ghrh–GH axis. Overexpression of Npy gene tended to increase the lifespan of rats, even in the absence of DR12. Activation of specific Npy receptors protects hippocampal neurons from kainate-induced excitotoxicity and ischemia13. Extended lifespan and resistance to harmful stresses are hallmarks of DR14. Additionally, Npy was recently reported to be involved in the antineoplastic effects of DR in mice15. Therefore, we hypothesized that Npy plays a key role in the effects of DR. We tested this hypothesis by comparing the effects of DR on lifespan, pathology, oxidative stress resistance, neuroendocrine systems, energy metabolism and gene expression between Npy-null (Npy−/−) and wild-type (WT) mice. We suspected that the absence of Npy may compromise the neuroendocrine equilibrium induced by DR and thus minimize the effects of DR in mice.
Results
The effects of DR on food intake, body weight and energy metabolism in Npy−/− mice
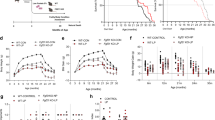
Npy is a potent orexigenic and energy-conserving neuropeptide7. Genetic disruption of Npy signaling, however, exerts subtle effects on feeding and weight gain in young mice16,17, most likely due to compensatory changes in the neuroendocrine system that normalizes feeding and energy expenditure in the absence of Npy. In the longevity groups in this study, male Npy−/−-AL mice consumed 5% less food, whereas female Npy−/−-AL mice had approximately 10% greater amounts of food during the lifespan study, although these differences were statistically insignificant (p = 0.1182 in males and p = 0.0624 in females by 2-f ANOVA (Genotype); FC data collected from 3 to 9 cages; Figure 1A and B). Accordingly, the male Npy−/−-DR mice received 5% less food, whereas the female Npy−/−-DR mice ate on average 10% more food than mice in the corresponding WT-DR groups. However, dietary restriction was generally maintained at 30% of AL intake in each group.
Food consumption (FC, g/day/mouse), body weight (BW) and food efficiency (FC/BW) in male and female mice.
(A, B) The FC in male and female Npy−/−-AL and WT-AL mice. The data for the control mice fed ad libitum (AL) are presented as the mean ± SE (n = 3 to 9 cages). The food allotments for the 30% dietary restricted (DR) groups were adjusted every 2 weeks between 12 and 32 weeks; the allotments were fixed between 32 and 72 weeks and from 72 weeks and onward. (C) The BW in the male mice. *, **, ***, p < 0.05, p < 0.01, p < 0.001 vs WT-DR at each age point by Bonferroni post hoc test. The data are presented as the mean ± SE. The initial numbers of mice as follows; n = 23, 21, 13 and 12 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively. (D) The BW in female mice. #, p < 0.05 vs. WT-AL at each age point by Bonferroni post hoc test. Data are presented as the mean ± SE. The initial numbers of mice were as follows; n = 25, 21, 12, 12 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively. (E) The FC/BW in male mice. Data represent means; the SEs are not drawn (n = 17, 16, 6 and 5 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively) (F) The FC/BW in female mice. ##, ###, p < 0.01, p < 0.0001 vs. WT-AL mice by Bonferroni post hoc test. **, ***, p < 0.01, p < 0.001, respectively, vs. WT-DR by Bonferroni post hoc test. The data represent the means; the SEs are not drawn (n = 10, 17, 5 and 9 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively).
Body weight (BW) did not statistically differ between male WT-AL and Npy−/−-AL mice (p = 0.0845 by 2-f ANOVA (Genotype): initial numbers were 23 male WT-AL and 13 Npy−/−-AL mice); BW was greater in male Npy−/−-AL than in WT-AL mice between 12 and 36 weeks, but tended to be lower in male Npy−/−-AL mice beyond 36 weeks (p = 0.0287 by 2-f ANOVA (Genotype × Age interaction); Figure 1C). BW was lower in female Npy−/−-AL mice than in WT-AL mice (p < 0.0001 by 2-f ANOVA (Genotype): initial numbers were 25 female WT-AL and 12 Npy−/−-AL; Figure 1D). BW was 8 to 16% lower in male Npy−/−-DR mice than in WT-DR mice (p < 0.0001 by 2f-ANOVA (Genotype): initial numbers were 21 male WT-DR and 12 Npy−/−-DR; Figure 1C); BW in female Npy−/−-DR and WT-DR mice did not differ (initial numbers were 21 female WT-DR and 12 Npy−/−-DR; Figure 1D).
Although the absolute values of FC and BW in Npy−/− and WT mice were similar or slightly different under AL or DR conditions (Figure 1A, B, C and D), FC normalized to BW was significantly less in WT-DR than in WT-AL males and females (p < 0.0001 in males and p < 0.0001 in females by 2-f ANOVA (Genotype); Figure 1E and F), indicating that DR increases food efficiency, as an index of how efficiently body weight (i.e., the lean body and fat mass) is maintained by food utilization. Weindruch and Wolford18 proposed a model of metabolic efficiency, in which DR retards aging by reducing mitochondrial proton leakage, which in turn increases ATP production efficiency and reduces body temperature and the generation of reactive oxygen species. In fact, recent studies in recombinant inbred strains of mice support the model19,20. Therefore, food efficiency as defined here could be used as a predictor of DR efficacy. The FC/BW was also lower in Npy−/−-DR than in Npy−/−-AL males and females (p < 0.0001 in males and p < 0.0001 in females by 2-f ANOVA (Genotype); Figure 1E and F). Therefore, the metabolic trait of DR was maintained in Npy−/− mice, although the FC/BW in male Npy−/−-AL mice was greater than in male WT-AL mice at 76 weeks of age and later (p < 0.0001 by 2-f ANOVA (Genotype × Age); Figure 1E). The FC/BW was 6% greater in male Npy−/−-DR versus WT-DR mice, particularly at 72 weeks of age and later (p < 0.0001 by 2-f ANOVA (Genotype); p = 0.0544 by 2-f ANOVA (Genotype × Age); Figure 1E). FC/BW was greater in female Npy−/−-AL mice than in WT-AL females at many, but not all, age points (p < 0.0001 by 2-f ANOVA (Genotype, Genotype × Age respectively; Figure 1F). The FC/BW was 10% greater Npy−/−-DR females than in WT-DR females between 16 and 68 weeks of age; thereafter, FC/BW was similar (p < 0.0001 by 2-f ANOVA (Genotype, Genotype × Age respectively); Figure 1F). Thus, by this measure, food efficiency was slightly reduced in Npy−/− versus WT mice in the lifelong, but not entire, period under AL and DR conditions, although the aging-related changes were sexually dimorphic in the DR groups.
The FC and BW data prompted us to analyze energy homeostasis under DR conditions. In the daily DR regimen, DR mice finish food allotments within a few hours and remain without food for approximately 20 hours thereafter. A metabolic shift from carbohydrate to fatty acid oxidation in the fed-and-fasted feeding cycle characterizes DR rodents21. In contrast, AL animals constantly oxidize carbohydrates according to the composition of standard chow. Due to bypassing complex I by electrons into the electron transport chain, the DR-specific preference for fatty acid oxidation to produce ATP contributes to the extension of lifespan through reduced generation of reactive oxygen species22. Npy enhances the utilization of carbohydrates as an energy source when food is supplied23. Npy also inhibits an excess loss of body energy, such as by heat and physical activity, under conditions of negative energy balance7. To eliminate the possibility that energy metabolism was compromised in the Npy−/−-DR mice, we analyzed the respiratory quotient (RQ) and energy expenditure (EE) in 7-month-old male mice in a metabolic cage with an indirect open-circuit calorimeter (Columbus Instruments, Columbus, OH). We suspected that the fraction of adaptive thermogenesis in the total EE might be increased in Npy−/− versus WT mice, given that Npy competes with β-adrenergic-induced thermogenesis in the brown adipose tissue (BAT)24.
The findings can be summarized as follows; 1) the diurnal variation in RQ was comparable in WT-DR and Npy−/−-DR mice (the number of mice in each group was 6; Fig. 2A), although the RQ was lower in Npy−/−-AL mice than in WT-AL mice; 2) the EE per unit body mass did not differ, particularly between male Npy−/−-DR and WT-DR mice (Fig. 2B); 3) there were no differences in body temperature (BT) between WT-DR and Npy−/−-DR mice (Fig. 2C), although the preprandial BT was lower in the DR groups than in the AL groups; 4) Consistently, mRNA expression of Ucp1, 2 and 3 in the BAT did not significantly differ between Npy−/−-DR and WT-DR mice (see Supplementary Table S1). The BAT weight was also comparable in the Npy−/−-DR and WT-DR mice (see Supplementary Table S2). Considering our results on energy metabolism, we concluded that Npy−/−mice were not metabolically impaired in comparison to WT mice when treated with long-term 30% DR, although food efficiency was slightly lower inNpy−/−-DR mice.
DR regulates fuel utilization and energy expenditure in Npy−/− mice in a manner similar to that in WT mice.
(A) Respiratory quotient (RQ) and (B) Energy expenditure (EE) in male mice at 7 months of age. The values represent the mean ± SE (n = 6). (C) Scatterplots of body weight and total EE within 21 h after feeding. R2 = 0.240 for WT-DR (solid line) and R2 = 0.468 for Npy−/−-DR (dotted line). (D) Body temperature during the pre- and post-prandial phases of feeding in male mice at 7 months of age. Values represent the means ± SE (n = 6). *, p < 0.05 vs. the preprandial phase in each group. #, p < 0.05, ###, p < 0.001, vs. the AL group during the preprandial phase.
The effects of DR on lifespan, cancer and stress resistance in Npy−/− mice
Lifespans in male and female Npy−/−-AL mice did not significantly differ from those in WT-AL groups (the initial number of mice for the longevity study (n = male/female): WT-AL (23/25), Npy−/−-AL (13/12); Fig. 3A, B). DR significantly extended lifespan in WT mice (p = 0.0028 in male and p = 0.0004 in female WT-DR vs. WT-AL mice by log-rank test; Fig. 3A, B) but not in Npy−/− mice (p = 0.7151 in male and 0.3852 in female Npy−/−-DR vs. Npy−/−-AL mice by log-rank test; WT-DR (21/21), Npy−/−-DR (12/12)).
The effects of dietary restriction (DR) on lifespan and stress response are diminished in Npy−/− mice.
Kaplan-Meier survival curves of WT and Npy−/− mice fed AL or the 30% DR diets, beginning at 12 weeks of age. (A) Survival curves in male mice. The initial numbers of mice as follows; n = 23, 21, 13 and 12 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively. Comparisons of survival curves by log-rank test; Npy−/−-AL vs. WT-AL, p = 0.0736; WT-DR vs. WT-AL, p = 0.0028; Npy−/−-DR vs. Npy−/−-AL, p = 0.7151. (B) Survival curves in female mice. The initial numbers of mice as follows; n = 25, 21, 12, 12 for the WT-AL, WT-DR, Npy−/−-AL and Npy−/−-DR groups, respectively. The longevity study was concluded at 176 weeks of age because only the female WT-DR group survived between 163 and 176 weeks of age and the proportion of surviving mice in the female WT-DR group decreased to below the 25th percentile survival point at 163 weeks. Comparisons of survival curves by log-rank test; Npy−/−-AL vs. WT-AL, p = 0.7648; WT-DR vs. WT-AL, p = 0.0004; Npy−/−-DR vs. Npy−/−-AL, p = 0.3852. (C) The proportion of surviving mice after lethal oxidative stress induced by daily injection of 3-nitropropionic acid (75 mg/kg BW) in 6-month-old male mice [the numbers of mice examined (n): WT-AL (11), WT-DR (15), Npy−/−-AL (15), Npy−/−-DR (10)]. Comparisons of survival curves by log-rank test; Npy−/−-AL vs. WT-AL, p = 0.2772; WT-AL vs. WT-DR, p < 0.0001; Npy−/−-AL vs. Npy−/−-DR, p = 0.0612.
The lifespan data are summarized in Table 1. In male WT mice, DR extended lifespan by 20.3% and 14.8% at the 50th and 25th percentile survival points; in male Npy−/− mice, these were −1.9% and 7.1%, respectively. In female WT mice, DR increased lifespan by 36.0% and 33.6% at the 50th and 25th percentile survival points; in female Npy−/− mice, DR increased lifespan by −1.0% and 19.5%. Female Npy−/−-DR mice received 10% more food compared to female WT-DR mice, this might cause the diminution of life-extending effect of DR, when compared to the extent of lifespan extension by DR in WT mice. However, daily allotments for male Npy−/−-DR mice were 5% less than those for male WT-DR mice; nonetheless, the life-extending effect was diminished in Npy−/−-DR mice. Thus, it is reasonable to conclude that the loss of Npy could minimize the effect of DR.
Five of twelve male Npy−/−-DR mice died before reaching the age of 52 weeks of life; in contrast, only a few WT mice died during this period (Fig. 3A). Post-mortem examination found no specific causes of death in these males. To eliminate bias resulting from these early deaths, we reanalyzed the lifespan data by censoring the deaths of these mice. In the reanalysis, we used a Cox hazards model with covariates, Genotype (WT vs. Npy−/−), Diet (AL vs. DR) and Sex (male vs. female). The results indicated that 1) WT mice lived longer than Npy−/− mice (p = 0.0004 [Genotype: Risk ratio (RR) = 0.418 (lower 95%: 0.262 ~ upper 95%: 0.671)], 2) the DR group also lived longer than the AL group (p < 0.0001 [Diet: RR = 0.339 (0.206 ~ 0.671)] and 3) the lifespan of male mice did not differ from that of female male (p = 0.5045 [Sex: RR = 0.864 (0.560 ~ 1.329)]). As suspected from the survival curves, WT and Npy−/− mice seemed to respond differently to the DR diet in terms of lifespan extension (p = 0.0578 [Genotype × Diet]). Then, we analyzed the lifespan data separately by the genotype. In WT mice, the DR group lived significantly longer than the AL group [p < 0.0001 [Diet: RR = 0.240 (0.126 ~ 0.440)]. In Npy−/− mice, DR mice also lived longer than AL mice (p = 0.0167 [Diet; RR = 0.329 (0.119 ~ 0.821)]. However, the RR seemed to be greater in Npy−/− mice than in WT mice. Based on the lifespan data, we concluded that the life-prolonging effect of DR was significantly diminished, if not completely abolished, in Npy−/− mice.
Post-mortem examination of the 72-week survivors revealed that 64.3% of the WT-AL mice had evidence of tumors at death, primarily malignant lymphoma, hepatocellular carcinoma, or lung adenocarcinoma (Table 2). The proportion of mice bearing tumors in the Npy−/−-AL group (55.6%) did not statistically differ in WT-AL mice (64.3%). The proportion was significantly reduced in WT-DR (35.1%) vs. WT-AL mice (Table 2), but did not differ between Npy−/−-DR (66.7%) and Npy−/−-AL mice.
Experimental animals subjected to DR are resistant to a range of stressors, such as oxidative and chemically toxic stresses14. This trait has been linked to the longevity induced by DR. We therefore assessed survival rates of 6-month-old male mice from different experimental groups subjected to oxidative stress induced by administration of 3-nitropropionic acid, an inhibitor of mitochondrial respiratory complex II25 [the numbers of mice examined (n): WT-AL (11), WT-DR (15), Npy−/−-AL (15), Npy−/−-DR (10)]. Most WT-AL and Npy−/−-AL mice died within the first 3 days (Fig. 3C). In contrast, the WT-DR mice exhibited notable stress resistance in comparison to WT-AL mice, as indicated by the significantly increased survival rate (p = 0.0009 by log-rank test; Fig. 3C). The DR effect was diminished in Npy−/− mice (p = 0.0612 by log-rank test; Fig. 3C); therefore, Npy may also be involved in the stress resistance induced by DR.
A limitation of this study is the fact that the genetic backgrounds of the Npy−/− (Npytm1Rpa/J, approximate to 129S1/SvImJ) and WT (129S6/SvEvTac) mice differed; the genetic background approximated those of the Npy-knockout mice originally generated by Erickson et al16. The responses to DR in RQ, neuropeptide mRNA expression and circulating hormones (as described below) were similar between WT and Npy−/− mice in this study, suggesting minimal effect of this difference in this respect. However, it is possible that the diminished salutary effects of DR were derived from the difference in the genetic backgrounds of the WT and Npy−/− mice. The small numbers of mice in the longevity study also limit our conclusions.
The effects of DR on hormonal and neuroendocrine systems
The loss of Npy signaling might compromise hormonal and neuroendocrine processes recruited to adapt to DR. We evaluated circulating insulin and IGF-1 concentrations, given that DR suppresses these hormones in mammals1. In contrast, inhibition of IGF-1-related signaling extends lifespan in mice, even if fed AL1; interference in insulin signaling also slightly extends lifespan in male but not female mice26. By definition, AL mice have free access to food. The DR mice ate quickly, fully consuming their food within 6 hours and then fasted until the next feeding. Blood samples were collected from the retro-orbital veins in the preprandial (4 hours prior to feeding in the DR groups) and postprandial phases (1.5 hours after feeding, the number of mice examined in each phase was 4 or 5). Plasma insulin concentrations did not show statistically significant differences between the preprandial and postprandial phases in the AL groups (Table 3); however, insulin concentrations remained low in the DR groups in the preprandial phase (Table 3) and significantly increased in the postprandial phase. Plasma insulin concentrations did not differ between WT-DR and Npy−/−-DR mice at 6 and 24 months of age (Table 3, see Supplementary Fig. S1). Blood glucose levels also did not significantly differ between WT and Npy−/− groups under AL or DR conditions at 6 and 24 months of age (see Supplementary Fig. S1).
Plasma IGF-1 concentrations, evaluated in the preprandial phase, did not significantly differ between WT-AL and Npy−/−-AL mice (9 mice per group; Table 3). DR reduced plasma IGF-1 by approximately 25% in WT and Npy−/− mice and there was no difference between WT-DR and Npy−/−-DR mice.
Anabolic signals including insulin and IGF-1 activate mammalian mTOR pathways1. The mTOR pathway plays a central role in the effects of DR in invertebrates27,28 and mammals29. We also investigated the levels of ribosomal protein S6 kinase polypeptide 1 (Rps6kb1), a component of the mTOR pathway, in the liver. The levels of phosphorylated (active) Rps6kb1 were significantly reduced by DR in WT and Npy−/− mice (the number of mice examined in each group was 3, 4, or 5 respectively; Table 3, see Supplementary Fig. S2); total protein abundance of Rps6kb1 did not differ between groups. We conclude that the insulin-, IGF-1- and mTOR-associated anabolic pathways are down-regulated by DR in the absence of Npy in mice.
DR may extend lifespan in part by reducing fat mass and altering adipokines30. Indeed, increased plasma adiponectin is associated with insulin-sensitizing, anti-diabetic and anti-atherosclerotic effects31. Transgenic overexpression of adiponectin genes extends lifespan in mice32. In this study, white adipose tissue (WAT) weight (the sum of inguinal and epididymal WAT weights) and WAT normalized to body weight (BW) were lower in the DR groups than in the AL groups (see Supplementary Table S2); the WAT/BW was even lower in Npy−/−-DR than in WT-DR mice at 6 months of age. The Plasma leptin and adiponectin concentrations were down- and up-regulated by DR in Npy−/− mice in a manner similar to that in WT mice (9 mice per group; Table 3).
Plasma corticosterone levels are increased by DR, particularly in the preprandial phase in rats33 and has been correlated with DR-induced stress resistance6. The present study also confirmed the modest increase in plasma corticosterone by DR in WT and Npy−/− mice (9 mice per group; Table 3); there was no difference between WT and Npy−/− mice.
Loss of Npy may alter hypothalamic neuropeptide expression to facilitate adaptation to DR. Therefore, we analyzed expression of hypothalamic neuropeptides by quantitative real-time PCR (qRT-PCR) in tissues collected during the preprandial phase in male mice at 6 months of age. The findings can be summarized as follows (4, 5 or 6 mice per group; Table 3): 1) loss of Npy significantly or marginally increased Agrp, Pomc, Ghrh and Crh transcript levels under AL feeding; 2) DR reduced Pomc and Ghrh levels in the absence of Npy. Loss of Npy had no influence on DR inhibition of Pomc and Ghrh transcript expression.
DR and gene expression profiles in the liver
Our results suggest that Npy-linked pathways mediating the effect of DR do not overlap with the insulin/IGF-1/mTOR, adipokine and corticosterone-associated pathways, which are thought to represent mechanisms of action of DR1,6,8,33. To characterize the putative Npy-linked pathways, we performed whole-genome microarray and pathway analyses in the liver with a gene set enrichment analysis (GSEA)34 using the Molecular Signatures Database (MsigDB) and NCBI BioSystems Database (n = 2 mice per group). Initially, we identified genes that were differentially expressed between 2 groups of mice, such as WT-DR vs. WT-AL or Npy−/−-DR vs. WT-DR, with filtering criteria set for the present study.
Based on the MsigDB, the GSEA delineated pathways involving differentially regulated genes that were significantly altered in WT-DR versus WT-AL mice (see Supplementary Tables S3 and S4). We focused on pathways that were down-regulated in Npy−/−-DR versus WT-DR mice (see Supplementary Table S5). These pathways might mediate the effects of DR and depend on Npy signaling. These procedures helped to identify four pathways (Fig. 4A). The gene sets of three of the pathways in this category reflect the gene expression profile of a superior survival group (versus poor survival of human hepatocellular carcinoma35) and that of relatively well-differentiated (versus poorly differentiated) states of head and neck cancers36. The remaining single gene set “MICROSOME” includes genes encoding the metabolism of drugs and xenobiotics. The pathways enriched in genes up-regulated in WT-DR (versus the WT-AL) but down-regulated in Npy−/−-DR mice likely reflect mechanisms underlying the inhibition or retardation of neoplastic processes by DR.
Summary of microarray data for the liver.
A) Gene set enrichment analysis (GSEA) identified 60 pathways enriched in the up-regulated (UP) genes of the WT-DR vs. WT-AL groups. Four of these pathways overlapped with pathways enriched in the down-regulated (DN) genes in the Npy−/−-DR vs. WT-DR groups. The pathway names of 4 gene sets are described in the rectangle and the references35,36 are noted in parentheses. These pathways reflect the anti-neoplastic effect of DR. In contrast, 10 pathways were enriched among the DN genes in WT-DR vs. WT-AL groups; one of these pathways overlapped with pathways enriched in the UP genes in Npy−/−-DR vs. WT-DR groups. The pathway37 reflects a stress response. B) The pathway analysis with NCBI BioSystems identified 16 pathways enriched in the UP genes of WT-DR vs. WT-AL mice. Twelve of these pathways overlapped with pathways enriched in the DN genes in Npy−/−-DR vs. WT-DR mice. Most of these are related to detoxification of chemical compounds. In contrast, 22 pathways were enriched in the DN genes of WT-DR vs. WT-AL groups; two of these pathways overlapped with pathways enriched in the UP genes in Npy−/−-DR vs. WT-DR groups. One pathway “Terminal pathway of complement” reflects activation of inflammation.
In contrast, the GSEA delineated 10 pathways associated with DR-down-regulated genes in WT mice (see Supplementary Table S6). One of these pathways was identified in the context of Npy deletion and its cancellation of the inhibitory effect of DR (Fig. 4A). This was the gene set “ICHIBA GRAFT VERSUS HOST DISEASE 35D DN”, which represents down-regulated genes associated with metabolic and endocrine functions in the liver where hepatic graft-versus-host diseases manifest and where genes related to inflammation were up-regulated37. Gene expression profiling in long-lived dwarf mice, mice subjected to DR and premature aging models has suggested that the down-regulation of metabolic and endocrine functions represents a protective mechanism against inflammatory and/or genotoxic stress during the aging process38. Therefore, loss of Npy might abolish some protections against cellular stresses.
Pathway analysis in the NCBI BioSystems Database identified pathways signified by up-regulated genes in WT-DR versus WT-AL mice (see Supplementary Table S7). These were mostly related to detoxification or degradation of chemical compounds. In contrast, pathways enriched in genes down-regulated by DR in WT mice included pathways relevant to lipid, cholesterol and steroid metabolism or biosynthesis (see Supplementary Table S8).
Comparison of gene expression profiles in Npy−/−-DR and WT-DR mice revealed that many of the pathways signified by DR-up-regulated genes in WT mice, i.e., pathways associated with detoxification, were enriched among the down-regulated genes in Npy−/−-DR mice (Fig. 4B, see Supplementary Table S9). Pathways enriched in up-regulated genes in Npy−/−-DR versus WT-DR mice included steroid biosynthesis and complement cascade (Fig. 4B, see Supplementary Table S10). The NCBI BioSystems pathway analysis suggests that detoxification or degradation of chemical compounds, which may be up-regulated by DR, is not activated properly in the absence of Npy. Up-regulation of the complement cascade also implies that Npy inhibits inflammatory processes.
Although our microarray analysis was limited to the liver, our analysis of hepatic gene expression profiles suggests Npy plays a key role in inhibiting the occurrence and/or progression of cancers through detoxification or degradation of potentially carcinogenic chemical compounds. The analysis also indicates Npy-linked pathways protect cells from inflammation.
Discussion
This study clearly showed that Npy is a neuropeptide that links DR to longevity in mammals. However, Npy is not required for many of the physiological adaptations to DR, as demonstrated by the changes in hypothalamic neuropeptide transcript levels, hormone concentrations and energy metabolism.
Among the neuroendocrine changes induced by DR, inhibition of anabolic signaling molecules, including insulin, GH/IGF-1 and mTOR and upregulation of adiponectin were found to extend lifespan in rodents without restricted food intake1,8,26,29,32. Thus, the effects of DR were attributed to these molecules or related signaling pathways based on the observed physiological adaptations. However, in the present study, the salutary effects of DR were significantly reduced in Npy−/− mice, even though they showed normal physiological adaptions to DR. Therefore, these neuroendocrine adaptations to DR may not be essential for longevity or cancer and stress resistance. Although the present findings do not directly support the evolutional view of the mechanisms of DR5,6, they reveal new roles of Npy as a lifespan and aging regulator.
Food efficiency was elevated by DR in WT and Npy−/− mice, although it was 6% and 10% lower in male and female Npy−/−-DR mice than in their respective WT-DR groups. Negative energy balance provokes adaptive responses to prevent an excess loss of body fat by decreasing energy expenditure, i.e., reducing thermogenesis and/or physical activity7. This adaptive response leads to elevated food efficiency and is induced by activation of hypothalamic Npy- and Agrp-synthesizing neurons and inhibition of signalling of α-melanocyte stimulating hormone or its precursor POMC by Agrp7. A recent study using the ILSXISS series of recombinant inbred strains of mice indicate a correlation between DR-induced longevity and greater fuel efficiency, defined by measures of weight maintenance and ability of maintaining hair and tail growth19. The life-extending effect of DR correlated inversely with fat reduction20. Therefore, the net reduction of food efficiency or a concomitant signal pathway might diminish the effects of DR.
The Npy pathways that induce the effects of DR remain elusive. Regarding the cancer-suppressing effects of DR, we speculate that Npy inhibits tumors by enhancing the detoxification or degradation of chemical compounds or xenobiotics (i.e., carcinogens and promoters) and inhibiting inflammatory processes (promoters), based on the hepatic gene expression profiles, because Npy could act in the liver via the sympathetic nerves and the circulation.
In Npy-overexpressing rats, it is thought that the sympatholytic and hypotensive effects of Npy protect the cardiovascular system from sympathetic hyperexcitation12. In this study, there were few cardiac lesions and they were not increased in Npy−/− mice. Thus, it seems unlikely that cardiac lesions affected the lifespan in Npy−/− mice. However, the potential role of Npy in the regulation of lifespan and ageing, via the sympathetic nervous system and/or circulation, remains to be elucidated.
Dysregulation of the hypothalamic-pituitary adrenal axis and increased noradrenergic activity causes neuropsychiatric disorders such as post-traumatic stress disorder39. In contrast, increased Npy levels in the brain and plasma are associated with resilience or improved recovery from the harmful effects of traumatic stress40. Thus, Npy−/− mice might remain under stressed conditions to an extent that diminishes the life-prolonging effect of DR. However, we observed no signs to indicate this kind of stress in Npy−/− mice. In fact, the plasma corticosterone level did not differ between Npy−/− and WT mice.
In the present study, the genetic backgrounds of the Npy−/− and WT (129S6/SvEvTac) mice were not identical. However, it should be emphasized that the life-extending and cancer-suppressing effects of DR are considerably diminished in Npy−/− mice when compared to the published data in a number of inbred strains as well as WT mice in the present study. Although a recent study indicated that some recombinant inbred strains do not respond to DR with lifespan extension41, to our knowledge, there has been no indication that the 129S substrains of mice are unresponsive to DR. In fact, the 129S1/SvImJ substrain, an approximate genetic match to Npytm1Rpa/J, is reported to respond to DR via physical fitness and antineoplastic effects15,42. Therefore, it is unlikely that genetic factors other than Npy confounded the present outcomes, although we cannot rule out this possibility.
Methods
Experimental animals and husbandry
The animal care and all experiments were performed in accordance with the guidelines approved by the Ethics Review Committee for Animal Experimentation at Nagasaki University. Male Npy−/− mice (129S-Npytm1Rpa/J) and female wild-type (WT) mice (129S6/SvEvTac), the strain used to derive the ES cell lines16, were obtained from Jackson Laboratory (Bar Harbor, ME, USA) and Taconic Farms, Inc. (Germantown, NY, USA). They were bred in a barrier facility at the Center for Frontier Life Sciences at Nagasaki University. After 2 generations of crossing male and female Npy+/− mice, offspring born between December 2006 and February 2007 were genotyped and set as longevity groups of Npy−/− and WT mice. Thus, Npy−/− and wild type (WT) control mice in this study were on a mixed 129S-Npytm1Rpa/J and 129S6/SvEvTac background.
Two or 3 mice (typically 3) were housed in individual cages in the barrier facility (temperature, 22–25°C; 12 h light/dark cycle) under specific pathogen-free conditions, which were maintained for the entire study. Sentinel animals were sent for bacterial and viral testing every 3 months43.
All mice were fed ad libitum (AL) with Charles River-LPF diet (Oriental Yeast Co. Ltd., Tsukuba, Japan). At 12 weeks of age, mice were divided into AL and DR groups. The DR groups received a food allotment consisting of 70% of the mean daily food intake of the AL groups of male and female Npy−/− and WT mice every day, 30 min before lights were turned off. The food allotments for the DR groups were adjusted every 2 weeks between 12 and 32 weeks; the allotments were fixed between 32 and 72 weeks and from 72 weeks and onward. Details of the feeding procedure have been described elsewhere44. Body weight was monitored every 2 weeks between 6 and 20 weeks of age and every 4 weeks thereafter. Dead mice were subjected to post-mortem examination.
Indirect calorimetry
In male 7-month-old mice, the respiratory quotient (RQ) and energy expenditure (EE) were measured in metabolic cages equipped with an indirect open-circuit calorimeter (Columbus Instruments, Columbus, OH). Experiments were initiated at 17:30, when an allotment was provided for each mouse in the DR group. Gas content was recorded for 10 seconds every 5 minutes between 18:00 and 15:00 on the following day, that is, for 21 hours. Data were averaged every 30 minutes. Lights were turned off at 20:00 and turned on at 8:00. The EE was calculated as the product of calorific value of oxygen and VO2 per kilogram body weight, where the calorific value of oxygen = 3.815 + 1.232*RQ. The EE was normalized to body weight45.
Body temperature
Body temperature was measured with rectal thermometers (TERUMO CTM-303: Terumo Corporation: Tokyo) following the procedure reported elsewhere46. The rectal probe (ME-PDK061: Terumo Corporation: Tokyo) was lubricated with glycerol and inserted ~3 cm into the rectum. The rectal temperature measurement procedure took 1 minute.
Enzyme-Linked Immunosorbent Assay (ELISA) for circulating hormone concentrations
Plasma samples collected from orbital blood were stored at −80°C. Plasma leptin and insulin concentrations were measured with ELISA kits (Shibayagi Co. Ltd., Gunma, Japan). Plasma adiponectin, IGF-1, Npy and corticosterone concentrations were measured by ELISA (adiponectin: Otsuka Pharmaceutical Co. Ltd., Tokyo, Japan, IGF-1 and Npy: Millipore, Billerica, MA, USA, corticosterone: Yanaihara Institute Inc., Fujinomiya, Shizuoka, Japan). All samples were tested in duplicate. All ELISA methods were performed according to manufacturer protocols.
Western blotting of ribosomal protein S6 kinase polypeptide 1 (Rps6kb1)
About 40 mg frozen liver tissues were homogenized in 800 μL T-PER buffer (PIERCE Biotechnology, Rockford, IL, USA) with a protease-inhibitor cocktail (P8340 Sigma-Aldrich, St. Louis, MO, USA) and a phosphatase-inhibitor cocktail (Nacalai Tesque Inc., Kyoto, Japan). The homogenates were centrifuged at 10000 × g for 5 min at 4°C and the supernatant was collected. Protein concentrations were measured using the BCA assay kit (PIERCE Biotechnology). All samples were mixed with Laemmli sample buffer and heated at 95°C for 5 min. Proteins (8 μg) were separated by 12.5% SDS-PAGE and transferred to PVDF membranes. The membranes were immediately placed in blocking solution (Blocking One-P for p-S6K or 3% non-fat dried milk in TBS-T buffer for Rps6kb1) for 60 min. The membranes were incubated with anti-phosphorylated Rps6kb1 (#2215; Cell Signaling Technology, Beverly, USA) or Rps6kb1 (#2217; Cell Signaling Technology) antibody diluted 1:1000 in TBS-T for 16 h at 4°C with gentle shaking and washed 3 times in TBS-T. Then, the membranes were incubated for 1 h with HRP-conjugated anti-rabbit IgG (Amersham Pharmacia Biotech, Little Chalfont, UK) diluted 1:10,000 in TBS-T. Immunoreactive proteins were visualized using ImmunoStar LD (Wako, Osaka, Japan) and quantitated using a densitometer (LAS-3000: Fuji Film, Tokyo, Japan) and MultiGauge software (Fuji Film). To minimize variations in signal intensity, a standard sample of p-Rps6kb1 was prepared from a WT-CR mouse liver and included in each blot.
Quantitative real-time (qRT) PCR for mRNA expression in tissues
Total RNA was purified from the hypothalamus, liver and brown adipose tissues47. The quality of extracted RNA was evaluated as the densitometric ratio of 28S and 16S ribosomal RNA. The extracted RNA was reverse-transcribed using a Bio-Rad reverse transcriptase reagent kit (Hercules, CA, USA) according to the manufacturer's instructions. Primers and probes were purchased from Applied Biosystems with reference to the TaqMan Gene Expression Assay library (Applied Biosystems, Tokyo, Japan) and TAKARA with reference to the Perfect Real Time support system (TAKARA BIO INC., Shiga, Japan) (see Supplementary Table S11). In PCR reactions with Takara primers, SYBR Green (ThunderBird SYBR qPCR Mix: TOYOBO Co. Ltd., Osaka, Japan) was used up to a final volume of 10 μL and reactions were performed according to the manufacturer's protocol. Control housekeeping genes [beta-actin (Actb), 18S ribosomal RNA (Rn18scc)] were also analyzed using TaqMan or SYBR Green primers and probes. All samples and standard curves were tested in triplicate. Amplification, data quantification and analysis were performed on an ABI PRISM 7900HT Sequence Detector (Applied Biosystems).
Gene expression profiling
RNA that was extracted from live tissues and that passed the Agilent Technologies 2100 Bioanalyzer quality control test was used to generate biotin-labelled cRNA with a MessageAmpTM II-Biotin Enhanced Kit (Ambion, Inc., Austin, TX). Labelled cRNA was applied to the CodeLink Mouse Whole Mouse Genome Bioarrays (Applied Microarrays, Inc., Tempe, AZ) for 18 hours at 37°C. Individual samples from 2 mice of each group were run on separate microarrays. After incubation, slides were washed, stained with Cy5-streptavidin (GE Healthcare Bio-Science Corp., Piscataway, NJ) and scanned by GenePix4000B (Molecular Devices LLC., Sunnyvale, CA). Array images were processed using CodeLink ™ Expression Analysis v5.0 (Applied Microarray Inc.) and each slide was subjected to quantile normalization. To identify genes differentially expressed between 2 groups of mice, the following filtering criteria were used; Quality flag, G (signal intensity was above noise levels), p-value less than 0.100, ratio of expression levels between 2 groups, over ±1.5-fold). With differential regulated genes, pathway analyses were performed with a gene set enrichment analysis (GSEA)32 using MsigDB [a nominal p < 0.01 and false discovery rate (FDR) q < 0.25; see Supplementary Tables S3, S4, S5, S6] and the NCBI BioSystems Database (nominal p < 0.01: see Supplementary Tables S7, S8, S9, S10).
The data set for these DNA microarrays has been deposited at the National Center of Biotechnology Information Gene Expression Omnibus (GEO; http://WWW.ncbi.nlm.nih.gov/geo/) and is accessible through GEO Series accession number GSE42761.
Microarray data were verified by qRT-PCR of 19 selected genes using RNA samples extracted from mice in the same groups used for the microarray analysis (see Supplementary Table S11). A scatter plot between the fold changes (Npy−/−-DR versus WT-DR) calculated by the microarray data and qRT-PCR data was drawn and analysed with a linear regression model (Supplementary Fig. S3).
Statistical analysis
Data were expressed as the mean and standard error (SE). Food consumption (FC), body weight (BW) and FC/BW and RQ were analyzed by 2-factor (2-f) analysis of variance (ANOVA) for the effects of genotype and age and their interaction (Genotype, Age, Genotype × Age). The Bonferroni post hoc test was performed to compare data between groups at each age point. Survival curves were estimated using the Kaplan–Meier method and were compared with the Log-rank test. A multivariate analysis with the Cox proportional-hazards model was also used to estimate the hazard ratios for mortality as variables: Genotype (Npy−/− vs. WT), Diet (DR vs. AL) and Sex (male and female). The proportion of mice bearing tumors and the prevalence of selected lesions were analyzed using chi-square or Fisher's exact tests. The levels of mRNA expression, circulating hormones and protein abundance were analyzed by one-factor ANOVA and the post hoc (Newman-Keuls multiple comparison) test when needed. Regression analysis of the scatter plot of EE and body weight and analysis of covariance (ANCOVA) were performed to determine the relationship of EE and body mass within each group and to test whether this relationship differs between groups44. All statistical analyses were performed using Prism 5 for Mac OS X (Graphpad Software, Inc.) and STATVIEW 5.0 software (SAS Institute Inc., Cary, NC). P values < 0.05 were considered statistically significant.
References
Mair, W. & Dillin, A. Aging and survival: the genetics of life span extension by dietary restriction. Annu. Rev. Biochem. 77, 727–754 (2008).
Weindruch, R. & Walford, R. L. [Dietary restriction: Effects on survivorship]. [31–72] The Retardation of Aging and Disease by Dietary Restriction. (Charles C Thomas Pub. Ltd., Springfield, 1988).
Colman, R. J. et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 325, 201–204 (2009).
Mattison, J. A. et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 489, 318–322 (2012).
Holliday, R. Food, reproduction and longevity: is the extended lifespan of calorie-restricted animals an evolutionary adaptation? Bioessays 10, 125–127 (1989).
Masoro, E. J. & Austad, S. N. The evolution of the antiaging action of dietary restriction: a hypothesis. J. Gerontol. A Biol. Sci. Med. Sci. 51, B387–91 (1996).
Sainsbury, A. & Zhang, L. Role of the hypothalamus in the neuroendocrine regulation of body weight and composition during energy deficit. Obes. Rev. 12, 234–257 (2011).
Shimokawa, I., Chiba, T., Yamaza, H. & Komatsu, T. Longevity genes: insights from calorie restriction and genetic longevity models. Mol. Cells 26, 427–435 (2008).
Luque, R. M., Park, S. & Kineman, R. D. Severity of the catabolic condition differentially modulates hypothalamic expression of growth hormone-releasing hormone in the fasted mouse: potential role of neuropeptide Y and corticotropin-releasing hormone. Endocrinology 148, 300–309 (2007).
Okada, K., Sugihara, H., Minami, S. & Wakabayashi, I. Effect of parenteral administration of selected nutrients and central injection of gamma-globulin from antiserum to neuropeptide Y on growth hormone secretory pattern in food-deprived rats. Neuroendocrinology 57, 678–686 (1993).
Tamura, H. et al. The effect of agouti-related protein on growth hormone secretion in adult male rats. Regul Pept 125, 145–149 (2005).
Michalkiewicz, M., Knestaut, K. M., Bytchkova, E. Y. & Michalkiewicz, T. Hypotension and reduced catecholamines in neuropeptide Y transgenic rats. Hypertension 41, 1056–1062 (2003).
Smiałowska, M. et al. Neuroprotective effects of neuropeptide Y-Y2 and Y5 receptor agonists in vitro and in vivo. Neuropeptides 43, 235–249 (2009).
Masoro, E. J. Influence of caloric intake on aging and on the response to stressors. J. Toxicol. Environ. Health B Crit. Rev. 1, 243–257 (1998).
Minor, R. K. et al. The arcuate nucleus and neuropeptide Y contribute to the antitumorigenic effect of calorie restriction. Aging Cell 10, 483–492 (2011).
Erickson, J. C., Hollopeter, G. & Palmiter, R. D. Attenuation of the obesity syndrome of ob/ob mice by the loss of neuropeptide Y. Science 274, 1704–1707 (1996).
Gunapala, K. M., Gallardo, C. M., Hsu, C. T. & Steele, A. D. Single gene deletions of orexin, leptin, neuropeptide Y and ghrelin do not appreciably alter food anticipatory activity in mice. PLoS ONE 6, e18377 (2011).
Weindruch, R. & Walford, R. L. [Mechanisms: How does dietary restriction retard aging?]. [231–294] The Retardation of Aging and Disease by Dietary Restriction. (Charles C Thomas Pub. Ltd., Springfield, 1988).
Rikke, B. A. et al. Genetic dissection of dietary restriction in mice supports the metabolic efficiency model of life extension. Exp Gerontol. 45, 691–701 (2010).
Liao, C. Y. fat maintenance is a predictor of the murine lifespan response to dietary restriction. Aging Cell 10, 629–639 (2010).
Bruss, M., Khambatta, C., Ruby, M., Aggarwal, I. & Hellerstein, M. Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates. Am. J. Physiol. Endocrinol. Metab. 298, E108 (2010).
Guarente, L. Mitochondria–a nexus for aging, calorie restriction and sirtuins? Cell 132, 171–176 (2008).
Currie, P. J., Mirza, A., Fuld, R., Park, D. & Vasselli, J. R. Ghrelin is an orexigenic and metabolic signaling peptide in the arcuate and paraventricular nuclei. Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R353–R358 (2005).
Granneman, J. G., Li, P., Zhu, Z. & Lu, Y. Metabolic and cellular plasticity in white adipose tissue I: effects of beta3-adrenergic receptor activation. Am. J. Physiol. Endocrinol. Metab. 289, E608–16 (2005).
Fu, Y. T., He, F. S., Zhang, S. L. & Zhang, J. S. Lipid peroxidation in rats intoxicated with 3-nitropropionic acid. Toxicon. 33, 327–331 (1995).
Nelson, J. F., Strong, R., Bokov, A., Diaz, V. & Ward, W. Probing the relationship between insulin sensitivity and longevity using genetically modified mice. J. Gerontol. A Biol. Sci. Med. Sci. 67, 1332–1338 (2012).
Walker, G., Houthoofd, K., Vanfleteren, J. R. & Gems, D. Dietary restriction in C. elegans: from rate-of-living effects to nutrient sensing pathways. Mech. Ageing Dev. 126, 929–937 (2005).
Kapahi, P. et al. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr. Biol. 14, 885–890 (2004).
Selman, C. et al. Ribosomal protein S6 kinase 1 signaling regulates mammalian life span. Science 326, 140–144 (2009).
Yamaza, H. et al. Involvement of insulin-like growth factor-1 in the effect of caloric restriction: regulation of plasma adiponectin and leptin. J. Gerontol. A Biol. Sci. Med. Sci. 62, 27–33 (2007).
Hui, X., Lam, K. S. L., Vanhoutte, P. M. & Xu, A. Adiponectin and cardiovascular health: an update. Br. J. Pharmacol. 165, 574–590 (2012).
Otabe, S. et al. Overexpression of human adiponectin in transgenic mice results in suppression of fat accumulation and prevention of premature death by high-calorie diet. Am. J. Physiol. Endocrinol. Metab. 293, E210–8 (2007).
Sabatino, F., Masoro, E. J., McMahan, C. A. & Kuhn, R. W. Assessment of the Role of the Glucocorticoid System in Aging Processes and in the Action of Food Restriction. J. Gerontol. 46, B171–B179 (1991).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Lee, J.-S. et al. Application of comparative functional genomics to identify best-fit mouse models to study human cancer. Nat. Genet. 36, 1306–1311 (2004).
Rickman, D. S. et al. Prediction of future metastasis and molecular characterization of head and neck squamous-cell carcinoma based on transcriptome and genome analysis by microarrays. Oncogene 27, 6607–6622 (2008).
Ichiba, T. et al. Early changes in gene expression profiles of hepatic GVHD uncovered by oligonucleotide microarrays. Blood 102, 763–771 (2003).
Schumacher, B. et al. Delayed and accelerated aging share common longevity assurance mechanisms. PLoS Genet. 4, e1000161 (2008).
Skelton, K. et al. PSTD and gene variants: New pathways and new thinking. Neuropharmacology 62, 628–637 (2012).
Sah, R. & Geracioti, T. D. Neuropeptide Y and posttraumatic stress disorder. Mol. Psychiatry 18, 646–655 (2013).
Liao, C. Y. et al. Genetic variation in the murine lifespan response to dietary restriction: from life extension to life shortening. Aging Cell 9, 92–95 (2010).
Minor, R. K. et al. Calorie restriction alters physiological performance but not cognition in two models of altered neuroendocrine signaling. Behav. Brain Res. 189, 202–211 (2008).
Shimokawa, I. et al. Life span extension by reduction in growth hormone-insulin-like growth factor-1 axis in a transgenic rat model. Am. J. Pathol. 160, 2259–2265 (2002).
Yamaza, H. et al. FoxO1 is involved in the antineoplastic effect of calorie restriction. Aging Cell 9, 372–382 (2010).
Arch, J. R. S., Hislop, D., Wang, S. J. Y. & Speakman, J. R. Some mathematical and technical issues in the measurement and interpretation of open-circuit indirect calorimetry in small animals. Int. J. Obes. Relat. Metab. Disord. 30, 1322–1331 (2006).
Saegusa, Y. & Tabata, H. Usefulness of infrared thermometry in determining body temperature in mice. J. Vet. Med. Sci. 65, 1365–7 (2003).
Chiba, T. et al. Similar metabolic responses to calorie restriction in lean and obese Zucker rats. Mol. Cell. Endocrinol. 309, 17–25 (2009).
Acknowledgements
We are grateful to the staff at the Laboratory Animal Center for Biomedical Research at the Center for Frontier Life Sciences, Nagasaki University for their animal care and technical assistance. We also thank Yutaka Araki, Yuko Moriyama and Rieko Tahara for their excellent technical assistance. The study was supported by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (I.S., nos. 19590227; T.C., nos. 24659181 and 25282027), the 2012 Danone Institute of Japan Foundation Research Grant (T.C.) and the JSPS Asian CORE Program (I.S., nos. FY2010-5).
Author information
Authors and Affiliations
Contributions
T.C. and T. Komatsu performed most of the experiments. Y.T. and R.F. performed most of the RT-PCR analysis. D.P. and H.Y.C. analyzed the microarray data. T. Kusudo and H.Y. conducted the indirect calorimetry experiment. S.J.P. measured the rectal temperature and served as advisor for the qRT-PCR in fat tissues. S.E.K., H.H. and R.M. served as advisors for the pathological analysis, animal care and provide knowledge of genotyping and western blots. I.S. was the overall director of the research, provided funds for the research (T.C., H.H. and R.M. also provided some funds) and contributed to the statistical analysis and writing and editing of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplemental Information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported license. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Chiba, T., Tamashiro, Y., Park, D. et al. A key role for neuropeptide Y in lifespan extension and cancer suppression via dietary restriction. Sci Rep 4, 4517 (2014). https://doi.org/10.1038/srep04517
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04517
This article is cited by
-
Comparing the Effects of Melatonin with Caloric Restriction in the Hippocampus of Aging Mice: Involvement of Sirtuin1 and the FOXOs Pathway
Neurochemical Research (2018)
-
The pathophysiology of defective proteostasis in the hypothalamus — from obesity to ageing
Nature Reviews Endocrinology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.