Abstract
Mouse embryonic stem (ES) cell cultures exhibit heterogeneity and recently are discovered to sporadically enter the 2-cell (2C)-embryo state, critical for ES potency. Zscan4 could mark the sporadic 2C-state of ES cells. However, factors that regulate the Zscan4+/2C state remain to be elucidated. We show that Tbx3 plays a novel role in regulation of Zscan4+/2C state. Tbx3 activates 2-cell genes including Zscan4 and Tcstv1/3, but not vise versa. Ectopic expression of Tbx3 results in telomere elongation, consistent with a role for Zscan4 in telomere lengthening. Mechanistically, Tbx3 decreases Dnmt3b and increases Tet2 protein levels and reduces binding of Dnmt3b to subtelomeres, resulting in reduced DNA methylation and derepression of genes at subtelomeres, e.g. Zscan4. These data suggest that Tbx3 can activate Zscan4+/2C state by negative regulation of DNA methylation at repeated sequences, linking to telomere maintenance and self-renewal of ES cells.
Similar content being viewed by others
Introduction
Mouse ES cells are derived from the inner cell mass (ICM) of blastocysts and thought to be functionally equivalent to inner cell mass (ICM), harboring similar gene expression patterns compared to ICM cells1. Mouse ES cells are featured with pluripotency and self-renewal2,3. Another unique hallmark of mouse ES cells is their ability to defy cellular senescence and maintain exceptional genomic stability undergoing many cell divisions compared to other cell types4. It is also to note that ES cell cultures are a heterogeneous mixture of metastable cells with fluctuating expression of genes such as Zscan4, Zfp42/Rex1, Rest, Nanog, Stella (also known as Dppa3) and Esrrb1,5,6. Furthermore, mouse ES cells fluctuate with activation of 2-cell embryo specific genes (2C-genes) and endogenous transposable element (TE) activities7, suggesting that ES cells in the 2C-state might resemble the totipotent zygotes/2C-stage embryos. In this regard, the 2C-state was postulated as a “super” state of ES cells8. Interestingly, a large number of the genomic locations encoding the 2C-genes are enriched for TE-derived sequences. In addition, the 2C-genes are co-regulated with TEs7.
Zscan4, expressed specifically in 2-cell embryos and transiently in sporadic ES cells (1–5%) at any given time, marks a transient 2C-state of mouse ES cells4 and is required for preimplantation embryonic development and lengthening telomeres promptly by recombination-based mechanisms and maintaining genomic stability4,9,10. Zscan4+ and Zscan4− ES cells can interconvert to each other and nearly all ES cells activate Zscan4 at least once during nine passages4. Interestingly, Zscan4 can activate early embryonic genes, including 2C-genes and maintain genomic stability during generation of induced pluripotent stem (iPS) cells11,12. Moreover, Zscan4 levels affect pluripotency of ES cells13. Without intermittent activation of Zscan4, embryos delay preimlantation development and ES cells lose their ability to proliferate indefinitely4,9, suggesting that the equilibrium between the 2C-state and canonical ES state is essential for proper embryonic development. However, how Zscan4/2-cell state is regulated in ES cell cultures remains elusive and presumably multiple regulators may be involved in the regulation of 2-cell state. Here, we have identified Tbx3 as an epigenetic regulator of Zscan4+/2C state in ES cell cultures.
Results
Tbx3 is upregulated in 2C-state of mouse ES cell cultures
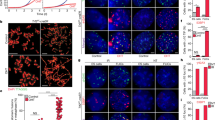
We hypothesized that the 2C-state regulators may exist within Zscan4+ cells/2-cell state and that Zscan4+ cells contain factors regulating Zscan4+ cell status. To search for factors that potentially regulate 2C-state, we thought to select genes prominently up-regulated in 2-cell embryos and 2C::tomato+ mouse ES cells compared to oocytes and 2C::tomato− mouse ES cells, respectively7. Zscan4, previously identified as a specific marker for two-cell embryo and mouse ES cells, highly expressed in 2C::tomato+ mouse ES cells while absent in 2C::tomato− mouse ES cells, can faithfully represent the 2C-state of mouse ES cells4,7,9. We also generated a Zscan4 promoter-driven EGFP (Zscan4-EGFP) ES cell line shown to closely recapitulate the endogenous Zscan4 expression4. Zscan4 was only expressed in a small subset of ES cells (Fig. 1a). ES cells were then sorted into Zscan4+ and Zscan4− populations by flow cytometry, the Zscan4-EGFP+ 2C-cell pool was limited to 1–5% (Fig. 1b), consistent with the recent report4. The selected genes including Tcstv1, Tcstv3, Dub1, Dazl, Ott and Tbx3 expressed at much higher levels in sorted Zscan4+ than in Zscan4− ES cells (Fig. 1c). These genes might be the potential regulators of Zscan4+/2C-state or downstream targets of Zscan4+/2C-state.
Tbx3 regulates Zscan4/2C genes.
(a) Zscan4-promoter-driven EGFP reporter in J1 ES cells shows that only a subpopulation of ES cells express Zscan4 at a given time. Nuclei stained by Hoechst 33342. Scale Bar = 5 μm. (b) Zscan4+ and Zscan4− ES cells were sorted by FACS. (c) Zscan4, Tcstv1, Tcstv3, Dub1, Dazl, Ott and Tbx3 up-regulated in Zscan4+ ES cells compared to Zscan4− ES cells. (d) Over-expression of Zscan4 in ES cells did not alter expression of above six genes. (e) Over-expression of Tcstv1, Tcstv3, Dub1, Dazl and Ott in ES cells did not change Zscan4 expression. (f) Tbx3 over-expression in ES cells up-regulated expression of Zscan4, Tcstv1, Tcstv3 and Dazl. (g) Immunostaining of Tbx3 (red) in Zscan4-EGFP ES cells. Nuclei stained by Hoechst 33342. Zscan4 positive (2C-state) ES cells showed higher expression of Tbx3. Scale Bar = 10 μm. Error bars indicate mean ± SEM (n = 3). **, p < 0.01; ***, p < 0.001, compared to controls.
To determine whether the six genes are downstream targets of Zscan4+/2C-state, we over-expressed Zscan4 in ES cells for 48 h and found that expression of genes Tcstv1, Tcstv3, Dub1, Dazl, Ott and Tbx3 did not differ between Zscan4 over-expressed ES cells and the mock ES cells (Fig. 1d), suggesting that Zscan4 itself does not activate Tcstv1, Tcstv3, Dub1, Dazl, Ott and Tbx3. To examine whether these six genes positively regulate Zscan4/2C-state in mouse ES cells, we transiently over-expressed each of the six genes alone in mouse ES cells and looked at expression levels of Zscan4. Overexpression of Tcstv1, Tcstv3, Dub1, Dazl or Ott did not change Zscan4 relative expression levels (Fig. 1e). However, forced expression of Tbx3 in mouse ES cells elevated expression levels of Zscan4 as well as Tcstv1, Tcstv3, Dub1 and Dazl (Fig. 1f). Tcstv1, Tcstv3, Dub1 all reportedly are 2-cell specific markers and Dazl also up-regulated in two-cell embryos7. Tbx3 was expressed in ES cells and expressed more in Zscan4-EGFP positive ES cells by immunofluorescence microscopy (Fig. 1g). Previous study also showed that Tbx3 are heterogeneously expressed in ES cell cultures by immunofluorescence5,14,15. These data suggest that Tbx3 might be a novel regulator of Zscan4/2C-state in mouse ES cells.
Furthermore, Tbx3 was up-regulated in zygotes and 2-cell embryos during mouse early embryo development (Supplementary Fig. 1a), consistent with previous report that Tbx3 was elevated in 2-cell embryos compared with oocytes7. We also verified that the 2-cell embryo specific genes Zscan4, Tcstv1 and Tcstv3 were elevated in 2-cell embryos, but greatly reduced after the 2-cell stage (Supplementary Fig. 1b–1d). It seemed that Tbx3 is expressed earlier, despite at relatively lower levels, than did other 2-cell genes.
Ectopic expression of Tbx3 activates Zscan4/2-cell state of mouse ES cell cultures
Transient ectopic expression of Tbx3 up-regulates Zscan4 (Fig. 1f). We also generated Tbx3 stable overexpression (OE) cell lines by electroporation. Morphologically, Tbx3 OE ES cells showed compacted cell colonies like mock ES cells electroporated with empty vector (Fig. 2a). Increased expression levels of Tbx3 and Zscan4 in Tbx3 OE cells were confirmed by immunofluorescence microscopy, quantitative real time PCR and western blot (Fig. 2b–2d). To examine the dynamics of Zscan4+/2C pool upon Tbx3 over-expression, ES cells were then sorted into Zscan4+ and Zscan4− populations by flow cytometry. Tbx3 over-expression only slightly increased Zscan4+/2C cell population, but notably more the fluorescence intensity of Zscan4+ cells (p < 0.0001) (Fig. 2e–2g). Tbx3 overexpression did not impact the cell cycle progression (Fig. 2h), nor Oct4 expression by immunofluorescence relative quantification estimated using ImageJ software (Fig. 2i, 2j). Furthermore, ectopic expression of Tbx3 did not alter expression of other pluripotency-associated genes by qPCR and immunofluorescence (Supplementary Fig. 2a, 2b), nor differentiation by standard in vitro embryoid body formation tests (Supplementary Fig. 3).
Tbx3 up-regulates Zscan4 and maintains normal cell cycle.
(a) Morphology of Tbx3 stable over-expressed ES cells and mock ES cells. (b–d) Confirmation of the over-expression of Tbx3 and Zscan4 in Tbx3 OE ES cells by immunofluorescence intensity (b), qPCR (c) and western blot (d). Full-length gel images are available in Supplementary Figure 5. ***, p < 0.001, compared to mocks. Scale bar = 100 μm. (e) Flow cytometry of Zscan4+ cells in transient Tbx3 over-expression in Zscan4-EGFP ES cells. Percentage and mean fluorescence intensity of Zscan4+ cells are indicated. (f) Tbx3 over-expression slightly increased proportion of Zscan4+ cells in ES cell population by FACS. **, p < 0.01, compared to mocks. (g) The mean fluorescence intensity (MFI) of Zscan4+ cells increased remarkably after Tbx3 over-expression. (h) Cell cycle analysis shows no significance difference between Tbx3 OE cells and mock ES cells. Error bars indicate mean ± SEM (n = 3 or 4). (i) Co-immunostaining of Tbx3 and Oct4 in Tbx3 OE ES cells compared with mock ES as controls. Scale bar = 10 μm. (j) Quantification of relative mean fluorescence intensity of Oct4 estimated by ImageJ software. n, number of cells counted.
Tbx3 plays a role in telomere length maintenance of mouse ES cells
Zscan4 is a specific marker for ES cells and the 2-cell embryos and required for telomere lengthening and genomic stability of ES cells by activating telomere sister chromatid exchange (T-SCE)4. Remarkably, telomeres lengthened rapidly in one- to two-cell stage embryos presumably through telomere recombination or T-SCE16. Both transient and stable Tbx3 overexpression up-regulates Zscan4 (Fig. 1f, Fig. 2). We hypothesized that Tbx3-induced activation of Zscan4+/2C-state genes may function in the telomere length regulation of ES cells. By telomere quantitative fluorescence In situ hybridization (Q-FISH) analysis17, telomeres were significantly (p < 0.0001) lengthened in Tbx3 OE ES cells compared to the mock ES cells following culture for 13 passages (83.65 ± 18.01 TFU in Tbx3 OE1 and 82.82 ± 17.00 TFU in Tbx3 OE2 cells vs 70.64 ± 16.49 TFU in mock ES cells) (Fig. 3a). The telomere QFISH data on telomere elongation in Tbx3 OE ES cells was further validated by quantitative real time PCR shown as T/S ratio18 (Fig. 3b).
Tbx3 elongates telomeres of ES cells and up-regulates Zscan4 in telomerase-deficient ES cells.
(a) Distribution histogram showing relative telomere length of Tbx3 overexpression (OE) ES cell lines compared to mock ES cells by telomere Q-FISH and the TFL-TELO software. TFU, arbitrary telomere fluorescence unit. Green line, median telomere length, which also is indicated on right hand corner. (b) qPCR analysis of telomere length shown as T/S ratio in Tbx3 OE ES cells compared with mock ES cells. (c) qPCR shows no significance difference in expression levels of Terc and Tert between Tbx3 OE ES cells and mock ES cells. (d) Zscan4 activation by Tbx3 over-expression is independent of telomerase involvement. Tbx3 over-expression up-regulates Zscan4 in WT ESC, G1 Terc−/− ESC and G4 Terc−/− ESC. (e) Expression levels of Tbx3 and Zscan4 in stable Tbx3 OE G1 Terc−/− ES cells by qPCR. (f) qPCR analysis of telomere length shown as T/S ratio of stable Tbx3 OE G1 Terc−/− ES cells compared with mock ES cells. Error bars indicate mean ± SEM (n = 3). *, p < 0.05; **, p < 0.01; ***, p < 0.001, compared to mocks.
Expression of telomerase subunit Tert and Terc remained at similar levels between Tbx3 OE ES cells and mock ES cells (Fig. 3c). Moreover, over-expression of Tbx3 in WT, telomerase-null G1 or G4 ES cells19 also led to increased expression of Zscan4 (Fig. 3d). These data suggest that Tbx3 overexpression does not significantly increase telomerase activity and that Tbx3 can still up-regulates Zscan4 in ES cells without telomerase. To determine whether telomere lengths are altered in Terc−/− ES cells by overexpressing Tbx3, we generated stable Tbx3 overexpression G1 Terc−/− ES cell lines. Expression levels of Tbx3 and Zscan4 were elevated in Tbx3 overexpression G1 Terc−/− ES cells relative to mock controls (Fig. 3e), while telomeres were notably lengthened in stable Tbx3 overexpression G1 Terc−/− ES cells following culture for 11 passages (Fig. 3f). Taken together, Zscan4-mediated telomere recombination may contribute to telomere elongation following Tbx3 overexpression. However, we cannot exclude other possibilities responsible for telomere elongation in Tbx3 OE ES cells, including increased accessibility of telomerase to telomeres, or up-regulation of telomeric non-coding RNAs recently shown to promote telomere elongation20.
Tbx3 regulates Zscan4 promoter activity
To understand potential mechanisms underlying regulation of Zscan4 expression by Tbx3, we performed dual luciferase assay using the 2570 bp of full length of Zscan4c promoter4. Zscan4c promoter activity was significantly increased following Tbx3 over-expression (p < 0.0001) (Supplementary Fig. 4a). Over-expression of Tbx3 resulted in nearly five fold enhanced luciferase activity compared to mock controls. Coincidently, Tbx3 increases the expression levels of Zscan4 in Zscan4+ cells (Fig. 2e, 2g). These data suggest that Tbx3 might act on the Zscan4 promoter to regulate its expression. We divided the promoter of Zscan4c into 7 regions (Supplementary Fig. 4b), with the initiation codon located at +811 bps4. The promoter activity was low in Luc1/Luc2, but rapidly increased from Luc3 and the Luc5 with full length of Zscan4c promoter the highest. To further narrow down possible location(s) of Zscan4c promoter affected by Tbx3, we analyzed the promoter activity following Tbx3 overexpression. Luc1 showed increased activity resulting from Tbx3 overexpression (Supplementary Fig. 4b), suggesting that Tbx3 might influence +608–+811 of Zscan4c promoter to regulate Zscan4 promoter activity. Luc2-Luc5 regions also exhibited increased Zscan4 promoter activity by Tbx3 overexpression. Luc6 and Luc7 did not contain Luc1 and Luc2, respectively, but also showed increased promoter activity after Tbx3 overexpression, suggesting that −1759–+608 regions also might be influenced by Tbx3 overexpression. Taken together, the full length of Zscan4c promoter can be influenced by Tbx3 overexpression.
Next, we asked whether Tbx3 as a transcription factor binds to Zscan4 promoter regions and directly regulates Zscan4 expression. We performed chromatin immunoprecipitation (ChIP)-qPCR in ES cells using Tbx3 antibody and 13 primer pairs 9 kb upstream of Zscan4c translational initiation code and primer for subtelomere upstream of Zscan4c locus. Contrary to speculation, Tbx3 was not enriched at Zscan4c promoter regions nor subtelomeric region upstream of Zscan4c locus (Supplementary Fig. 4c). Thus, Tbx3 overexpression also did not increase Tbx3 binding at these regions. Consistently, Tbx3 ChIP-seq data also showed no enrichment of Tbx3 at Zscan4 promoter regions in mouse ES cells21. These data do not appear to suggest a direct regulation of Zscan4 by Tbx3, although Tbx3 over-expression influences the Zscan4 promoter activity. Tbx3 might indirectly activate Zscan4/2-cell genes.
Tbx3 over-expression reduces DNA methylation level
Further analysis of the up-regulated genes activated by Tbx3 in Fig. 1f identified that some of the up-regulated genes, including Zscan4, Tcstv1 and Tcstv3 are located at subtelomeric regions and Zscan4 gene clusters are located at subtelomeric regions of chromosome 7. Telomeres and subtelomeres are densely compacted with repressive DNA methylation and histone modifications that are important regulators of mammalian telomere lengths22. The repressive H3K9me3 is detected at satellite, telomere, subtelomere and active long-terminal repeats and can spread into proximal unique sequences23. Mouse primary cells deficient for Suv39h1 and Suv39h2, which govern methylation of histone H3 Lys9 in heterochromatin regions, exibit abnormal telomere lengthening and increased telomere recombination24, suggesting the essential role of H3K9me3 in the negative regulation of telomere length. Other than the repressive histone modifications, telomeric H3 and H4 histones are generally hypoacetylated25,26. DNA methyltransferases (DNMTs) deficiency can lead to reduced global DNA methylation, increased telomere recombination as indicated by sister-chromatid exchanges involving telomeric sequences and dramatic telomere elongation27. These repressive DNA methylation and histone modification at subtelomeric regions form an inhibitory effect to the genes nearby, known as Telomere Position Effect (TPE)28,29.
Tbx3 over-expression did not alter the histone modification levels of H3K9me3 and acetylated H3 (AcH3) by western blot (Fig. 4a). We then asked whether Tbx3 inhibits DNA methylation, resulting in derepression of Zscan4 at subtelomeres. DNA methyltranferases Dnmt3a and Dnmt3b can methylate hemimethylated and unmethylated DNA, whereas the recently discovered ten-eleven translocation (Tet) family proteins have the ability to remove methyl group in cytosine by gradual oxidization, leading to DNA demethylation30,31,32. An elegant study clearly demonstrate that maternal origin Dnmt3a is notably expressed in oocytes, zygotes, 2-cell and 4-cell early mouse embryos and dramatically reduced from 8-cell stage to blastocysts, while Dnmt3b is undetectable in zygote and 2-cell embryos, but expressed only from 4 cell -to blastocysts33, further supporting that Dnmt3a and Dnmt3b also exhibit non overlapping functions in development34,35. Dnmt3b is specifically expressed in pluripotent embryonic cells, such as inner cell mass, epiblast and embryonic ectoderm cells, whilst Dnmt3a is significantly and ubiquitously expressed after E10.535. Minimal levels of Dnmt3b in zygote and 2-cell embryos coincide with activation of 2-cell genes Zscan4, Tcstv1/3 and Eif1α. Western blot analysis proved the decreased protein levels of Dnmt3b and increased levels of Tet2 following Tbx3 over-expression (Fig. 4a). Accordingly, DNA modification of 5-methylcytosine (5mC) was reduced and 5-hydroxymethylcytosine (5hmC) increased in Tbx3 OE ES cells analyzed by flow cytometry (Fig. 4b, 4c), suggesting that Tbx3 can reduce global DNA methylation levels by regulation of enzymes for methylation or demethylation. Tbx3 is a transcriptional repressor that belongs to the Tbx2/3/4/5 subfamily of T-box transcriptional regulators36,37 and binds to 5′ proximal regions of Dnmt3a and Dnmt3b by ChIP-Seq analysis (1581bp to 5′ proximal of Dnmt3a and 6199 bp to 5′ proximal of Dnmt3b, respectively)21. Furthermore, we performed ChIP-qPCR using anti-Dnmt3b and showed the reduced binding of Dnmt3b to Zscan4 promoter regions, Tcstv1 and Tcstv3 promoter locus, as well as subtelomeres of chromosome 7 following Tbx3 overexpression (Fig. 4d). Finally, knockdown of Dnmt3b with different shRNA sequences raised the protein levels of Zscan4 in ES cells (Fig. 4e, 4f). These data suggest that Tbx3 represses Dnmt3b (and possibly Dnmt3a), reducing DNA methylation level and leading to up-regulation of Zscan4.
Tbx3 reduces global DNA methylation of ES cells.
(a) Tbx3 over-expression did not change levels of H3K9me3 and AcH3 but reduced Dnmt3b and increased Tet2 protein levels. Full-length gel images are available in Supplementary Figure 5. (b) Global DNA 5-methylcytosine/5-hydroxymethylcytosine level in Tbx3 OE ES cells and mock ES cells by specific labeling with anti-5-MeC and 5hmC antibodies analyzed by flow cytometry. (c) Relative 5-methylcytosine/5-hydroxymethylcytosine level in Tbx3 OE ES cells compared with mock ES cells. The fluorescence intensity for mock ES cells was arbitrarily set as 1 and the fluorescence intensity of Tbx3 OE ES cells was expressed relative to mock ES cells. *, p < 0.05; **, p < 0.01, compared to mocks. Error bars indicate mean ± SEM (n = 3). (d) ChIP-qPCR assay of Dnmt3b binding on proximal Zscan4 and Tcstv1-Tcstv3 promoters and subelomere of chromosome 7 in mock and Tbx3 OE ES cells. Error bars indicate mean ± SEM (n = 2). (e) Dnmt3b knockdown increased Zscan4 protein level in ES cells. Two different Dnmt3b RNAi sequences were used. (f) Relative protein quantity normalized to β-actin by Bio-Rad Quantity One software.
Discussion
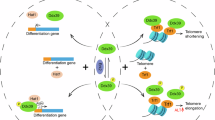
Our data show that Tbx3 activates Zscan4+/2C-state in mouse ES cells likely by reducing global DNA methylation level, which in turn leads to derepression of genes at subtelomeres, including Zscan4 and Tcstv1/3. Telomere length maintenance by Zscan4, despite that the precise underlying mechanism remains elusive, is critical for unlimited self-renewal and pluripotency of ES/iPS cells4,15,17. Dnmt3b preferentially targets to repetitive sequences38 and is specifically required for methylation of pericentromeric minor satellite repeats34 and also telomere repeats39,40. Subtelomeric regions are CG-rich and contain repetitive sequences41,42. Zscan4 promoter activity was activated by Tbx3. However, no direct binding to Zscan4 promoter was found for Tbx3 and this data is consistent with the ChIP-seq data21. We find that Tbx3 appears to inhibit Dnmt3b and reduce DNA methylation, indirectly regulating genes located in repetitive sequences including Zscan4 at subtelomeres, which contributes to telomere recombination and elongation, providing evidence supporting telomere position effects influenced by Tbx3. A speculative model on the role of Tbx3 in epigenetic regulation of telomere length in ES cells is proposed in Figure 5.
A speculative model for role of Tbx3 in ES cell cultures by epigenetic regulation of Zscan4/2C-state.
Tbx3 plays a novel role in maintaining self-renewal and pluripotency of mouse ES cells. Tbx3 represses Dnmt3b, reduces DNA methylation and increases hypomethylation, de-repressing genes at repeated sequences, including Zscan4 at subtelomeres, which contributes to telomere elongation and self-renewal of ES cells. (?) Tbx3 also may regulate Zscan4/2C state and telomeres via other mechanisms that remain to be identified.
Tbx3 belongs to T- box (TBX) transcription factors family43, is known to function generally as a transcriptional repressor, although Tbx3 has also been shown to have an activation domain44,45. Tbx3 mediates the leukemia inhibitory factor (LIF) signaling to core circuitry of pluripotency in mouse ES cells14. Phosphoinositide 3-kinase (PI3K)-dependent signaling has been implicated in the regulation of ES cell fate15. Tbx3, Zscan4 and Nanog are all regulated by PI3K signaling pathway14,15. Zscan4 and Nanog are decreased within 24 h following PI3K inhibition by LY294002 treatment, while Tbx3 is decreased after 48 h treatment15, demonstrating that Tbx3 is delayed in response to PI3K inhibition compared to Nanog and Zscan4. Tbx3 acts upstream of Nanog in PI3K pathway to mediate LIF independent self-renewal14 and Tbx3 over-expression up-regulates Zscan4, suggesting that Zscan4 may be downstream target of Tbx3 in PI3K signaling pathway.
Notably, Tbx3 enhances reprogramming efficiency and quality of iPS cells21. Expression of Tbx3 is directly repressed by binding of Tcf3 to its promoter and up-regulated by Tcf3 knockdown21. Furthermore, Tcf3 depletion increases somatic cell reprogramming46. Interestingly, Zscan4 enhances the reprogramming efficiency, promotes genomic stability during reprogramming and dramatically improves the quality of iPS cells as demonstrated by tetraploid complementation11,12. We found a novel role for Tbx3 in regulation of ES pluripotency and self-renewal by regulating 2C-state and Zscan4 expression that may elongate telomeres by recombination. Tbx3 reduces Dnmt3b and increases Tet2 levels, which synergistically results in reduced DNA methylation level and elevated Zscan4 promoter activity and gene expression. It has been shown that Dnmt3b and DNA methylation block iPS induction47,48,49, whereas Tet2 and hypomethylation facilitates iPS generation50,51. It is possible that reduced levels of DNA methylation together with increased expression of Zscan4 and other 2C genes induced by Tbx3 also may contribute to enhanced iPS reprogramming efficiency and improved iPS cell quality through activation of Zscan4/2C genes during reprogramming.
Tbx3 plays critical roles in mouse embryogenesis and ES cell fate decision52. Tbx3-deficient embryos have limb defects and fail to form mammary glands and die between E12.5 and E15.544,53,54. Tbx3 is upregulated during early embryo development and involved in telomere length maintenance. Telomere length was associated with authentic pluripotency of ES/iPS cells19. Telomeres are critical for highly proliferative cell types and organs and telomere shortening leads to defective embryonic development.
In addition to its key roles in embryonic development, TBX3 also is implicated in tumorigenesis. TBX3 was shown to be over-expressed in variety types of cancers and implicated in cancer stem cell proliferation and metastasis, including breast, ovarian, pancreas, melanoma, liver, cervical and lung cancers36. Telomere maintenance also is critical for cancer growth and metastasis55,56 and telomere has been a target for cancer therapy. Using ES cell model, we suggest that Tbx3 plays a novel role in promoting telomere elongation by epigenetic regulation and activation of Zscan4.
Methods
Mouse ES cells
BF10 and F1 ES cell lines derived from B6C3F1 mice and early and late generation telomerase-deficient ES cells and control ES cells were generated and characterized as described previously19,57. J1 ES cells were cultured without feeder. The ES cell culture medium consisted of knock-out DMEM (Gibco) with 15% FBS (Hyclone), 1000 U/ml mouse leukemia inhibitory factor (LIF) (ESGRO, Chemicon), 0.1 mM non-essential amino acids, 0.1 mM β-mercaptoethanol, 1 mM L-glutamine and penicillin (100 U/ml) and streptomycin (100 μg/ml). For culture of ES cell lines, the medium was changed daily and cells routinely were passaged every two days. Animals were cared for and treated according to guidelines set by U.S. National Research Council and the use of mice for the research approved by Nankai Animal Care and Use Committee.
Vector construction
Murine Tcstv1, Tcstv3, Ott and Tbx3 CDS were cloned into expression vector pCAGIpuro; murine Zscan4c CDS was cloned into pCMV-Tag2B and Dazl CDS was cloned into pEF6 expression vector. A putative Zscan4c promoter containing the 2570 bp upstream sequences from the Zscan4c start codon4, was amplified from mouse F1 ES cell genomic DNA with TransStar Fastpfu polymerase (Transgene) using the following primers: forward: AGAGATGCTTCTGCATCTGT; reverse: TGTGGTGACAATGGTGTGAAAG. The PCR product was inserted into pEGFP-1 vector at SalI/KpnI sites. The 2570 full length putative Zscan4c promoter was then cut using SacI/SmaI from pEGFP-1-Zscan4c and inserted into pGL3-basic vector containing several base pairs from pEGFP-1 vector. The other six Zscan4 promoter fragments were amplified from the full length Zscan4 promoter and inserted into pGL3-basic vector at KpnI/SalI sites.
Generation of Tbx3 OE, pZscan4-EGFP and stable Dnmt3b knockdown ES cells
BF10 ES cells were transfected by electroporation with pCAGIpuro-Tbx3 expression vector or empty vector served as control and selected with 1.5 μg/ml puromycin for two weeks to achieve stable Tbx3 over-expression or mock ES cell lines. G1 Terc−/− ES cells were infected with pMSCV-Tbx3 overexpression or control retrovirus, selected with G418 and clones were picked for further experimental analysis. The pEGFP-1-Zscan4 vector was linearized by Xho I digestion and purified by PCR purification kit (Transgene). Feeder-free J1 ES cells were transfected with 2 μg linearized vector using lipofectamine 2000 (Invitrogen) and selected with 400 μg/ml G418 (Invitrogen) for 2 weeks and a clone with bright green fluorescence was picked and expanded for further experiments. Zscan4-EGFP ES cells were sorted by fluorescence-activated cell sorting using BD FACSAria. The sorted Zscan4+ (positive) and Zscan4− (negative) ES cells were used for RNA extraction. Control and two different shRNA sequences against Dnmt3b mRNA were used for Dnmt3b knockdown experiment. The sequences were cloned into pSIREN-RetroQ (Clontech) with puromycin replaced by neomycin and the resultant vectors introduced into Plat-E cells to package retrovirus. Feeder-free J1 ES cells were then infected with control and Dnmt3b RNAi retrovirus, ES cells were then selected with 500 μg/ml G418 for about 10 days and the resistant clones were picked. The RNAi sequences were as following:
Control shRNA forward:
GATCCGGCGTTCAATTAGCAGACCATTCAAGAGATGGTCTGCTAATTGAACGCCTTTTTTAAGCTTG;
reverse:
AATTCAAGCTTAAAAAAGCGTTCAATTAGCAGACCATCTCTTGAATGGTCTGCTAATTGAACGCCG.
Dnmt3b shRNA1 forward:
GATCCGGAGTTGGGTATTAAAGTGTTCAAGAGACACTTTAATACCCAACTCCTTTTTTAAGCTTG;
Reverse:
AATTCAAGCTTAAAAAAGGAGTTGGGTATTAAAGTGTCTCTTGAACACTTTAATACCCAACTCCG.
Dnmt3b shRNA4 forward:
GATCCGCTTGAAGTAGGTAGTAAGATGTTCAAGAGACATCTTACTACCTACTTCAAGCTTTTTTAAGCTTG.
Reverse:
AATTCAAGCTTAAAAAAGCTTGAAGTAGGTAGTAAGATGTCTCTTGAACATCTTACTACCTACTTCAAGCG.
FACS analysis of Zscan4 positive ES cells
1 × 105 Zscan4-EGFP J1 ES cells per 12-well were transfected with 1 μg mock or Tbx3 over-expression (OE) vector and analyzed for percentage of Zscan4 positive cells 48 h later by FACS.
Luciferase reporter assay
1 × 105 feeder-free J1 ES cells per 24 well were transfected with 0.8 μg pGL3-Zscan4c vector (containing Luc1 to Luc7) and 8 ng pRL-SV40 vector (as control) and 1 μg mock or Tbx3 over-expression vector using lipofectamine 2000 (Invitrogen) according to manufacturer's instructions. Transfected J1 ES cells were lysed 24 h later with 1 × PLB (positive lysis buffer, Promega), shaken for 15 min and then centrifuged at 13000 rpm for 10 min at 4°C. The supernatants were collected and analyzed for luciferase activity by dual reporter assay according to manufacturer's instructions.
Immunofluorescence microscopy
Cells were washed twice in phosphate buffered saline (PBS), then fixed in freshly prepared 3.7% paraformaldehyde in PBS (pH 7.4) for 15 min on ice cube, permeabilized in 0.1% Triton X-100 in blocking solution (3% goat serum plus 0.5% BSA in PBS) for 30 min at room temperature, washed three times (each for 15 min) and left in blocking solution for 1 h. Cells were incubated overnight at 4°C with primary antibodies against Tbx3 (sc-31657, Santa Cruz), Nanog (ab80892, Abcam), Oct4 (sc5279, Santa Cruz), SMA (ab5694-100, Abcam), AFP (DAK-N1501, DAKO), Nestin (ab6142, Abcam), washed three times (each for 15 min) and incubated for 1 h with secondary antibodies, bovine anti-goat IgG-FITC (sc2348, Santa Cruz), Alexa Fluor 594 donkey anti-goat IgG (A-11058, MP), Alexa Fluor 568 Goat anti-rabbit (A-11011, MP), or Alexa Fluor 488 Goat anti-mouse (A-11001, MP), diluted 1:200 with blocking solution. Samples were washed and counterstained with 0.5 μg/ml Hoechst33342 (H1398, MP) in Vectashield mounting medium. Fluorescence was detected and imaged using a fluorescence microscope (Zeiss Axio Imager Z1).
Telomere quantitative fluorescence in situ hybridization (QFISH)
Telomere length and function (telomere integrity and chromosome stability) was estimated by QFISH. Cells were incubated with 0.5 μg/ml nocodazole for 1.5 h to enrich cells at metaphases. Chromosome spreads were made by a routine method. Metaphase-enriched cells were exposed to hypotonic treatment with 75 mM KCl solution, fixed with methanol: glacial acetic acid (3:1) and spread onto clean slides. Telomere FISH and quantification were performed as described previously58, except for FITC-labeled (CCCTAA) peptide nucleic acid (PNA) probe used in this study. Telomeres were denatured at 80°C for 3 min and hybridized with telomere specific PNA probe (0.5 μg/ml) (Panagene, Korea). Chromosomes were counter-stained with 0.5 μg/ml DAPI. Fluorescence from chromosomes and telomeres was digitally imaged on a Zeiss microscope with FITC/DAPI filters, using AxioCam and AxioVision software 4.6. For quantitative measurement of telomere length, telomere fluorescence intensity was integrated using the TFL-TELO program (gift kindly provided by P. Lansdorp, Terry Fox Laboratory, Vancouver, Canada) and calibrated using standard fluorescence beads.
Telomere measurement by quantitative real-time PCR
Cells were washed in PBS and stored at −20°C until subsequent DNA extraction. Genome DNA was prepared using DNeasy Blood & Tissue Kit (Qiagen, Valencia, CA). Average telomere length was measured from total genomic DNA using a real-time PCR assay, modified for measurement of mouse telomeres18. PCR reactions were performed on the iCycler iQ real-time PCR detection system (Bio-Rad, Hercules, CA), using telomeric primers, primers for the reference control gene (mouse 36B4 single copy gene) (Table S2) and PCR settings as previously described16. For each PCR reaction, a standard curve was made by serial dilutions of known amounts of DNA. The telomere signal was normalized to the signal from the single copy gene to generate a T/S ratio indicative of relative telomere length. Equal amounts of DNA were used for each reaction.
Gene expression analysis by quantitative real-time PCR
Total RNA was isolated from cells using RNeasy mini kit (Qiagen). 2 μg of RNA were subjected to cDNA synthesis using M-MLV Reverse Transcriptase (Invitrogen). Real-time quantitative PCR reactions were set up in duplicate with the FastStart Universal SYBR Green Master (ROX) (Roche) and run on the iCycler iQ5 2.0 Standard Edition Optical System (Bio-Rad). Each sample was repeated 3 times and analyzed with β-actin as the internal control. Most primers were designed using the IDT DNA website (http://www.idtdna.com/Home/Home.aspx). The primers used are in Table S1.
Global DNA methylation analysis
DNA methylation was analyzed by immunostaining with 5-MeC (NA81, Calbiochem) and 5hmC (39769, active motif) antibodies as previously described59,60. ES cells were dissociated in trypsin–EDTA and washed in PBS twice and fixed in ice-cold 3.7% PFA for 30 min, washed in 0.05% PBT, permeabilized in 0.2% Triton X-100 solution for 30 min. Then, cells were depurinated in 4N HCl, 0.1% Triton X-100 for 10 min, washed and then blocked in 2% BSA in PBT. Samples were incubated with anti-5-MeC or anti-5hmC antibody overnight at 4°C, washed and then incubated with FITC conjugated goat anti-mouse IgG (A-11001, Molecular Probes) or FITC conjugated anti-rabbit IgG (554020, BD Biosciences) in blocking solution for 1 h at RT, washed and then ES cells were placed in the fluorescence-activated cell sorting for analysis and sorting by flow cytometry (BD Biosciences). Cells with immunostaining only with normal IgG antibody served as negative controls. Fluorescence distribution and intensity of cell populations following immunostaining with anti-5-MeC and 5hmC antibodies were quantified by FACS analysis. The fluorescence intensity for mock ES cells was arbitrarily set as 1 and the fluorescence intensity of Tbx3 OE ES cells expressed relative to that of mock ES cells.
Western blot
Cells were washed twice in PBS, collected, lysed and boiled in SDS Sample Buffer at 99°C for 5 min; 35 μg total proteins of each cell extracts were resolved by 10–12% Bis-Tris SDS-PAGE and transferred to polyvinylidene difluoride membrane (PVDF, Millipore). Nonspecific binding was blocked by incubation in 5% skim milk in TBST at room temperature for 1–2 h. Blots were then probed with various primary antibodies, Tbx3 (sc-31657, Santa Cruz), Zscan4 (custom-made), Dnmt3b (ab13604, Abcam), Tet2 (kind gift from Jinsong Li at SIBS), Histone H3 (ab1791, Abcam), H3K9me3 (07-442, Millipore), AcH3 (06-599, Millipore) and β-actin (P30002, Abmart) by overnight incubation at 4°C in 5% skim milk in TBST. Immunoreactive bands were then probed for 1–2 h at room temperature with the appropriate horseradish peroxidase-conjugated secondary anti-Rabbit IgG -HRP (GE Healthcare, NA934V), or anti-mouse IgG-HRP (Santa Cruz, sc-2031), or anti-goat IgG-HRP (Santa Cruz, sc-2020). The protein bands were detected by Enhanced ECL AmershamTM prime western blotting detection reagent (GE Healthcare, RPN2232).
ChIP-qPCR analysis
Mock and Tbx3 OE ES cells (each 3 × 107) were used for ChIP experiment. Briefly, cells were fixed with freshly prepared 1% paraformaldehyde for 10 min at room temperature. Cells were harvested and their nuclei extracted, lysed and sonicated. DNA fragments were then enriched by immunoprecipitation with 8 μg Tbx3 antibody (sc31657, Santa Cruz) and 4 μg Dnmt3b antibody (ab13604, Abcam), respectively. The eluted protein:DNA complex was reverse-crosslinked at 65°C overnight. DNA was recovered after proteinase and RNase A treatment. ChIP-enriched DNA was analyzed by real-time PCR using primers for Zscan4c loci, sub-telomeres (Table S3). β-actin served as negative control.
Statistical analysis
Percentage data and other number were analyzed by analysis of variance (ANOVA) and means were compared by Fisher's protected least-significant difference (PLSD) test using StatView software from SAS Institute Inc. (Cary, NC). Significant differences were defined as p < 0.05, 0.01, or lower.
References
Carter, M. G. et al. An in situ hybridization-based screen for heterogeneously expressed genes in mouse ES cells. Gene Expr Patterns 8, 181–198 (2008).
Niwa, H. How is pluripotency determined and maintained? Development 134, 635–646 (2007).
Boyer, L. A., Mathur, D. & Jaenisch, R. Molecular control of pluripotency. Curr Opin Genet Dev 16, 455–462 (2006).
Zalzman, M. et al. Zscan4 regulates telomere elongation and genomic stability in ES cells. Nature 464, 858–863 (2010).
Lanner, F. et al. Heparan sulfation-dependent fibroblast growth factor signaling maintains embryonic stem cells primed for differentiation in a heterogeneous state. Stem Cells 28, 191–200 (2010).
Yoshikawa, T. et al. High-throughput screen for genes predominantly expressed in the ICM of mouse blastocysts by whole mount in situ hybridization. Gene Expr Patterns 6, 213–224 (2006).
Macfarlan, T. S. et al. Embryonic stem cell potency fluctuates with endogenous retrovirus activity. Nature 487, 57–63 (2012).
Surani, A. & Tischler, J. Stem cells: a sporadic super state. Nature 487, 43–45 (2012).
Falco, G. et al. Zscan4: a novel gene expressed exclusively in late 2-cell embryos and embryonic stem cells. Dev Biol 307, 539–550 (2007).
Sander, T. L. et al. The SCAN domain defines a large family of zinc finger transcription factors. Gene 310, 29–38 (2003).
Jiang, J. et al. Zscan4 promotes genomic stability during reprogramming and dramatically improves the quality of iPS cells as demonstrated by tetraploid complementation. Cell Res 23, 92–106 (2013).
Hirata, T. et al. Zscan4 transiently reactivates early embryonic genes during the generation of induced pluripotent stem cells. Sci Rep 2, 208 (2012).
Amano, T. et al. Zscan4 restores the developmental potency of embryonic stem cells. Nat Commun 4, 1966 (2013).
Niwa, H., Ogawa, K., Shimosato, D. & Adachi, K. A parallel circuit of LIF signalling pathways maintains pluripotency of mouse ES cells. Nature 460, 118–122 (2009).
Storm, M. P. et al. Characterization of the phosphoinositide 3-kinase-dependent transcriptome in murine embryonic stem cells: identification of novel regulators of pluripotency. Stem Cells 27, 764–775 (2009).
Liu, L. et al. Telomere lengthening early in development. Nat Cell Biol 9, 1436–1441 (2007).
Wang, F. et al. Molecular insights into the heterogeneity of telomere reprogramming in induced pluripotent stem cells. Cell Res 22, 757–768 (2012).
Callicott, R. J. & Womack, J. E. Real-time PCR assay for measurement of mouse telomeres. Comp Med 56, 17–22 (2006).
Huang, J. et al. Association of telomere length with authentic pluripotency of ES/iPS cells. Cell Res 21, 779–792 (2011).
Cusanelli, E., Romero, C. A. & Chartrand, P. Telomeric Noncoding RNA TERRA Is Induced by Telomere Shortening to Nucleate Telomerase Molecules at Short Telomeres. Mol Cell 51, 780–791 (2013).
Han, J. et al. Tbx3 improves the germ-line competency of induced pluripotent stem cells. Nature 463, 1096–1100 (2010).
Blasco, M. A. The epigenetic regulation of mammalian telomeres. Nat Rev Genet 8, 299–309 (2007).
Mikkelsen, T. S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Garcia-Cao, M., O'Sullivan, R., Peters, A. H., Jenuwein, T. & Blasco, M. A. Epigenetic regulation of telomere length in mammalian cells by the Suv39h1 and Suv39h2 histone methyltransferases. Nat Genet 36, 94–99 (2004).
Benetti, R., Garcia-Cao, M. & Blasco, M. A. Telomere length regulates the epigenetic status of mammalian telomeres and subtelomeres. Nat Genet 39, 243–250 (2007).
Wong, L. H. Epigenetic regulation of telomere chromatin integrity in pluripotent embryonic stem cells. Epigenomics 2, 639–655 (2010).
Gonzalo, S. et al. DNA methyltransferases control telomere length and telomere recombination in mammalian cells. Nat Cell Biol 8, 416–424 (2006).
Ottaviani, A., Gilson, E. & Magdinier, F. Telomeric position effect: from the yeast paradigm to human pathologies? Biochimie 90, 93–107 (2008).
Slijepcevic, P. Telomere length regulation--a view from the individual chromosome perspective. Exp Cell Res 244, 268–274 (1998).
He, Y. F. et al. Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 333, 1303–1307 (2011).
Ito, S. et al. Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature 466, 1129–1133 (2010).
Ito, S. et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333, 1300–1303 (2011).
Hirasawa, R. et al. Maternal and zygotic Dnmt1 are necessary and sufficient for the maintenance of DNA methylation imprints during preimplantation development. Genes Dev 22, 1607–1616 (2008).
Okano, M., Bell, D. W., Haber, D. A. & Li, E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99, 247–257 (1999).
Watanabe, D., Suetake, I., Tada, T. & Tajima, S. Stage- and cell-specific expression of Dnmt3a and Dnmt3b during embryogenesis. Mech Dev 118, 187–190 (2002).
Lu, J., Li, X. P., Dong, Q., Kung, H. F. & He, M. L. TBX2 and TBX3: the special value for anticancer drug targets. Biochim Biophys Acta 1806, 268–274 (2010).
Rallis, C., Del Buono, J. & Logan, M. P. Tbx3 can alter limb position along the rostrocaudal axis of the developing embryo. Development 132, 1961–1970 (2005).
Chen, T. & Li, E. Structure and function of eukaryotic DNA methyltransferases. Curr Top Dev Biol 60, 55–89 (2004).
Deng, Z., Campbell, A. E. & Lieberman, P. M. TERRA, CpG methylation and telomere heterochromatin: lessons from ICF syndrome cells. Cell Cycle 9, 69–74 (2010).
Yehezkel, S., Segev, Y., Viegas-Pequignot, E., Skorecki, K. & Selig, S. Hypomethylation of subtelomeric regions in ICF syndrome is associated with abnormally short telomeres and enhanced transcription from telomeric regions. Hum Mol Genet 17, 2776–2789 (2008).
Brock, G. J., Charlton, J. & Bird, A. Densely methylated sequences that are preferentially localized at telomere-proximal regions of human chromosomes. Gene 240, 269–277 (1999).
de Lange, T. et al. Structure and variability of human chromosome ends. Mol Cell Biol 10, 518–527 (1990).
Papaioannou, V. E. & Silver, L. M. The T-box gene family. Bioessays 20, 9–19 (1998).
Davenport, T. G., Jerome-Majewska, L. A. & Papaioannou, V. E. Mammary gland, limb and yolk sac defects in mice lacking Tbx3, the gene mutated in human ulnar mammary syndrome. Development 130, 2263–2273 (2003).
He, M., Wen, L., Campbell, C. E., Wu, J. Y. & Rao, Y. Transcription repression by Xenopus ET and its human ortholog TBX3, a gene involved in ulnar-mammary syndrome. Proc Natl Acad Sci U S A 96, 10212–10217 (1999).
Lluis, F. et al. T-cell factor 3 (Tcf3) deletion increases somatic cell reprogramming by inducing epigenome modifications. Proc Natl Acad Sci U S A 108, 11912–11917 (2011).
Guo, X. et al. microRNA-29b is a novel mediator of Sox2 function in the regulation of somatic cell reprogramming. Cell Res 23, 142–156 (2013).
Huangfu, D. et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nat Biotechnol 26, 795–797 (2008).
Shi, Y. et al. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3, 568–574 (2008).
Costa, Y. et al. NANOG-dependent function of TET1 and TET2 in establishment of pluripotency. Nature 495, 370–374 (2013).
Doege, C. A. et al. Early-stage epigenetic modification during somatic cell reprogramming by Parp1 and Tet2. Nature 488, 652–655 (2012).
Weidgang, C. E. et al. TBX3 directs cell-Fate decision toward mesendoderm. Stem Cell Reports 1, 248–265 (2013).
Hoogaars, W. M. et al. Tbx3 controls the sinoatrial node gene program and imposes pacemaker function on the atria. Genes Dev 21, 1098–1112 (2007).
Bamshad, M. et al. The spectrum of mutations in TBX3: Genotype/Phenotype relationship in ulnar-mammary syndrome. Am J Hum Genet 64, 1550–1562 (1999).
Blasco, M. A. Telomeres and human disease: ageing, cancer and beyond. Nat Rev Genet 6, 611–622 (2005).
Xu, L., Li, S. & Stohr, B. A. The role of telomere biology in cancer. Annu Rev Pathol 8, 49–78 (2013).
Chen, Z. et al. Birth of parthenote mice directly from parthenogenetic embryonic stem cells. Stem Cells 27, 2136–2145 (2009).
Poon, S. S., Martens, U. M., Ward, R. K. & Lansdorp, P. M. Telomere length measurements using digital fluorescence microscopy. Cytometry 36, 267–278 (1999).
Li, C. et al. Correlation of expression and methylation of imprinted genes with pluripotency of parthenogenetic embryonic stem cells. Hum Mol Genet 18, 2177–2187 (2009).
Santos, F., Hendrich, B., Reik, W. & Dean, W. Dynamic reprogramming of DNA methylation in the early mouse embryo. Dev Biol 241, 172–182 (2002).
Acknowledgements
We thank Dr. Peter Lansdorp for the TFL-TELO software. This work was supported by China MOST National Major Basic Research Program (2012CB911202, 2011CBA01002), National Natural Science Foundation of China (31271587) and Natural Science Foundation of Tianjin (12JCZDJC24800).
Author information
Authors and Affiliations
Contributions
J.D., M.L., M.O., X.Y., J.Y. and J.L. performed experiments and data analysis. J.D. designed the experiments and wrote the manuscript; L.L. designed and advised the experiments and revised manuscripts.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Figures S1-5; Tables S1-3
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Dan, J., Li, M., Yang, J. et al. Roles for Tbx3 in regulation of two-cell state and telomere elongation in mouse ES cells. Sci Rep 3, 3492 (2013). https://doi.org/10.1038/srep03492
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep03492
This article is cited by
-
The role of telomere-binding modulators in pluripotent stem cells
Protein & Cell (2020)
-
Novel HDAC5-interacting motifs of Tbx3 are essential for the suppression of E-cadherin expression and for the promotion of metastasis in hepatocellular carcinoma
Signal Transduction and Targeted Therapy (2018)
-
Feeders facilitate telomere maintenance and chromosomal stability of embryonic stem cells
Nature Communications (2018)
-
Telomere chromatin establishment and its maintenance during mammalian development
Chromosoma (2018)
-
The T-box transcription factor TBX3 drives proliferation by direct repression of the p21WAF1 cyclin-dependent kinase inhibitor
Cell Division (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.