Abstract
Lithium elemental and isotopic compositions of olivines in peridotite xenoliths from Hebi in the North China Craton provide direct evidence for the highly variable δ7Li in Archean lithospheric mantle. The δ7Li in the cores of olivines from the Hebi high-Mg# peridotites (Fo > 91) show extreme variation from −27 to +21, in marked deviation from the δ7Li range of fresh MORB (+1.6 to +5.6) although the Li abundances of the olivines are within the range of normal mantle (1–2 ppm). The Li abundances and δ7Li characteristics of the Hebi olivines could not have been produced by recent diffusive-driven isotopic fractionation of Li and therefore the δ7Li in the cores of these olivines record the isotopic signature of the subcontinental lithospheric mantle. Our data demonstrate that abnormal δ7Li may be preserved in the ancient lithospheric mantle as observed in our study from the central North China Craton, which suggest that the subcontinental lithospheric mantle has experienced modification of fluid/melt derived from recycled oceanic crust.
Similar content being viewed by others
Introduction
Lithium has proven to be an important geochemical tracer for fluid-related processes in the Earth's surface as well as crust-mantle recycling1,2 because of its moderate incompatibility during mantle melting3, strong fluid mobility4 and the large mass fractionation, with up to 80‰ variation of 7Li/6Li in terrestrial samples1. In contrast to the large variations in Li isotope compositions of the Earth's surface environments, the δ7Li of mantle is more restricted, in the range of about +4 ± 2 as inferred from relatively pristine olivines and fresh MORB5,6,7,8.
The Li isotopic heterogeneity in the mantle caused by fluid/melt-rock interaction can be rehomogenized by diffusion over geologically short time periods due to the fast diffusion of Li7,9,10,11,12,13,14 and thus the mantle is broadly characterized by homogeneous Li isotope composition. However, several studies have suggested the existence of abnormal δ7Li in the mantle, such as high δ7Li (up to +7.9) of HIMU lavas15,16 and low δ7Li (lower than −7.0) of mantle-derived minerals17,18,19,20 and lavas21, which are considered to result from isotope fractionation during the dehydration of subducted oceanic crust22,23. To date, the data on abnormal δ7Li are mainly recorded from indirect samples of the mantle or directly on clinopyroxene separates in peridotites, although the latter are susceptible to recent diffusive fractionation of Li isotopes during the entrainment and transportation of the xenoliths by host magma due to the much faster diffusion of Li in pyroxene than in olivine9,24,25. In contrast, the Li isotope compositions in the cores of large olivine grains from peridotite xenoliths, which represent direct samples of the upper mantle, provide more robust signature of the lithospheric mantle.
In this study, we report the anomalous Li isotope compositions of olivines in a suite of peridotite xenoliths from the Hebi locality in the central North China Craton, one of the world's oldest continental nuclei26 (Fig. 1). Our data bring out the largest variation in δ7Li values among those published for olivines from peridotite xenoliths7,27,28 and demonstrate the existence of abnormal δ7Li in the ancient subcontinental lithospheric mantle.
Sketch map showing the major tectonic units of the North China Craton and the localities of Hebi xenolith and Paleozoic kimberlites (tectonic units revised after Zhao et al.48.
The inset shows location of the North China Craton (NCC).
Results
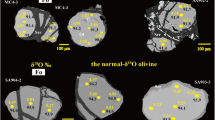
The peridotite xenoliths in the Hebi Cenozoic basalts (about 4 Ma) of present study are remarkably fresh and about 5 cm in diameter. They are coarse-grained with olivine grains typically ranging in size from 3 to 6 mm in diameter, with a maximum up to 10 mm. Compositionally, the rocks are dominantly high-Mg# (Fo > 91) harzburgites with one low-Mg# (Fo < 91) lherzolite (HB29; Supplementary Table 1). The high-Mg# harzburgite xenoliths have been interpreted as shallow relics of the Archean cratonic mantle beneath the North China Craton and the low-Mg# lherzolite may represent the newly-formed29 or modified lithospheric mantle30. The olivines in the high-Mg# harzburgites show highly variable δ7Li. The grain cores have Li abundances of 0.9 to 2.2 ppm and δ7Li ranging from −27 to +22 and of the rims show 0.9 to 2.3 ppm and δ7Li of −38 to +20, respectively. The Li abundance and δ7Li of olivine in the low-Mg# lherzolite are 1.9 ppm and +1.2 in the core and 3.6 ppm and −13 in the rim, showing high-δ7Li core with lower Li abundance as compared to the low-δ7Li rim (Fig. 2).
Variation of δ7Li with Li abundances of olivines in the Hebi xenoliths compared to the published data for olivines from worldwide peridotites.
Data sources: Seitz et al.5, Magna et al.6, Jeffcoate et al.7, Rudnick and Ionov10, Halama et al.34, Tang et al.18,19,20, Wagner and Deloule49, Ionov and Seitz12, Aulbach and Rudnick13, Zhang et al.46, Gao et al.36. The field for normal mantle is based on the compositions of relatively pristine olivines and fresh MORB5,6,7,8. The dashed lines link the core and rim of each olivine.
Discussion
The 6Li is predicted to have high diffusivity, about 3% faster than 7Li based due to a mass difference of 16.7%31. Thus, large Li isotopic fractionations can be produced by diffusion during processes of fluid/melt-rock reaction7,10,32. Hence, diffusion-driven fractionation of Li isotopes has been invoked to explain the Li isotopic variations in peridotites, a process that could occur during rock-melt/fluid reaction prior to or coincident with the entrainment into host magmas and the transport of the mantle xenolith to the surface10,13,33,34,35, as well as during the cooling of peridotites12,14,36. The results of diffusive fractionation are reflected in the variations of δ7Li from meter to micron scale7,9,19,32,37. Thus, an important point to evaluate is whether the δ7Li values obtained from the olivines provide robust signature of the lithospheric mantle.
Modeling of diffusive fractionation of Li isotopes generally assumes ingress of Li into rocks or minerals from a source of Li, such as Li diffusing into peridotite from melt32, Li diffusing into country rocks from Li-rich pegmatite37, or into clinopyroxene from coexisting olivine during cooling of peridotites14. In the case of Li addition to peridotites, the mantle minerals first become enriched in 6Li because of its greater diffusion rate than 7Li, leading to low δ7Li relative to its precursor. This mechanism can account for the observations of isotopically heavy cores with lower Li concentrations than the light rims of minerals7,10,25, such as the case of the low-Mg# sample HB29 in our present study (Fig. 2) and suggest recent diffusive ingress of Li into the rim of olivine. However, the model of diffusive ingress of Li does not adequately explain the characteristics of Li abundances and δ7Li of olivines in the high-Mg# peridotites because (1) the Li content does not show enrichment and the values are close to that of the normal mantle of 1–2 ppm38,39; (2) there is no apparent difference in Li abundances and δ7Li between the rims and the cores of most olivines from the high-Mg# samples; and (3) olivine from the sample 05HB72 has a Li abundance of 1 ppm and much higher δ7Li (+20) than the normal mantle, further excluding the possibility of recent diffusive ingress of Li.
In the case of Li diffusion into clinopyroxene from coexisting olivine during the cooling of peridotites, the olivine should have a δ7Li value that is significantly higher than the normal mantle36. However, most of the samples in our study show very low δ7Li in olivines. This contradiction excludes the possibility that the δ7Li of olivines are the results of diffusive fractionation of Li isotopes during the cooling of peridotites.
We therefore conclude that the δ7Li in the cores of large olivine grains from the Hebi peridotites reflect the signatures of the lithospheric mantle beneath the North China Craton. Compared to the published δ7Li of olivines from worldwide peridotites (Fig. 3), the δ7Li in the cores of olivines from the Hebi peridotites show extreme variation from −27 to +21, far beyond the δ7Li range (+1.6 to +5.6) of fresh MORB8. These observations suggest the existence of abnormal δ7Li in the old lithospheric mantle beneath the craton.
Histograms of δ7Li for olivines in the Hebi xenoliths and those from worldwide peridotites.
Data sources as in Fig. 2.
Another important question to be addressed relates to whether the survival of δ7Li anomaly in the mantle over a long time scale is theoretically possible. Equilibration temperatures for the Hebi peridotites have been estimated using two-pyroxene thermometer of Wells40 as recommended in Zheng et al.29. The estimated equilibrium temperatures of the harzburgites vary from about 900 to 1150°C and that of the lherzolite is 1020°C, which are consistent with those estimated previously for the peridotites from the same locality29. The partitioning experiments of Li between plagioclase and clinopyroxene showed that Li preferentially partitions into clinopyroxene as temperature decreases between 900 and 1200°C41. As a result, we infer that the estimated equilibrium temperatures exceed the closure temperatures for Li in clinopyroxene and the system of Li in the peridotite remains open although the closure temperature for Li in olivine is unknown. At the estimated mantle temperatures, Li heterogeneities are likely to be rehomogenized through time11. However, based on experimental studies, Dohmen et al.24 proposed two mechanisms of Li diffusion, a fast (interstitial site) mechanism and a slow (vacancy, octahedral site) mechanism and suggested that the former is unlikely to be dominant in most natural systems. The diffusion of Li in the slow mechanism is about an order of magnitude faster than that of Fe, Mg and most other divalent cations in olivine; such diffusion of Li in olivine will be much slower than that in clinopyroxene at the same conditions24. Another study of an olivine xenocryst suggested that the diffusion coefficients of Li in olivine fell within a factor of three of the Mg-Fe interdiffusion coefficient42. This is generally in agreement with the experimental observations that the diffusion of Li into olivine is similar to that of divalent cations9,24,43. Calculations based on the diffusion coefficient of Li in clinopyroxene suggested that a kilometer-thick altered oceanic crust could lose most of the excess Li in a few tens of millions years11. On the other hand, the rocks can preserve 7Li/6Li distinctive from the ambient mantle over a time period over 1,500 Ma16 regardless of the model based on the fast mechanism of diffusion41. A recent experimental study shows that very large Li isotopic fractionations persisted after the Li concentration had become effectively homogenized, which suggests that it takes much longer for the isotopic composition to become uniform compared to the time it takes for diffusion to homogenize the Li concentration25. Therefore, the survival of δ7Li anomaly in the mantle over a relatively long time scale is possible, as also evidenced from Li isotopic variations in the Trinity Ophiolite32 and peridotite xenoliths from the northeastern North China Craton20.
It has been proposed that abnormal δ7Li in lithospheric mantle could be produced by infiltration of mantle peridotites by isotopically normal fluid/melt derived from the asthenosphere10,13,32 and δ7Li-anomaly melt/fluid related to subducted slab16,17,18,20,21. For the first possibility, the normal mantle-derived fluid/melt could produce the δ7Li of olivines higher than −10 (published data in Figs. 2 and 3), which cannot account for the low δ7Li of −30 in the cores of olivines. Furthermore, olivines in the peridotites/pyroxenites metasomatized by the normal mantle-derived fluid/melt should have higher Li contents than that of the normal mantle20, which is contrast to our observations. Therefore, the δ7Li-anomaly can be related to the subducted slab because the fluids released by the dehydration of subducted slab are rich in 7Li, whereas the slab tends to be depleted in 7Li. The low-δ7Li component is subducted into the deeper mantle to form a low-δ7Li reservoir, which could be the potential source of low δ7Li22,23. However, it has been proposed that dehydration can only produce δ7Li decrease of up to 3‰ and that the deeply subducted eclogites possess higher δ7Li than the mantle44. Thus, the isotopic composition of the upper mantle with contributions of recycled components by subduction remains speculative and the mechanism of origin of abnormal δ7Li in the lithospheric mantle is equivocal. However, experiments have shown that δ7Li of recycled components depend on the compositions of subducted materials and concomitant dehydration of metabasic oceanic crust, containing chlorite and clinopyroxene with Li in six-fold coordination, releases fluids enriched in Li and 7Li into the fore-arc mantle and thus introduces a low δ7Li component into the deeper mantle23,45. Therefore, the abnormal δ7Li of olivines in the harzburgites indicate that the subcontinental lithospheric mantle experienced the modification of fluid/melt derived from recycled oceanic crust.
Methods
Li concentrations and isotopic ratios were measured on gold-coated thin sections using Cameca IMS-1280 ion microprobe at the Institute of Geology and Geophysics, Chinese Academy of Sciences, following the procedures described in Zhang et al.46. Li isotope compositions are expressed as δ7Li ( = [(7Li/6Li)sample/(7Li/6Li)standard − 1] × 1000) relative to the National Institute of Standards and Technology standard L-SVEC with 7Li/6Li of 12.0192 and shown in Supplementary Table 1. The olivine sample BZ2947 and in-house standard of our Lab, Ol334, were used as standards to correct the measured data. For the two standards our δ7Li are +4.9 ± 0.9 and −3.1 ± 0.9, respectively, well consistent with the recommended values (+4.4 and −3.2) within analytical error. The external 2σ errors of the isotope compositions for both the standards and the samples are less than 2.7‰, with the majority less than 2.0‰. The measured olivine grains in our samples are homogeneous in major elemental compositions at a mineral scale and show Fo values similar to that of the standards. Thus, matrix effect can be excluded in our measurements and the obtained data are reliable.
References
Tomascak, P. B. Developments in the understanding and application of lithium isotopes in the earth and planetary sciences. in Geochemistry of non-traditional stable isotope: Reviews in Mineralogy and Geochemistry, Vol. 55 (eds. Johnson, C. M., Beard, B. I. & Albarede, F.) 153–195 (Mineral Soc. Am., Washington, D.C., 2004).
Elliott, T., Thomas, A., Jeffcoate, A. & Niu, Y. L. Lithium isotope evidence for subduction-enriched mantle in the source of mid-ocean-ridge basalts. Nature 443, 565–568 (2006).
Brenan, J. M. et al. Behaviour of boron, beryllium and Lithium during melting and crystallization: constraints from mineral-melt partitioning experiments. Geochim. Cosmochim. Acta 62, 2129–2141 (1998).
Brenan, J. M., Ryerson, F. J. & Shaw, H. F. The role of aqueous fluids in the slab-to-mantle transfer of boron, beryllium and lithium during subduction: experiments and models. Geochim. Cosmochim. Acta 62, 3337–3347 (1998).
Seitz, H. M., Brey, G. P., Lahaye, Y., Durali, S. & Weyer, S. Lithium isotopic signatures of peridotite xenoliths and isotopic fractionation at high temperature between olivine and pyroxenes. Chem. Geol. 212, 163–177 (2004).
Magna, T., Wiechert, U. & Halliday, A. N. New constraints on the lithium isotope compositions of the Moon and terrestrial planets. Earth Planet. Sci. Lett. 243, 336–353 (2006).
Jeffcoate, A. B. et al. Li isotope fractionation in peridotites and mafic melts. Geochim. Cosmochim. Acta 71, 202–218 (2007).
Tomascak, P. B., Langmuir, C. H., le Roux, P. J. & Shirey, S. B. Lithium isotopes in global mid-ocean ridge basalts. Geochim. Cosmochim. Acta 72, 1626–1637 (2008).
Parkinson, I. J., Hammond, S. J., James, R. H. & Rogers, N. W. High-temperature lithium isotope fractionation: Insights from lithium isotope diffusion in magmatic systems. Earth Planet. Sci. Lett. 257, 609–621 (2007).
Rudnick, R. L. & Ionov, D. A. Lithium elemental and isotopic disequilibrium in minerals from peridotite xenoliths from far-east Russia: Product of recent melt/fluid-rock reaction. Earth Planet. Sci. Lett. 256, 278–293 (2007).
Halama, R., McDonough, W. F., Rudnick, R. L. & Bell, K. Tracking the lithium isotopic evolution of the mantle using carbonatites. Earth Planet. Sci. Lett. 265, 726–742 (2008).
Ionov, D. A. & Seitz, H. M. Lithium abundances and isotopic compositions in mantle xenoliths from subduction and intra-plate settings: Mantle sources vs. eruption histories. Earth Planet. Sci. Lett. 266, 316–331 (2008).
Aulbach, S. & Rudnick, R. L. Origins of non-equilibrium lithium isotopic fractionation in xenolithic peridotite minerals: Examples from Tanzania. Chem. Geol. 258, 17–27 (2009).
Gallagher, K. & Elliott, T. Fractionation of lithium isotopes in magmatic systems as a natural consequence of cooling. Earth Planet. Sci. Lett. 278, 286–296 (2009).
Chan, L. H., Lassiter, J. C., Hauri, E. H., Hart, S. R. & Blusztajn, J. Lithium isotope systematics of lavas from the Cook-Austral Islands: Constraints on the origin of HIMU mantle. Earth Planet. Sci. Lett. 277, 433–442 (2009).
Vlastélic, I., Koga, K., Chauvel, C., Jacques, G. & Télouk, P. Survival of lithium isotopic heterogeneities in the mantle supported by HIMU-lavas from Rurutu Island, Austral Chain. Earth Planet. Sci. Lett. 286, 456–466 (2009).
Nishio, Y. et al. Lithium isotopic systematics of the mantle-derived ultramafic xenoliths: implications for EM1 origin. Earth Planet. Sci. Lett. 217, 245–261 (2004).
Tang, Y. J. et al. Lithium isotopic systematics of peridotite xenoliths from Hannuoba, North China Craton: Implications for melt-rock interaction in the considerably thinned lithospheric mantle. Geochim. Cosmochim. Acta 71, 4327–4341 (2007).
Tang, Y. J., Zhang, H. F., Nakamura, E. & Ying, J. F. Multistage melt/fluid-peridotite interactions in the refertilized lithospheric mantle beneath the North China Craton: constraints from the Li–Sr–Nd isotopic disequilibrium between minerals of peridotite xenoliths. Contrib. Mineral. Petrol. 161, 845–861 (2011).
Tang, Y. J. et al. Slab-derived lithium isotopic signatures in mantle xenoliths from northeastern North China Craton. Lithos 149, 79–90 (2012).
Agostini, S., Ryan, J. G., Tonarini, S. & Innocenti, F. Drying and dying of a subducted slab: Coupled Li and B isotope variations in Western Anatolia Cenozoic Volcanism. Earth Planet. Sci. Lett. 272, 139–147 (2008).
Zack, T., Tomascak, P. B., Rudnick, R. L., Dalpé, C. & McDonough, W. F. Extremely light Li in orogenic eclogites: the role of isotope fractionation during dehydration in subducted oceanic crust. Earth Planet. Sci. Lett. 208, 279–290 (2003).
Wunder, B. et al. Lithium isotope fractionation between Li-bearing staurolite, Li-mica and aqueous fluids: An experimental study. Chem. Geol. 238, 277–290 (2007).
Dohmen, R., Kasemann, S. A., Coogan, L. & Chakraborty, S. Diffusion of Li in olivine. Part I: Experimental observations and a multi species diffusion model. Geochim. Cosmochim. Acta 74, 274–292 (2010).
Richter, F., Watson, B., Chaussidon, M., Mendybaev, R. & Ruscitto, D. Lithium Isotope Fractionation by Diffusion in Minerals. Part 1: Pyroxenes. Geochim. Cosmochim. Acta 126, 352–370 (2014).
Liu, D. Y., Nutman, A. P., Compston, W., Wu, J. S. & Shen, Q. H. Remnants of ≥3800 Ma crust in the Chinese part of the Sino-Korean craton. Geology 20, 339–342 (1992).
Bell, D. R., Hervig, R. L. & Buseck, P. R. Li isotope heterogeneity in mantle-derived xenoliths. EOS 85, V51C–0591 (2004).
Bell, D. R., Hervig, R. L., Buseck, P. R. & Aulbach, S. Lithium isotope analysis of olivine by SIMS: Calibration of a matrix effect and application to magmatic phenocrysts. Chem. Geol. 258, 5–16 (2009).
Zheng, J. P. et al. Relict refractory mantle beneath the eastern North China block: significance for lithosphere evolution. Lithos 57, 43–66 (2001).
Tang, Y. J. et al. Highly heterogeneous lithospheric mantle beneath the Central Zone of the North China Craton evolved from Archean mantle through diverse melt refertilization. Gondwana Res. 23, 130–140 (2013).
Richter, F. M., Davis, A. M., Depaolo, D. J. & Watson, E. B. Isotope fractionation by chemical diffusion between molten basalts and rhyolite. Geochim. Cosmochim. Acta 67, 3905–3923 (2003).
Lundstrom, C. C., Chaussidon, M., Hsui, A. T., Kelemen, P. & Zimmerman, M. Observations of Li isotopic variations in the Trinity Ophiolite: Evidence for isotopic fractionation by diffusion during mantle melting. Geochim. Cosmochim. Acta 69, 735–751 (2005).
Aulbach, S., Rudnick, R. L. & McDonough, W. F. Li-Sr-Nd isotope signatures of the plume and cratonic lithospheric mantle beneath the margin of the rifted Tanzanian craton (Labait). Contrib. Mineral. Petrol. 155, 79–92 (2008).
Halama, R., Savov, I. P., Rudnick, R. L. & McDonough, W. F. Insights into Li and Li isotope cycling and sub-arc metasomatism from veined mantle xenoliths, Kamchatka. Contrib. Mineral. Petrol. 158, 197–222 (2009).
Pogge von Strandmann, P. A. E. et al. Variations of Li and Mg isotope ratios in bulk chondrites and mantle xenoliths. Geochim. Cosmochim. Acta 75, 5247–5268 (2011).
Gao, Y., Snow, J. E., Casey, J. F. & Yu, J. Cooling-induced fractionation of mantle Li isotopes from the ultraslow-spreading Gakkel Ridge. Earth Planet. Sci. Lett. 301, 231–240 (2011).
Teng, F.-Z., McDonough, W. F., Rudnick, R. L. & Walker, R. J. Diffusion-driven extreme lithium isotopic fractionation in country rocks of the Tin Mountain pegmatite. Earth Planet. Sci. Lett. 243, 701–710 (2006).
Seitz, H. M. & Woodland, A. B. The distribution of lithium in peridotitic and pyroxenitic mantle lithologies - an indicator of magmatic and metasomatic processes. Chem. Geol. 166, 47–64 (2000).
Woodland, A. B., Seitz, H. M. & Yaxley, G. M. Varying behaviour of Li in metasomatised spinel peridotite xenoliths from western Victoria, Australia. Lithos 75, 55–66 (2004).
Wells, P. R. A. Pyroxene thermometry in simple and complex systems. Contrib. Mineral. Petrol. 62, 129–139 (1977).
Coogan, L. A., Kasemann, S. A. & Chakraborty, S. Rates of hydrothermal cooling of new oceanic upper crust derived from lithium-geospeedometry. Earth Planet. Sci. Lett. 240, 415–424 (2005).
Qian, Q., O'Neill, H. S. C. & Hermann, J. Comparative diffusion coefficients of major and trace elements in olivine at ~950°C from a xenocryst included in dioritic magma. Geology 38, 331–334 (2010).
Spandler, C. & O'Neill, H. Diffusion and partition coefficients of minor and trace elements in San Carlos olivine at 1,300°C with some geochemical implications. Contrib. Mineral. Petrol. 159, 791–818 (2010).
Marschall, H. R., Pogge von Strandmann, P. A. E., Seitz, H. M., Elliott, T. & Niu, Y. The lithium isotopic composition of orogenic eclogites and deep subducted slabs. Earth Planet. Sci. Lett. 262, 563–580 (2007).
Wunder, B., Meixner, A., Romer, R. L. & Heinrich, W. Temperature-dependent isotopic fractionation of lithium between clinopyroxene and high-pressure hydrous fluids. Contrib. Mineral. Petrol. 151, 112–120 (2006).
Zhang, H. F., Deloule, E., Tang, Y. J. & Ying, J. F. Melt/rock interaction in remains of refertilized Archean lithospheric mantle in Jiaodong Peninsula, North China Craton: Li isotopic evidence. Contrib. Mineral. Petrol. 160, 261–277 (2010).
Decitre, S. et al. Behavior of Li and its isotopes during serpentinization of oceanic peridotites. Geochem. Geophy. Geosys. 3, 10.1029/2001GC000178 (2002).
Zhao, G. C., Wilde, S. A., Cawood, P. A. & Sun, M. Archean blocks and their boundaries in the North China Craton: lithological, geochemical, structural and P-T path constraints and tectonic evolution. Precam. Res. 107, 45–73 (2001).
Wagner, C. & Deloule, E. Behaviour of Li and its isotopes during metasomatism of French Massif Central lherzolites. Geochim. Cosmochim. Acta 71, 4279–4296 (2007).
Acknowledgements
This work was financially supported by the National Science Foundation of China (grants: 91214203, 91014007 and 41073028). We thank Xian-Hua Li, Guo-Qiang Tang, Yu Liu and Yan Hu for their assistance in measurement of Li isotopes.
Author information
Authors and Affiliations
Contributions
Y.J.T., H.F.Z. and E.D. contributed equally to idea development. Y.J.T. wrote the main text. B.X.S., J.F.Y., M.S. and Y.X. contributed equally to the intellectual growth of this paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary table 1
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Tang, YJ., Zhang, HF., Deloule, E. et al. Abnormal lithium isotope composition from the ancient lithospheric mantle beneath the North China Craton. Sci Rep 4, 4274 (2014). https://doi.org/10.1038/srep04274
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep04274
This article is cited by
-
Geochemical Behaviour and Influencing Factors of Salt-Forming Elements in Lithium-Rich Salt Lake Region: A Case Study from the Nalenggele River Basin, Qaidam Basin
Aquatic Geochemistry (2024)
-
Review on in situ Isotopic Analysis by LA-MC-ICP-MS
Journal of Earth Science (2023)
-
Widespread refertilization of cratonic lithospheric mantle related to circum-craton plate subduction: evidence from peridotite xenoliths from the central North China Craton
Contributions to Mineralogy and Petrology (2022)
-
Geochronological and geochemical characteristics of continental basalts of the eastern North China Craton: insights into crust–mantle interaction induced by continental subduction
Contributions to Mineralogy and Petrology (2022)
-
Nature and secular evolution of the lithospheric mantle beneath the North China Craton
Science China Earth Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.