Abstract
We report the unprecedented square-planar coordination of iridium in the iron iridium arsenide Ca10(Ir4As8)(Fe2As2)5. This material experiences superconductivity at 16 K. X-ray photoemission spectroscopy and first-principles band calculation suggest Ir(II) oxidation state, which yields electrically conductive Ir4As8 layers. Such metallic spacer layers are thought to enhance the interlayer coupling of Fe2As2, in which superconductivity emerges, thus offering a way to control the superconducting transition temperature.
Similar content being viewed by others
Introduction
Platinum exhibits a rich variety of coordination geometries. For instance, all of the basic polyhedral forms, including octahedral1, triangle-planar2, tetrahedral3 and square-planar4,5,6,7, can be seen in platinum arsenides. The diversity of coordination chemistry allows us to synthesize many functional materials, such as superconductors. The following are prominent platinum-arsenide superconductors: SrPt2As2, which consists of PtAs4 tetrahedra3, exhibits superconductivity at a transition temperature of Tc = 5.2 K8, in which a charge transfer from donor to acceptor layers9 and subsequent emergence of charge-density waves has been discussed3,8; SrPtAs, which consists of PtAs3 triangles2, shows superconductivity at 2.4 K10, for which a broken time-reversal symmetry in a locally noncentrosymmetric structure has been proposed11; Ca10(Pt4As8)(Fe2−xPtxAs2)5, which consists of PtAs4 planar squares, exhibits superconductivity at 38 K4,5,6,7 and therefore constitutes a member of the iron-based superconductors12,13,14. Palladium exhibits similar coordination chemistry15,16,17,18; Ca10(Pd3As8)(Fe2−xPdxAs2)5 with PdAs4 planar squares was reported very recently to exhibit superconductivity at 17 K18.
In contrast, iridium shows limited coordination geometries; only octahedral and tetrahedral coordination are known in arsenides, as in IrAs319 and SrIr2As23. In this paper, we report the occurrence of square-planar coordination of iridium in a novel iron iridium arsenide Ca10(Ir4As8)(Fe2As2)5. This is the first inorganic compound that includes square-planar coordination of iridium. This compound exhibits superconductivity at Tc = 16 K. First-principles calculations and X-ray photoelectron spectroscopy (XPS) suggest the presence of iridium (II) oxidation state. The resultant metallic nature of Ir4As8 spacer layers will be discussed.
Results
Crystal structure
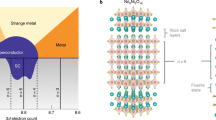
Single-crystal structure analysis revealed that the compound, discovered in this study, crystallizes in the tetragonal structure with the space group P4/n (#85) with a chemical composition of Ca10(Ir4As8)(Fe2As2)5 (see the Supplementary Tables S1 and S2 for crystallographic data) (CCDC 962099). The atomic ratios of Ca:Fe:Ir:As = 10:10:4:18 are consistent with the results of energy dispersive X-ray spectrometry, 10:9.8:5.8:20.1. The structure consists of alternating stacking of (Fe2As2)5 and Ir4As8 layers with five Ca ions between them, as shown in Figure 1. This is isotypic to Ca10(Pt4As8)(Fe2−xPtxAs2)56 or α-(CaFe1−xPtxAs)10Pt4−yAs87. The Fe2As2 layers, composed of edge-sharing FeAs4 tetrahedra, are the common building block among iron-based superconductors12,13,14. The Ir4As8 layers are unique to the present compound and act as spacer layers. The size of the Ir square lattice (with an Ir-Ir distance of 4.411 Å) is larger than that of the Fe2As2 square lattice (3.860–3.924 Å). This lattice mismatch leads to the formation of the √5 × √5 superstructure in the ab-plane, as shown in Figure 1c.
Crystal structure of Ca10(Ir4As8)(Fe2As2)5 with tetragonal structure [space group P4/n (#85)].
The thick solid lines indicate the unit cell. (a), (b) and (c) show the schematic overviews, Ir4As8 layer and (Fe2As2)5 layer, respectively. The blue and dark-blue hatches in (b) indicate IrAs4 squares with coplanar Ir1 and non-coplanar Ir2, respectively. The dashed ellipsoids in (b) represent As2 dimers.
The characteristic square-planar coordination of Ir was found in the Ir4As8 layers. There are two Ir sites, as shown in Figure 1b. Ir1 adopts square-planar coordination, resulting in coplanar IrAs4 squares with a Ir1-As3 bond length of 2.414 Å. On the other hand, Ir2 is at a non-coplanar site with respect to the As4 square; Ir2 is displaced upward/downward by 0.676 Å toward the As4 ion at the adjacent Fe2As2 layer, as shown in Figure 1a. However, the distance between Ir2 and As4 (3.000 Å) is by far longer than the Ir2-As3 bond length (2.441 Å), thus Ir2 can be regarded as adopting square-planer coordination. The corner-sharing Ir1As34 and Ir2As34 squares constitute Ir4As8 layers, as shown in Figure 1b, where the As3 atoms form As2 dimers with an As-As bond length of 2.454 Å, which comparable to twice the covalent radius of arsenic that is 2.42 Å18. These bond lengths are similar to those in platinum analogue, Ca10(Pt4As8)(Fe2−xPtxAs2)57: Corresponding distances, Pt1-As3 = 2.484 Å, Pt2-As4 = 3.087 Å and Pt2-As3 = 2.415 Å, suggest that the valence state of Ir is similar to that of Pt.
Superconductivity
Figure 2 shows the temperature dependence of the in-plane electrical resistivity ρab of Ca10(Ir4As8)(Fe2As2)5. ρab(T) decreases with decreasing temperature and shows a kink at approximately 100 K. This kink is not due to antiferromagnetic ordering, which is widely observed in iron-based superconductors12,13,14, since the single-peak structure of the 57Fe-Mössbauer spectrum at 300 K remains unchanged down to 50 K, as shown in the upper inset of Figure 2. At low temperatures, ρab(T) exhibits a sharp drop below 20 K, the characteristic of the onset of superconductivity. Zero resistivity was observed below 17 K. The 10–90% transition width was estimated to be approximately 2 K. The bulk superconductivity was evidenced by the temperature dependence of the magnetization M, shown in Figure 3. M(T) exhibits diamagnetic behavior below 16 K. The shielding signal estimated at 5 K corresponds to 83% of perfect diamagnetism.
Discussion
The observed Tc of 16 K is relatively low among iron-based superconductors12,13,14. We suggest that Ca10(Ir4As8)(Fe2As2)5 is in an overdoped region. The lower inset of Figure 2 shows the temperature dependence of the Hall coefficient RH. The negative value suggests that the major carriers are electrons. The small value of RH as well as the small temperature dependence indicates the overdoping, as inferred from the RH of Ba(Fe1−xCox)2As220. This is consistent with the absence of antiferromagnetic ordering, which is characteristic of underdoped regions12,13,14. The consideration of charge neutrality based on the Zintl concept results in the same consequence. Assuming a divalent Ir2+, the present compound is written as Ca2+10(Ir2+4(As2)4−4)(Fe2+2As3−2)5·2e−; the excess charge 0.2e−/Fe is intrinsically injected into the superconducting Fe2As2 layers. This doping level corresponds to overdoping, judging from the data on doped BaFe2As221. We expect that a higher Tc can be realized by reducing the intrinsic charge carriers.
Iron-based superconductors reported to date can be characterized by the insulating spacer layers12,13,14, which include rare-earth oxides22 and alkaline-earth fluorides23 with a fluorite-type structure, alkali24 or alkali-earth25 ion and complex metal oxides with combined rock-salt and perovskite-type structures26,27,28,29,30. The insulating spacer layers are stacked in an alternating fashion with superconductive Fe2As2 layers, resulting in two-dimensional electronic Fermi surfaces that have been thought to be a key ingredient of high Tc superconductivity12,13,14. In contrast, the Ir4As8 spacer layers of the present compound can be metallic: Figure 4 shows the partial density of states (pDOS) projections of Fe 3d and Ir 5d of Ca10(Ir4As8)(Fe2As2)5 from first-principles calculations using the WIEN2k package31. Fe 3d predominates in the pDOS at the Fermi energy (EF), in common with the other iron-based superconductors32. A remarkable difference is noticeable in the pDOS of the spacer layers; a finite contribution of Ir 5d can be seen in the pDOS at EF, suggesting that the Ir4As8 spacer layers are metallic. This is in contrast with the negligible pDOS at EF of the spacer layers for the other iron-based superconductors12,13,14,32, including the platinum analogue Ca10(Pt4As8)(Fe2As2)5: The Pt4As8 spacer layers are semiconducting because of the opening of the gap in the pDOS of Pt 5d at EF7,33. The difference between the Pt4As8 and Ir4As8 layers might be attributed to that of the electron configurations; Pt2+ (5d8) forms a closed-shell configuration with a completely filled dxy orbital in the square-planar coordination, whereas dxy of Ir2+ (5d7) is formally half-filled, resulting in a metallic nature. The oxidation state of iridium (II) is suggested by first-principles calculations, which give an estimate of the total number of electrons of Ir1 and Ir2 (and thus the nominal oxidation states) to be 74.89 (Ir2.11+) and 74.91 (Ir2.09+) from the sum of pDOS up to EF, respectively. This is consistent with XPS results, as shown in Figure 5: The binding energy at the peak position of Ir 4f7/2 spectrum suggests that the valence of Ir in Ca10(Ir4As8)(Fe2As2)5 is close to 2+, if we refer to the binding energy of Ca3CoIrO634 with Ir4+ and assume that the binding energy is decreased by approximately 1 eV when the valence is decreased by 1 as inferred from the XPS data of K3IrBr6 and K2IrBr6.
In cuprates, it has been suggested that the interlayer coupling of superconducting CuO2 planes enhances Tc35. The metallic nature of the spacer layers of the present compound Ca10(Ir4As8)(Fe2As2)5 may give rise to an opportunity to engineer the interlayer coupling of superconducting Fe2As2 and to thus further enhance the superconducting transition temperature. To do so, we have to develop chemical methods of optimizing the carrier concentration of Ca10(Ir4As8)(Fe2As2)5.
The unusual square-planar coordination of Fe2+ has been reported for the oxide SrFeO236. It has been discussed that strong hybridization or covalent nature between Fe 3d and O 2p orbitals for Fe2+ in the square-planar coordination is the key ingredient for the stability of SrFeO237. Similar mechanism might be applicable to the formation of the square-planar coordination of Ir2+ of Ca10(Ir4As8)(Fe2As2)5 because of the strong hybridization between Ir 5d and As 4p orbitals.
In summary, we found the square-planar coordination of iridium in the Ir4As8 layers of the iron iridium arsenide Ca10(Ir4As8)(Fe2As2)5. This finding provided a novel iron-based superconductor with Tc = 16 K. The optimization of the metallic spacer layer might offer a way to further increase the superconducting transition temperature of iron-based materials.
Methods
Preparation and characterization of samples
Single crystals of Ca10(Ir4As8)(Fe2As2)5 were grown by heating a mixture of Ca, FeAs, IrAs2 and Ir powders in a ratio of Ca:Fe:Ir:As = 10:10:4:18 or 10:26:14:40. The mixture was placed in an alumina crucible and sealed in an evacuated quartz tube. The manipulation was carried out in a glove box filled with argon gas. The ampules were heated at 700°C for 3 h and then at 1100–1150°C for 10–40 h, after which they were quenched in ice water. The quenching procedure was found to be crucial to obtaining the Ca10(Ir4As8)(Fe2As2)5 phase. This process yielded Ca10(Ir4As8)(Fe2As2)5 together with a small amount of powder mixture of CaFe2As2 and IrAs2. Plate-like single crystals of Ca10(Ir4As8)(Fe2As2)5 with typical dimensions of 0.5 × 0.5 × 0.02 mm3 were separated from the mixture. The crystals were characterized by synchrotron radiation X-ray diffraction38, energy dispersive X-ray spectrometry and conventional transmission Mössbauer spectroscopy with a 57Co/Rh source.
Electrical resistivity and magnetization measurements
The electrical resistivity (parallel to the ab-plane) and Hall coefficient were measured using the Quantum Design PPMS. Magnetization was measured using the Quantum Design MPMS.
X-ray photoelectron spectroscopy (XPS) measurements
The single crystals were cleaved under the ultrahigh vacuum for the XPS measurements that were carried out using JEOL JPS9200 analyzer and a Mg Kα source (1253.6 eV). The total energy resolution was set to about 1.0 eV. The binding energy was calibrated using the Au 4f core level of the gold reference sample.
References
Thomassen, L. Crystallization of Binary Compounds of Metals of Platinum Group. Z. Phys. Chem. B 2, 349–379 (1929).
Wenski, G. & Mewis, A. Trigonal-planar koordiniertes Platin: Darstellung und Struktur von SrPtAs (Sb), BaPtP (As, Sb), SrPtxP2−x, SrPtxAs0.90 und BaPtxAs0.90 . Z. Anorg. Allg. Chem. 535, 110–122 (1986).
Imre, A. et al. Inkommensurabel modulierte Kristallstrukturen und Phasenumwandlungen - Die Verbindungen SrPt2As2 und EuPt2As2 . Z. Anorg. Allg. Chem. 633, 2037–2045 (2007).
Kakiya, S. et al. Superconductivity at 38 K in Iron-Based Compound with Platinum–Arsenide Layers Ca10(Pt4As8)(Fe2−xPtxAs2)5 . J. Phys. Soc. Jpn. 80, 093704 (2011).
Nohara, M. et al. Iron–platinum–arsenide superconductors Ca10(PtnAs8)(Fe2−xPtxAs2)5 . Solid State Commun. 152, 635–639 (2012).
Ni, N., Allred, J. M., Chan, B. C. & Cava, R. J. High Tc electron doped Ca10(Pt3As8)(Fe2As2)5 and Ca10(Pt4As8)(Fe2As2)5 superconductors with skutterudite intermediary layers. Proc. Natl. Acad. Sci. 108, E1019–E1026 (2011).
Löhnert, C. et al. Superconductivity up to 35 K in the Iron Platinum Arsenides (CaFe1−xPtxAs)10Pt4−yAs8 with Layered Structures. Angew. Chem. Int. Ed. 50, 9195–9199 (2011).
Kudo, K., Nishikubo, Y. & Nohara, M. Coexistence of Superconductivity and Charge Density Wave in SrPt2As2 . J. Phys. Soc. Jpn. 79, 123710 (2010).
Zheng, C. & Hoffmann, R. Donor-Acceptor Layer Formation and Lattice Site Preference in the Solid: The CaBe2Ge2 Structure. J. Am. Chem. Soc. 108, 3078–3088 (1986).
Nishikubo, Y., Kudo, K. & Nohara, M. Superconductivity in the Honeycomb-Lattice Pnictide SrPtAs. J. Phys. Soc. Jpn. 80, 055002 (2011).
Goryo, J., Fischer, M. H. & Sigrist, M. Possible pairing symmetries in SrPtAs with a local lack of inversion center. Phys. Rev. B 86, 100507(R) (2012).
Ishida, K., Nakai, Y. & Hosono, H. To What Extent Iron-Pnictide New Superconductors Have Been Clarified: A Progress Report. J. Phys. Soc. Jpn. 78, 062001 (2009).
Paglione, J. & Greene, R. L. High-temperature superconductivity in iron-based materials. Nat. Phys. 6, 645–658 (2010).
Johnston, D. C. The puzzle of high temperature superconductivity in layered iron pnictides and chalcogenides. Adv. Phys. 59, 803–1061 (2010).
Brese, N. E. & von Schnering, H. G. Bonding trends in pyrites and a reinvestigation of the structures of PdAs2, PdSb2, PtSb2 and PtBi2 . Z. Anorg. Allg. Chem. 620, 393–404 (1994).
Johrendt, D. & Mewis, A. Darstellung und Kristallstrukturen der Verbindungen CaPdAs, CaPdSb und CaPdBi. Z. Anorg. Allg. Chem. 618, 30–34 (1992).
Mewis, A. The ThCr2Si2-Type and Related Structures of APd2X2 Compounds (A = Ca, Sr, Ba; X = P, As). Z. Naturforsch. B 39, 713–720 (1984).
Hieke, C. et al. Superconductivity and crystal structure of the palladium–iron–arsenides Ca10(Fe1−xPdxAs)10Pd3As8 . Phil. Mag. 93, 3680–3689 (2013).
Kjekshus, A. & Pedersen, G. The Crystal Structures of IrAs3 and IrSb3 . Acta. Cryst. 14, 1065–1070 (1961).
Katayama, N., Kiuchi, Y., Matsushita, Y. & Ohgushi, K. Variation in Electronic State of Ba(Fe1−xCox)2As2 Alloy as Investigated in Terms of Transport Properties. J. Phys. Soc. Jpn. 78, 123702 (2009).
Canfield, P. C., Bud'ko, S. L., Ni, N., Yan, J. Q. & Kracher, A. Decoupling of the superconducting and magnetic/structural phase transitions in electron-doped BaFe2As2 . Phys. Rev. B 80, 060501(R) (2009).
Kamihara, Y., Watanabe, T., Hirano, M. & Hosono, H. Iron-Based Layered Superconductor La[O1−xFx]FeAs (x = 0.05–0.12) with Tc = 26 K. J. Am. Chem. Soc. 130, 3296–3297 (2008).
Matsuishi, S. et al. Superconductivity Induced by Co-Doping in Quaternary Fluoroarsenide CaFeAsF. J. Am. Chem. Soc. 130, 14428–14429 (2008).
Tapp, J. H. et al. LiFeAs: An intrinsic FeAs-based superconductor with Tc = 18 K. Phys. Rev. B 78, 060505(R) (2008).
Rotter, M., Tegel, M. & Johrendt, D. Superconductivity at 38 K in the Iron Arsenide (Ba1−xKx)Fe2As2 . Phys. Rev. Lett. 101, 107006 (2008).
Zhu, X. et al. Sr3Sc2Fe2As2O5 as a possible parent compound for FeAs-based superconductors. Phys. Rev. B 79, 024516 (2009).
Kawaguchi, N., Ogino, H., Shimizu, Y., Kishio, K. & Shimoyama, J. New Iron Arsenide Oxides (Fe2As2)(Sr4(Sc,Ti)3O8), (Fe2As2)(Ba4Sc3O7.5) and (Fe2As2)(Ba3Sc2O5). Appl. Phys. Express 3, 063102 (2010).
Zhu, X. et al. Transition of stoichiometric Sr2VO3FeAs to a superconducting state at 37.2 K. Phys. Rev. B 79, 220512(R) (2009).
Ogino, H. et al. A new homologous series of iron pnictide oxide superconductors (Fe2As2)(Can +2(Al, Ti)nOy) (n = 2, 3, 4). Supercond. Sci. Technol. 23, 115005 (2010).
Shirage, P. M. et al. Superconductivity at 28.3 and 17.1 K in (Ca4Al2O6−y)(Fe2Pn2) (Pn = As and P). Appl. Phys. Lett. 97, 172506 (2010).
Blaha, P., Schwarz, K., Madsen, G. K. H., Kvasnicka, D. & Luitz, J. Wien2k, An Augmented Plane Wave + Local Orbitals Program for Calculating Crystal Properties, Vienna University of Technology, Wien, (2001).
Singh, D. J. & Du, M.-H. Density Functional Study of LaFeAsO1−xFx: A Low Carrier Density Superconductor Near Itinerant Magnetism. Phys. Rev. Lett. 100, 237003 (2008).
Shein, I. R. & Ivanovskii, A. L. AB INITIO STUDY OF THE NATURE OF THE CHEMICAL BOND AND ELECTRONIC STRUCTURE OF THE LAYERED PHASE Ca10(Pt4As8)(Fe2As2)5 AS A PARENT SYSTEM IN THE SEARCH FOR NEW SUPERCONDUCTING IRON-CONTAINING MATERIALS. Theor. Exp. Chem. 47, 292–295 (2011).
Takubo, K. et al. Electronic structure of Ca3CoXO6 (X = Co, Rh, Ir) studied by x-ray photoemission spectroscopy. Phys. Rev. B 71, 073406 (2005).
Sterne, P. A. & Wang, C. S. Higher Tc through metallic inter-layer coupling in Bi2Sr2CaCu2O8 . J. Phys. C: Solid State Phys. 21, L949–L955 (1988).
Tsujimoto, Y. et al. Infinite-layer iron oxide with a square-planar coordination. Nature 450, 1062–1065 (2007).
Tassel, C. & Kageyama, H. Square planar coordinate iron oxides. Chem. Soc. Rev. 41, 2025–2035 (2012).
Sugimoto, K. et al. Extremely High Resolution Single Crystal Diffractometory for Orbital Resolution using High Energy Synchrotron Radiation at SPring-8. AIP Conf. Proc. 1234, 887–890 (2010).
Acknowledgements
Part of this work was performed at the Advanced Science Research Center, Okayama University. It was partially supported by Grants-in-Aid for Scientific Research (A) (23244074) and (C) (25400372) from the Japan Society for the Promotion of Science (JSPS) and the Funding Program for World-Leading Innovative R&D on Science and Technology (FIRST Program) from the JSPS. The synchrotron radiation experiments performed at BL02B1 and BL02B2 of SPring-8 were supported by the Japan Synchrotron Radiation Research Institute (JASRI; Proposal No. 2012A0083, 2012B0083, 2013A0083 and 2013A1197).
Author information
Authors and Affiliations
Contributions
K.K. and M.N. conceived and planed the research. D.M., M.T. and K.K. synthesized single crystals. Y.S., K.S., N.K. and H.S. performed single-crystal structural analysis using synchrotron radiation X-ray diffraction. D.M. and K.K. measured electrical resistivity and magnetization. T.F. carried out Mössbauer spectroscopy. H.S.K., K.T. and M.I. conducted first-principles calculations. T.M. carried out X-ray photoelectron spectroscopy. K.K. and M.N. discussed the results and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Accession codes: The crystal structure of Ca(Ir4As8)(Fe2As2)5 has been deposited at the Cambridge Crystallographic Data Centre (http://www.ccdc.cam.ac.uk). Deposition number is CCDC 962099.
Electronic supplementary material
Supplementary Information
Supplementary Tables S1 and S2
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Kudo, K., Mitsuoka, D., Takasuga, M. et al. Superconductivity in Ca10(Ir4As8)(Fe2As2)5 with Square-Planar Coordination of Iridium. Sci Rep 3, 3101 (2013). https://doi.org/10.1038/srep03101
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep03101
This article is cited by
-
Slow Spin Dynamics in Superconducting Ca0.9Ce0.1Fe2As2
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.