Abstract
The contamination of Japan after the Fukushima accident has been investigated mainly for volatile fission products, but only sparsely for actinides such as plutonium. Only small releases of actinides were estimated in Fukushima. Plutonium is still omnipresent in the environment from previous atmospheric nuclear weapons tests. We investigated soil and plants sampled at different hot spots in Japan, searching for reactor-borne plutonium using its isotopic ratio 240Pu/239Pu. By using accelerator mass spectrometry, we clearly demonstrated the release of Pu from the Fukushima Daiichi power plant: While most samples contained only the radionuclide signature of fallout plutonium, there is at least one vegetation sample whose isotope ratio (0.381 ± 0.046) evidences that the Pu originates from a nuclear reactor (239+240Pu activity concentration 0.49 Bq/kg). Plutonium content and isotope ratios differ considerably even for very close sampling locations, e.g. the soil and the plants growing on it. This strong localization indicates a particulate Pu release, which is of high radiological risk if incorporated.
Similar content being viewed by others
Introduction
The accident at the Fukushima Daiichi nuclear power plant (FDNPP) in 2011 caused massive contaminations of the Japanese land surface and the Pacific Ocean with mainly volatile fission products. In the course of the venting operations large amounts of radionuclides escaped from the reactor pressure vessels. The release was enhanced by three massive hydrogen explosions that occurred in Units 1, 3 and 4. In the course of the accident, large areas in Japan were contaminated with 131I, 132Te, 134Cs, 137Cs and other volatile radionuclides1,2,3,4. Numerous studies revealed these airborne radionuclides in the entire northern hemisphere. Radioisotopes of refractory elements such as strontium5,6,7, barium and lanthanides were monitored only in rare occasions8. Actinides, however, proved to be the most severely understudied group of elements, despite the fact that many of them are long-lived α-emitters: most importantly 239Pu (half-life 24,110 y) and 240Pu (half-life 6,561 y). Schwantes et al.9 estimated releases of only 0.002% (±0.003%) of the overall plutonium inventory (>5.6 kg in units 1 and 3)9 into the environment. Recent analyses by Zheng et al. indicated even lower plutonium releases of only 0.00002% of the core inventories10. Only few studies targeted the monitoring of actinides (most importantly plutonium) in environmental media (including ocean and river water)7,11,12,13,14,15. Most profound evidence for atmospheric releases of plutonium from Fukushima was presented in the study by Zheng et al.11.
Since the atmospheric nuclear weapon tests of the 20th century, plutonium has become a ubiquitous element in the environment. However, using the atomic ratio 240Pu/239Pu as an isotopic signature, it is possible to distinguish between the weapons' fallout (240Pu/239Pu ca. 0.18)16 and plutonium releases from a nuclear reactor (240Pu/239Pu ca. 0.4–0.6). Due to the nuclides' very similar α-particle energies, radioanalytical methods can usually not be used to distinguish the isotopic ratios between 239Pu and 240Pu. Instead, low-ionization MS techniques17,18 or accelerator mass spectrometry (AMS) have established as powerful alternatives. AMS is currently regarded as the most sensitive method for the determination of the isotopic composition of environmental plutonium.
Previous indications for the release of actinides from Fukushima, in particular environmental abundance of 239Np (the parent nuclide of 239Pu), were presented by Shozugawa et al.19. In that study, 239Np was detected using gamma spectrometry. Spectral overlap of the most intense peak of 239Np with the fission product 129mTe, however, made it necessary to investigate both soil and vegetation samples from selected hot spots in the present study using AMS and to search for the isotopic plutonium-signatures at these spots.
Results
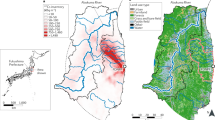
Soil and vegetation samples taken from several Japanese hot spots in 2011 (Fig. 1 and Table 1) were investigated with special focus on anthropogenic actinides, in particular plutonium.
Locations of Fukushima Daiichi nuclear power plant (FDNPP) and samples of this study using the codes of Table 1.
The α-spectrometric and AMS results with respect to the atom ratios 240Pu/239Pu as well as detectable activity concentrations of 239Pu and 240Pu of soil and plant samples are listed in Table 2. Just in a few samples, plutonium could be detected using α-spectrometry. Also, for most samples, AMS revealed no detectable amounts of plutonium. For signals below the decision threshold (according to ISO 11929)20 only an upper limit for the 240Pu/239Pu ratio is given. If 239Pu was above the decision threshold, but 240Pu was not, an upper limit for the atomic ratio is given. If neither of the isotopes could be detected by AMS the respective table entry reads “n.d” (not detected). The small amounts available did not allow for splitting of the samples and hence a 242Pu spike concentration was added forming a compromise between high alpha-activity required by alpha spectroscopy and low atom-concentration desirable for AMS measurements. Hence, AMS results of the present work suffer from a very high background count rate.
Soil sample H-S2 revealed a 240Pu/239Pu ratio and AMS signal strength characteristic for global fallout; hence no quantification was undertaken for this sample. However, vegetation sample, A-V revealed a significantly higher 240Pu/239Pu atomic ratio (0.381 ± 0.046). With a much higher analytical uncertainty, sample G-V also may carry an isotopic signature (0.64 ± 0.37) that is higher than the omnipresent fallout from the nuclear weapons tests. Such high ratios are characteristic for nuclear reactor plutonium and hence evidence the release of small but detectable amounts of plutonium from the damaged reactors of FDNPP.
Discussion
Of 20 samples investigated in this study, 17 did not exceed the detection limit for plutonium. One soil sample was contaminated only by global fallout plutonium with its characteristic isotopic ratio of 240Pu/239Pu < 0.2. However, at least one (A-V) or two (G-V; higher uncertainty) of the vegetation samples showed detectable amounts of reactor derived plutonium (isotopic ratio 240Pu/239Pu > 0.2). One can assume that the ubiquitous fallout plutonium background masked the minute contribution of Fukushima-derived plutonium in soil, as illustrated by Zheng et al.11 However, given the low mobility and bioavailability of plutonium, one can expect that plant uptake of fallout plutonium will be negligible. Consequently, dry or wet deposition of airborne plutonium on the surface of the plants will be highly visible for sensitive analytical techniques such as AMS. This probably explains why plants proved to be such suitable bioindicators for airborne plutonium from Fukushima in the present study.
It is remarkable to note that distance alone is no sufficient factor to estimate the findings of refractory elements such as plutonium. Although the vegetation sample taken closest to the reactors (~0.9 km away) exhibited detectable amounts of reactor-plutonium, no other sample in close vicinity of the reactors (1.5, 1.9,… km away) did so. However, a plant sample as far as 16 km away in north-northwestern direction (G-V) is suspected to contain plutonium from Fukushima. If this observation was confirmed, it would indicate a very nonuniform distribution of plutonium, most probably in particulate form. This may also have health physical implications because the inhalation of such plutonium-rich particles may result in high local dose delivery to the lung tissue.
There is common agreement among the scientific community that only minute amounts of actinides have been released in the course of the Fukushima nuclear accident. Schwantes et al.9 calculated the average activity inventories of Units 1 and 3, from which a 240Pu/239Pu atomic ratio of 0.441 can be derived. A similar value can be derived from the activity inventory data published by Kirchner et al.21, in particular 0.393. Sakaguchi et al.12 estimated the ratio to more than 0.4. All these predicted, calculated or estimated values are in good agreement with the ratio observed in sample A-V (0.381 ± 0.046) and also within the analytical uncertainty observed in G-V (0.64 ± 0.37). Zheng et al. observed somewhat lower values for soil and litter11 (between 0.30 and 0.33) and concluded that mixing with global fallout plutonium shifted the ratio to lower numbers. However, the actual composition of a particle also can vary from average activity inventory of an entire reactor, depending on its nuclear “history”21. Also some deviations between uranium-operated Unit 1 and MOX-operated Unit 3 can be expected. Sakaguchi et al. found an increased 240Pu/239Pu isotopic ratio (0.308 ± 0.176) in water from Abukuma River, which fostered the need for further analysis due to relatively large analytical uncertainties.
In any case, the sector field ICP-MS used by Zheng et al. proved to produce results for the isotopic ratio that are quite comparable with AMS of this study. The absolute activity concentrations for 239+240Pu are also comparable to previously published data11,13, only slightly higher. These values, however, are still within the range of plutonium concentrations found before the Fukushima nuclear accident (0.15–4.31 mBq/g)11,22. The sum activity of 239+240Pu alone, therefore, provides no sufficient evidence for plutonium releases from FDNPP.
In this context, it is interesting to note that only vegetation samples but none of the soil samples exhibited detectable traces of reactor plutonium. Since the Fukushima nuclear accident seemingly did not significantly increase the environmental plutonium inventory, one can hypothesize that the global plutonium background in soil dilutes the isotopic ratio and blurs the characteristic isotopic fingerprint of much smaller amounts of freshly deposited plutonium particles. Given the low environmental mobility and bioavailability of plutonium, vegetation collects airborne particles on its surface with a negligible background due to “old” plutonium uptake. Future findings of plutonium particles on plant material are increasingly unlikely, because not only atmospheric releases of actinides have ceased since 2011, but also rainfall will have washed down particles from the plant surface or vegetation cycles will probably have renewed the exposed leaf surface. In any case, the plutonium activities found in the vegetation samples (grass, leaves) sampled in late 2011 were (already?) so low that, assuming they were agricultural plants, the plutonium activity concentrations would not have exceeded any of the early regulatory limits for α-emitting radionuclides (actinides) in food23.
The results of this study confirm the very low release of refractory elements from the Fukushima reactors. The plutonium concentrations found herein and reported by Zheng et al.11 are partly more than three orders of magnitude lower than the values obtained in environmental samples around the Chernobyl site after 198624. This is also true for the semi-volatile radionuclide 90Sr that has been monitored only occasionally after the Fukushima accident and revealed relatively low activity concentrations in environmental samples in Japan. As described in a previous study6, a vegetation sample from spot G (a spot that is also suspected to be contaminated with reactor plutonium according to Table 2) also carried a comparatively high 90Sr contamination, but a rather low radiocesium activity concentration (activity ratio 90Sr/137Cs approximately 0.1). This 90Sr over 137Cs activity ratio was found to be much smaller with all the other samples investigated in the previous study6. Spot G is located outside the main “contamination strip” that goes from the reactor in northwestern direction, which makes the presence of reactor plutonium even more unexpected. If the findings of reactor plutonium at spot G are confirmed, one may speculate what the reason for this unusual radionuclide pattern is (high concentrations of refractory radionuclides, but relatively low in volatile radionuclides). One possible explanation could be that this spot was contaminated with fuel particles that have experienced temperatures high enough to volatilize most of their radiocesium content, before or while they were emitted from the reactors. However, this hypothesis needs further investigations. In any case, it seems that there is not necessarily a correlation between the levels of radiocesium (and other volatile radionuclides) and the presence of reactor plutonium in the environment. This observation makes it likely that the release of plutonium was a more singular event, whereas the volatile radionuclides were released from the pressure vessels over several days in the course of the early venting operations.
In summary, our study evidenced the release of plutonium from the damaged FDNPP via its isotopic fingerprint. Two vegetation samples exhibited 240Pu/239Pu isotopic ratios of 0.381 ± 0.046 and 0.64 ± 0.37, respectively, both of which are higher than the global fallout background. The 239+240Pu activity concentrations, however, were relatively low (0.49 and 0.17 Bq·kg−1, respectively), confirming early predictions of a low plutonium release from Fukushima. The fact that reactor plutonium has not been found in more than two samples (one of which remains more in question) indicates that plutonium releases and fallout from FDNPP occurred in the form of particulates causing nonuniform plutonium contaminations. Future investigations will aim at a comprehensive screening for plutonium containing fuel particles in larger samples and if applicable detailed investigation of particles by single particle analytical techniques. Detection limits and decision thresholds will be lowered by use of high purity spikes. Furthermore, a 244Pu spike can be used if no alpha measurements are performed on the same sample. This can further improve the performance of AMS on such environmental samples.
If confirmed, a release of plutonium-rich hot particles is of potential health concern upon inhalation or incorporation. Our findings demonstrate the need for more detailed investigations on plutonium distribution and speciation in order to assess potential radiological consequences for the public. In any case, our study supports previous findings that indicated that the environmental plutonium inventory in Japan has not significantly increased after the Fukushima nuclear accident.
Methods
Sampling
Samples were taken between October and December 2011 from several sites on the eastern Japanese coast (Fig. 1). For the sampling in restricted areas permission was obtained from the mayor of Okuma town. Some of the spots have already been investigated previously with respect to short-lived γ-emitting radionuclides19 and 90Sr6. On most spots two types of samples were taken: top soil and vegetation (leaves, grass, conifer needles etc.). The sampling sites included highly contaminated spots such as the gate of the FDNPP (Fukushima I), locations in the close vicinity of the NPP including Fukushima Daini NPP (Fukushima II), but also more remote areas such as Yokohama or Kashiwa (some 200 km south of FDNPP). Exact locations and sampling dates are given in Table 1.
Sample preparation and measurement
All samples were dried at 105°C. All given specific-activities and masses are referring to the dry mass of the respective sample.
Soil was available in relatively low quantities, ranging typically between 1–2 g, with the exception of the samples H-S1 and H-S2, which were available in greater amounts. Hence, we refrained from separating the soil skeleton to avoid additional sample losses. The samples were ashed with a low heating-up rate over 103 h up to 450°C. The ash was spiked with about 30 mBq 242Pu and digested using concentrated nitric acid and concentrated hydrofluoric acid. The dry residue was then taken up in a solution of 1 M aluminum nitrate and 3 M nitric acid and filtered. The chemical separation of plutonium was performed by extraction chromatography using Eichrom® TEVA resin25. After the separation the sample was transferred to a stainless steel plate via electrodeposition25,26. The electrodeposition took 2 h at a constant current of 950 mA. After alpha spectrometry had been conducted, the electrodeposited material was re-dissolved in hot nitric acid (3 M) from the alpha-plate for AMS measurement. To optimize the separation from uranium, a second separation step was applied. Plutonium was subsequently co-precipitated by ferric hydroxide and converted into ferric oxide by heating up to 800°C25. Finally, the sample was mixed with aluminum powder in a ratio of 1:1 and pressed into an AMS target holder.
Basically the same procedure was applied for plant samples with some slight changes. The ashing procedure was performed with a heating program over 20 h up to 600°C. We could not perform alpha-spectrometric measurements, due to the very low sample quantities of 26–230 mg of the sample ash (this fact is referred to as “N/A” (not applicable) in Table 2). For the same reason reduced amounts of only 5 mBq 242Pu were spiked. Furthermore, the digestion procedure was changed to microwave-pressure-digestion using concentrated nitric (8 mL), hydrochloric (5 mL) and hydrofluoric acid (2 mL) at 210°C. For the AMS-target, the aluminum powder was replaced by niobium because recent tests have shown that uranium and plutonium samples show higher counting rates when dispersed in a niobium matrix.
Accelerator mass spectrometry (AMS)
The AMS measurement was performed at the Laboratory of Ion Beam Physics of ETH Zurich, Switzerland, using the compact, low energy 0.6 MV AMS facility “Tandy”27. Over the past few years, the Tandy system was upgraded with an additional magnet on the high energy side and equipped with an improved ion source28. Since 2011, helium is used as a stripper gas for all measurements on the Tandy29. With a transmission of up to 40% for uranium and thorium29, an abundance sensitivity of up to 10−12 and detection limits in the sub-femtogram range for plutonium isotopes30, the compact (lab-sized) ETH Zurich AMS system Tandy is well suited to detect ultra-trace amounts of actinides.
The setup for plutonium measurements is summarized in ref. 30. Briefly, negative PuO ions are extracted from the ion source on the low energy side and accelerated by a terminal voltage of about 300 kV. At the terminal, He stripping is used to destroy any interfering molecular ions and Pu3+ is selected by the magnetic and electrostatic filters on the high energy side. Finally, plutonium ions are identified in a dedicated low noise gas ionization detector31. Generally, different plutonium isotopes are injected and counted sequentially. The measurement time for each plutonium isotope can be adjusted before the run according to the expected counting rate of the samples.
In this study, 239Pu and 240Pu were measured for 10 s and 20 s each, while the 242Pu tracer was counted for 8 s. In addition, the background counting rate on atomic mass unit (amu) 239 (caused by “tailing” of 238U) was monitored for 5 s on each sample by injecting 238U16O− into the accelerator while leaving the high energy side tuned to amu 239. The above sequence was repeated five times (i.e. one pass) before measuring the next sample. The isotopic ratios of interest were calculated from the average counting rates acquired during this pass. The whole measurement procedure as described above was repeated 9 –14 times (passes) for each sample. The final plutonium isotopic ratios of the samples were calculated from the error weighted mean of all single measurements (passes). In total, each sample was measured between 35 and 60 minutes.
All plutonium isotopic ratio measurements were normalized to the ETH in-house plutonium standard “CNA”32. Corrections for 238U induced background on amu 239 were below 1% and therefore negligible. The (one sigma) uncertainty of the measured 240Pu/239Pu ratios was typically 5% for all the soil samples and about 10% for the vegetation samples. Three vegetation samples, however, revealed relatively low counting rates on the spike (probably due to low chemical recovery) so that the uncertainty of these samples was higher (10–20%).
Unfortunately, the 242Pu spike material used in this study also contained significant amounts of 239Pu and 240Pu so that a correction for spike-supported Pu-isotopes was necessary. To minimize the uncertainty of this correction 3 and 7 spike samples were measured together with the soil samples and the vegetation samples, respectively. The average 239/242 and 240/242 ratios of the spike were used to correct the corresponding isotopic ratios of the samples after each run. The final 240/239 ratios (as reported in Table 2) were calculated from the spike-corrected 239/242 and 240/242 ratios of the samples. Although several spike samples were averaged, the final (one sigma) uncertainty of the 240/239 ratio (as given in Table 2) is still dominated by the propagated uncertainty of the spike correction. For this reason only upper limits can be reported for most of the 240/239 ratios in Table 2. To avoid this problem in future studies a cleaner spike material has to be used. It would also be advantageous if greater sample amounts were available in future studies. Alternatively and if only the ratio 240/239 is of interest, samples can be prepared and measured without the addition of any spike material.
The above method was validated using IAEA reference materials (IAEA-375, IAEA-Soil-6 and IAEA-384).
Alpha spectrometry
Alpha-spectrometry was performed using the Canberra Model 7200 Alpha Analyst Integrated Alpha Spectrometer. A passivated implanted planar silicon (PIPS) detector with an active area of 450 mm2 (Canberra, Model A450-18AM) was used. The samples were measured for 7 days and the received spectra evaluated using the software program Genie 2000, Alpha Acquisition & Analysis, V 2.0, Mar2, 2001 from Canberra Industries. The method was validated by IAEA reference material (IAEA-375, IAEA-Soil-6 and IAEA-384).
References
Yasunari, T. J. et al. Cesium-137 deposition and contamination of Japanese soils due to the Fukushima nuclear accident. Proc. Natl. Acad. Sci. USA 108, 19530–19534 (2011).
Kinoshita, N. et al. Assessment of individual radionuclide distributions from the Fukushima nuclear accident covering central-east Japan. Proc. Natl. Acad. Sci. USA 108, 19526–19529 (2011).
Tagami, K. et al. Specific activity and activity ratios of radionuclides in soil collected about 20 km from the Fukushima Daiichi Nuclear Power Plant: Radionuclide release to the south and southwest. Sci. Total Environ. 409, 4885–4888 (2011).
Stohl, A., Seibert, P. & Wotawa, G. The total release of xenon-133 from the Fukushima Dai-ichi nuclear power plant accident. J. Environ. Radioact. 112, 155–159 (2012).
Povinec, P. P., Hirose, K. & Aoyama, M. Radiostrontium in the Western North Pacific: Characteristics, Behavior and the Fukushima Impact. Environ. Sci. Technol. 46, 10356–10363 (2012).
Steinhauser, G., Schauer, V. & Shozugawa, K. Concentration of strontium-90 at selected hot spots in Japan. PLoS One 8, e57760 (2013).
Kim, C.-K. et al. Radiological impact in Korea following the Fukushima nuclear accident. J. Environ. Radioact. 111, 70–82 (2012).
Loaiza, P. et al. Air radioactivity levels following the Fukushima reactor accident measured at the Laboratoire Souterrain de Modane, France. J. Environ. Radioact. 114, 66–70 (2012).
Schwantes, J. M., Orton, C. R. & Clark, R. A. Analysis of a Nuclear Accident: Fission and Activation Product Releases from the Fukushima Daiichi Nuclear Facility as Remote Indicators of Source Identification, Extent of Release and State of Damaged Spent Nuclear Fuel. Environ. Sci. Technol. 46, 8621–8627 (2012).
Zheng, J., Tagami, K. & Uchida, S. Release of plutonium isotopes into the environment from the Fukushima Daiichi nuclear power plant accident: what is known and what needs to be known. Environ. Sci. Technol. 47, 9584–9595 (2013).
Zheng, J. et al. Isotopic evidence of plutonium release into the environment from the Fukushima DNPP accident. Sci. Rep. 2, 304 (2012).
Sakaguchi, A. et al. Isotopic determination of U, Pu and Cs in environmental waters following the Fukushima daiichi nuclear power plant accident. Geochem. J. 46, 355–360 (2012).
Yamamoto, M. et al. An early survey of the radioactive contamination of soil due to the Fukushima Dai-ichi Nuclear Power Plant accident, with emphasis on plutonium analysis. Geochem. J. 46, 341–353 (2012).
Zheng, J., Aono, T., Uchida, S., Zhang, J. & Honda, M. C. Distribution of Pu isotopes in marine sediments in the Pacific 30 km off Fukushima after the Fukushima Daiichi nuclear power plant accident. Geochem. J. 46, 361–369 (2012).
Lujaniene, G., Valiulis, D., Bycenkiene, S., Sakalys, J. & Povinec, P. P. Plutonium isotopes and 241Am in the atmosphere of Lithuania: A comparison of different source terms. Atmos. Environ. 61, 419–427 (2012).
Krey, P. W. et al. Mass isotopic composition of global fall-out plutonium in soil. in IAEA-SM-199-39. 671–678 (IAEA).
Walther, C. & Denecke, M. A. Actinide Colloids and Particles of Environmental Concern. Chem. Rev. 113, 995–1015 (2013).
Kersting, A. B. et al. Migration of plutonium in ground water at the Nevada Test Site. Nature 397, 56–59 (1999).
Shozugawa, K., Nogawa, N. & Matsuo, M. Deposition of fission and activation products after the Fukushima Dai-ichi nuclear power plant accident. Environ. Pollut. 163, 243–247 (2012).
International Organization of Standardization. Determination of the characteristic limits (decision threshold, detection limit and limits of the confidence interval) for measurements of ionizing radiation — Fundamentals and application, <https://www.iso.org/obp/ui/#iso:std:iso:11929:ed-1:v1:en> (2010); accessed May 2013.
Kirchner, G., Bossew, P. & De, C. M. Radioactivity from Fukushima Dai-ichi in air over Europe; part 2: what can it tell us about the accident? J. Environ. Radioact. 114, 35–40 (2012).
Muramatsu, Y., Yoshida, S. & Tanaka, A. Determination of Pu concentration and its isotope ratio in Japanese soils by HR-ICP-MS. J. Radioanal. Nucl. Chem. 255, 477–480 (2003).
Merz, S., Steinhauser, G. & Hamada, N. Anthropogenic Radionuclides in Japanese Food: Environmental and Legal Implications. Environ. Sci. Technol. 47, 1248–1256 (2013).
Steinhauser, G., Brandl, A. & Johnson, T. E. Comparison of the Chernobyl and Fukushima nuclear accidents: a review of the environmental impacts. Sci. Total Environ., in press; 10.1016/j.scitotenv.2013.10.029.
Bisinger, T., Hippler, S., Michel, R., Wacker, L. & Synal, H. A. Determination of plutonium from different sources in environmental samples using alpha-spectrometry and AMS. Nucl. Instrum. Meth. B 268, 1269–1272 (2010).
Lee, M. H. & Lee, C. W. Preparation of alpha-emitting nuclides by electrodeposition. Nucl. Instrum. Methods Phys. Res., Sect. A 447, 593–600 (2000).
Stocker, M. et al. A universal and competitive compact AMS facility. Nucl. Instrum. Meth. B 240, 483–489 (2005).
Müller, A. M., Christl, M., Lachner, J., Suter, M. & Synal, H. A. Competitive 10Be measurements below 1 MeV with the upgraded ETH–TANDY AMS facility. Nucl. Instrum. Meth. B 268, 2801–2807 (2010).
Vockenhuber, C. et al. The potential of He stripping in heavy ion AMS. Nucl. Instrum. Meth. B 294, 382–386 (2013).
Dai, X., Christl, M., Kramer-Tremblay, S. & Synal, H.-A. Ultra-trace determination of plutonium in urine samples using a compact accelerator mass spectrometry system operating at 300 kV. J. Anal. At. Spectrom. 27, 126–130 (2012).
Suter, M. et al. Advances in particle identification in AMS at low energies. Nucl. Instrum. Meth. B 259, 165–172 (2007).
Christl, M. et al. The ETH Zurich AMS facilities: Performance parameters and reference materials. Nucl. Instrum. Meth. B 294, 29–38 (2013).
Acknowledgements
This publication was supported by Grant Number T42OH009229-07 from CDC NIOSH Mountain and Plains Education and Research Center. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the CDC NIOSH and MAP ERC. GS gratefully acknowledges funding by the US Nuclear Regulatory Commission (NRC), grant number NRC-HQ-12-G-38-0044. The Laboratory of Ion Beam Physics is partially funded by its consortium partners EAWAG, EMPA and PSI.
Author information
Authors and Affiliations
Contributions
K.S. provided materials, S.S., C.W., S.B., V.S., M.C., H.A.S., G.S. performed the experiments, all authors discussed the data and results. S.S. and G.S. wrote the paper and prepared Fig. 1. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Schneider, S., Walther, C., Bister, S. et al. Plutonium release from Fukushima Daiichi fosters the need for more detailed investigations. Sci Rep 3, 2988 (2013). https://doi.org/10.1038/srep02988
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep02988
This article is cited by
-
Identification, isolation, and characterization of a novel type of Fukushima-derived microparticle
Journal of Radioanalytical and Nuclear Chemistry (2022)
-
Reference materials for quality assurance of environmental plutonium analysis
Journal of Radioanalytical and Nuclear Chemistry (2020)
-
Isotopic composition and source of plutonium in the Qinghai-Tibet Plateau frozen soils
Scientific Reports (2019)
-
First determination of Pu isotopes (239Pu, 240Pu and 241Pu) in radioactive particles derived from Fukushima Daiichi Nuclear Power Plant accident
Scientific Reports (2019)
-
Method for detecting and characterising actinide-bearing micro-particles in soils and sediment of the Fukushima Prefecture, Japan
Journal of Radioanalytical and Nuclear Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.